Abstract
Objectives
This study aimed to assess the diagnostic value of human epididymal protein 4 (HE4), a potential novel biomarker for lung cancer, and its combined detection with five other conventional biomarkers in lung cancer diagnosis and subtyping.
Methods
In this retrospective study, 115 lung cancer patients, 50 patients with benign pulmonary disease, and 50 healthy controls were included. Serum HE4, progastrin-releasing peptide (ProGRP), squamous cell carcinoma (SCC) antigen, cytokeratin-19 fragment (CYFRA21-1), neuron-specific enolase (NSE), and carcinoembryonic antigen (CEA) were analyzed using the electrochemiluminescence immunoassay and chemiluminescence immunoassay. The receiver operating characteristic curve was performed to analyze the diagnostic efficacy of individual biomarkers in identifying both lung cancer and its histologic subtypes.
Results
All six biomarkers showed significantly elevated levels in the lung cancer group compared to both benign pulmonary disease and control groups (P < 0.05). Among the biomarkers evaluated, HE4 exhibited the highest diagnostic performance for lung cancer, lung adenocarcinoma, and lung squamous cell carcinoma with area under the curve (AUC) values of 0.921, 0.891, and 0.937, respectively. ProGRP was the optimal biomarker for small cell lung cancer with an AUC of 0.973. The combination of all six biomarkers yielded the largest AUCs in the diagnosis of lung cancer subtypes (0.937 for lung adenocarcinoma, 0.998 for lung squamous cell carcinoma, and 0.985 for small cell lung cancer). Furthermore, specific combinations, such as HE4 + CEA, HE4 + SCC, and ProGRP + HE4 + NSE, showed strong diagnostic performance in lung cancer.
Conclusions
HE4 and its combined detection held substantial clinical significance in the diagnosis of lung cancer and its histologic subtyping, especially for lung adenocarcinoma and lung squamous cell carcinoma.
Keywords
Introduction
Lung cancer (LC) stands as a significant threat to human health and life, with both its incidence and mortality rates consistently on the rise. 1 According to global cancer statistics, in 2020, there were 1.7 million deaths from lung cancer worldwide, making it the leading cause of cancer-related deaths. 2 This prominence can be attributed, in part, to the insidious nature of early-stage lung cancer, as approximately 60% of patients are diagnosed only in the advanced stages. 3 Therefore, early diagnosis and treatment are pivotal in the management of lung cancer.
Pathologic tissue biopsy is a relatively accurate method for clinical lung cancer diagnosis. However, this diagnostic approach can be traumatic for patients and has low patient compliance. 4 In contrast, detecting serum tumor markers offers a simpler diagnostic method for lung cancer, with advantages such as minimal invasiveness and ease of repeatability. 4 Currently, commonly used tumor markers for diagnosing lung cancer include carcinoembryonic antigen (CEA), neuron-specific enolase (NSE), cytokeratin-19 fragment (CYFRA21-1), squamous cell carcinoma antigen (SCC), and progastrin-releasing peptide (ProGRP). However, their sensitivity and specificity often fall short of the desired requirements. 5 The emergence of human epididymis protein 4 (HE4) holds promise in filling this diagnostic gap.
HE4 is a small molecule secreted glycoprotein, which has been studied as a tumor marker primarily in patients with ovarian cancer. Back in 2006, Bingle et al. 6 found that this glycoprotein may play a potential role in LC development as a component of the innate immune system of the lung and the respiratory tract. As research progressed, the potential value of HE4 in the diagnosis and assessment of LC was becoming increasingly noticeable, but the results were subject to different possibilities. 7 For instance, the findings of Wang et al. 8 and Wojcik et al. 9 suggested that HE4 was a suitable diagnostic marker for patients with lung squamous cell carcinoma (LUSC) and small cell lung cancer (SCLC). On the other hand, several studies have shown that HE4 was expressed in lung adenocarcinoma (LUAD), but was not common in LUSC and SCLC.10,11 Therefore, more sufficient evidence is needed to support the clinical application of HE4 in LC diagnosis.
In this study, we analyzed and compared the efficacy of HE4 and five other biomarkers including SCC, CEA, CYFRA21-1, NSE, and ProGRP—alone or in combination—for the diagnosis of LC, with the aim of providing more evidence for the potential utility of HE4 in the clinical diagnosis of LC.
