Abstract
Background
The aim of this study is to explore the expression of beclin 1, an autophagy gene, in bladder cancer and to evaluate its clinical and prognostic significance in patients with bladder cancer.
Methods
Beclin 1 expression was examined at mRNA and protein levels by real-time quantitative polymerase chain reaction (RT-PCR), Western blotting, and immunohistochemistry in bladder cancer tissues and adjacent normal bladder tissues. The relationship between the expression of beclin 1 and clinicopathological characteristics and prognosis was statistically analyzed.
Results
mRNA level, protein expression and immunoreactivity of beclin 1 were decreased in bladder cancer tissues compared with adjacent normal tissues. Downregulation of beclin 1 was more frequent in tumors with higher histological grades (the expression of beclin 1 was reduced by 49.0% in G1 and G2, and by 71.8% in G3, p=0.010), and was also reduced by 69.5% in the muscle invasive type and by 51.1% in the non-muscle invasive type (p=0.04). Reduced beclin 1 expression was positively associated with higher histological grade and more advanced clinical stage (p<0.05). Kaplan-Meier survival analysis revealed that patients exhibiting lower beclin 1 expression experienced a shorter survival than those with higher expression (p=0.006). Cox proportional hazards regression analysis showed that beclin 1 protein is an independent predictor of survival (p=0.005).
Conclusion
Beclin 1 has an influence on the progression of bladder cancer and might serve as a potential prognostic factor for patients with bladder cancer.
Introduction
Urinary bladder carcinoma (UBC) is the 10th most common cancer in China, accounting for 17,365 deaths in 2005, and the mortality rate is steadily increasing between the years 1991 and 2005 (1). The incidence of bladder cancer rises with age, peaking between 50 and 70 years, and is 3 times more common in men than in women (2). More than 90% of bladder cancers are transitional cell carcinomas, 5% are squamous cell carcinomas, and less than 2% are adenocarcinomas. Histological staging is performed according to the tumor-node-metastasis (TNM) staging system, in which the tumor (T) stage of the primary tumor is based on the extent of penetration or invasion into the bladder wall (3). In addition, UBC occurrence is about 3 to 4 times higher in developed countries (4). Great efforts have been made to explore protein biomarkers and their role in the development and progression of bladder carcinomas. These markers include the expression of oncogenes, tumor suppressor genes, cell cycle regulators, growth factors, and cell adhesion molecules (5).
Autophagy, the major endogenous pathway for degradation, delivery and recycling of long-lived proteins and organelles, has gained more attention during recent years. Autophagy occurs through different pathways, including macro-autophagy, micro-autophagy, chaperone-mediated autophagy, mitophagy, and crinophagy (6, 7). Autophagy consists of several sequential steps: sequestration, transport to lysosomes, degradation, and utilization of degradation products; each of these steps may exert different functions (8). Although an increasing number of reports indicate the role of autophagy in tumor suppression (9), studies on autophagic genes are limited in the context of bladder cancer.
The human autophagy gene beclin 1, a mammalian orthologue of the yeast Apg6/Vps30, participates in the formation of the autophagosome, mediating the localization of other autophagy proteins to the pre-autophagosomal membrane (10). Recent research showed that beclin 1 also functions as a haploinsufficient tumor suppressor (11, 12) and is related to the clinicopathological features and prognosis in ovarian, breast, and brain carcinoma (13). The aim of the current study is to investigate the role of beclin 1 in bladder cancer and evaluate the clinical and prognostic significance of beclin 1 in patients with bladder cancer.
Materials and Methods
Specimen selection
In this retrospective study a total of 147 paraffin-embedded samples from 147 bladder cancer patients were collected between 2002 and 2009 and were clinically and histologically diagnosed at the Sun Yat-sen University Cancer Center (SYSUCC). The samples included 127 specimens of transitional cell bladder cancer and 20 specimens of adjacent normal bladder tissues. All cancer patients in the series were categorized in accordance to the 2002 American Joint Committee on Cancer (AJCC) staging system (14). Patients' age ranged between 15 and 87 years (median 63), and included 15 females and 112 males. Among these patients, 44 underwent radical cystectomy, 21 underwent partial cystectomy and 62 patients underwent transurethral resection of bladder tumor (TURBT). After partial cystectomy and TURBT, mitomycin C or pirarubicin were used in intravesical therapy as a weekly intravesical injection beginning within 24 hours after surgery. Nine patients had distant metastases and 28 had recurrence. The follow-up time for all patients ranged between 6 and 162 months, with a median of 56 months. Thirty patients died due to the disease at the time of analysis. In addition, 14 paired samples of transitional cell bladder cancers and adjacent normal tissues from the patients who underwent surgery in SYSUCC were used for Western blotting or real-time quantitative PCR analysis. The informed consent was obtained from each patient and the study was approved from the research ethics committee of the institute.
