Abstract
The programmed cell death-ligand 1 (PD-L1) protein expression on tumor cells predicts the efficacy of immunotherapy in patients with non-small cell lung cancer. However, the assessment of PD-L1 expression on tumor cells has limited power for selecting patients for immunotherapy due to intra-tumoral heterogeneity and inter-tumoral heterogeneity of PD-L1 expression, the inter-observer variability in scoring PD-L1 staining, and reproducibility. These difficulties and pitfalls in interpreting the PD-L1 assessment are discussed in detail in this review.
Introduction
The programmed cell death protein 1 (PD-1), a member of the CD28 family, is an immunosuppressive molecule that is expressed by tumor-specific activated T-cells, macrophages, monocytes, B-lymphocytes, dendritic cells, myeloid cells, and natural killer cells. 1 Its ligand is the PD-L1 (programmed cell death-ligand 1) that is physiologically expressed by various cell types, including macrophages, dendritic cells, and epithelial cells in various tissues as well as by tumor cells and tumor-infiltration immune cells, which enable tumor cells by binding to PD-1 to escape immune surveillance.
In the last decade, remarkable progress has been made in the treatment of non-small cell lung cancer (NSCLC) with the introduction of immune checkpoint inhibitors (ICI). ICIs target the PD-1, PD-L1, or the cytotoxic T lymphocyte antigen 4 (CTLA-4) and thus prevent the immune escape by tumor cells. Notably, patients with a PD-L1 expression of ≥50% by the tumor cells experience the most relevant benefit with ICIs. Therefore, the PD-L1 expression of tumor cells seems to be a promising biomarker for treatment response. Thus, the tumor proportion score (TPS) that represents the expression of PD-L1 on tumor cells and is defined as the percentage of immunoreactive tumor cells measured against the total number of tumor cells should be evaluated by immunohistochemistry (IHC) prior to ICI treatment in lung cancer patients.