Methods
Study design and population
This was a retrospective study conducted at Wenzhou Central Hospital. Clinical data were collected for analysis from patients with LC, patients with benign pulmonary diseases (BPD), and healthy controls from July 2021 to July 2023. All eligible LC patients in this study were newly diagnosed with LC and met the diagnostic criteria for LC in the Chinese Medical Association Clinical Guidelines 12 (with pathological tissue biopsy as gold standard) and had not undergone neoadjuvant chemotherapy or radiotherapy. Patients with concurrent malignancies in other organ systems, severe liver or kidney dysfunction, rheumatic diseases, immune system disorders, and those with incomplete clinical data that could affect the analysis were excluded from the study. Patients eligible for inclusion with BPD included those with chronic obstructive pulmonary disease, asthma, pneumonia, and benign lung tumors. 13 Healthy controls were apparently healthy individuals undergoing routine physical examination at our hospital.
As depicted in Figure 1, this study encompassed a cohort of 115 LC patients, with ages ranging from 28 to 81 years and an average age of 51 ± 16 years. Additionally, 50 patients with BPD, aged between 39 and 73 years with an average age of 53 ± 11 years, were included. Furthermore, 50 healthy individuals were recruited as the control, with ages from 33 to 71 years and an average age of 52 ± 10 years.
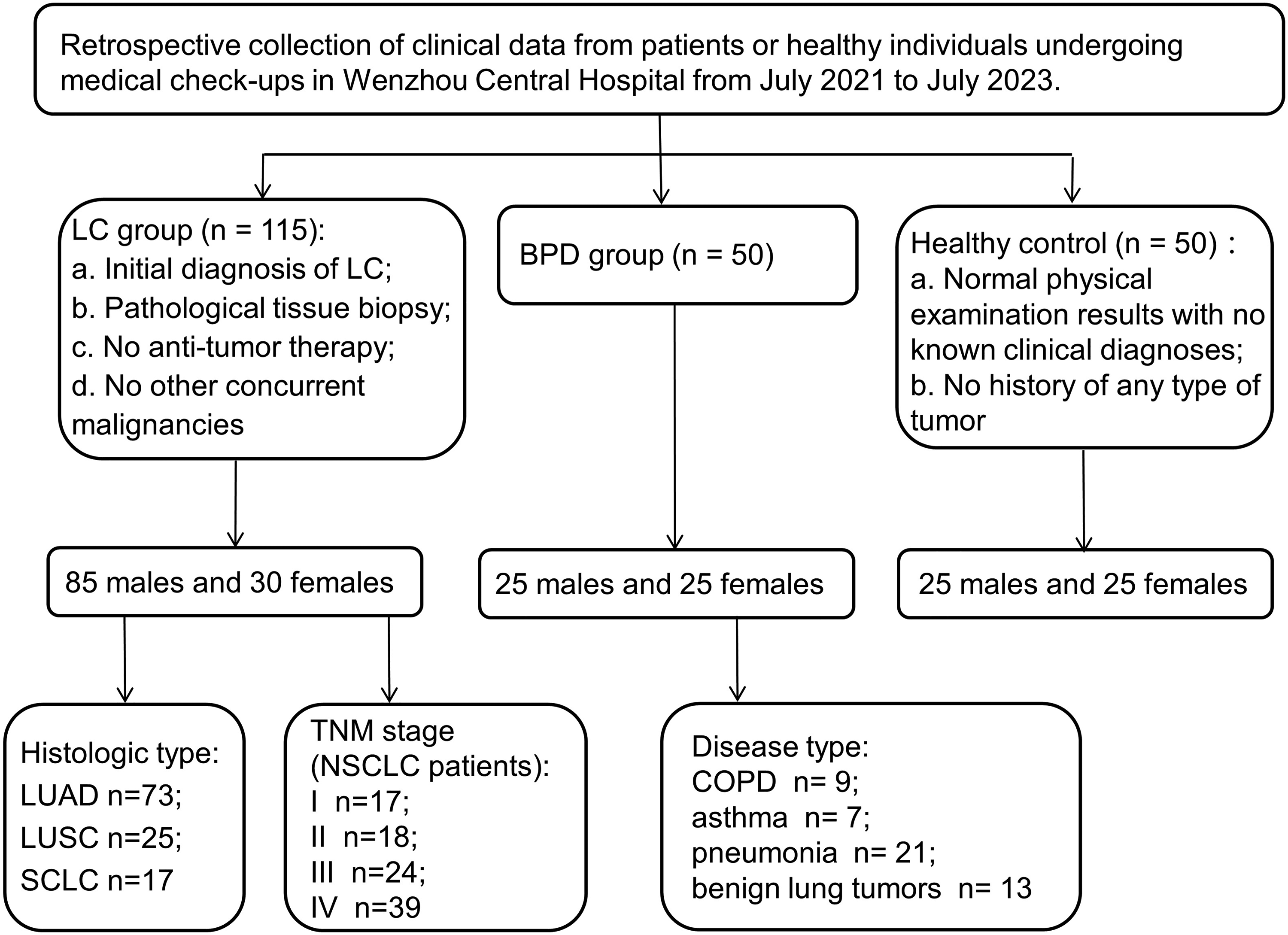
Patient enrollment flowchart.
