Abstract
Introduction
Breast cancer is a disease with high global prevalence. Clinical inflammatory biomarkers have been proposed as prognostic indicators in oncology. This research aims to determine the relationship between inflammatory markers and overall survival in breast cancer patients from four representative hospitals in Lima, Peru.
Methods
This is a multicentric, analytical, longitudinal retrospective cohort study with survival analysis in female patients with breast cancer, from 2015 to 2020, who had received at least one complete treatment regimen. The dependent variable was overall survival, and the independent variables were inflammatory markers neutrophil lymphocyte ratio, platelet lymphocyte ratio (PLR), albumin, and red cell distribution width; intervening variables included age, clinical stage, molecular subtype, and other known prognostic factors. The Kaplan–Meier method was applied to generate survival curves with the Log-Rank test, and finally, Cox regression, to find crude and adjusted hazard ratios (HR).
Results
Of 705 evaluated patients, 618 were analyzed. The mean age was 56.6 ± 12.3 years, 18.0% of patients were pure HER2 positive, 39.3% luminal A, 29.9% luminal B, 11.0% triple-negative, and 81.4% showed overweight and obesity. The average overall survival was 51.1 months. In the multivariate analysis, factors significantly related to lower overall survival were PLR > 150 (adjusted HR: 2.33; 95% confidence interval (CI): 1.22, 4.44) and stage III (adjusted HR: 4.15; 95% CI: 1.35, 12.83).
Conclusions
The Elevated Platelet-Lymphocyte Index and advanced clinical stage were associated with lower overall survival in breast cancer patients. Furthermore, PLR >150 proved to be an independent prognostic factor for mortality.
Keywords
Introduction
Breast cancer is the most frequently diagnosed neoplasm in women worldwide, representing approximately 2.3 million of all cancer cases, 1 thus it is the second leading cause of cancer death among women. 2
Several translational medicine studies have reported that the interaction between the tumor and the host microenvironment, including inflammation and immune response, plays a significant role in tumor progression and prognosis.3–7 Inflammatory biomarkers have been proposed as prognostic indicators in oncology, and several have been studied: neutrophil-lymphocyte ratio (NLR), neutrophil-monocyte ratio (NMR), platelet-lymphocyte ratio (PLR), red cell distribution width (RDW), among others.
Recent studies have confirmed that tumor-related inflammation is a critical component of the tumor microenvironment and that inflammatory cells can contribute to the onset, development, and prognosis of cancer.8,9 The systemic inflammatory response is associated with tumor progression and affects the host's immune response to malignant tumors. 10
Globally, the PLR is an informative marker that reflects changes in platelet and lymphocyte counts. An elevated Platelet-Lymphocyte Index is related to a worse prognosis, according to recent meta-analysis. 2
Recently, the PLR has become an attractive, convenient, and cost-effective blood-derived marker, as well as an inflammation- and immune system-related prognostic score for assessing the prognosis of various solid tumors.8–11
The association between platelets and breast cancer was first shown in 1968, 12 when a 50% reduction in tumor metastases was observed in an animal model after experimental thrombocytopenia. The antimetastatic effect was blocked with the infusion of platelet-rich plasma. A high platelet count was associated with better survival of cancer cells in circulation, enhanced adhesion of tumor cells to endothelial cells, penetration of cancer cells into the parenchyma of distant tissues, and increased metastasis to distant sites. 13
However, the correlation of PLR with the prognosis of breast cancer is still debatable, and it is not known whether it can predict the clinical–pathological characteristics of breast cancer.14–19
Few studies have been published regarding PLR in the Peruvian and Latin American population. In addition, current studies on breast cancer prognostic prediction using PLR are inconsistent, and sample sizes are small.12,20–22
Therefore, the objective of this study was to determine the relationship between inflammatory markers and overall survival (OS) in breast cancer patients from four representative hospitals in Lima, Peru.
Methods
Research method and design
This was a multicentric, analytical, longitudinal retrospective cohort study with survival analysis in female patients with breast cancer, during the period 2015–2020.