Immunohistochemistry
We performed immunohistochemistry for the autophagic protein beclin 1 on 20 adjacent normal specimens and 127 malignant bladder specimens. The staining procedures were carried out with classical protocols of the 2-step methods. Firstly, paraffin-embedded specimens were cut into 4 μm sections and baked at 65°C for 30 minutes. Deparaffinization of sections was performed with xylene 2 times, sections were then rehydrated with alcohol at different concentrations. Then, sections were immersed into citric acid antigenic retrieval buffer and microwaved for antigenic retrieval for 5 minutes at room temperature, followed by immersion in 3% hydrogen peroxide to quench the activity of endogenous peroxidases for 15 minutes.
The sections were incubated with a rabbit polyclonal anti-beclin 1 antibody (dilution, 1:800; NB 500-249; NAVUS Biologicals, LLC, USA) (1:800) overnight at 4°C and were cultivated with biotinylated anti-rabbit secondary antibody at 37°C for 30 minutes. The same isotype of the rabbit IgG was used as negative control. Sections were then incubated with a streptavidin-horseradish peroxidase complex. Then the chromogen reaction of sample sections was developed in 3,3-diaminobenzidine (DAB) tetrahydrochloride for about 1 minute at room temperature. Sections were then counterstained with Mayer's hematoxylin and mounted using Olympus CX31 microscope (Olympus, Center Valley, PA, USA).
To assess the beclin 1 immunoreactivity, the samples of formalin-fixed, paraffin-embedded sections were semiquantitatively evaluated by 2 independent observers, according to the percentage of positive tumor cells, within the number of total cells, and the intensity of the cytoplasmic staining. The 2 observers' scores were quite consistent with each other (κ=0.726), where Kappa = (P0 - Pe)/(1 - Pe). For the discrepant cases, both observers rescored the sections, discussed the scores, and took the final decision. The scores on the proportion of positive tumor cells were categorized as 0 (if <5%), 1 (if 6%-25%), 2 (if 26%-50%), 3 (if 51%-70%) or 4 (if >70%) whereas the intensity of the immunostaining was classified as 0 when absent, 1 when weak, 2 when moderate and 3 when strong, as previously described (15). The final score resulted from the sum of the 2 values, and ranged from 0 to 7. All bladder cancer patients were divided into 2 groups according to the median score of anti-beclin1 immunostaining (4) and a score of ≥4 was considered as a high expression.
Statistical analysis
Statistical analysis was performed using the SPSS 13.0 statistical software package (SPSS Inc., Chicago, IL, USA). The Mann-Whitney test was used to analyze any significant difference in beclin 1 protein and mRNA expression between bladder cancer tissues and the adjacent normal tissues. The association between the expression of beclin 1 protein, the clinicopathological characteristics, and the clinical prognosis was determined by the Kolmogorov-Smirnov test, showing a non-normal distribution of the data, and the χ 2 test. Survival curves were performed by the Kaplan-Meier method, and the log-rank test was used for comparison. A Cox proportional hazard model was used to determine the effect of the assessed parameters on death events. A value of <0.05 was considered statistically significant.
Western blotting analysis
Western blotting analysis was carried out with the proteins collected from the adjacent normal tissues and the cancer tissues according to standard methods, as described previously (16). Firstly, tissues were solubilized in 1×SDS sample buffer (2% SDS, 62.5 mmol/L Tris-HCl [pH 6.8], 5% 2-mercaptoethanol, and 10% glycerol). The concentration of the protein was measured by the BCA protein assay kit (PIERCE, Rockford, IL, USA). Equal amounts of protein (35 μg) were electrophoretically separated on a 9% SDS-polyacrylamide gel and then incubated with the primary polyclonal antibody against beclin 1 (dilution 1:1,000; NB 500-249; NAVUS Biologicals, LLC, USA) overnight at 4°C. At the same time, anti- glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (1:2,000, Santa Cruz, CA, USA) was used as a loading control. Anti-rabbit IgG antibodies (1:2,000, Santa Cruz Biotechnology, Santa Cruz, CA, USA) and anti-mouse IgG antibodies (1:3,000, Santa Cruz Biotechnology, Santa Cruz, CA, USA) were used as secondary antibodies. Protein signals were detected by the enhanced chemiluminescence (ECL) detection system (Amersham Biosciences Europe, Freiburg, Germany) according to the manufacturer's protocol.
