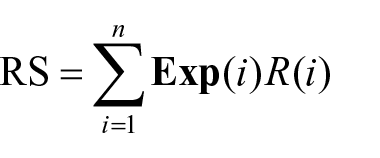Abstract
Objective:
The aim of this study was to develop and validate a reliable nomogram to estimate overall survival in bladder cancer.
Method:
Patients diagnosed with bladder cancer identified in the Surveillance, Epidemiology, and End Results database were randomly divided into training and validation cohorts. The powerful prognostic variables were examined using Cox regression analyses. A nomogram was developed on the prognostic factors.
Results:
The results suggested that age, sex, race, grade, histologic type, primary site, pathological stage, surgical treatment, and number of primary tumors, were the powerful prognostic factors. All these factors were integrated to construct the nomogram. The nomogram for predicting overall survival showed better discrimination power than the tumor-node-metastasis (TNM) stage system 8th edition.
Conclusion:
The nomogram has the potential to provide an individualized prediction of overall survival in patients with bladder cancer.
Introduction
Bladder cancer (BLCA) is one of the most common urologic tumors worldwide, which accounts for approximately 430,000 new cases and more than 165,000 deaths per year. 1 The main treatment for BLCA is surgery, but metastasis and recurrence are common in clinical practice. Patients with distant metastasis always have poor prognoses despite receiving chemotherapy, and their median overall survival (OS) is about 15 months. 2 Thus, it is imperative to construct an exact model to evaluate the prognosis of BLCA patients, enhancing individualization of treatment according to clinicopathologic factors, and facilitating the choice of the best treatment option.
At present, the American Joint Committee on Cancer (AJCC) Staging Manual (8th edition) is widely used to predict the prognosis for BLCA patients. 3 In this classification system, depth of invasion, number of metastasis nodes, and the status of distant metastasis are used to predict the outcomes of BLCA patients. However, patients with the same AJCC stage may have quite different prognoses. In fact, many other important variables, such as age, sex, race, number of primary tumors, tumor site and tumor differentiation, are also associated with prognosis in many cancers.4,5 Therefore, a comprehensive classification system including other relevant clinicopathological factors may provide a more credible prediction of prognosis than the AJCC staging manual.
A nomogram is a convenient and reliable statistical prediction tool, in which several important factors different from tumor node metastasis (TNM) variables are also used to predict the prognosis.6-9 Through comprehensive analysis of these clinicopathological factors, a nomogram could provide a patient’s individual prediction of OS. Compared with the AJCC TNM staging system, the nomogram for predicting OS could be more accurate because clinicopathological factors and AJCC stage are combined together.10,11 Some nomograms have been established for predicting the prognosis of BLCA patients. Xu et al. 12 developed a nomogram for predicting the first 2-year recurrence risk of bladder cancer by two independent predictors—muscle-invasive status and Rad-score (calculated using radiomics features)—on the basis of 71 BLCA patients (34 recurrent). Lu et al. 13 established a prognosis model for the prediction of lymph node metastasis (LNM) in bladder urothelial carcinoma using the LNM signature (including 48 differentially expressed genes in patients with lymph node positive and negative) and the mutation of MLL2 based on 196 bladder urothelial carcinoma samples from The Cancer Genome Atlas (TCGA). Kim et al. 14 reported nomograms to predict recurrence and progression in primary non-muscle-invasive bladder cancer (NMIBC) based on 970 diagnosed NMIBC patients after transurethral resection of bladder tumor (TURBT) in a single institution. However, the previous nomograms only focused on urothelial carcinoma, NMIBC, or MIBC, and the small number of patients affected the accuracy and applicability of their results. Thus, a simple and universal applicability nomogram for predicting prognosis in BLCA need to be developed.
In this study, we make great efforts to construct a prognostic nomogram, which is based on large population data selected from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) cancer database in order to provide more precise and personalized OS in BLCA patients.
Patients and methods
Patient selection
In this study, the data were obtained from the SEER cancer database, which is one of the largest cooperative projects by the National Cancer Institute. The SEER project collects and publishes approximately 28% of the American population’s cancer incidence and mortality. The following inclusion criteria was used for data screening: (a) patients were diagnosed as BLCA; (b) the histologic type (derived Histologic Type ICD-O-3) of patients should be available, including transitional cell carcinoma, papillary transitional cell carcinoma, and non-transitional carcinoma (including squamous cell carcinoma, adenocarcinoma, and any other types); (c) it should include clinicopathological information for the age at diagnosis, race, primary site (trigone, dome, lateral wall, anterior wall, and posterior wall of bladder, and bladder neck), grade (well differentiated, grade I; moderately differentiated, grade II; poorly differentiated, grade III; undifferentiated, grade IV), number of primary tumors, surgical treatment (RX Summ-Surg Prin Site (1998+)) (including no surgery, (TURBT) and other surgeries, such as partial, radical, and complete cystectomy with reconstruction) and tumor stage (derived from the 8th edition of the AJCC Staging Manual (2017+)); and (d) the survival time and vital status should be available for survival analyses. Patients who did not meet the above criteria or lacked enough details for clinicopathological information would be excluded. Patients with urothelial tumors in the upper urinary tract were also excluded.