Nevertheless, the assessment of PD-L1 protein expression on tumor cells has limited power for selecting patients due to intra-tumoral heterogeneity, inter-tumoral heterogeneity, inter-observer variability in scoring PD-L1 staining, and reproducibility (Figure 1).
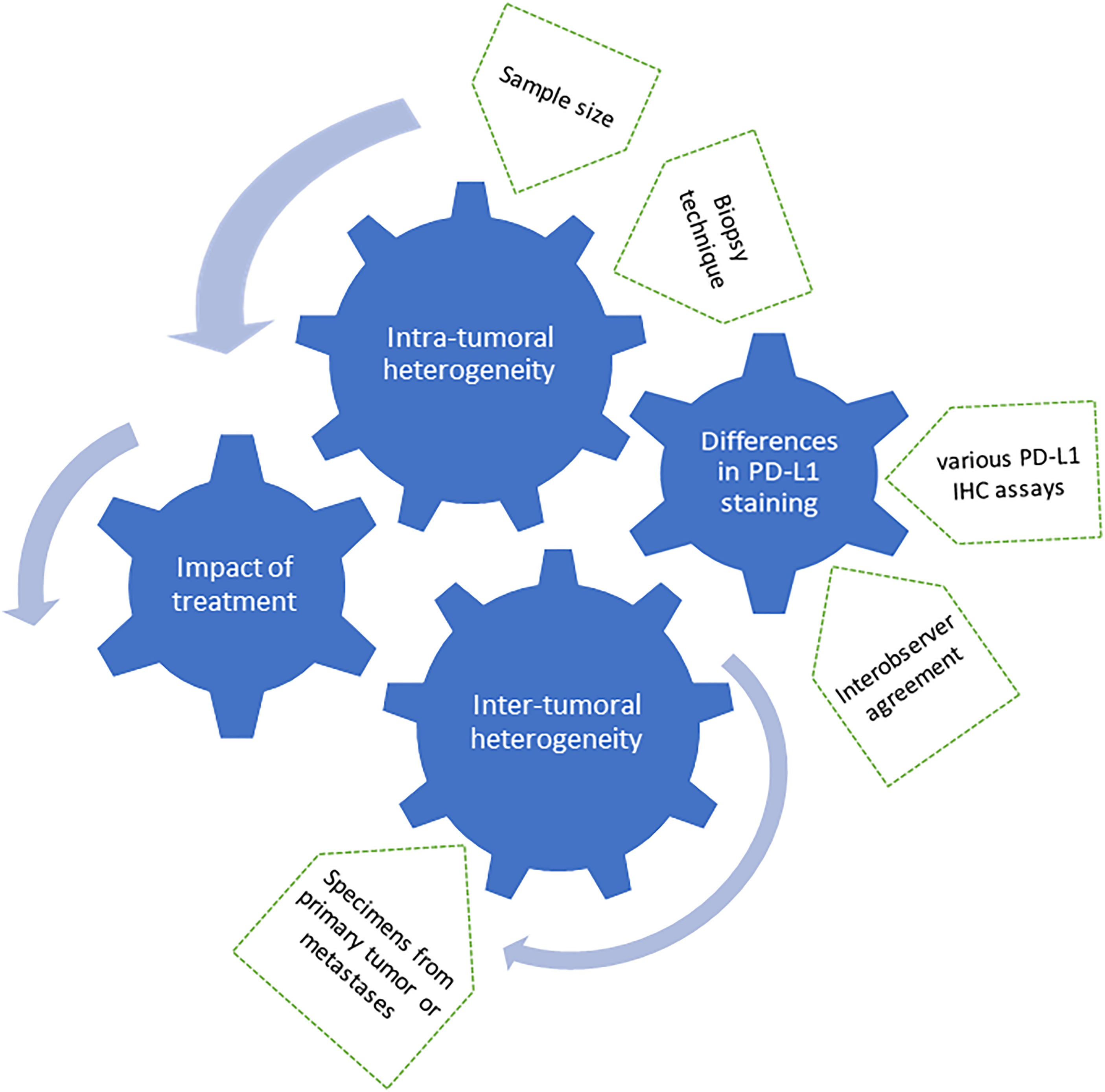
Factors influencing the PD-L1 assessment.
These difficulties and pitfalls in interpreting the PD-L1 assessment are discussed in detail in this review.
Intra-tumoral heterogeneity of PD-L1 expression
A large proportion of patients with lung cancer present with inoperable status at the time of diagnosis. Therefore, PD-L1 testing is performed mainly on biopsy specimens and not on surgical specimens. However, it is questionable whether the small amount of tissue analyzed in biopsies may represent the PD-L1 expression as the majority of tumors demonstrate a considerable intra-tumoral heterogeneity of PD-L1 expression (Figure 2).
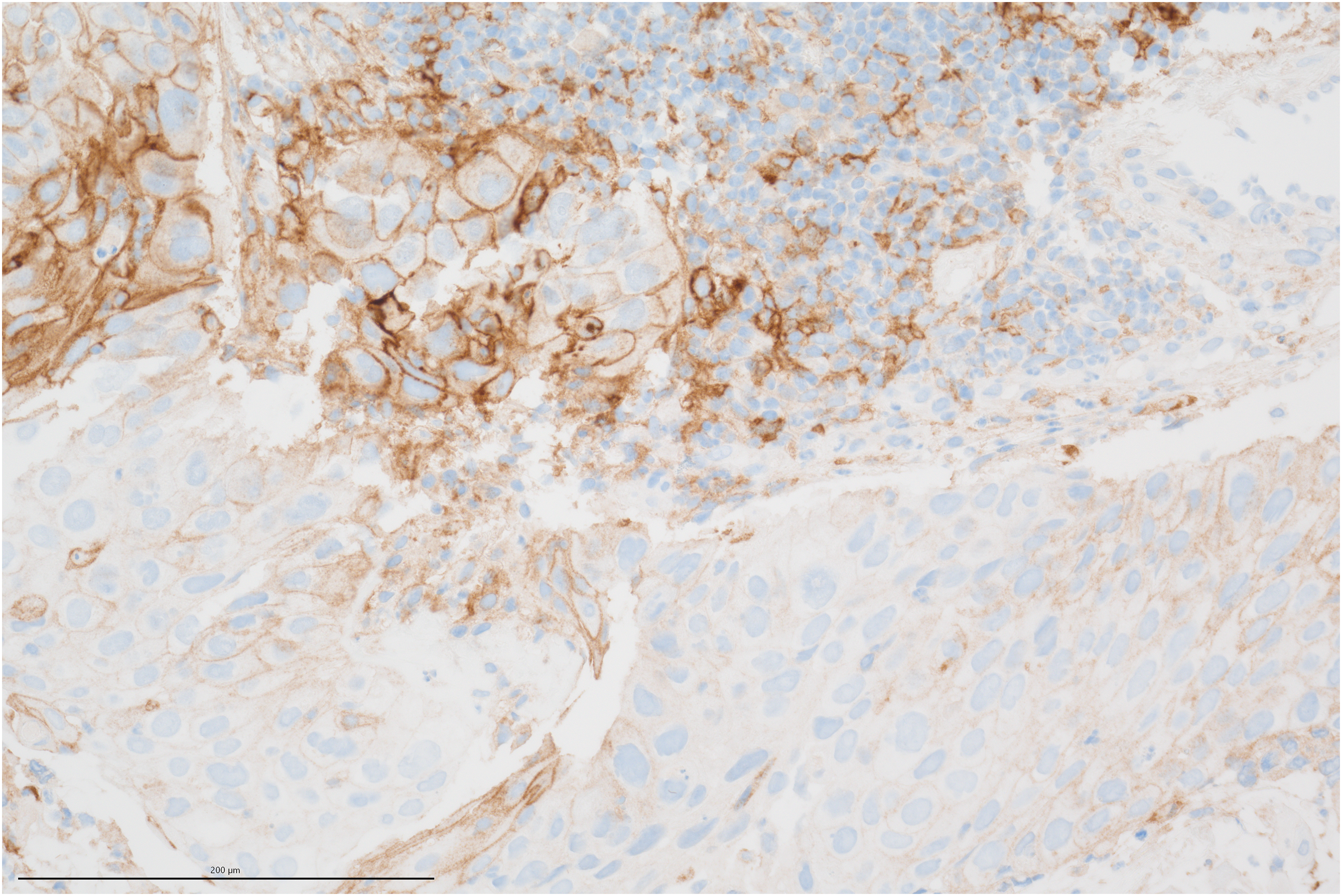
Intra-tumoral heterogeneity of PD-L1 expression (PD-L1 Klon BSR90 Firma BioSite).
Haragan et al. 2 described this intra-tumoral heterogeneity in different sites of the tumor. Comparing 1 cm² areas of a tumor, a PD-L1 tumor proportion score (TPS) change of ≥1% was found in 56% of the tumors, whereby three specimens presented a PD-L1 TPS change of ≥10%. This intra-tumoral heterogeneity is especially evident in various studies comparing PD-L1 expression of biopsies with surgical resections that resulted in controversial findings. Kitazono et al. 3 compared PD-L1 expression between biopsies from the primary tumor and matched surgical specimens in 79 lung cancer patients. 