Population or universe, setting or context of study
The patients included in the study were diagnosed with breast cancer between 2015 and 2020, in four reference hospitals in Lima, Peru.
Sample or participants
Of 705 evaluated patients, all those who met the selection criteria were included: 618 patients. Statistical power was calculated, obtaining 85% according to the PLR variable
23
with a
Characteristics of the sample or participants
Inclusion criteria were:
Patients over 18 years of age, who were hospitalized in the oncology departments of the participating hospitals; Patients with a complete blood count (inflammatory indices) and a full hepatic profile dosage at the time of diagnosis and prior to the initiation of treatment; Patients who had received at least one complete chemotherapy treatment regimen. (All included patients received neoadjuvant or adjuvant chemotherapy with/without trastuzumab regardless of subsequent complementary hormonal treatment). Patients with breast cancer without a report by immunohistochemistry or fluorescent in-situ hybridization (FISH); Medical histories that did not contain all the data of interest for the research; Pregnant women diagnosed with breast cancer; Patients who had active infectious diseases, autoimmune diseases, and chronic steroid administration.
Exclusion criteria were:
Variables used
The dependent variable was OS, and the independent variables were the inflammatory markers NLR with a cutoff point ≥2.44 and below, 13 PLR with a cutoff point ≥150 and <150, 14 albumin with a cutoff value of 3.5 g/dL 15 and RDW ≥15 vs. <15 16 ; intervening variables were age (≥60 vs. >60), 17 clinical stage (locally vs. locally advanced), 18 molecular subtype, 19 and other known prognostic factors.
Data collection instruments
Information was gathered from the clinical histories of patients diagnosed with breast cancer from the oncology department of the hospitals. The instrument used was the data collection form, which consisted of four items; the study variables were collected from the individual forms:
• Item one: Patient identification data;
• Item two: Clinical-pathological data of breast cancer;
• Item three: Laboratory data;
• Item four: Treatment received.
Data processing techniques
Data were collected using Microsoft Excel, and then analyzed with STATA software version 12.
Procedures
The participating centers obtained and sent their data based on the information from the clinical histories in the data collection form developed by the research authors.
Ethical aspects
The project was approved by the Ethics Committee for Research of the Faculty of Human Medicine at Ricardo Palma University, code PG 103-2022, as well as the respective approvals in the participating institutions. Letter 09 AA-11/5/15.00 (HMC), PG- 49-021 (HN2 M) Letter 0210-202-DG-HNDM, Approval letter (HMA/CIEI/044/2021) (HMA).
Results
Out of 705 evaluated patients, 618 breast cancer patients were included for analysis. The average age was 56.6 ± 12.3 years; 18.0% of the patients were pure HER2 positive, 39.3% luminal A, 29.9% luminal B, and 11.0% triple negative; 81.4% of the patients showed overweight and obesity. General data are mentioned in Table 1 and Table 2.
Clinical-pathological characteristics of patients with breast cancer.