This study adhered to the principles of the Helsinki Declaration and received approval from the Ethics Committee of Wenzhou Central Hospital [REC number: L2023-04-084]. Informed consent was obtained from all participants in this study.
Sample collection and detection
All subjects had venous blood collected upon admission in accordance with specimen collection protocols. Blood samples were obtained from the LC group and BPD group before surgery or treatment, while healthy controls provided samples during medical check-ups. The collected blood samples were promptly stored at −80.0°C within 2 hours after collection and remained at that temperature until testing.
Serum CEA concentrations in all samples were analyzed by chemiluminescence using a Beckman Coulter UniCel DXI 800 analyzer and CEA kit (Beckman Instruments Inc., Brea, CA, USA), following the instructions on the instrument and reagent manual. The remaining five tumor markers (ProGRP, NSE, CYFRA21-1, SCC, and HE4) were detected using the Cobase602 analyzer and its accompanying kits (Roche Diagnostics GmbH, Mannheim, Germany) by following the manufacturer's instructions, and were analyzed by electrochemiluminescence immunoassay to analyze the serum concentration in the samples.
Statistical analyses
SPSS 20.0 statistical software (IBM Corp., Armonk, NY, USA) was used for data analysis. Normally distributed continuous variables were expressed as mean ± standard deviation, and intergroup comparisons were conducted using one-way analysis of variance. Non-normally distributed continuous variables were expressed as median (quartiles), and two-group comparisons were made using the Mann–Whitney U test, while multiple-group comparisons were conducted using the Kruskal–Wallis H test.
Receiver operating characteristic (ROC) curves were generated using pathology diagnosis results as the gold standard to evaluate the diagnostic performance of serum HE4, ProGRP, NSE, CYFRA21-1, SCC, and CEA, both individually and in combination. The optimal cut-off values were determined by calculating the Youden Index. A difference was considered statistically significant when P < 0.05.
Results
Comparison of serum levels of six biomarkers in three groups of subjects
A total of 115 LC patients, 50 patients with BPD, and 50 healthy individuals were included in this study. In the LC group, serum levels of HE4, ProGRP, CYFRA21-1, SCC, NSE, and CEA were all significantly higher than those in the BPD group and the healthy control (P < 0.05). There were no statistical differences in serum levels of six biomarkers (HE4, ProGRP, NSE, CYFRA21-1, SCC, and CEA) between the BPD group and the healthy controls (all P > 0.05, as shown in Table 1). Moreover, in LC patients with varying histological types, significant differences were observed in the serum levels of all biomarkers (Table 2).
Comparison of serum levels of six biomarkers from LC, BPD, and healthy controls groups [M (Q1, Q3)].