3 The authors used a hybrid score to describe PD-L1 expression on tumor cells and differed between PD-L1 positivity and PD-L1 negativity. They found a satisfying concordance rate of PD-L1 status of 92.4% between the biopsies and resected tumors. Similar results were reported by Heymann et al. 4 who evaluated a concordance rate of PD-L1 expression of 91% in 23 patients with NSCLC and by Gradecki et al. 5 who described a concordance rate of 92.2% between core biopsies and resection PD-L1 assessments. In contrast to these findings, Ilie et al. 6 who used a scoring system from 0 to 3 depending on PD-L1 expression (≥50%, ≥ 5% and < 5%) described a poor agreement between surgically resected and matched biopsy specimens in 160 patients with an overall discordance rate of PD-L1 expression of 48%. In another study of 113 lung cancer patients in a total, a quantitative comparison between the diagnostic biopsy of the primary tumor with corresponding resected surgical specimens revealed a statistically significant correlation of PD-L1 expression on tumor cells. However, when using a cut-off value of ≥50% PD-L1 TPS the concordance rate with 78% was only moderate. 7
These controversial results of various trials demonstrate that the intra-tumoral heterogeneity remains a clinical problem, which can potentially preclude precise treatment planning for patients with advanced NSCLC. Therefore, criteria such as the required number of biopsies are defined to minimize the impact of intra-tumor heterogeneity. 8
Inter-tumoral heterogeneity and impact of biopsy techniques
Besides the intra-tumoral heterogeneity, various studies evaluated the inter-tumoral heterogeneity of PD-L1 expression of the primary tumor and its metastases in NSCLC patients.
