Abstract
Objective
Radiation-induced trismus (RIT), one of the rare but serious side effects of concurrent chemoradiotherapy (C-CRT), is difficult to predict with high accuracy. We aimed to examine whether the pretreatment pan-immune-inflammation value (PIV) measures predict RIT in patients with locally advanced nasopharyngeal carcinoma (LA-NPC) receiving C-CRT.
Methods
Data of patients with LA-NPC who underwent C-CRT and had maximum mouth openings (MMO) > 35 mm were reviewed. Any MMO of 35 mm or less after C-CRT was considered RIT. All PIV values were computed using the complete blood count test results: PIV = (Platelets × Monocytes × Neutrophils) ÷ Lymphocytes. The receiver operating characteristic analysis was employed to dissect a possible association between pre-treatment PIV readings and RIT status. Confounding variables were tested for their independent relationship with the RIT rates using logistic regression analysis.
Results
The research comprised 223 participants, and RIT was diagnosed in 46 (20.6%) at a median time from C-CRT to RIT of 10 months (range: 5–18 months). Pre-C-CRT PIV levels and RIT rates were analyzed using receiver operating characteristic curve analysis, with 830 being the optimal cutoff (area under the curve: 92.1%; sensitivity: 87.5%; specificity: 85.5%; Youden index: 0.730). RIT was significantly more prevalent in the PIV > 830 cohort than its PIV ≤ 830 counterpart (60.3% vs. 5%; hazard ratio 5.79; P < 0.001). Multivariate logistic regression analysis revealed that advanced T-stage (P = 0.004), masticatory apparatus dose V58Gy≥%32 (P = 0.003), and PIV > 830 (P < 0.001) were independently linked with significantly elevated rates of RIT.
Conclusion
The presence of elevated pre-C-CRT PIV is a unique biological marker that independently predicts increased RIT rates in LA-NPC undergoing C-CRT.
Keywords
Introduction
Combining radiation therapy and chemotherapy, as opposed to each modality alone or sequentially, has increased the cure rates for locally advanced nasopharyngeal carcinoma (LA-NPC) over the past several decades. Individual patient data from the Meta-Analysis of Chemotherapy (MAC) in NPC clearly showed that adding concurrent chemotherapy (C-CRT) to radiotherapy (RT) improves overall survival, progression-free survival, locoregional control, and distant control rates in this patient group. 1 Hence, RT is the backbone of the curative therapy of LA-NPCs and is also beneficial as a salvage re-irradiation or palliative intervention in carefully chosen patients.1,2 Despite the widespread use of intensity-modulated RT (IMRT) in treatment algorithms, studies have shown that there is still a high risk of acute and long-term local-regional side effects from RT and C-CRT that can exceed 50%. 3 Some of the most common side effects are dry mouth, trouble swallowing, dental caries, periodontal disease, mucositis, skin vascular abnormalities, and scarring of the skin or mucosa.
Radiation-induced trismus (RIT), one of the most dreadful side effects of C-CRT, is difficult to predict with a high degree of accuracy and has a terrible impact on the quality of life of those who experience it.4–6 RIT may result in many adverse health consequences, including dysphagia, dysgeusia, ageusia, dental disease, orofacial pain, oral infections, and even osteoradionecrosis and difficulties with intubation.7,8 Although much remains unknown about the process, it is commonly accepted that radiation-induced inflammation, endothelial damage, hypoxia, and fibrosis are the primary reasons for its onset and development.7,8 Tissues experience elevated oxidative stress owing to the increased release of radiation-induced tissue damage-associated enzymes, leading to tissue ischemia and vascular thrombosis, 9 which further exacerbates local tissue damage and triggers the release of inflammatory cytokines and chemokines. 10 Because of hypoxia and radiation-induced fibrosis of the masticatory apparatus, this inflammatory response ultimately leads to RIT. 11 This information makes it plausible to hypothesize that some biomarkers may be able to predict RIT rates after C-CRT with accuracy. This hypothesis is supported by earlier research that suggests the neutrophil-to-platelet ratio (NLR) and hemoglobin-to-platelet ratio (HPR) may be crucial in the development of RIT and precise prevalence prediction.12,13 Somay et al. 12 recently examined the impact of NLR on the prevalence of RIT in 51 parotid cancer patients who received RT and found that it was more prevalent in the patient group with NLR higher than the established cutoff value of 2.7 compared to the other group (35.2% vs. 5.8%; P < 0.001). The same authors also assert that low pre-C-CRT hemoglobin levels and pre-treatment HPR values were highly potent in predicting the prevalence of RIT in NPC patients after C-CRT.12,13 These studies support the idea that RT-induced inflammation activates blood-borne markers and possibly related proinflammatory cytokines, which in turn leads to the formation of RIT.