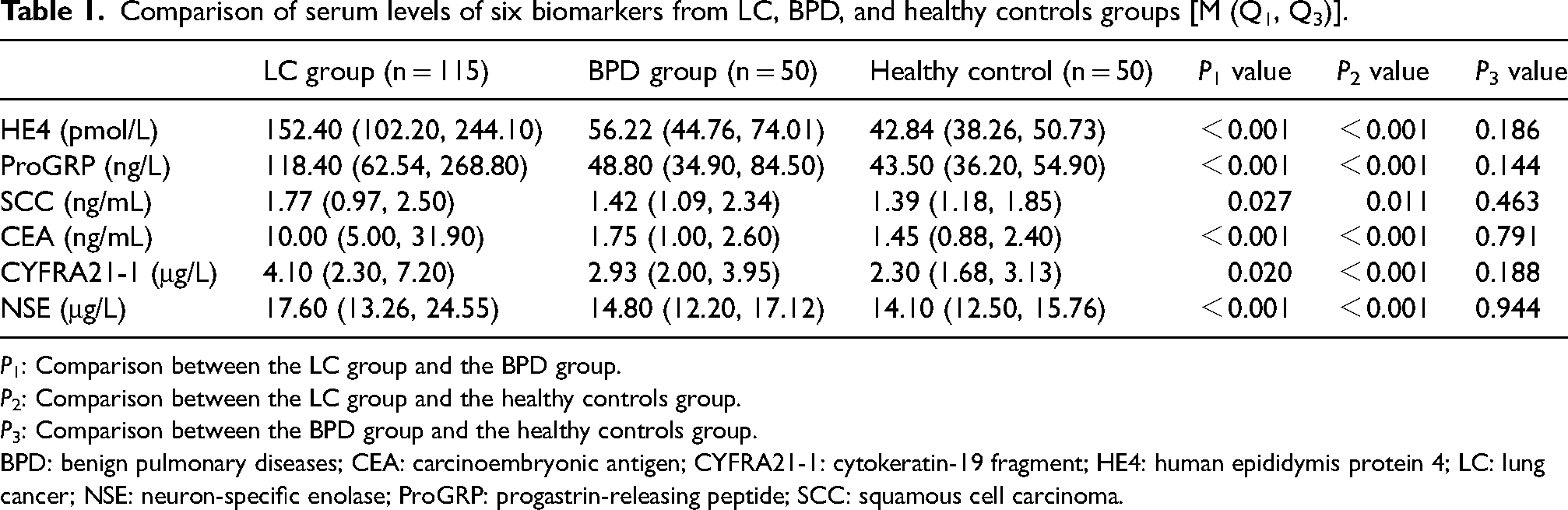
P1: Comparison between the LC group and the BPD group.
P2: Comparison between the LC group and the healthy controls group.
P3: Comparison between the BPD group and the healthy controls group.
BPD: benign pulmonary diseases; CEA: carcinoembryonic antigen; CYFRA21-1: cytokeratin-19 fragment; HE4: human epididymis protein 4; LC: lung cancer; NSE: neuron-specific enolase; ProGRP: progastrin-releasing peptide; SCC: squamous cell carcinoma.
Comparison of serum levels of six biomarkers in patients with different histological types of lung cancer [M (Q1, Q3)].
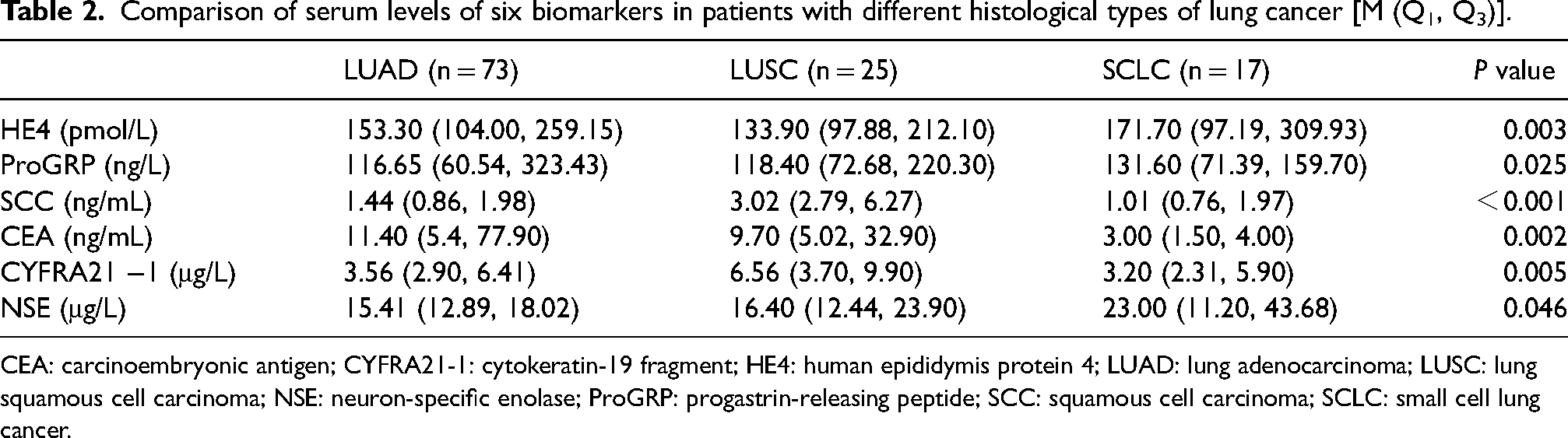
CEA: carcinoembryonic antigen; CYFRA21-1: cytokeratin-19 fragment; HE4: human epididymis protein 4; LUAD: lung adenocarcinoma; LUSC: lung squamous cell carcinoma; NSE: neuron-specific enolase; ProGRP: progastrin-releasing peptide; SCC: squamous cell carcinoma; SCLC: small cell lung cancer.