Real time reverse transcription-polymerase chain reaction (RT-PCR) analysis
Total RNAs were purified from tissues using TRIzol Reagent (Invitrogen, Carlsbad, CA, USA), and 2 μg RNA of each sample were reverse transcribed using the Super Script RT kit (Invitrogen Life Technologies, Carlsbad, CA, USA). RT-PCR primers were designed with the assistance of the primer premier 5.0 software (PREMIER Biosoft International, Palo Alto CA, USA). The sequences of the primers used were the following: for beclin 1, 5-GAGGTGAAGAGCATCGGGG-3′ (forward), 5′-TTCGCCTGGGCTGTGGTA-3′(reverse), and for GAPDH, 5′-GACTCATGACCACAGTCCATGC-3′ (forward) 5′-AGAGGCAGGGATGATGTTCTG-3′ (reverse). We used the SYBR Green kit (Invitrogen Life Technologies, Carlsbad, CA, USA) to perform the amplification of the cDNA. The real time PCR cycling parameters were as follows: denaturation at 95°C for 15 seconds, annealing at 55°C for 30 seconds, and extension at 72°C for 30 seconds. The expression data were normalized to the geometric mean of the housekeeping gene GAPDH to control the difference in expression levels, and were analyzed using the 2-delta delta Ct method described by the previous report (17).
Results
Beclin 1 expression in bladder cancer tissues
Real-time PCR and Western blotting analysis showed that beclin 1 expression was significantly downregulated in bladder cancer tissues compared to the paired adjacent normal tissues at both mRNA (Fig. 1) and protein levels (Fig. 2).
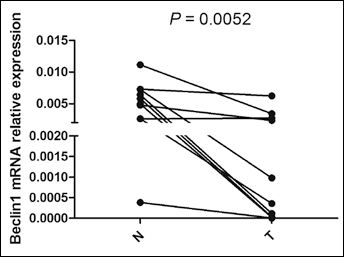
The mRNA level of beclin 1 in bladder cancer tissues. The mRNA level of beclin 1 was examined in 10 cancer tissues and their paired adjacent normal tissues by real-time PCR and was normalized to GAPDH. Beclin 1 mRNA was significantly downregulated in tumor tissues (T) compared with the adjacent normal tissues (N) (p=0.0052).
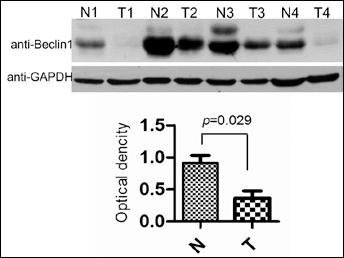
The protein level of beclin 1 in bladder cancer tissues. The protein expression of beclin 1 was analyzed by Western blotting in 4 bladder cancer tissues (T) and their paired adjacent normal tissues (N) (upper panel). The densitometric data analysis (lower panel) showed a decrease in beclin 1 protein expression in bladder cancer tissues compared with the adjacent normal tissues (p=0.029). GAPDH served as an internal loading control.
We then determined the expression of beclin 1 in paraffin embedded tissues. Immunohistochemistry analysis showed that high beclin 1 protein expression was detected in all 20 (100%) normal bladder tissues, while high beclin 1 expression was observed in only 47 (37.01%) of the 127 bladder cancer tissues. Beclin 1 was widely distributed in the epithelial lining of the mucosa within the bladder normal tissue cells and all showed a moderate to strong immunoreactivity (dark brown) in the cytoplasm and occasionally in the nucleus (Figs. 3A and 3B). However, most of the bladder cancer tissue cells showed negative (Figs. 3E and 3F) to moderate staining (Figs. 3C and 3D).