The pan-immune-inflammation value (PIV) is a recently developed immune-inflammatory blood-based composite biomarker integrating neutrophils, platelets, monocytes, and lymphocytes. 14 The novel PIV, which functions as a mirror of systemic inflammation and immune activation, is a factor that is an indicator of aggravated inflammation in patients with disorders like vascular occlusion, atherosclerosis, and different types of cancer.15–17 Arthritis and venous occlusion leading to fibrosis have been linked to inflammation in prior research.18,19 It has also been hypothesized that inflammatory mediators such as tumor necrosis factor-alpha, vascular endothelial growth factor (VEGF), transforming growth factor - beta 1 (TGF-β1), and interleukin-10 (IL-10) contribute to systemic inflammation's fibrosis-causing effects.20–22 Based on these theories, it is conceivable that high PIV levels may accelerate the fibrosis process, leading to excessive tissue scarring and ultimately to RIT.20,15 Interestingly, despite the existence of such solid data, no study has yet looked into how pre-C-CRT PIV levels affect the prevalence of RIT in any head and neck cancer location. Therefore, the current retrospective study sought to determine whether the pre-treatment PIV had a reliable predictive value for the occurrence of RIT in LA-NPC patients receiving C-CRT.
Patients and methods
Ethics, consent, and permissions
The retrospective study was performed following the principles outlined in the Helsinki Declaration and its later revisions and was authorized by the Institutional Review Board of the Baskent University Medical Faculty (Project no.: DKA 19/39). Before the C-CRT began, all participants provided written informed consent for data collection and dissemination of associated results in line with our institution's norms.
Patient population
The Baskent University Medical Faculty's Department of Radiation Oncology and the Oral and Dental Health Clinics collaborated on this retrospective investigation. Patients were identified by reviewing the medical records of LA-NPC patients who visited our dentistry clinic for comprehensive oral and dental exams before and after C-CRT between January 2012 and January 2022. Patients with LA-NPC who did not have temporomandibular joint disorders (TMD) prior to C-CRT were included in the study using the established criteria for TMD (Diagnostic Criteria/TMD). 17
Patients were considered eligible if they were between the ages of 18 and 80 years, had an ECOG performance status of 0 or 1, had histopathologic evidence of squamous cell NPC and locally advanced disease per the American Joint Committee on Cancer (AJCC) 8th edition (T3–4aN0-3M0), had no prior history of cancer, had not previously received systemic chemotherapy or RT to the head and neck, and had received definitive C-CRT with at least one course of concurrent chemotherapy. Eligibility also required the availability of a RIT-free oral examination and complete blood counts before the C-CRT, as well as electronic records of RT dosimetry and post-C-CRT oral exams. According to the widely acknowledged research of Dijkstra and colleagues, a maximum mouth opening (MMO) of 35 mm or less was considered “trismus,” 23 and patients who were below this limit at the time of admission were disqualified from the study. Edentulous individuals, patients with missing maxillary and/or mandibular central incisors, prior TMJ surgery, TMD, TMJ ankylosis, TMJ dislocation, head and neck trauma, muscle-related pain or myofascial pain syndrome, or primary tumor, or nodal invasion in the masticatory muscles were also excluded.
Pre- and post-treatment MMO measures for the assessment of trismus
Oral and dental evaluations were completed 2 weeks before the start of C-CRT. Post-treatment evaluations continued until the last visit, the patient’s passing, or the follow-up loss. The interincisal distance was used to work out pre- and post-C-CRT MMOs: the distance between the matching incisal edges of the upper and lower central incisors, measured with the Therabite® motion scale (Atos Medical AB, Hörby, Sweden) while the mouth was opened as wide as it could go.24,25 Three MMO readings were taken in succession from each patient by an experienced oral and maxillofacial surgeon (Efsun Somay), and the mean of those readings was noted. For the diagnosis of trismus, MMO measures were taken before C-CRT and again at 1, 3, 6, 9, and 12 months after C-CRT, then at each planned visit or as required, using preset trismus criteria of an MMO of 35 mm or less. 23
Measurements of PIV values
The pre-C-CRT PIV values were derived from routine complete blood count test results obtained on the first day of C-CRT, using the original PIV formula: PIV = (P × M × N) ÷ L, where P, M, N, and L represent the pretreatment platelet, monocyte, neutrophil, and lymphocyte counts, respectively. 14
Treatment protocol
Our standard institutional RT practice advises simultaneous integrated boost IMRT, as used here, for all LA-NPC patients. All RT target volumes were defined using pretreatment co-registered computed tomography (CT), 18-FDG-PET-CT, and/or magnetic resonance imaging (MRI) scans of the affected primary site and the entire neck. 26 The target volumes and corresponding RT doses were as previously reported. 26 Briefly, for high-, intermediate-, and low-risk planning target volumes, the doses were 70.0, 59.4, and 54 Gy over 33 daily fractions administered in a 5-days-a-week schedule. 26 One to three cycles of cisplatin and 5-fluorouracil chemotherapy were administered concurrently with RT (every 21 days), depending on tolerance. The same chemotherapy regimen was recommended for two courses for all patients receiving adjuvant therapy.
Statistical analysis
For this study, the primary endpoint was the possibility of a relationship between pre-C-CRT PIV levels and the rates of RIT. The RIT diagnosis times were determined as the interval between the last day of C-CRT and the established RIT diagnosis. Percentage frequency distributions were used to illustrate the distribution of categorical variables, while medians and ranges were used to characterize continuous variables. Continuous variables that could divide the entire study group into two groups with significantly different outcomes were estimated before and after C-CRT using receiver-operating characteristic (ROC) curve analysis. Only those variables that were significant in the univariate analysis were used in the multivariate logistic regression analysis. All P-values were two-tailed, and a value of < 0.05 was considered statistically significant.