Diagnostic efficacy of six biomarkers alone and combined for LC diagnosis
Among individual biomarkers, HE4 had the highest area under the curve (AUC) in distinguishing LC from healthy controls (0.921; 95% CI 0.876, 0.966), followed by CEA (0.886; 95% CI 0.837, 0.935). Subsequently, we attempted to combine the optimal two to three biomarkers or all six indicators for diagnostic purposes. ROC analysis results demonstrated that the combination of six biomarkers yielded the most favorable diagnostic performance (AUC = 0.939; 95% CI 0.903, 0.974), as depicted in Table 3 and Figure 2(a).
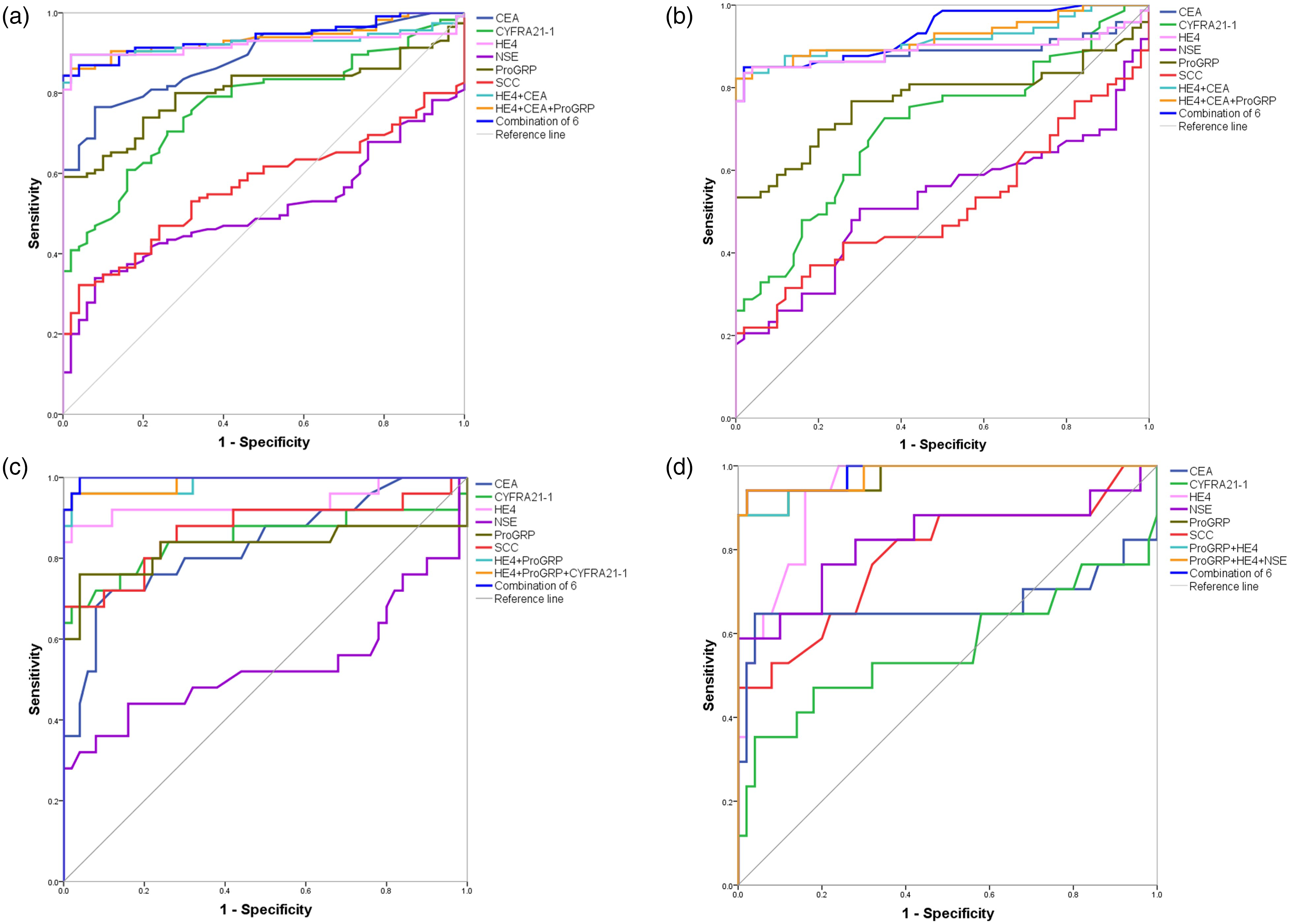
ROC curves of six biomarkers and their combinations in: (a) LC diagnosis; (b) LUAD diagnosis; (c) LUSC diagnosis; and (d) SCLC diagnosis.