Study variables and endpoints
The variables from the selected cohorts were listed as follows: the age at diagnosis, sex, race (black, white, American Indian/Alaska (AK) native, Asian/Pacific Islander), primary site, histologic type, grade, surgical treatment, 8th edition of the AJCC Staging Manual tumor stage including Ta/Tis/T1/T2a/T2b/T3a/T4b /N0 /N1/N2/N3/M0/M1 stage and number of primary tumors (solitary and multiple). The value of age at diagnosis and number of primary tumors were transformed into categorical variables to facilitate the following analysis. OS was considered as an endpoint.
Validation and visualization of the nomogram
Both internal and external validation of this nomogram were performed on the basis of the training cohort and validation cohort. First, the discrimination was evaluated by Concordance index (C-index), which was proved to be better for censored data. 15 The C-index was used to access the nomogram as well as the 8th edition of the AJCC Staging Manual system. The value of C-index statistics was from 0.5 (no discrimination) to 1 (perfect discrimination), and the greater the value the better the prognostic model. Next, the consistency between predicted survival and observed survival was accessed using calibration curves of the nomogram. To decease the overfit bias, bootstrap analyses with 1000 resamples were used. 10
Statistical analyses
Univariate and multivariate Cox regression analyses were carried out to identify independent prognostic variables associated with OS in BLCA patients. Then, the following formula was used to estimate the risk score (RS) of the multiple Cox regression model:
where Exp(i) is the value of each variable; n is the number of variables; and R(i) is the regression coefficient in multivariate Cox regression analysis. When the RS of a sample was more than the mean RS, the sample was considered high risk. In contrast, the sample was considered low risk. Then, the Kaplan-Meier method was used to estimate survival curves in both the high- and the low-risk groups. Additionally, the area under the curve (AUC) value and receiver operating characteristic (ROC) curve were used to estimate specificity and sensitivity.
In the current study, a P-value of less than 0.05 was considered statistically significant. All the statistical analyses were performed by R software version 3.4.3.
Result
Clinicopathological characteristics of the patients
According to our inclusion criteria, 31,374 eligible patients as the primary cohort of BLCA were included in this study. The primary cohort was then randomly divided into a training cohort (n=18,826) and a validation cohort (12,548), which was used to validate the nomogram. The characteristics of these cohorts are shown in Table 1. Overall, the majority of patients were male (24,264; 77.34%), more than 50 years old (30,128; 96.03%) and white (28,177; 89.81%) with Ta (15,927; 50.76%), N0 (30,333; 96.68%), and M0 (30,850; 98.33%) stages. The main site of BLCA were at the lateral wall of bladder, accounting for 46.1%. Papillary transitional cell carcinoma of BLCA was 74.20%. Most patients had a single tumor (20,122; 64.14%) and received TURBT (21,722; 69.24%). A total of 7,415 (23.63%) patients (4427 in the training cohort and 2988 in the validation cohort) had died by the end of the follow-up. The average OS was 27 months for both the training and the validation cohort. There was no significant difference in patient composition between the two cohorts at different levels.
Patient demographics and clinical characteristics (Part I).
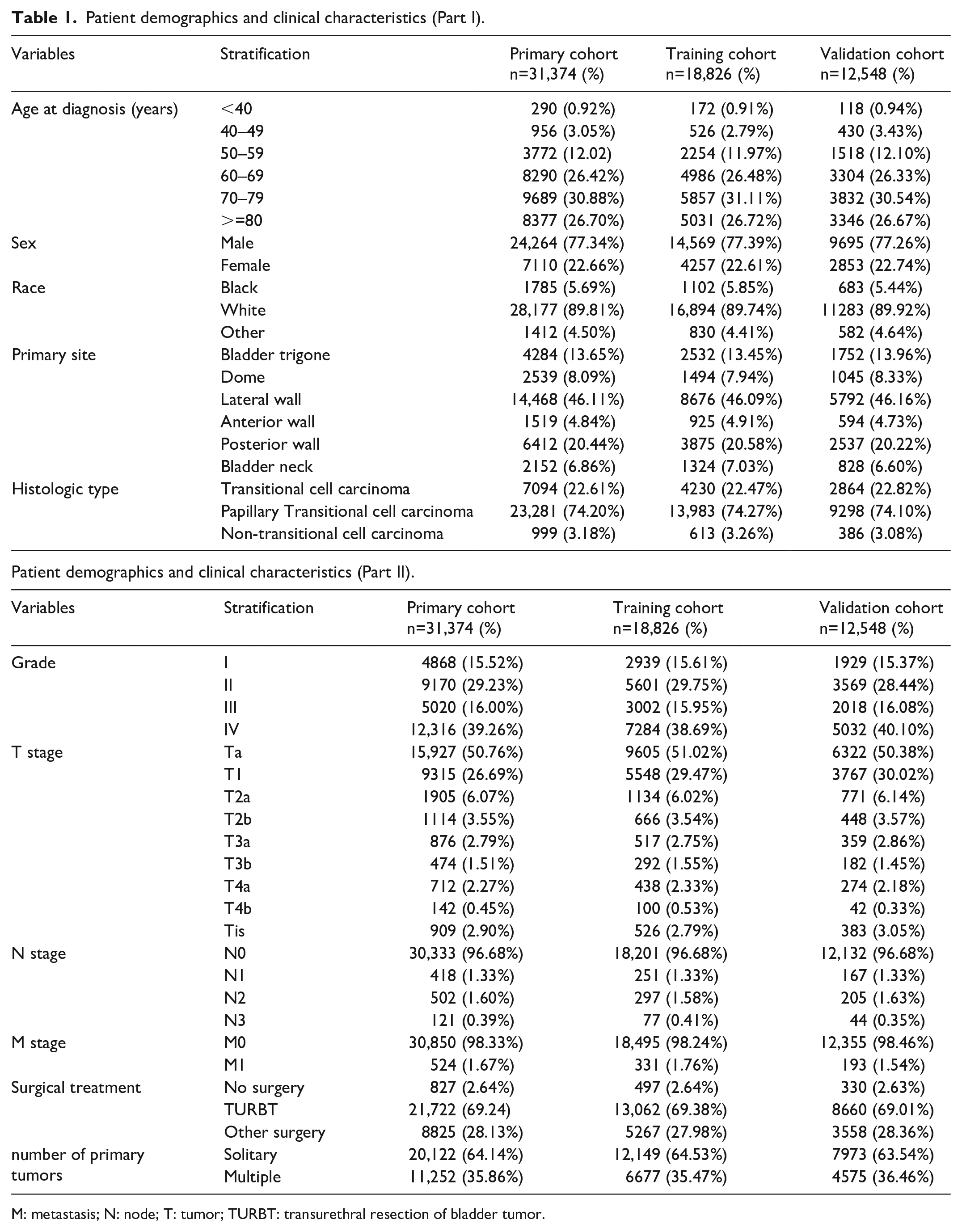