Results
Data from 223 patients stood eligible for investigation after a retrospective review of the institutional data records. The median age of the entire cohort was 57 years old (range: 18–76), and 155 (69.5%) were male, as shown in Table 1. A total of 62.3% and 32.3% of patients had past alcohol and tobacco use, respectively. Most patients were at T3–4 (N = 162, 72.6%) or N2–3 (N = 180, 80.6%) clinical stages. The median pre-C-CRT MMO was 41.4 mm (range: 37.8–46.8 mm), which decreased by 3.1 mm (7.5%) during final post-CCRT measurements to 38.3 mm (range: 25–44 mm) (Table 1 and Table 2).
Baseline and treatment characteristics of the whole study cohort per pan-immune-inflammation value group.
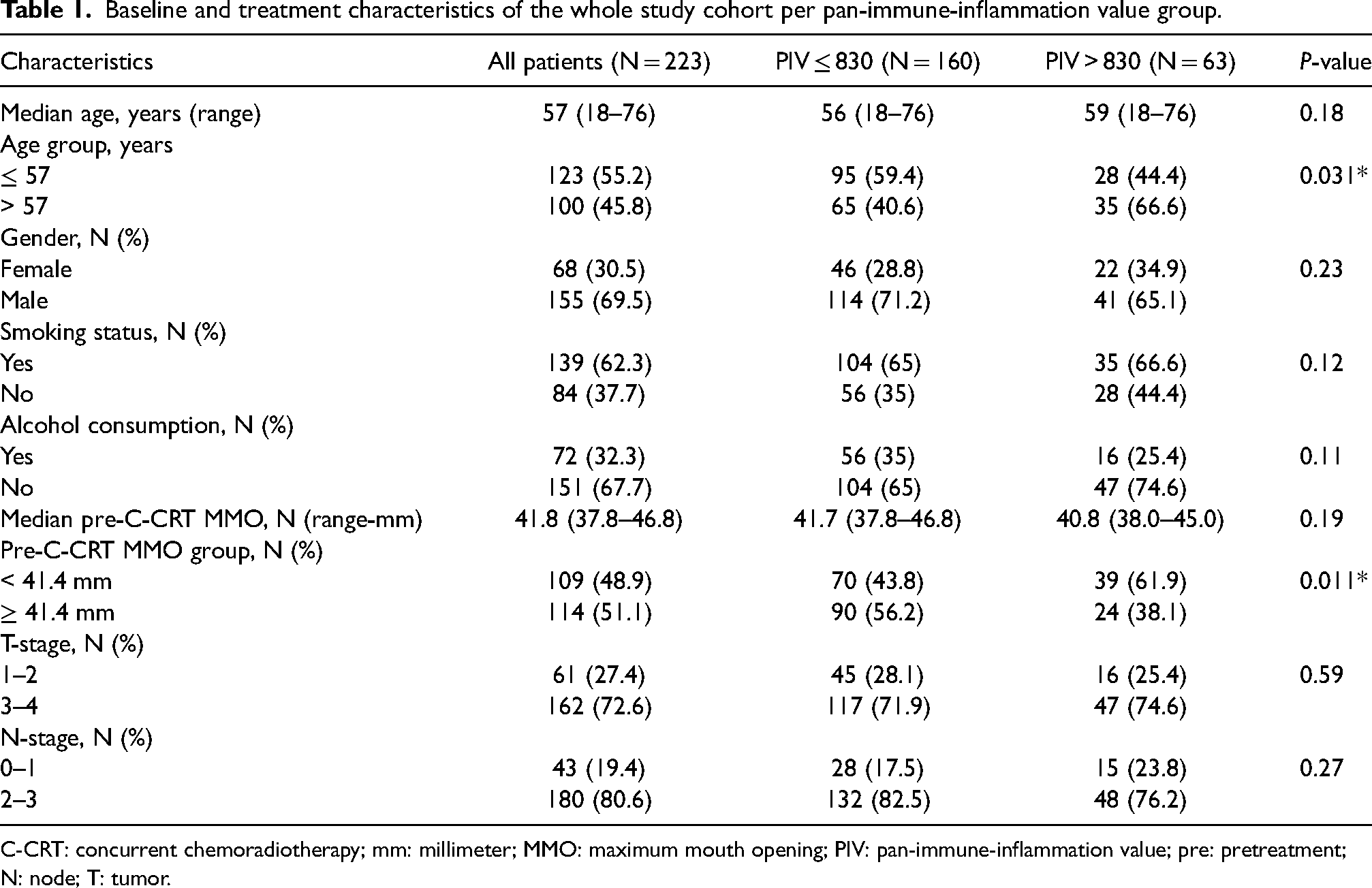
C-CRT: concurrent chemoradiotherapy; mm: millimeter; MMO: maximum mouth opening; PIV: pan-immune-inflammation value; pre: pretreatment; N: node; T: tumor.
Treatment characteristics of the entire study cohort per pan-immune-inflammation value group.