Diagnostic efficacy of six biomarkers alone and combined in lung cancer.
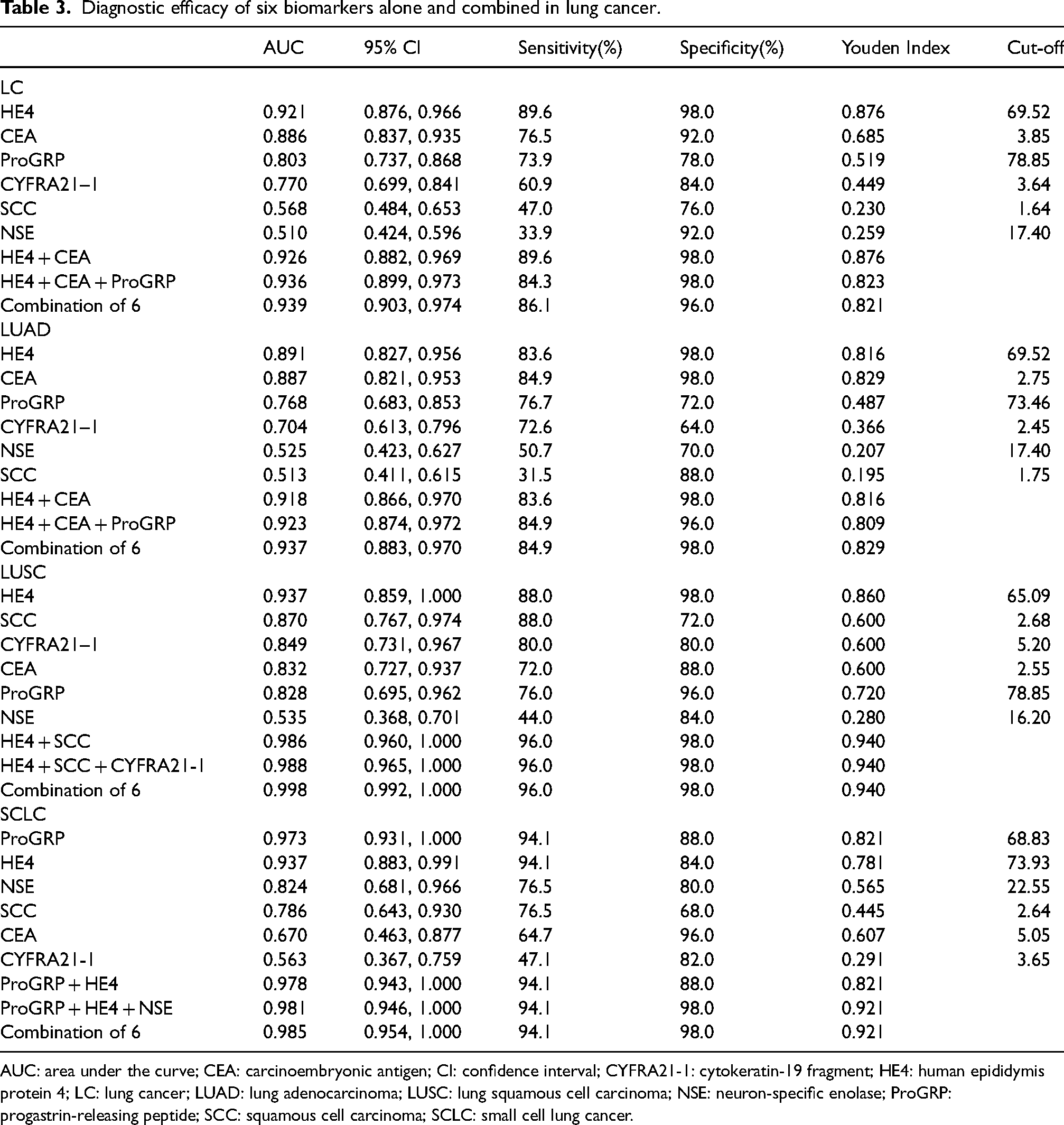
AUC: area under the curve; CEA: carcinoembryonic antigen; CI: confidence interval; CYFRA21-1: cytokeratin-19 fragment; HE4: human epididymis protein 4; LC: lung cancer; LUAD: lung adenocarcinoma; LUSC: lung squamous cell carcinoma; NSE: neuron-specific enolase; ProGRP: progastrin-releasing peptide; SCC: squamous cell carcinoma; SCLC: small cell lung cancer.
Diagnostic efficacy of six biomarkers alone and combined for diagnosis of LUAD, LUSC and SCLC
The identification of histological subtypes played a pivotal role in guiding the treatment for LC. As previously mentioned, the elevation of serum biomarkers has been associated with distinct histological subtypes, so we investigated the effectiveness of histological subtyping based on these biomarkers.
Among the six biomarkers, HE4 exhibited the most robust diagnostic performance with the highest AUC and 98.0% specificity in distinguishing LUAD and LUSC from healthy controls (LUAD: AUC = 0.891; 95% CI 0.827, 0.956; LUSC: AUC = 0.937; 95% CI 0.859, 1.000). ProGRP emerged as the optimal biomarker for SCLC diagnosis (AUC = 0.973; 95% CI 0.931, 1.000), demonstrating a sensitivity of 94.1% and a specificity of 88.0%.
