Abstract
BACKGROUND:
The optimal timing of combined chemotherapy with radiotherapy for locally advanced nasopharyngeal carcinoma (LA-NPC) is undetermined.
OBJECTIVE:
This study aimed to compare the therapeutic efficacy of neoadjuvant chemotherapy (NACT) followed by radiotherapy (RT) and concurrent chemoradiotherapy (CCRT).
METHODS:
Five hundred and thirty-eight patients diagnosed with LA-NPC and treated with NACT
RESULTS:
Compared with the CCRT alone regimen, the NACT + RT regimen showed a significantly better OS rate with a 62% decreased risk of death in a subgroup of patients aged
CONCLUSIONS:
Age is a significant biomarker when selecting NACT
Introduction
Nasopharyngeal carcinoma (NPC) is a common type of head and neck cancer that originates from epithelial cells of the head and neck. NPC demonstrates an unbalanced endemic distribution and is associated with the Epstein-Barr virus (EBV) [1, 2]. Globally, China has one of the highest incidence and mortality rates for NPC, especially in the southern region [3]. More than 70% of patients with NPC are initially diagnosed with locally advanced disease [4].
Radiotherapy (RT) is the main treatment for NPC as it is suited to the complex anatomy of NPC and its high sensitivity to radiation [5, 6]. With the introduction of intensity-modulated radiation therapy (IMRT) and the use of chemotherapy in patients with advanced diseases, rates of successful local control have improved, and the risk of distant metastasis has also improved greatly [7, 8, 9]. RT is the main treatment for early-stage disease, and the 5-year overall survival (OS) rates in patients with stage I–II NPC can reach 95–100%. However, the 5-year OS rate of patients with stage III–IVA NPC remains unsatisfactory [10], with approximately 30% of patients at high risk of disease recurrence and metastasis. Distant metastasis represents the main mode of failure [11, 12], which may be related to metastatic cancer with a relatively long natural history. For locally advanced NPC (LA-NPC), concurrent chemoradiotherapy (CCRT) can prolong OS. CCRT is considered the standard treatment for LA-NPC [13]. A meta-analysis by Langendijk et al. reported that the most effective way to introduce chemotherapy is to perform it simultaneously with RT, which increases the 5-year survival rate by 20% [14].
In recent decades, numerous trials have studied the value of adding chemotherapy to RT for NPC [15, 16, 17]. Compared with adjuvant chemotherapy (ACT), neoadjuvant chemotherapy (NACT) has the advantages of increasing radiosensitivity, killing micro-metastasis, and reducing tumor stage, as well as better tolerance [18]. There is increasing evidence that patients with LA-NPC can benefit from NACT [19, 20]. Furthermore, compared with CCRT, the incidence of acute side effects of RT after sequential NACT is significantly reduced, and patient compliance is improved [21, 22].
At present, NACT or CCRT are considered superior to RT alone [16], but a long-term follow-up study has shown that there is no significant difference in the survival rate between the two regimens [23]. Therefore, the optimal time to combine chemotherapy with RT remains undetermined. Therefore, this study aimed to compare the relative survival benefits of CCRT and NACT
Materials and methods
Patient characteristics
This was a secondary retrospective study, and data were collected from patients who received treatment at Sun Yat-Sen University Cancer Center between October 2009 and February 2012. A total of 538 patients satisfied the inclusion criteria and were included in the analysis. Tumor characteristics, histology, age, sex, tumor (T) stage, nodal (N) stage, OS, and treatment regimen (NACT followed by RT and CCRT alone) were recorded.
Inclusion criteria
Patients were enrolled in the study if they satisfied all the following criteria:
Proven nasopharyngeal carcinoma. No evidence of distant metastasis. Treatment modality: Treatment with NACT Complete follow-up data.
As this was a retrospective analysis, the requirement for informed consent was waived.
Treatment regimens: Patients were treated with neoadjuvant chemotherapy followed by radical IMRT or concurrent chemoradiotherapy alone. The specific regimes of each treatment were as follows: (1) NACT: including paclitaxel in combination with cisplatin (TP) or cisplatin in combination with 5-fluorouracil (PF); (2) CCRT: chemotherapy of cisplatin combined with intensity-modulated radiotherapy – two cycles of each treatment regime should be accomplished (Supplement 1).
The 7
The endpoint was OS, which was defined as the time from diagnosis to death for NPC or the last outpatient visit.