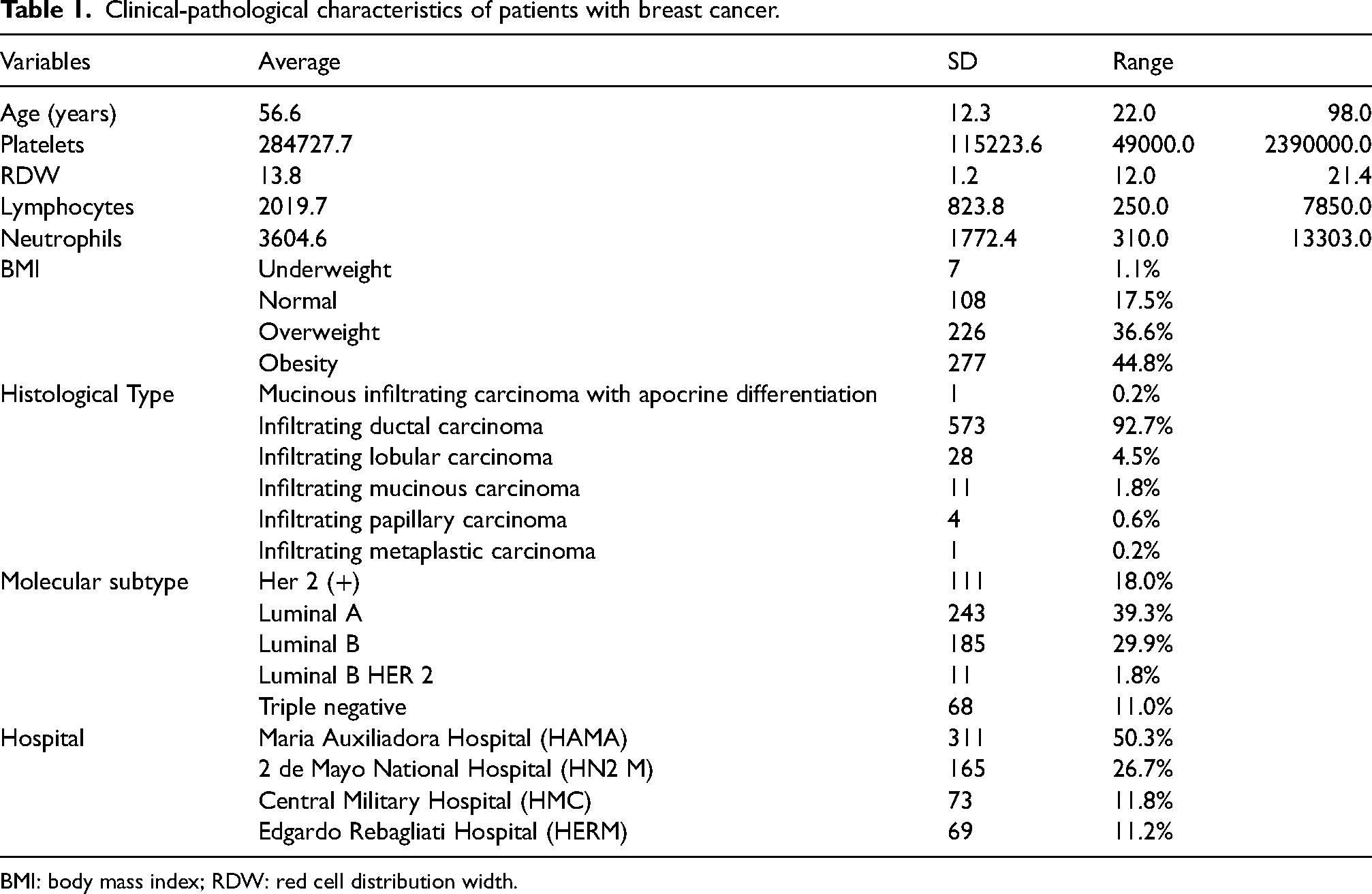
BMI: body mass index; RDW: red cell distribution width.
Stage and treatment of patients with breast cancer.