M: metastasis; N: node; T: tumor; TURBT: transurethral resection of bladder tumor.
Univariate and multivariate Cox regression of the training cohort
The univariate and multivariate Cox regression analyses were performed to analyze variables that influence OS. For quantifying the effect of each variable, hazard ratio was used. The following variables were used for Cox regression analysis: age at diagnosis, race, sex, histologic type, grade, primary site, pathological stage (TNM, AJCC Staging Manual, 8th edition), surgical treatment and number of primary tumors. The results are presented in Table 2. All variables had statistical significance both in the univariate and multivariate Cox regression analyses for OS. This suggests that all the variables included in the present study were the independent prognostic factors.
Univariable and multivariable Cox regression model analyses of overall survival in nomogram cohort (Part I).
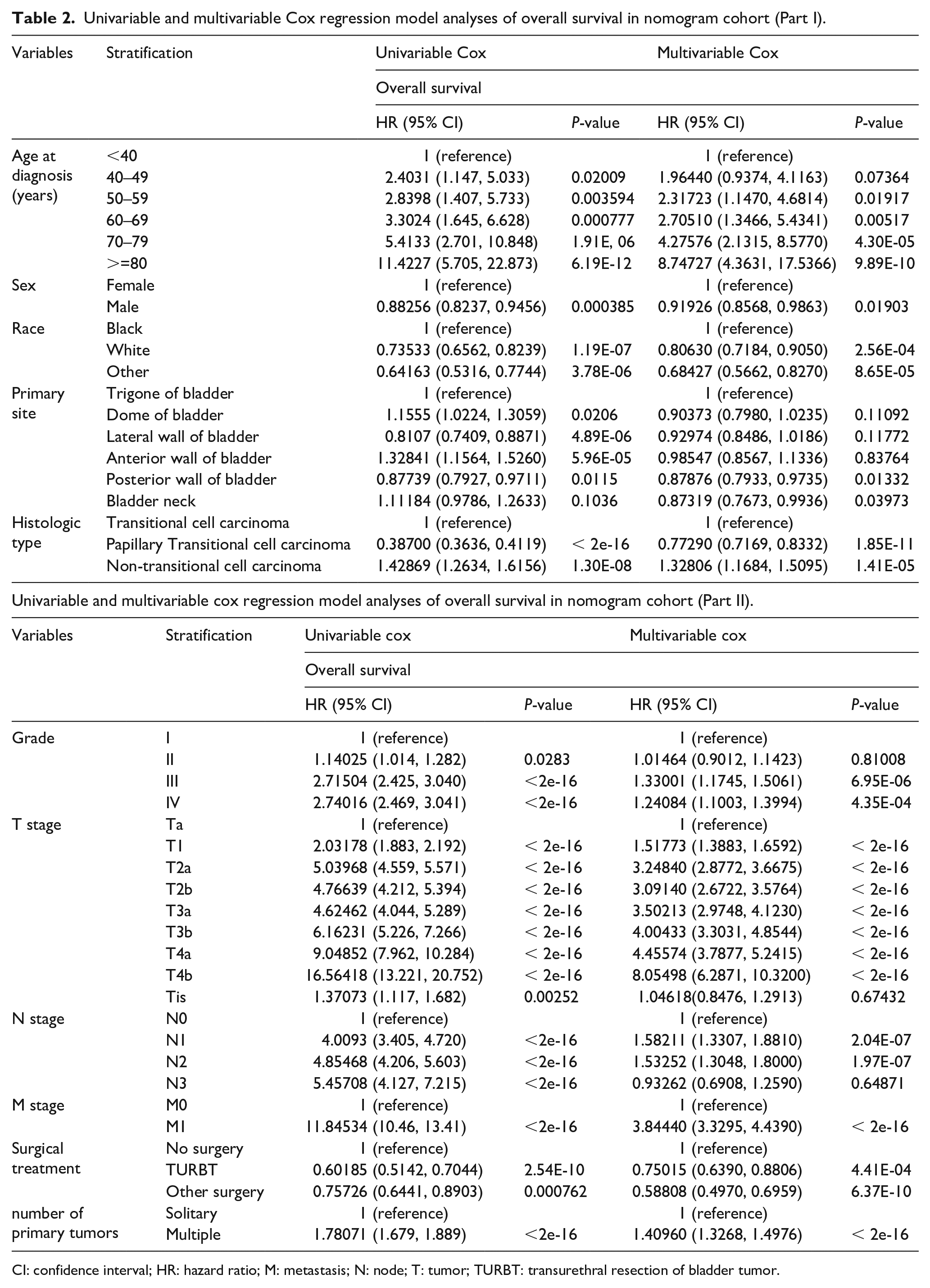
CI: confidence interval; HR: hazard ratio; M: metastasis; N: node; T: tumor; TURBT: transurethral resection of bladder tumor.
According to risk score values of each variable in the multiple Cox regression model, the patients in the training cohort were assigned into either the high- or low-risk group. The high-risk group had significantly shorter survival time compared with the low-risk group (Figure 1). In order to estimate the specificity and sensitivity of the survival prediction, time-dependent ROC analysis was adopted, and AUC values were determined. AUC values for 3-year and 5-year survival predictions were 0.771 and 0.77, respectively (Supplementary Figure 1(a) for 3 years and Figure1(b) for 5 years), indicating high prediction performances.
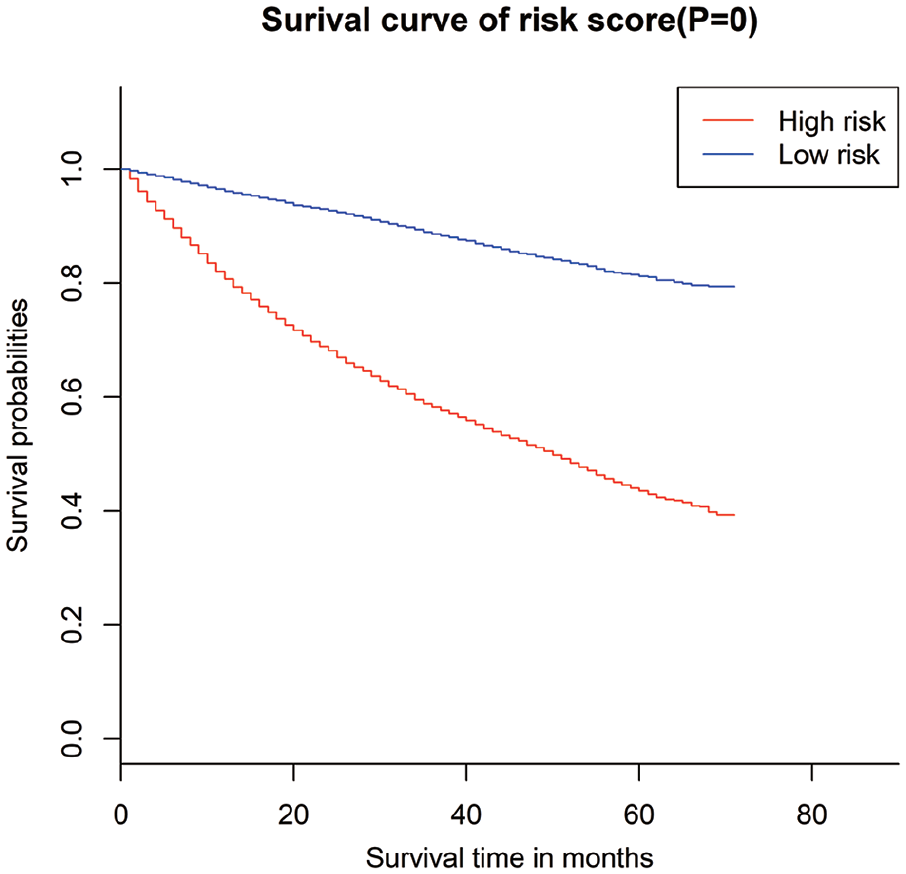
Kaplan-Meier survival curves for risk associated with OS in BLCA.