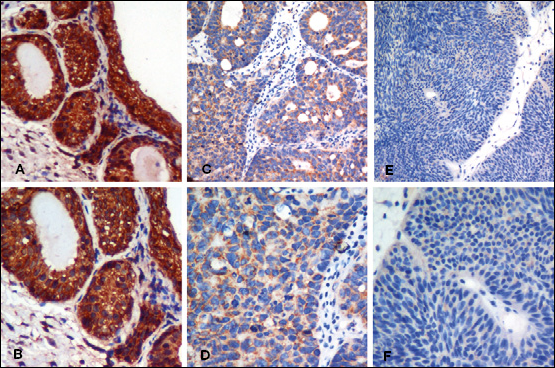
Immunoreactivity of beclin 1 in bladder cancer tissues. The immunoreactivity of the beclin 1 protein was examined by immunohistochemistry in bladder cancer tissues and matched normal tissues. A, B) Beclin 1 has a strong cytoplasmic expression in normal bladder tissues, partially in nuclei (A, ×200; B, ×400). C, D) A moderate cytoplasmic immunoreactivity of beclin 1 expression in bladder cancer cells (C, ×200; D ×400). E, F) A negative beclin 1 expression in bladder cancer tissue (E, ×200; F ×400).
Relationship between beclin 1 protein expression and clinicopathological parameters
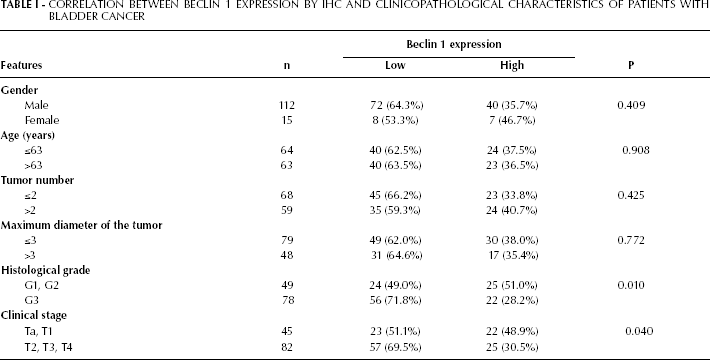
The association between beclin 1 expression and clinical parameters are summarized in Table I. Based on the median immunoreactivity score (4), all patients were divided into 2 groups. Higher beclin 1 expression (≥4) was observed in 47 cases (37.01%) and lower beclin 1 expression (<4) in 80 cases (62.99%). The expression of the beclin 1 protein was reduced by 49.0% in G1 and G2 bladder carcinomas and by 71.8% in G3 bladder carcinomas (p<0.05, with the χ 2 test). In addition, the expression of the beclin 1 protein was decreased by 69.5% in muscle invasive bladder cancer and by 51.1% in nonmuscle invasive bladder cancer (p<0.05). Reduced beclin 1 expression was significantly correlated with higher histological grade (p=0.010) and more advanced clinical stage (p=0.040), but was not associated with gender, age, tumor number, the maximum diameter of the tumor, or recurrence (p>0.05).
Correlation Between Beclin 1 Expression BY IHC AND Clinicopathological Characteristics of Patients with Bladder Cancer
Correlation of beclin 1 expression with overall survival
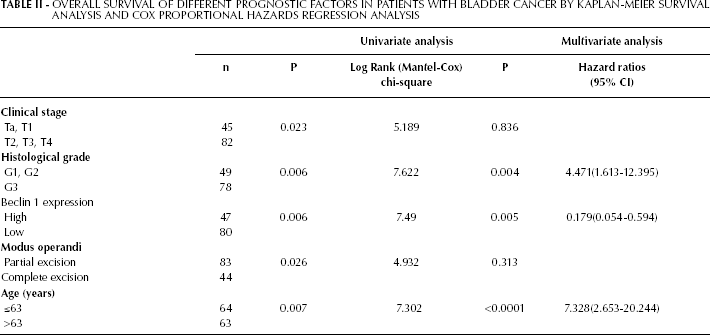
The 5-year survival rate in the lower beclin 1 expression group was 68.78% and was 90.21% in the higher expression group (p=0.005) (Fig. 4). We also performed multivariate survival analysis by the Cox proportional hazard model to test the effects of independent factors (such as beclin 1 expression level, histological classification, clinical stage, age, gender, tumor number and modus operandi) on survival. As presented in Table II, beclin 1 expression level, histological classification and age were independent predictors of survival, suggesting that beclin 1 protein level has a significant association with clinicopathological characteristics. In addition, beclin 1 is probably a protective factor in this type of tumor (β=-1.720, Exp(β)=0.179). Thus, beclin 1 may be a potential prognostic marker for patients with bladder cancer.