Follow-up and measurement
Patients were required to return for a general physical condition assessment every 3 months in the first 2 years, every 6 months in years 2–5, and annually thereafter. If patients failed to return to the clinic, we obtained their review data by contacting the patient or their families. The follow-up period was defined as the time of first diagnosis or first treatment to death or the last outpatient visit.
Routine tests, including serum tumor markers, electronic nasopharyngoscopy, chest computed tomography, transabdominal color Doppler scan, brain magnetic resonance imaging, bone scan, and positron emission tomography-computed tomography, were performed to evaluate the systemic tumor load.
Endpoint and statistical analysis
The primary objective of the analysis was to compare the OS of patients with LA-NPC after treatment with NACT followed by RT or CCRT.
Baseline characteristics were compared using the Fisher’s test for categorical variables. Restricted cubic spline regression (RCS) was used to determine the relationship between age and the hazard ratio of death. To explore the association between different treatment regimens with clinical outcome benefits in patients with LA-NPC, the survival rate was estimated using the Kaplan-Meier method; univariate comparisons were performed using the log-rank test. Cox proportional hazards models were used to adjust for potential confounders. The subgroup analysis was performed on five covariates to evaluate the prognosis for NACT followed by RT vs. CCRT, including age, sex, histology, clinical T stage, and clinical N stage; hazard ratios (HRs) and 95% confidence interval (CI) were used to present the results.
All statistical tests were two-sided.
Results
Clinical characteristics of NPC patients
The data for this analysis was published in PLOS One in 2016 and included 538 patients who were newly diagnosed with LA-NPC at SYSUCC from October 2009 to February 2012. Among them, 236 (43.9%) received CCRT and 302 (56.1%) received NACT
Basic characteristics of the patients in the NACT
RT vs. CCRT groups
Basic characteristics of the patients in the NACT