Based on the results in Table 2, the survival curves of these variables were obtained by Kaplan-Meier curve analysis (results are shown in Supplementary Figure 2). However, to facilitate the analysis, the 8th edition of the AJCC Staging Manual tumor stage was roughly considered as Ta, Tis, T1 (Ta, Tb), T2 (T2a, T2b), T3 (T3a, T3b), T4 (T4a, T4b). The results of the Kaplan-Meier curve analysis suggested that age, sex, race, primary site, histologic type, grade, TNM classifications, number of primary tumors, and surgical treatment were the independent risk factors influencing the prognosis.
Prognostic nomogram for OS
Considering the results of univariate and multivariate Cox regression analysis, the above-mentioned variables were included in the nomogram for 3- and 5-year OS (Figure 2). Each subtype of the variables was assigned a certain score. By summing the scores related to each variable, and locating the total scores on the bottom scales, probabilities could be evaluated for 3- and 5-year OS. The nomogram indicated that age more than 80 was the largest contributor to prognosis, followed by AJCC TNM stage. However, race, surgical treatment, and tumor histologic type also showed a moderate effect on OS.
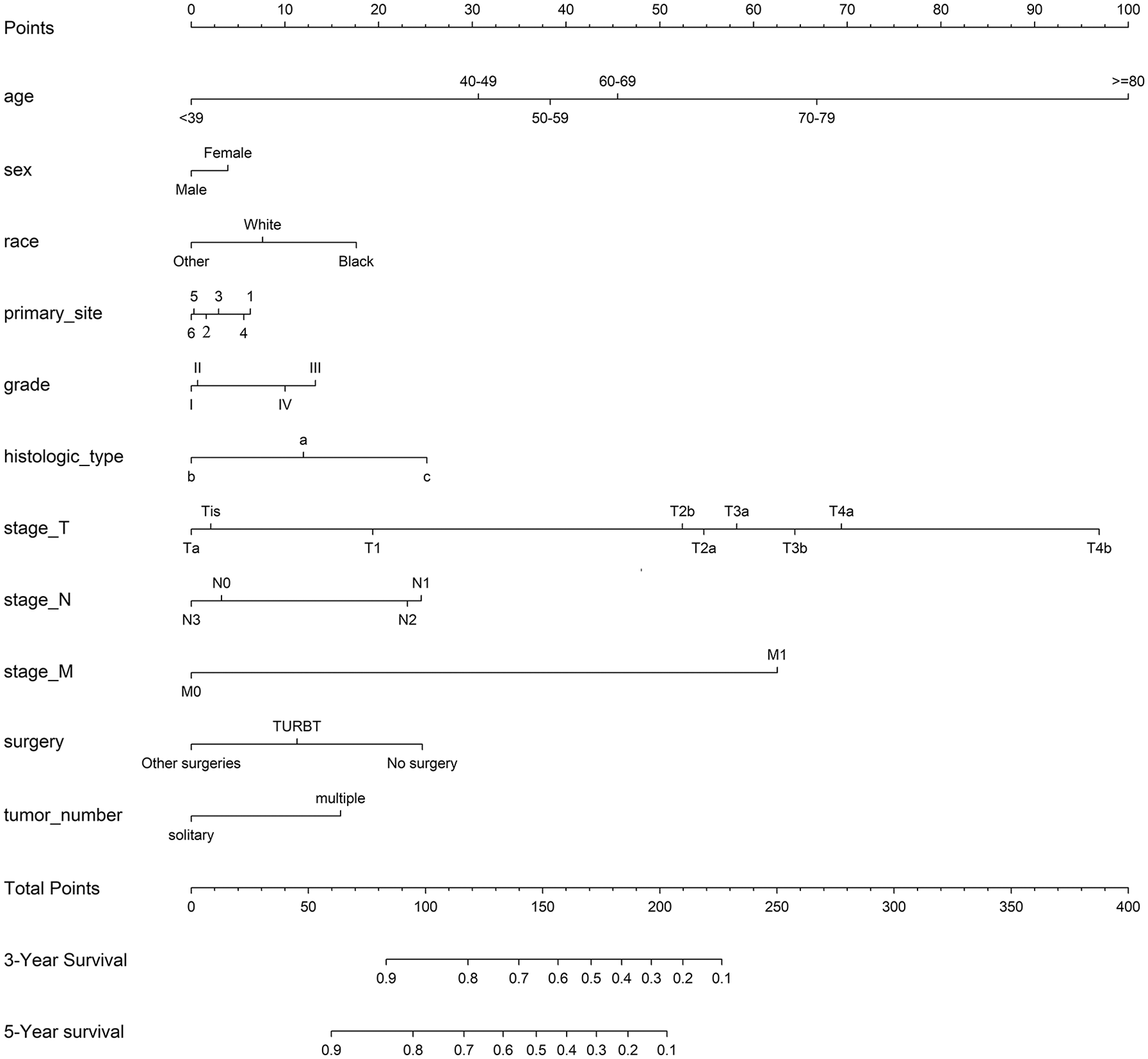
Nomograms for predicting the 3- and 5-year OS of BLCA patients. Race: other represents American Indian/AK Native, Asian/Pacific Islander; primary site: 1–6 represent trigone of bladder, dome of bladder, lateral of bladder, anterior of bladder, posterior of bladder and bladder neck, respectively; histologic type:
To better understand the use of this nomogram, we can take a patient with BLCA as an example. A 75-year-old male black patient with a tumor located on the bladder neck, grade IV, T2aN0M0 stage who received TURBT would score > 150 points, which means that the patient has approximately 60% and 45% survival probability at the third and fifth year, respectively.