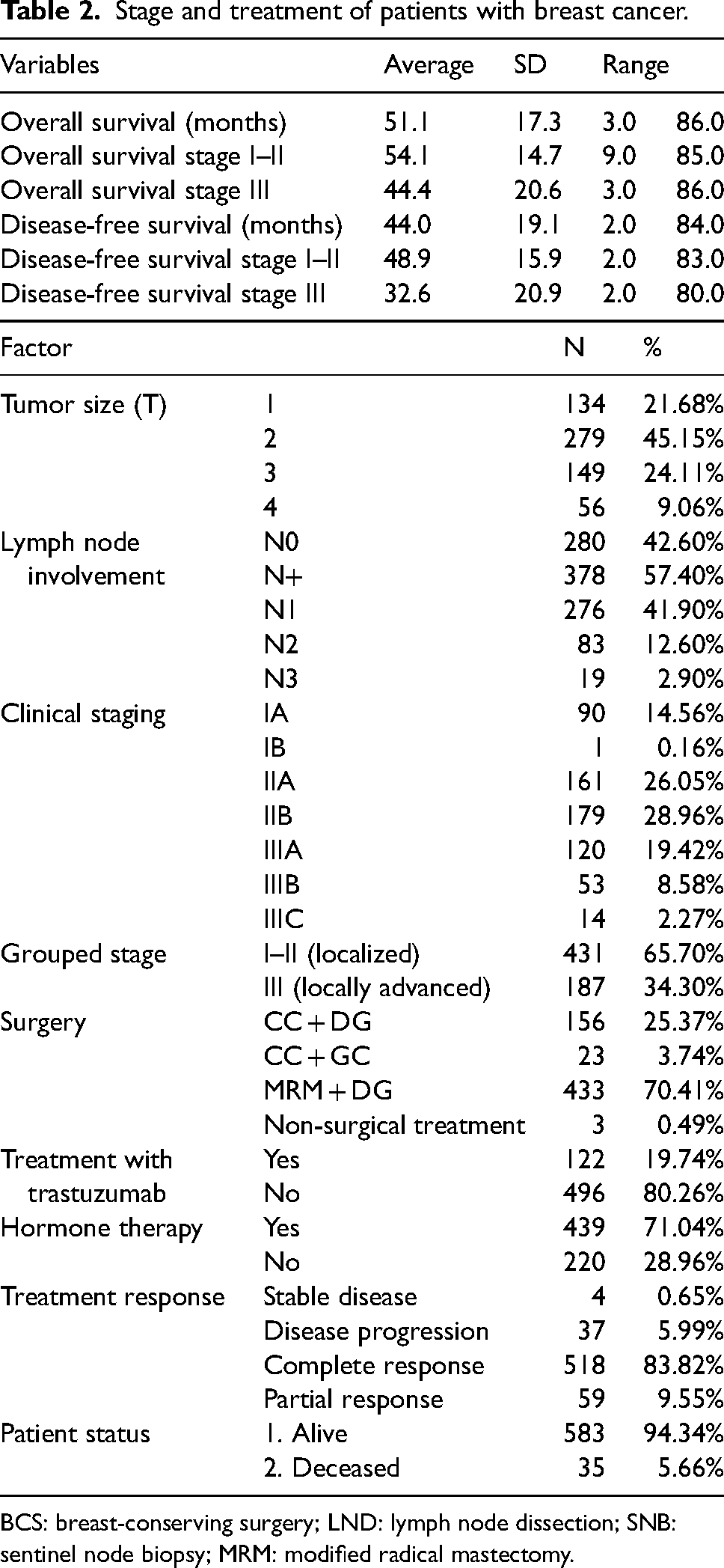
BCS: breast-conserving surgery; LND: lymph node dissection; SNB: sentinel node biopsy; MRM: modified radical mastectomy.
By multivariate analysis (Table 3), the factors significantly related to OS were PLR > 150 (adjusted HR: 2.33 95% CI: 1.22, 4.44) and stage III (adjusted HR: 4.15 95% CI: 1.35, 12.83).
Factors associated with mortality in breast cancer patients (mortality related to cancer).