Data are presented as numbers (percentage). NACT, neoadjuvant chemotherapy; CCRT, concurrent chemoradiotherapy; RT, radiotherapy; WHO, World Health Organization.
The median follow-up time of this study was 48.0 months (5.0–73.0 months). A total 22.49% (121/538) of the patients died until the final follow-up.
Kaplan-Meier curves of overall survival in patients with NPC treated with NACT 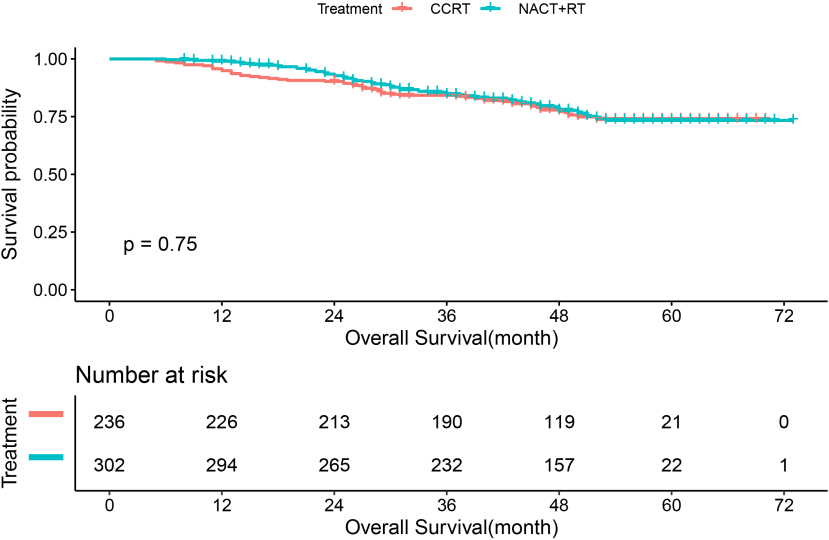
Kaplan-Meier curves of overall survival in patients with NPC stratified by age. NACT, neoadjuvant chemotherapy; CCRT, concurrent chemoradiotherapy; RT, radiotherapy; NPC, nasopharyngeal carcinoma.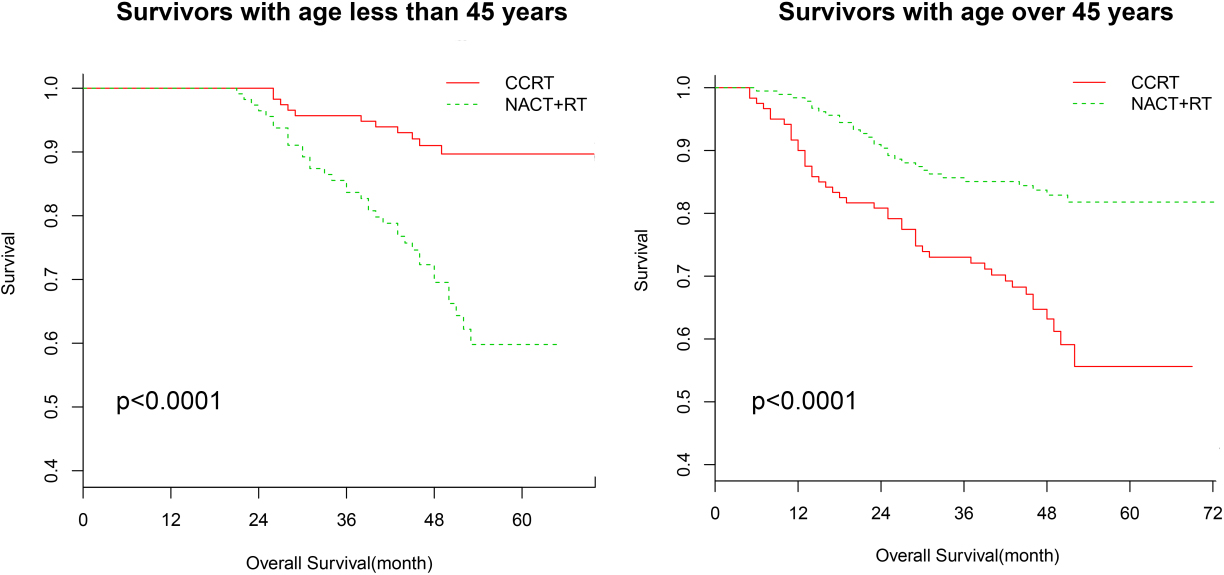
Survival analysis showed that there was no difference between the 5-year survival rate between patients who received NACT
NACT
RT treatment (vs. CCRT) and multivariate HR of overall survival with 95% CIs in NPC
NACT
Data are presented as numbers or hazard ratios (95% confidence interval).
Stratified analysis for unadjusted HR in the subgroups of age, sex, histology, T stage, and N stage HR (vs. continued) 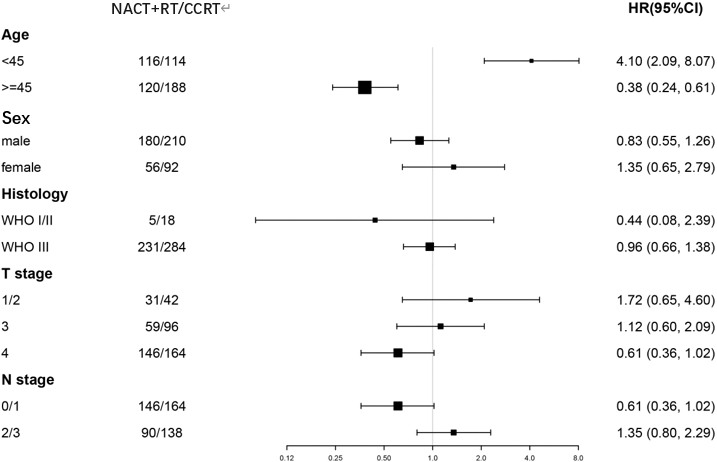
Compared with the CCRT alone regimen, the NACT
The number of death patients in NACT
RT and CCRT
The number of death patients in NACT
The relationship between age and the HR of overall mortality.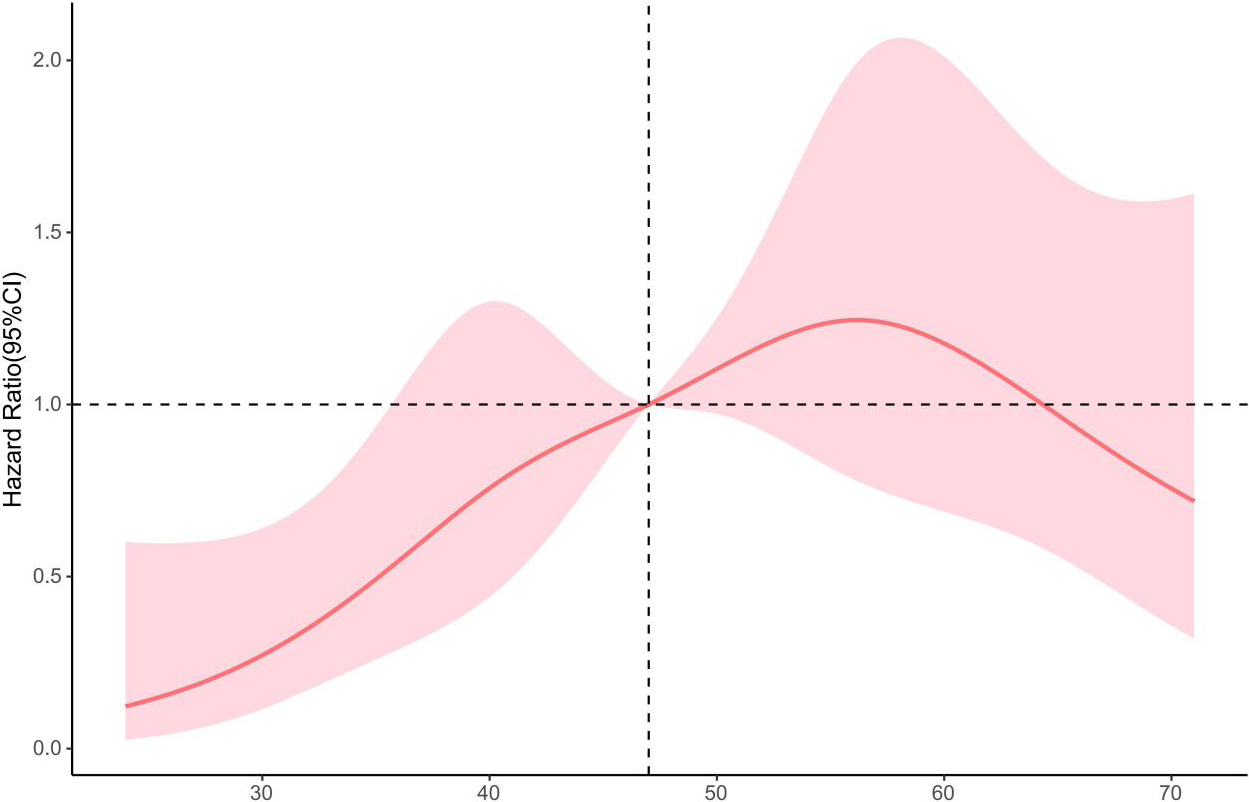
Finally, multivariate analysis was performed to explore the association between different treatment regimens and overall survival (NACT
RCS indicated that the HR of overall mortality increased with age, and was highest at the age of 57, then decreased gradually (Fig. 4). To further explore the relationship between age and the survival of patients with LA-NPC, the study patients were divided into three groups according to the median age and the highest HR of overall mortality ( age
Kaplan-Meier curves of overall survival in patients with NPC stratified by age a: the survival of patients with 45–57 years; b:the survival of patients with 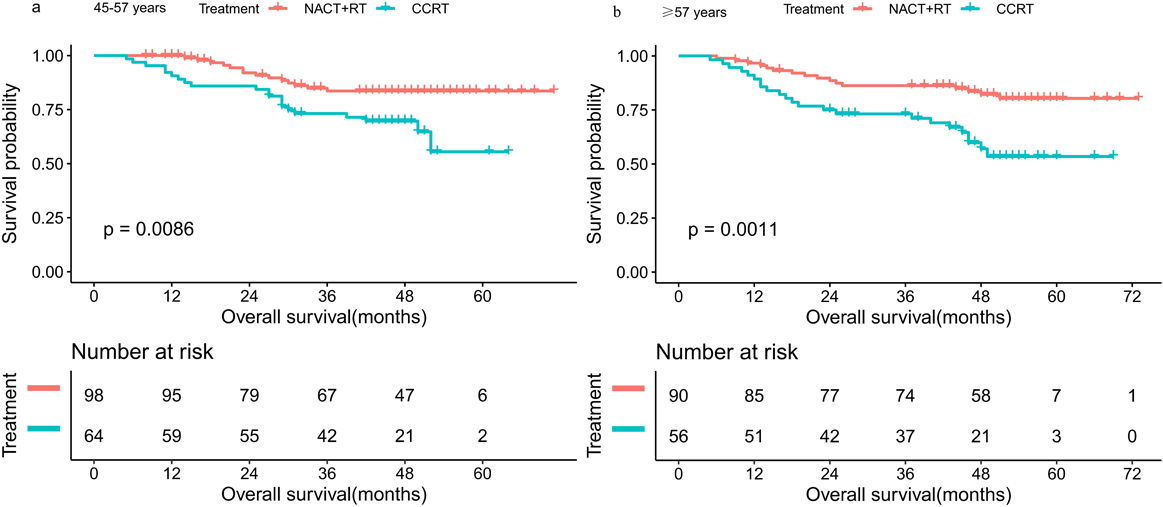
In our study, we reported the results of 538 newly diagnosed patients with LA-NPC who received CCRT or NACT