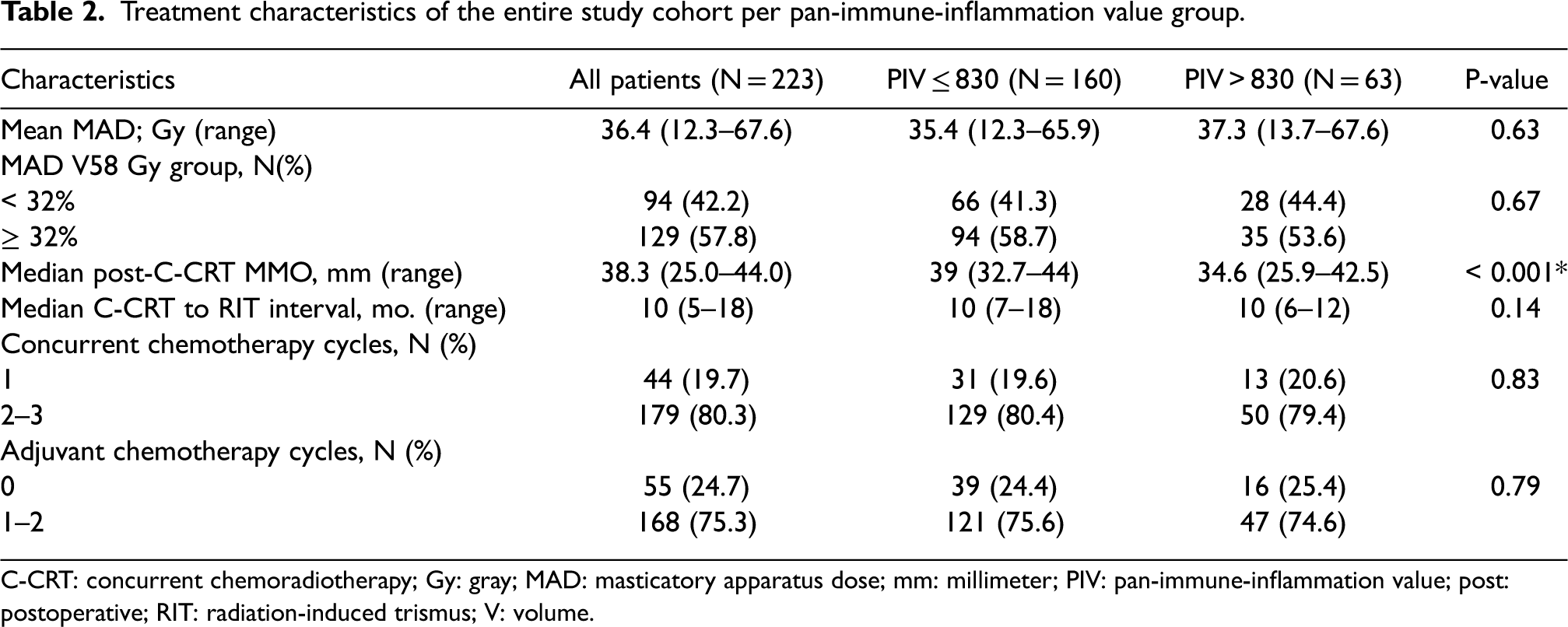
C-CRT: concurrent chemoradiotherapy; Gy: gray; MAD: masticatory apparatus dose; mm: millimeter; PIV: pan-immune-inflammation value; post: postoperative; RIT: radiation-induced trismus; V: volume.
According to Dijkstra et al.'s 23 well-respected MMO ≤ 35 mm criterion, RIT was diagnosed in 46 individuals with a prevalence of 20.6%. The median duration between the completion of C-CRT and the diagnosis of RIT was 10 months (range: 5–18 months). Using ROC analysis (Figure 1), we investigated the prospect of a connection between pre-C-CRT PIV levels and RIT rates and found that 830 was the optimal cutoff value (area under the curve (AUC): 92.1%; sensitivity: 87.5%; specificity: 85.5%; Youden index: 0.730). This cutoff consequently divided the entire research population into two groups: Group 1: PIV ≤ 830 (N = 160) and Group 2: PIV > 830 (N = 63). A cross-tabulation using a Chi-square test between the PIV groups and RIT status revealed that the prevalence of RIT was significantly higher in the PIV > 830 cohort than in the PIV ≤ 830 cohort (60.3% vs. 5.0%; odds ratio (OR); 5.79; P < 0.001], despite the comparable total and per-fraction RT doses among the groups (Figure 1). The volume of the masticatory apparatus dose (MAD) of V58 Gy received by 32% (AUC: 77.2%; sensitivity: 70.6%; and specificity: 69.7%; Youden index: 0.412) of the masticatory apparatus and the median pre-C-CRT MMO of 41.4 mm (AUC: 74.4%; sensitivity: 58.2%; and specificity: 76.1%; Youden index: 0.34) were shown to have a significant connection with RIT incidence rates among all other continuous covariates (Table 3). A comparison of the V58 Gy groups indicated that RIT was considerably more prevalent in the MAD V58 Gy ≥ 32% group than in the MAD V58 Gy < 32% group (28.2% vs. 8.2% for MAD V58 Gy < 32%; OR: 4.23; P = 0.002). Similarly, comparisons between the two pre-C-CRT MMO groups revealed that MMO ≤ 41.4 mm group had a higher RIT incidence risk (31.1% vs. 9.6%; OR: 5.14; P < 0.001) than its MMO < 41.4 mm counterpart (Table 3).