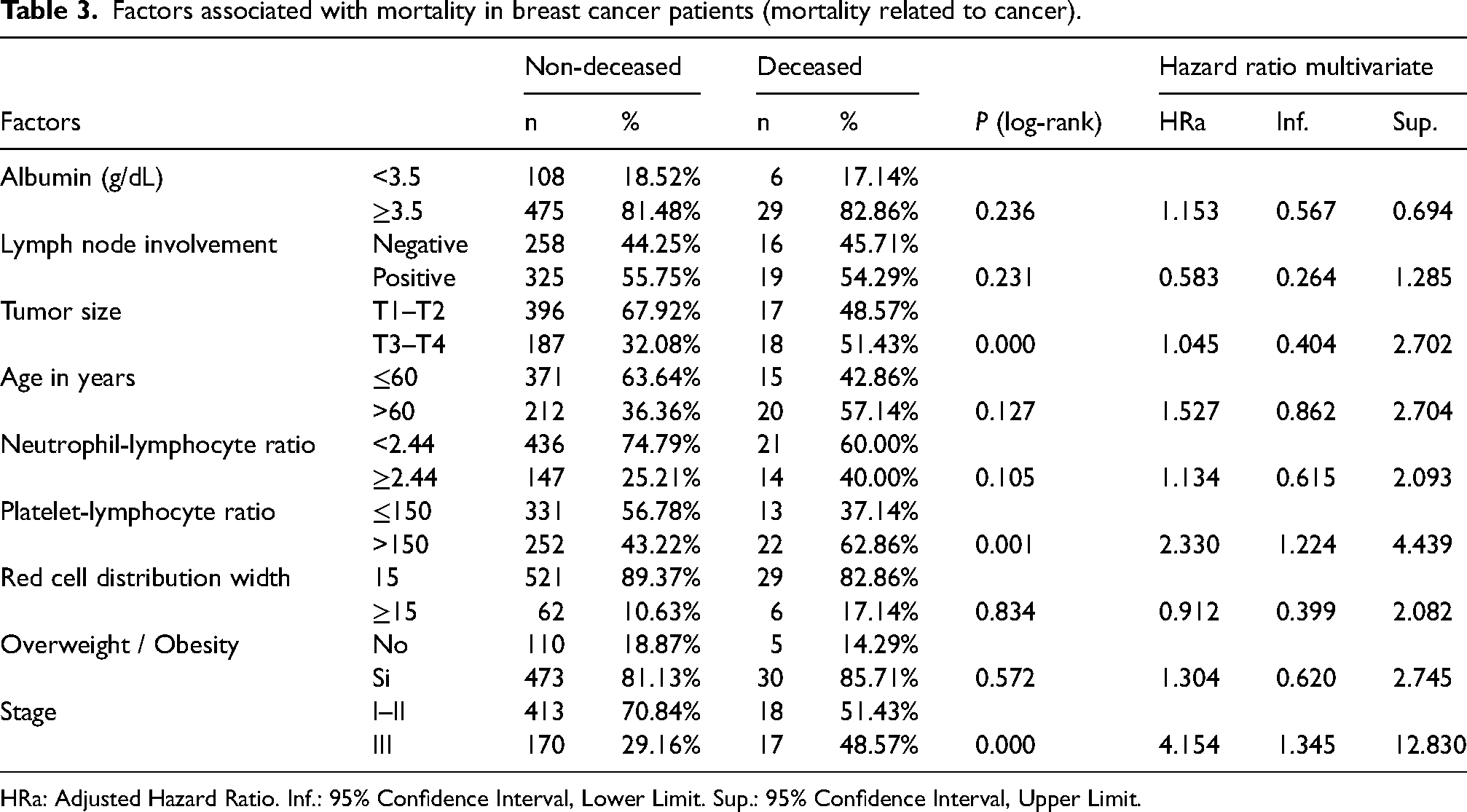
HRa: Adjusted Hazard Ratio. Inf.: 95% Confidence Interval, Lower Limit. Sup.: 95% Confidence Interval, Upper Limit.
The analysis of survival curves was conducted, observing that patients with platelet/lymphocyte ratio <150 showed greater survival, which was statistically significant—similar to the survival curves according to tumor stages I and II (see Figure 1).