A prospective observational study by Chang et al. [26] showed that CCRT could not provide 5-year estimated OS, disease-free survival (DFS), recurrence-free survival (RFS), or metastasis-free survival (MFS) benefits for patients with locally advanced NPC based on a large number of patients treated with NACT
Moreover, through the subgroup analysis (NACT
For patients in the T1/2 stage and N 2/3 stage, the efficacy of CCRT was better than that of NACT
Our study had some limitations. First, this was a retrospective study with a relatively small sample size from a single institution. The use of data from randomized trials performed at different centers could produce more persuasive results. Second, previous studies have shown that the difference in NACT regimens has an impact on the survival rate of patients, and regimens of docetaxel, cisplatin, and fluorouracil can bring greater benefits in local control and distant metastasis than TP [33, 34]. Third, in terms of EBV detection, a previous study found that maintaining a level of EBV deoxyribonucleic acid (DNA) after NACT was a predictor of poor prognosis for patients with LA-NPC [35, 36]. However, our retrospective study did not consider the status of EBV expression, which may have an impact on the evaluation of disease progression and therapeutic effects. Therefore, further studies on EBV expression and EBV DNA replication in tumor tissues are needed. Finally, there was no further analysis of adverse reactions in the two groups. According to previous research [26, 37], compared with CCRT groups, non-CCRT groups have a lower proportion of patients with nausea, vomiting, and oral mucositis, and the incidence of grade 3/4 acute toxicity with CCRT was much higher and more serious. Therefore, the incidence of toxicity may also be a factor influencing the choice of the treatment plan for patients. NACT
Conclusions
Age has predictive value for the prognosis of patients with LA-NPC; so the age should be considered when selecting NACT
Funding
This work was supported by the Natural Science Foundation of China (Nos 81302067 and 81502360); The Natural Science Foundation of Fujian Province (Nos 2016J01576 and 2020J011147); The Science and Technology Innovation Joint Foundation of Fujian Province (No 2017Y9125).
Authors’ contributions