ROC analysis showed that the combination of the six biomarkers exhibited the largest AUCs in the diagnosis of three pathological types of LC (0.937 for LUAD, 0.998 for LUSC, and 0.985 for SCLC). Additionally, the combinations of HE4 + CEA and HE4 + CEA + ProGRP had AUCs of 0.918 and 0.923, respectively, in discriminating between LUAD and healthy controls. For LUSC, HE4 + SCC and HE4 + SCC + CYFRA21-1 exhibited sensitivities and specificities comparable to the combination of all six, albeit with slightly lower AUCs. Furthermore, the diagnostic efficacy of the combination ProGRP + HE4 + NSE in distinguishing SCLC from healthy individuals was closely aligned with the performance of the combination of the six (AUC = 0.98; 94.1% sensitivity, 98.0% specificity), suggesting it as an ideal simplified combination. More details are referred to in Table 3 and Figure 2(b) to (d).
Discussion
This study demonstrated that HE4, ProGRP, CEA, NSE, CYFRA21-1, and SCC possessed good or moderate capabilities in characterizing at least one subtype of LC. Among these biomarkers, HE4 emerged as the most effective biomarker for distinguishing LUAD and LUSC from healthy controls, while ProGRP exhibited superior diagnostic performance for SCLC. The combined diagnosis of histologic subtypes using all six biomarkers yielded the largest AUCs. Furthermore, specific combinations, such as HE4 + CEA, HE4 + SCC, and ProGRP + HE4 + NSE, showed strong diagnostic performance, suggesting their potential as simplified patterns for combined detection of the six biomarkers.