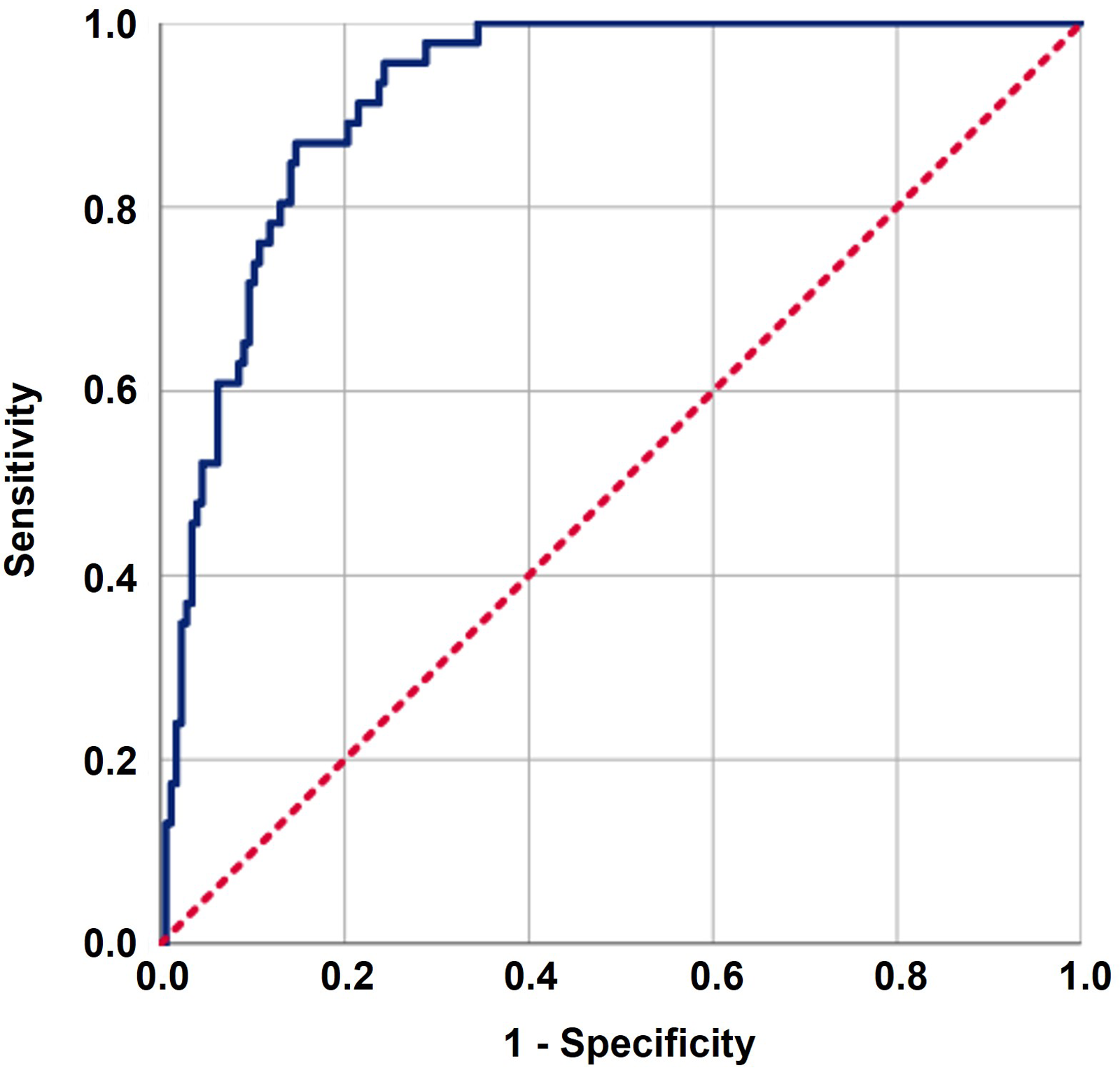
The outcomes of a receiver operating characteristic (ROC) curve analysis examining the correlation between pan-immune-inflammation values (PIV) and post-concurrent-chemoradiotherapy (C-CRT) radiation-induced trismus rates following (PIV cutoff: 830; area under the curve (AUC): 92.1%; sensitivity: 87.5%; specificity: 85.5%; Youden index: 0.730).
Radiation-induced trismus outcomes of the entire study group.