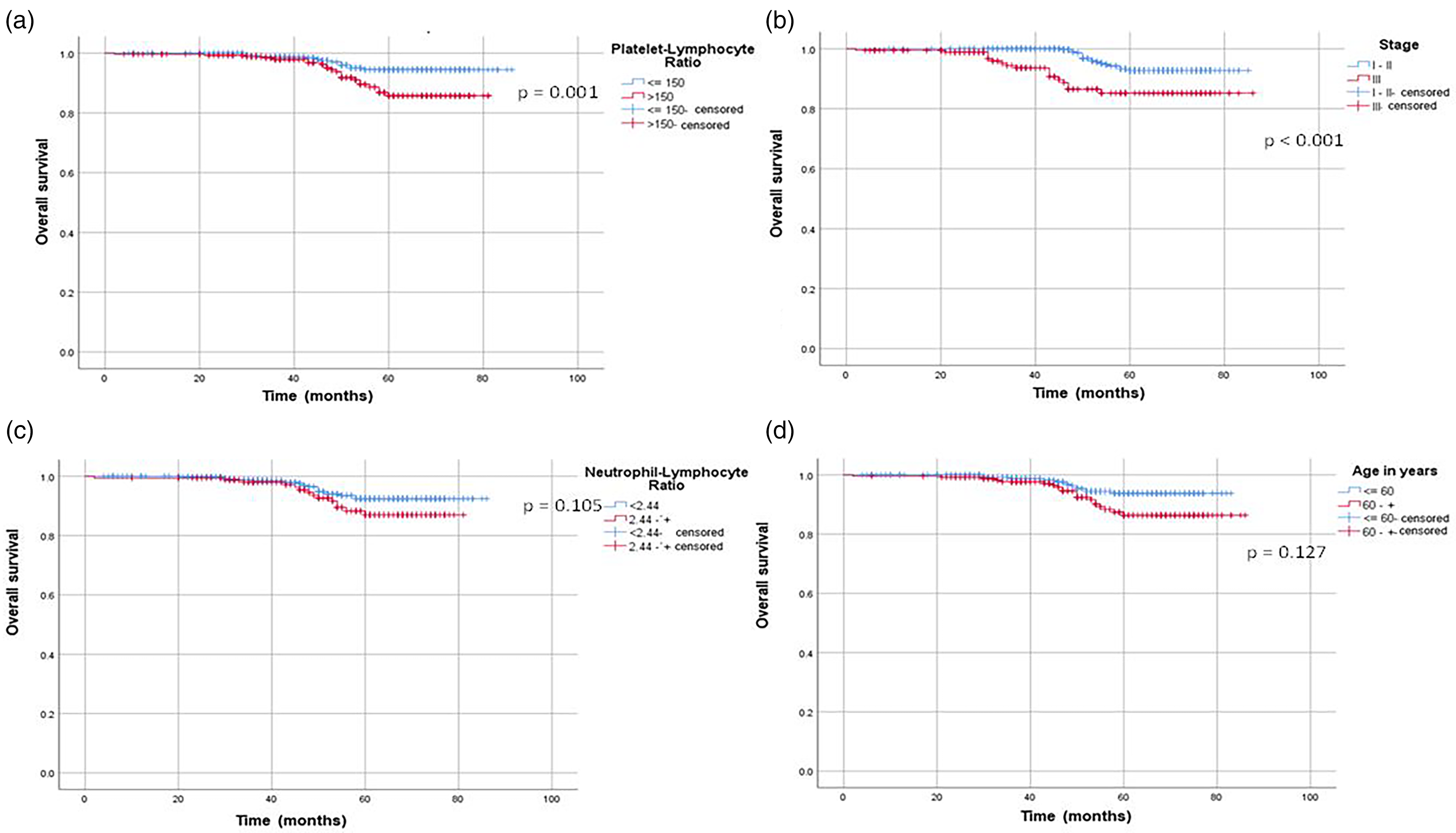
Overall survival according to (a) PLR. (b) Stages. (c) NLR. (d) Age.
Finally, considering the results of the bivariate analysis (Table 3), the research team completed the adjusted analysis of OS by the Cox regression model including the variables NLR, PLR, age, and clinical stage in the model. (see Supplementary Material 1).
Discussion
The inflammatory marker, PLR, assessed at the time of diagnosis and before the start of treatment, showed prognostic value for the OS of breast cancer patients in the four hospitals in Lima-Peru. Survival curves according to stage I and II vs. stage III and PLR <150 vs. >150 corresponded and were similar, with higher survival for stages I and II and PLR < 150. As expected, patients with breast cancer and the presence of advanced stage showed lower survival.