According to selected previous studies, the AUC of HE4 in distinguishing LC from healthy controls ranged from 0.761 to 0.998. Similarly, the cut-off values determined based on the Youden Index exhibited some variation.14–17 Our study yielded results (AUC = 0.921; 69.52 pmol/L) close to those reported by Iwahori et al. 14 (AUC = 0.988; 65.6 pmol/L). However, the cut-off value in the present study was slightly lower than the findings of Wang et al. 18 (76.89 pmol/L) and higher than those of Li et al. 7 (60.14 pmol/L). The variations in the aforementioned outcomes may be attributed to differences in the demographic characteristics of the study cohorts, including gender, age, and tumor type. Prior research has shown that the serum HE4 level was higher in males than in females, and elevated HE4 levels exhibited a positive correlation with age progression and tumor stage advancement. 19 Specifically, in Li et al’s 7 and Wang et al's 18 LC groups, the male-to-female ratios were approximately 1.3 and 1.6, while it was 2.8 in this study, aligning more closely with the Chinese male-to-female incidence of LC, which stands at 2.5. The mean age of our subjects was significantly younger than Wang et al's 18 (51 ± 16 vs. 61 ± 13). In addition, Li et al's 7 findings had a larger proportion (70.7%) of patients with early-stage LC, whereas the proportion of patients with stage III–IV LC in this study was 64.3%.
This study further investigated the diagnostic efficacy of the six biomarkers in histologic subtyping of LC. As previously noted, HE4 and ProGRP were found to be the optimal biomarkers for histologic subtyping, with HE4 being identified for LUAD and LUSC, and ProGRP for SCLC. The results were consistent with those obtained by Zeng et al. 15 and Tang et al. 20 ProGRP, the precursor form of gastrin-releasing peptide, was widely recognized for its clinical significance in both the diagnosis and prognosis of SCLC. Numerous studies have shown the high sensitivity of ProGRP in the diagnosis of SCLC, especially when utilized in combination with established tumor markers such as NSE and CYFRA21-1.21,22
Combined detection typically enhanced overall diagnostic reliability compared to individual biomarkers due to the complementary nature of different indicators, a consensus supported by most of the previous reports.23,24 Our study revealed that the combined detection of all six biomarkers yielded an impressive AUC ranging from 0.937 to 0.998 in diagnosing the three subtypes of LC, coupled with a high specificity of 98.0% (Table 3). This suggested that the combined detection of these six biomarkers held substantial potential for application in the precise diagnosis of LC subtypes. To provide more practical options for clinical diagnosis, we incorporated two- and three-biomarker combinations in our analysis. For instance, in SCLC, we observed that the AUC of the ProGRP + HE4 + NSE closely mirrored that of the combination of the six (0.981 vs. 0.985), with equivalent sensitivity and specificity. Remarkably, this combination also emerged as the most effective set for diagnosing SCLC patients in a study conducted by Li and colleagues. 19 Therefore, this combination may stand out as an ideal choice for SCLC identification, and be considered a viable alternative to the six-biomarkers-combined detection, particularly under limited conditions.
There were some limitations of this study:
1. Lack of subgroup analysis for demographic characteristics: Considering the potential impact of variables such as age, gender, or tumor type, future investigations could aim to conduct comprehensive subgroup analyses with larger samples, to better understand the nuances of biomarker utility in diverse patient populations.
2. Relatively few patients with early-stage NSCLC were included in this study, with a proportion of 36%. Despite the high diagnostic efficacy of the combination of the six biomarkers in this study, the sensitivity and specificity may be lower than the above values in identifying some NSCLC patients with small lesions. Furthermore, given the current challenges in early LC diagnosis, a combination of multiple tests remains a more prudent recommendation. For example, liquid-biopsy-based testing has proven to be highly beneficial for identifying tumor markers, especially when solid tissue biopsies are insufficient or unattainable. 25 Existing radiomics techniques for early diagnosis of LC, such as 18F-FDG-positron emission tomography/computed tomography imaging,26,27 are also more established. We support future studies exploring more robust methods for primary LC diagnosis from the perspective of combining biomarkers such as HE4 with the above assays.
Conclusions
HE4 was a promising biomarker for LC diagnosis, particularly for LUAD and LUSC. Its combined use with other biomarkers demonstrated significant clinical value in the histological subtyping of LC.
Footnotes
Acknowledgements
Not applicable.
Author contributions
LW and XC researched literature and conceived the study. TP and ET were involved in protocol development, gaining ethical approval, patient recruitment, and data analysis. WB and LC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The Ethics Committee of Wenzhou Central Hospital approved this study (REC number: L2023-04-084).
Funding
This research was funded by the University-Industry Collaborative Education Program [grant number: 202102242018].
University-Industry Collaborative Education Program, (grant number 202102242018).