All authors made a significant contribution to data collection, study design, statistical analysis, the interpretation of results, writing of the manuscript.
Conception: The idea were conceived by the Yihong lin and Xiongbin Yu.
Interpretation Or Analysis Of Date: The interpretation or analysis of data was analyzed by Linbin Lu, Yihong Lin, Junxian Wu.
Preparation Of The Manuscript: The manuscript was written by Yihong Lin.
Revision For Important Intellectual Content: The revision of important intelligence content was completed by Hong Chen, Xuewen Wang, Yayin Chen, Qin Lin.
Supervision: The manuscript was completed under the supervision of Xiong Chen, Xi Chen.
Than, all the uthors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Ethics approval
Ethical approval was obtained from the Department of Ethics Committee of SYSUCC.
Availability of data and material
The data for this analysis was published in PLOS One with a title that “Neoadjuvant and Concurrent Chemotherapy Have Varied Impacts on the Prognosis of Patients with the Ascending and Descending Types of Nasopharyngeal Carcinoma Treated with Intensity-Modulated Radiotherapy” in 2016.
Publication ethics
This study was a retrospective analysis, not a particular case study, the ethical approval was obtained from the Department of Ethics Committee of SYSUCC. All authors read and approved the final manuscript and consent for publication.
Consent to participate
This study was a retrospective analysis, so the requirement for informed consent was waived.
Consent for publication
All authors read and approved the final manuscript and consent for publication.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210357.
sj-docx-1-cbm-10.3233_CBM-210357.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-210357.docx
Footnotes
Acknowledgments
We are grateful for the raw data collected by Professor Jijin Yao at Sun Yat-Sen University Cancer Center. We are also thankful for the statistical analyses of the study supported by the Empower U team of the Department of Epidemiology and Biostatistics, X & Y Solutions Inc., Boston. Last, we would like to thank Editage (www.editage.cn) for English language editing.
Conflict of interest
All authors declare no conflict of interest.