Internal and external validation of the nomograms
The C-indexes of this nomogram for OS prediction were 0.780 (95% CI 0.770, 0.790) and 0.776 (95% CI 0.765, 0.788) in the training and the validation cohort—all of which were greater than 0.7, suggesting the suitability of our nomogram for BLCA patients. In addition, we compared the difference of the nomogram with that of the AJCC TNM staging system (8th edition) in the training cohort and validation cohort. The C-indexes of the TNM staging were 0.705 (95% CI 0.697, 0.713) and 0.695 (95% CI 0.685, 0.704) in training and validation cohort, respectively, which were significantly lower than that of the nomogram mentioned above. The results demonstrated that the nomogram discrimination for OS was better than AJCC TNM staging. Furthermore, internal and external calibration plots for 3-year and 5-year OS are presented in Figure 3, showing an excellent correlation between nomogram prediction and observed outcomes in both the training cohort and the validation cohort, revealing great reliability of the nomogram.
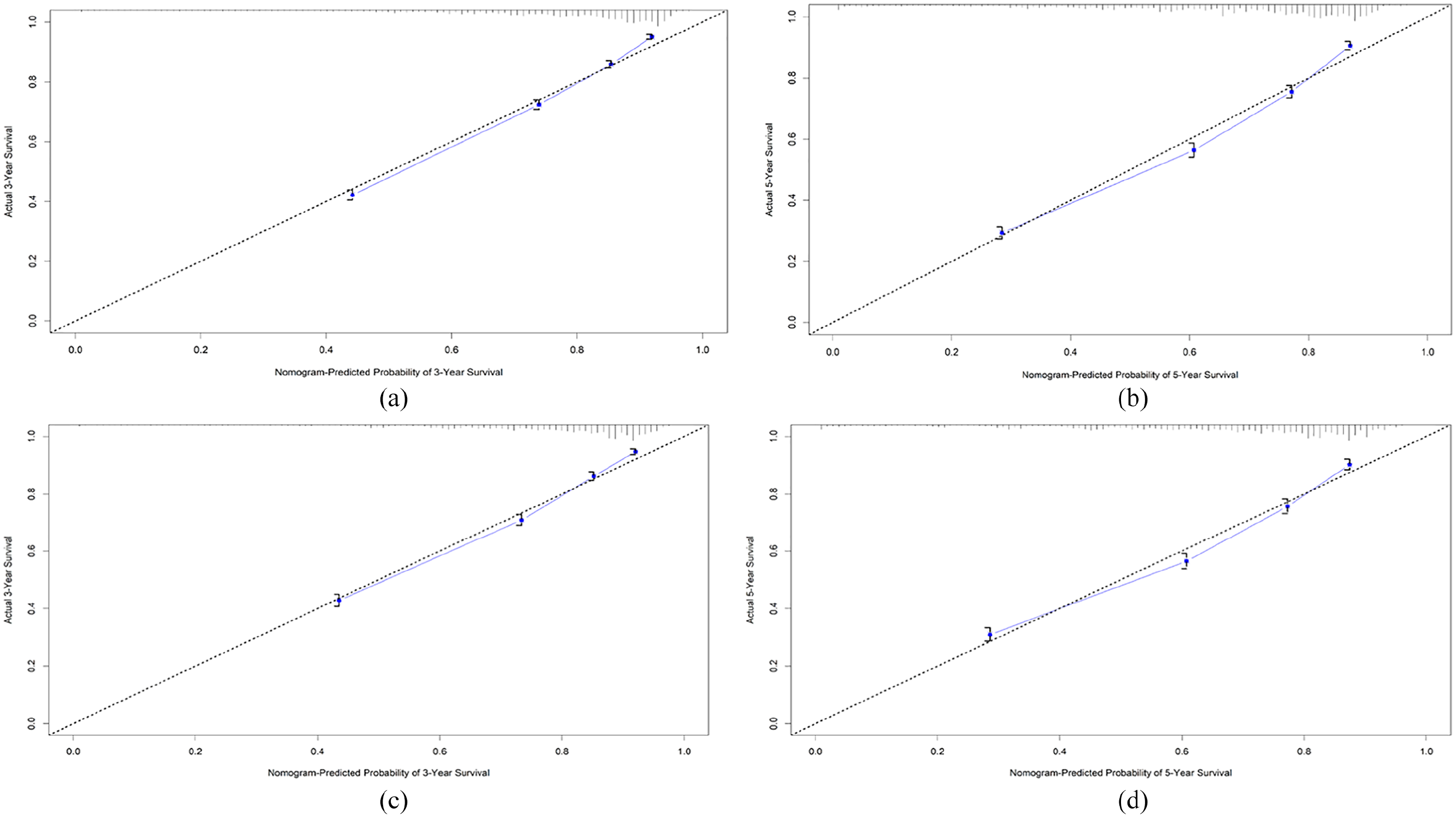
The calibration plot for training cohort (a) and (b) and validation cohort (c) and (d). (a) and (c) 3- and (b) and (d) 5-year overall survival (OS) nomogram calibration curves.