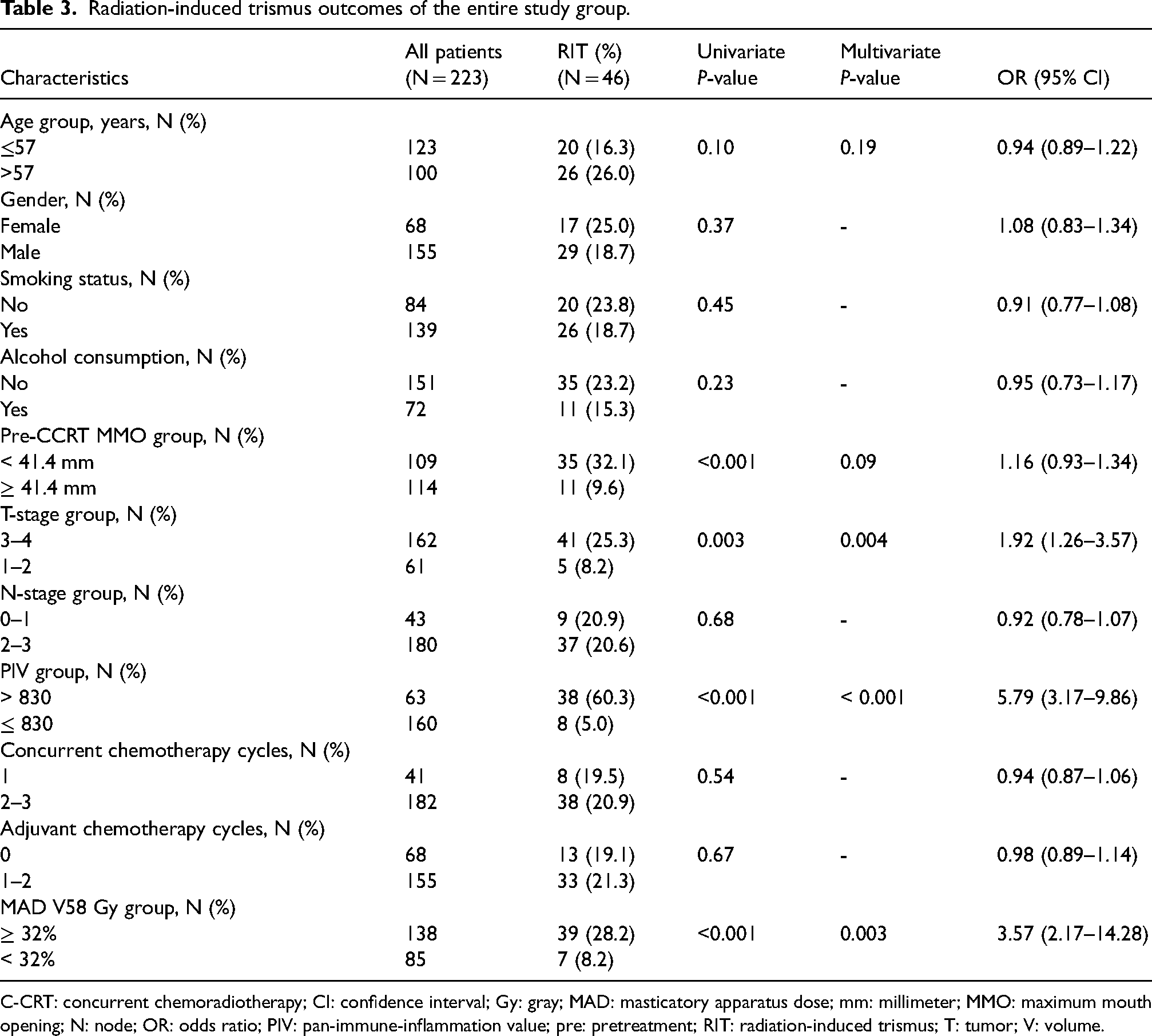
C-CRT: concurrent chemoradiotherapy; CI: confidence interval; Gy: gray; MAD: masticatory apparatus dose; mm: millimeter; MMO: maximum mouth opening; N: node; OR: odds ratio; PIV: pan-immune-inflammation value; pre: pretreatment; RIT: radiation-induced trismus; T: tumor; V: volume.
Among the baseline characteristics, patients older than the median age of 57 (66.6% vs. 40.6% for PIV ≤ 830; P = 0.031) and with a pre-C-CRT MMO ˂ 41.4 mm (61.9% vs. 43.8% for PIV ≤ 830; P = 0.031) were significantly more prevalent in the PIV ≤ 830 cohort (Table 1). In univariate analyses (Table 3), we found a significant relationship between RIT and pre-C-CRT MMO (P < 0.001), T-stage (P = 0.003), MAD V58 (P < 0.001), and pre-treatment PIV grouping (P < 0.001). Accordingly, T3–T4 tumor stage (25.3% vs. 8.2% for T1–T2; OR: 1.92; P = 0.004), MAD V58 Gy ≥ 32% (28.2% vs. 8.2 for MAD V58 Gy < 32%; OR: 3.57; P < 0.001), and PIV >830 (60.3% vs. 5.0% for PIV ≤830 groups; OR: 5.79; P < 0.001) cohorts had significantly higher rates of RIT than their more favorable counterparts. As shown in Table 3 and Figure 2, the results of multivariate analyses demonstrated that all four characteristics remained independent and significant predictors of RIT in LA-NPC patients treated with definitive C-CRT (P < 0.05 for each).