Inflammation plays a vital role in the onset and development of tumors. 24 Neutrophils, monocytes, platelets, and lymphocytes in peripheral blood are closely related to tumor cell invasion, metastasis, and angiogenesis. 23 In the inflammatory cascade, cytokine secretion causes the release of inflammatory cells, thus increasing cancer growth and metastasis.23,25 Analyzing the PLR, mounting evidence supports the role of inflammation in cancer development, progression, metastasis, and treatment resistance. 26
In breast cancer patients, thrombocytosis was associated with poor prognosis, suggesting a key role of platelets in the pathogenesis, development, and progression of breast cancer. 27 A study evaluated 165 breast cancer patients and found that pre-treatment thrombocytosis was a significant negative prognostic factor in multivariate analysis, 28 and these patients were more likely to have metastasis at the time of breast cancer diagnosis. Also advanced age, negative hormone receptors, and higher staging were associated with thrombocytosis and lower OS. 28
The study by Cho et al. 29 evaluated PLR as an independent prognostic marker for survival in all breast cancer patients. They developed a nomogram that incorporated PLR and accurately predicted the individualized survival probability in breast cancer. This practical model could support clinicians and patients in clinical decision-making and treatment optimization.
A meta-analysis study focused on the prognostic value of PLR on breast cancer and analyzed seven published articles. The combined results demonstrated that PLR was a valid prognostic biomarker for poor OS and disease-free survival (DFS) without significant publication bias meta-analisis.
30
High PLR was associated with poor OS (HR = 1.55, 95% CI: 1.07, 2.25,
The present research emphasizes the importance of the PLR inflammatory marker in Peruvian patients with breast cancer, in terms of OS and risk of mortality, similar to that observed with a well-known and established prognostic variable in the international oncology community: the stage of the cancer.
First, this study provides evidence of the importance of the inflammatory marker PLR related to OS through survival analysis by Kaplan–Meier, taken variable by variable and dichotomized by their respective cutoff points. Second, it shows the importance of PLR as an independent factor for breast cancer mortality, by the Cox regression model, obtaining significant adjusted HRs. Finally, the adjusted analysis of OS by the Cox regression model confirms high PLR and advanced cancer stage as independent predictive factors of lower OS, adjusted for the other factors. These data validate the value of PLR as a prognostic biomarker in breast cancer and align with the results of previous breast cancer studies.
Our research carries the inherent limitations of a retrospective cohort design, with variables that could not be fully evaluated, such as molecular and genomic markers of breast cancer. Future prospective analytical studies in different hospitals in Peru and Latin American hospitals are necessary to validate this marker in the Latin American population. Taking specific groups according to the molecular classification of breast cancer and assessing the value of the PLR in these groups would be a significant challenge. Deepening the knowledge of clinical inflammatory markers under investigation in breast cancer is attractive not only for their potential prognostic value, but also their possible therapeutic targets in the era of immuno-oncology. Finally, the importance of continuing to study inflammatory markers and indices lies in their potential application in public health with an emphasis on middle- and low-income countries, due to the ease, accessibility, availability, and low cost of these markers in any primary healthcare center.
Conclusions
The inflammatory biomarker, elevated PLR, and advanced clinical stage, are associated with lower OS in breast cancer patients. Furthermore, a PLR > 150 proved to be an independent prognostic factor for mortality.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155241283480 - Supplemental material for Inflammatory markers related to survival in breast cancer patients: Peru
Supplemental material, sj-docx-1-jbm-10.1177_03936155241283480 for Inflammatory markers related to survival in breast cancer patients: Peru by Jhony Alberto De La Cruz-Vargas, Luis Roldán-Arbieto , Robert Malpartida Palomino, Diana Ferrer Ponce, Cesar Fernández Anccas, Lucy Correa López, Brady Ernesto Beltrán Gárate and José Manuel Vela Ruiz in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155241283480 - Supplemental material for Inflammatory markers related to survival in breast cancer patients: Peru
Supplemental material, sj-docx-2-jbm-10.1177_03936155241283480 for Inflammatory markers related to survival in breast cancer patients: Peru by Jhony Alberto De La Cruz-Vargas, Luis Roldán-Arbieto , Robert Malpartida Palomino, Diana Ferrer Ponce, Cesar Fernández Anccas, Lucy Correa López, Brady Ernesto Beltrán Gárate and José Manuel Vela Ruiz in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