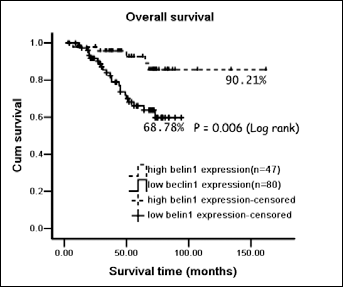
Kaplan-Meier overall survival analysis of bladder cancer patients. All patients were stratified into 2 groups according to the anti-beclin 1 immunoreactivity. Kaplan-Meier overall survival analysis showed that the group with lower beclin 1 expression (n=80) had a lower 5-year survival rate (68.78%) compared to the higher beclin 1 expression group (n=47) (90.21%), p=0.006. The longest follow-up time was 162 months.
Overall Survival of Different Prognostic Factors in Patients with Bladder Cancer BY Kaplan-Meier Survival Analysis and Cox Proportional Hazards Regression Analysis
Discussion
Autophagy, an evolutionary conserved process, has been reported to be involved in many physiological and pathological processes such as development, cell death, tumorigenesis, etc (18, 19). Autophagy is generally considered to be a tumor-suppressive process. Decreased expression of beclin 1, an autophagy gene, has been associated with tumor progression in ovarian, breast, and brain carcinomas, as well as with a lower survival rate in colorectal, esophageal, hepatocellular, and brain carcinomas (13, 20-22), thus suggesting that beclin 1 acts as a tumor suppressor. In our study, we focused on the role of beclin 1 in bladder cancer tissues and found that the expression of beclin 1 in bladder was significantly decreased in terms of protein and mRNA levels compared to the adjacent normal tissues. This finding was further confirmed by immunohistochemistry, which showed a clear difference in the immunoreactivity of beclin 1 in bladder cancer tissues and the adjacent normal tissues, indicating that beclin 1 is involved in bladder cancer progression. A recent report demonstrated that beclin 1 can also control the level of p53 through regulation of deubiquitinating activities of 2 ubiquitin-specific peptidases (USP), USP10 and USP13. These findings provided an important molecular mechanism of beclin 1 as a haploid-insufficient tumor suppressor, which is frequently monoallelically lost in human breast, ovarian, and prostate cancers (12, 23, 24). However, a recent study in Atg5 and Atg7 knockout mice showed that autophagy deficiency might lead to benign tumors in liver, but not in other tissues (25), suggesting that the tumor suppressor function of autophagy-related genes may be various in different tissues. A recent study indicated that autophagy could inhibit cell proliferation after a short period (48 hours) of Ras overexpression and enhance cell proliferation after a long period (2 weeks) of Ras overexpression. H-rasval12 induces different autophagic responses depending on the duration of Ras overexpression (26).
In 2010, Chen and colleagues preliminarily reported the relationship between autophagy and bladder cancer. Pterostilbene, a naturally occurring phytoalexin, could induce autophagy in sensitive and chemoresistant human bladder cancer cells (27), confirming the involvement of autophagy in bladder cancer. However, the exact role and mechanisms of autophagy in bladder cancer still need further exploration. In addition, Huang YC et al reported that para-Phenylenediamine (p-PD), also called 1,4-diaminobenzene or 1,4-phenylenediamine, can induce urothelial cell death. This process referred to the activation of ERK1/2 signaling pathway via mutant p53, and led to stimulation of autophagy in SV-HUC-1 cells (28).
The majority of transitional cell (urothelial) carcinomas (TCCs) occur as papillary non-muscle invasive carcinomas. However, approximately 80% of urothelial bladder cancer patients suffer from recurrence within 1 to 2 years of initial treatment (29). Despite radical cystectomy and systemic therapy, 50% of patients with invasive tumors die from metastasis (30, 31). In recent years, tremendous advances have been made at the molecular level in the discovery of new markers, which have shown considerable clinical relevance in the areas of diagnosis, tumor classification, prognosis, and prediction of individual patient's response to treatment. In this study, beclin 1 was found to be a potential prognostic factor in bladder cancer. The 5-year survival was significantly different between the lower beclin 1 expression group and the higher beclin 1expression group, suggesting that the beclin 1 protein is correlated with the survival rate in bladder cancer. In addition to histological grade and TNM stage, beclin 1 expression level was also an independent prognostic factor for bladder cancer, since lower expression of beclin 1 protein correlated with poor prognosis for patients with bladder cancer. This correlation is consistent with previous studies of beclin 1 in other human cancers, including ovarian, breast, and brain malignancies (13, 20-22).
In conclusion, this study showed that decreased expression of beclin 1 is correlated with higher histopathological grade, advanced TNM stages, and a poor overall survival in bladder cancer patients. This suggests that beclin 1 is involved in human bladder cancer and that downregulation of beclin 1 might serve as a potential prognostic factor in bladder cancer.