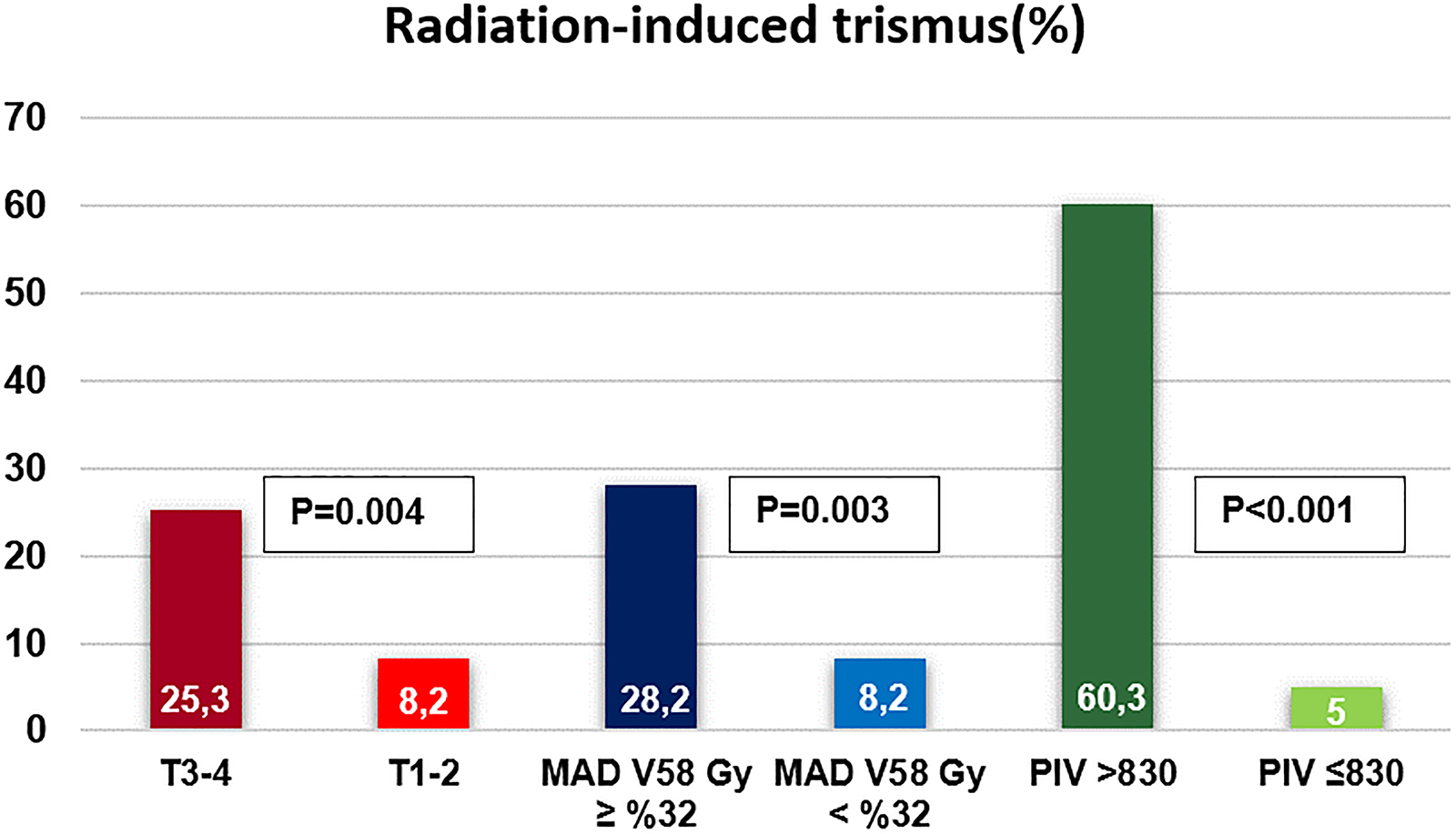
The bar graph displays the frequency of radiation-induced trismus based on significant variables in multivariate analyses: MAD V58 Gy 32% groups, T-stage groups, and pre-C-CRT PIV groups.
Discussion
The primary objective of this retrospective study was to examine the feasibility of using pre-C-CRT PIV levels as a biomarker for the prediction of RIT incidence in patients with LA-NPC. Our study's most notable finding was that the incidence of RIT was significantly higher in patients with a PIV > 830 before C-CRT compared to those with a PIV ≤ 830 (60.3% vs. 5%; OR; 5.79; P < 0.001). Other remarkable findings were T3–T4 tumor stage (25.3% vs. 8.2% for T1–T2; OR: 1.92; P = 0.004) and MAD V58 Gy ≥ 32% (28.2% vs. 8.2 for MAD V58 Gy < 32%; OR: 3.57; P < 0.001).
Researchers have pinpointed a wide variety of patient, disease, clinical, and dosimetric factors as contributors to RIT occurrence.27–30 Our current findings corroborated the potent predictive power of advanced T-stage (25.3% for T3–T4 vs. 8.2% for T1–T2; OR: 1.92; P = 0.004) and larger MAD V58 (28.2% for 32% vs. 8.2% for ≥ 32%; OR: 3.57, P = 0.003) for RIT, while pre-C-CRT MMO showed a trend towards statistical significance (32.1% for < 41.4 mm vs. 9.6% for ≥ 41.4 mm; OR: 1.16; P = 0.09). The likely correlation between advanced T-stage and increased RIT rates can be explained by the fact that larger radiation volumes are used in more advanced cancers, which may cause trismus due to the unavoidably higher RT doses exposed by the masticatory apparatus. 31 The research results on MAD are scarce, but the mean dose (a dosimetric parameter for 100% volume) to the components of the masticatory apparatus is shown to be related to RIT rates.12,13 Together, these data and our findings show that the mean dose and MAD are whole and partial volume representations of a general or partial destructive process in the masticatory apparatus, which functions like a parallel organ. Therefore, it is reasonable to recommend mean dose and MAD as appropriate metrics to incorporate in dose planning without a threshold and to keep the masticatory apparatus dosages as low as possible to reduce the likelihood of RIT.
We have previously shown that the neutrophil-to-platelet ratio (NLR) and the hemoglobin-to-platelet ratio (HPR) are also reliable indicators of immune inflammation and hypoxia plus inflammation in predicting the prevalence of RIT in LA-NPC and parotid cancers that underwent C-CRT and RT, respectively.12,13 Jointly, these equations comprise the three of four cells that formulate PIV. Because all four cells of PIV are directly or indirectly involved in the inflammatory, hypoxic, hypovascular, and fibrotic pathogenesis and progression of RIT,32,33 we hypothesized that PIV might also be a relevant biomarker worth investigation in the RIT setting, which may be more potent than the NLR due to its all-inclusive nature.