Discussion
Nomograms are graphical presentation of statistical prediction models16,17 and play important roles in modern medical decision making. 18 Currently, several nomograms have been developed for predicting the prognosis in patients with muscle invasive bladder carcinoma (MIBC). Specifically, three research studies19-21 tried to construct nomograms to predict oncological outcomes of patients receiving radical cystectomy by using clinical variables. They reported predictors such as biomarkers of systemic inflammatory response, 19 sarcopenia status, 21 and lymph node radiologic status 20 for predicting nomogram confined disease status, 19 OS, 21 and pathological lymph node involvement. 20 In contrast, three studies22-24 developed nomograms based on pathological variables for predicting the prognosis of patients with locally advanced and/or recurrent MIBC. One study on metastatic patients was also reported. 25 Based on a cohort of 201 BLCA patients, Green et al. 26 previously built a nomogram for preoperative prediction of lymph node metastases in bladder cancer without internal validation. Stojadinovic et al. 27 validated their model and concluded that the Green et al. nomogram seemed suitable for patients from Europe, despite various risk factors in the validation cohort. Aziz et al. 28 identified age, American Society of Anesthesiologists score, hospital volume, clinically lymphatic metastases, and clinically distant metastases as independent factors, and developed a nomogram for predicting 90-day mortality after radical cystectomy for bladder cancer. Recently, this nomogram was validated by Osawa et al. 29 using 834 patients who received radical cystectomy at 21 Japanese institutions, and concluded that Aziz A’ nomogram was suitable for both preoperative patient counseling and perioperative care in Europe and Japan. Wakai et al. 30 developed and validated a predictive model for predicting high-grade papillary bladder cancer before first-time TURBT based on macrohematuria, tumor size, multiple tumors, appearance, and positive urinary cytology in 424 patients. They also identified number of primary tumors significantly associated with high-grade papillary bladder cancer, which is in accordance with ours. In contrast to ours, age and tumor location were not significantly associated with high-grade papillary bladder cancer in their study. This may be because of the different scale of samples and patients with different disease status. Dong et al. 11 established nomograms focusing on primary small cell carcinoma of bladder. Huang et al. 31 constructed a nomogram for predicting cancer-specific survival in bladder transitional cell carcinoma on the basis of the data from the SEER database. The nomogram included independent risk factors, listed as: insurance, histological grade, T stage, SEER stage, tumor size, and lymph node ratio. However, most of the studies mentioned above were based on small patient cohorts, just one histologic type, or a lack of external validations, which decreased the power of their findings. Furthermore, we know that some clinical variables, such as tumor histological type, grade, tumor primary site, number of primary tumors, and surgical treatment, are closely associated with the prognosis of patients with BLCA. Hence, the universal applicability of a nomogram to BLCA needs further validation with the added benefit that the additional variables above would increase the accuracy of our nomogram.
In this study, we developed a nomogram for predicting individual OS based on a large patient cohort of BLCA from the SEER database. The nomogram was constructed on the basis of individual demographic, and pathologic and surgical treatment data, and it revealed well discrimination in both internal and external validation, indicating good clinical applicability of this nomogram for BLCA. Additionally, the nomogram showed more satisfactory accuracy and robustness than the AJCC TNM staging system (8th edition). Also the OS associated with all the variables as independent prognostic factors was calculated via the Kaplan-Meier method, and the results showed favorable correlation between each variable and OS with statistical significance.