The most noteworthy and novel finding of our study was the significantly higher incidence of RIT in the pre-C-CRT PIV > 830 group than in the PIV ≤ 830 group (60.3% vs. 5.0%; OR; 5.79; P < 0.001). It is challenging to discuss the effects of PIV on RIT in a fact-based manner in the absence of comparable studies. Yet, our research findings are consistent with two recent studies exploring the association between RIT rates and the NLR and HPR.12,13 In the first study, 51 patients with parotid gland cancer receiving postoperative RT were examined for the relationship between pretreatment NLR values and RIT incidence rates. 12 The incidence rate of RIT was noted to be significantly higher in the NLR > 2.7 than in the NLR ≤ 2.7 group (35.2% vs. 5.8%; P < 0.001). The predictive power of pretreatment HPR levels on RIT rates was examined in the second study, which comprised 198 LA-NPC patients treated with C-CRT. 13 Despite the essentially identical distribution of pretreatment characteristics across the two cohorts, the incidence of RIT was significantly higher in the HPR ≤ 0.54 group than in the HPR > 0.54 group (34.1% vs. 12.9%; P < 0.001). In this context, the novel PIV appears to be an additional biomarker with a high level of discriminatory ability between the two RIT risk categories.
It is still unclear exactly how high pre-C-CRT PIV levels and other biological markers are related to higher RIT rates at the molecular levels. Yet, given that RIT is a consequence of a radiation-induced hyperinflammatory, hypoxic, and hyper-fibrotic condition, 33 it is possible to draw some insightful observations by analyzing the well-established functions of the PIV cells in this state. High platelet counts can cause vascular occlusion and hypoxia by interfering with blood flow at the tissue and vascular levels. 34 In addition, platelets may trigger the secretion of numerous factors, including TGF-β, which is involved in various fibrotic processes including the RIT. 35 Neutrophils are the first cells to arrive at the site of inflammation and play a crucial role in the innate immune response, 36 which can trigger various cascades leading to inflammation and fibrogenesis by themselves or via their byproducts. 37 In addition to the “effector” cells such as fibroblasts and myofibroblasts, monocytes have been shown to have a role in fibrosis by activating a number of different fibrotic pathways. In contrast, the PIV formula’s fourth and final component—lymphocytes—may slow or even prevent the continuing fibrotic process because of their well-recognized anti-inflammatory properties. 38 In summary, increased counts of proinflammatory platelets, monocytes, and neutrophils may induce a hyperinflammatory and hypoxic tissue state, which may cause the release of inflammatory mediators such as IL-1, IL-6, VEGF, HIF-1, and TGF-β, triggering the fibrosis process of RIT.39,40 Conversely, a decrease in lymphocyte counts indicates that the hypoxic and hyper-fibrotic processes are continuing out of control. Therefore, although the precise mechanisms may be more complex, our findings suggest that the increased rates of RIT in the high PIV cohort may be attributable to increased levels of proinflammatory and immunosuppressive platelets, monocytes, and neutrophils, and decreased levels of anti-inflammatory and immune-competent lymphocytes.
Several factors contribute to the strength of the current study. First, the initial staging of NPC in all patients was performed using a head and neck MRI and positron emission tomography (PET)-CT combination as the gold standard. These imaging tools were also employed to ascertain and delineate the target volumes. Second, the response and toxicity assessments for all qualified patients were identical. Third, earlier studies examining the effects of other inflammation biomarkers, like NLR and HPR, supported our findings.12,13 However, there are some limitations to the present study as well. First, since this is a retrospective study carried out by a single institution, our findings could be affected by unintended biases typical of these kinds of studies. Second, our results were based on a single snapshot of pre-C-CRT PIV data, which may not accurately represent the optimal outcomes. Nevertheless, since PIV is an intrinsically dynamic biomarker generated from blood components, it may fluctuate substantially during and after C-CRT owing to changes in host immunity, systemic inflammatory status, and tumor load. Therefore, before definitive conclusions can be reached about the causal relationship between pretreatment PIV levels and RIT rates, the current results should be seen as exploratory and validated by subsequent studies.
Conclusion
The current study examined the pre-treatment PIV values to predict RIT in LA-NPC patients receiving definitive C-CRT. Our findings demonstrated that such patients could be successfully divided into two RIT groups using a cutoff value of 830. It may be possible to stratify these patients and develop risk-based follow-up algorithms based on these results if additional research confirms them.
Footnotes
List of abbreviations
Acknowledgements
Not applicable.
Consent for publication
Each participant signed an informed consent form for the collection, analysis, and publication of their results, either individually or through legally authorized representatives.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author (Efsun Somay) on special request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Before acquiring any information from the patient, the study design was approved by the Institutional Review Board of the Baskent University School of Medicine and complies with the Declaration of Helsinki. We ensured that all patients signed an informed consent form before the beginning of the evaluation, either themselves or their legally authorized representatives for acquisition and analysis of the patients’ sociodemographic, dental, and medical records, blood samples, and publication of the outcomes.
Authorship contribution statement
All authors contributed significantly and equally; and all authors approved the final form of the manuscript.