Our model was composed of 11 independent prognostic variables from clinical practice: age; race; sex; primary site; tumor grade; histologic type; number of primary tumors; T, N, and M stage; and surgical treatment. Age has been reported to be an important prognostic factor for OS in several research studies,32,33 although the underlying mechanism remains unknown. Our results from the Kaplan-Meier curve analysis showed that patients older than 40 years have a lower OS than young patients, and that patients older than 80 years have the worst prognosis. Evidence suggests that race was also associated with prognosis in patients with cancer, and black patients had a worse prognosis than other races, 34 which was in accordance with our result. In terms of tumor histologic type, Vetterlein et al. 35 demonstrated that pure squamous cell and neuroendocrine carcinoma histologic types were associated with worse OS relative to pure urothelial carcinoma. Our results revealed that non-transitional cell carcinoma histologic types had a worse prognosis than transitional cell carcinoma. It is widely known that a high-grade tumor is always associated with a poor prognosis, which is in accordance with our result. Dobruch et al. 36 also reported that women with BLCA have higher cancer-specific mortality than men. This may be attributed to a significantly greater delay resulting from urologic referral and guideline-concordant imaging, which is performed less frequently in women. Interestingly, according to the results of the Kaplan-Meier curve analysis, we also found that male patients with a single tumor located on the posterior wall of the bladder were related to better prognosis. Additionally, patients receiving TURBT seemed to have higher OS than patients receiving no surgery or other surgery. This may be because most of the patients receiving TURBT belonged to an earlier TNM stage.
Compared with the TNM staging, our model is not only easier to follow but also offers a quantitative prognosis for each patient. For example, consider two patients with BLCA (tumor located on bladder neck: papillary transitional carcinoma: T2aN0M0: grade IV): case A— a 75-year-old black patient with multiple tumors who received TURBT would score approximately 190 points; and case B—a 65-year-old other race patient with a solitary tumor who received TURBT would score approximately 130 points. Using the nomogram established by us, the two patients have a 3-year OS probability of 35% and 75%, respectively, and a 5-year OS probability of less than 20% and 60%, respectively. In contrast, both patients would be thought to have the same traditional TNM staging system, which means similar outcomes. These results clearly indicate the shortcomings of TNM classification in predicting individual prognosis. Thus, the nomogram could be crucial for estimating patients prognosis in the follow-up period and making treatment strategy. For instance, younger patients with well-differentiated histology and an earlier TNM stage might undergo surgery because of their good prognosis, while older patients with poorly differentiated histology and later TNM stage who have low life expectancy might receive non-surgical treatment. However, the choice of treatment for patients only on the basis of TNM staging could be unclear, and physicians need to depend on their clinical experience. Therefore, through our nomogram, which consists of many clinicopathological variables, physicians can make a better choice of the optimal treatment for patients with BLCA.
Generally speaking, our nomogram has some advantages over earlier ones. First, to the best of our knowledge, this is the first study to construct a nomogram based on a large, diverse, population-based cohort for predicting the OS of patients with BLCA. Second, the present study included several variables taken into account for the first time, such as primary site, histologic type, and number of primary tumors, which are likely to increase the accuracy of nomogram. Third, the calibration plots and C-indexes were also performed to estimate the accuracy of the model. All C-indexes of the nomogram were more than 0.7, indicating excellent accuracy to predict OS. Fourth, the Kaplan-Meier curve analyses were also performed in our study and the results showed good correlation between the variables and OS. Finally, the 11 clinical variables used in our nomogram are easily accessible and can better exhibit patient and tumor characteristics.
Although the present nomogram has good accuracy, some potential limitations should be noted. First, some important clinicopathological variables, such as chemotherapy, surgical margin status, and vascular invasion, are unavailable in the SEER database. Moreover, information on some important clinicopathological variables are not complete, resulting in some cases being excluded. In addition, the nomogram was constructed on the basis of retrospective data, which could bring the risk of treatment selection bias. The nomogram must be further validated based on a prospective cohort. Finally, as a user-friendly tool for physicians to make treatment strategy decisions, the nomogram did not take all prognostic variables into account and could not provide exact prognoses in clinical practice.
Conclusion
Our study suggested that sex, age, race, tumor primary site, histologic type, number of primary tumors, grade, and TNM stage were independent risk variables for OS in BLCA patients. A nomogram was constructed to better predict the 3- and 5-year OS rate of BLCA patients on the basis of specific characteristics. The C-index was satisfactory in both internal and external validation. The predictive tool could be beneficial for physicians to evaluate individualized survival predictions and could provide better treatment allocation for patients with BLCA.
Supplemental Material
Supplementary_figure_1 – Supplemental material for Nomogram for predicting overall survival of patients with bladder cancer: A population-based study
Supplemental material, Supplementary_figure_1 for Nomogram for predicting overall survival of patients with bladder cancer: A population-based study by Jiawu Wang, Yan Wu, Weiyang He, Bo Yang and Xin Gou in The International Journal of Biological Markers
Supplemental Material
Supplementary_figure_2 – Supplemental material for Nomogram for predicting overall survival of patients with bladder cancer: A population-based study
Supplemental material, Supplementary_figure_2 for Nomogram for predicting overall survival of patients with bladder cancer: A population-based study by Jiawu Wang, Yan Wu, Weiyang He, Bo Yang and Xin Gou in The International Journal of Biological Markers
Footnotes
Acknowledgements
The authors would like to than SEER program for providing open access to the database.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data access
We obtained permission to access the SEER database with the reference number 14581-Nov2017. Extraction of data from the SEER database does not require informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.