In a trial published by Kim et al., 9 surgical resection specimens of the primary tumor and the nodal metastases of 146 patients with pulmonary adenocarcinoma were used for PD-L1 assessment. After dichotomizing cases into PD-L1 negative and PD-L1 positive groups using cut-off values of 1% and 50%, respectively, the concordance rate was 80.1% and 90.7%, respectively, resulting in a moderate agreement (k = 0.492 and 0.598). In contrast to this study, Saito et al. 10 reported a serious discrepancy in PD-L1 staining between resected primary tumors and their metastatic lymph nodes in 35 patients with NSCLC. In approximately 70% of the patients, no correlation was found between PD-L1 expression in the primary tumor and metastatic lymph nodes. Haragan et al. 2 also found a significant inter-tumoral heterogeneity between the primary tumor and nodal metastases in 53% of 107 patients with resected NSCLC.
In these above-mentioned studies, surgical specimens of the primary tumor and lymph node metastases were used for the comparison of the PD-L1 status. Other studies compared the PD-L1 expression of primary tumors and their metastases obtained by different biopsy techniques, which may in addition also have an impact on inter-tumoral heterogeneity. This is of great importance, as in some inoperable patients, histological diagnosis is confirmed from biopsy of metastatic sites.
In a small patient cohort, Sakakibara et al. 11 compared the PD-L1 expression obtained by endobronchial-ultrasound transbronchial needle aspiration (EBUS-TBNA) from lymph node metastases, transbronchial biopsies from the primary tumor, and resection specimens from the primary and secondary tumors. Overall, they found a moderate correlation. Interestingly, the authors described a higher diagnostic yield and better quality of samples derived from EBUS-TBNA resulting in a good concordance of PD-L1 positivity between EBUS-TBNA and the corresponding primary tumors.
Besides PD-L1 staining, some trials investigated the PD-L1 copy number, which is one molecular determinant of PD-L1 tumor levels. The PD-L1 gene is located on chromosome 9p24.1, and amplification of this region was frequently observed in patients with NSCLC. 12 Yoshimura et al. 13 evaluated the PD-L1 protein expression with ≥1% as the cut-off and the PD-L1 copy number (PD-L1 CNA) from EBUS-TBNA specimens and other specimens (transbronchial biopsy from the primary tumor and resected specimens from primary and metastatic sites). Overall, they found a moderate concordance of PD-L1 protein expression of 71 EBUS-TBNA specimens and other specimens. The concordance of PD-L1 CNA status was comparable with that of PD-L1 expression. However, when the PD-L1 copy number was assessed as a continuous variable, the correlation of PD-L1 CNAs was superior to that of PD-L1 expression. Also, the intra-tumoral heterogeneity of the PD-L1 copy number was less heterogeneous than the protein expression in whole sections of resected tumors. Similar results were found by Aujla et al. 14 and Leong et al. 15 who described low heterogeneity in copy number values across different anatomical sites. Summarizing, PD-L1 copy number values seem to be highly stable across different tumor regions; thus, their evaluation may represent a potential for a robust and complimentary biomarker for predicting response to immunotherapy.
In other respects, different studies described the concordance of PD-L1 expression between the primary tumor and brain metastases. Mansfield et al., 16 who investigated the PD-L1 expression of 73 paired primary lung cancers and brain metastases, found agreement in 86%. Regarding the temporal heterogeneity, there were fewer discordant pairs when obtained less than 6 months apart than 6 or more months apart. In another study, Zhou et al. 17 found higher PD-L1 protein detection in brain metastases compared to the primary tumor in 25 patients with NSCLC. The inter-tissue concordance was only 60%. Surprisingly, this discordance was more prominent in cases with simultaneous diagnosis of lung cancer and brain metastases. A higher concordance rate in PD-L1 expression between the paired primary lung tumor and brain metastases with 79% was described by Batur et al. 18
Impact of different IHC assays
For non-small cell lung cancer, different PD-L1 IHC assays were used within clinical trials that evaluated the efficacy of different checkpoint inhibitors; thus, the various PD-L1 IHC assays are linked to a different cancer treatment. However, real-world biopsies are sent to pathology with no information as to which immune checkpoint inhibitor the patient is planned to be treated with. Furthermore, the biopsy specimens are often not sufficient for multiple PD-L1 IHC assays. Therefore, the question of the interchangeability of different PD-L1 IHC assays was raised, and several studies were conducted to evaluate the concordance between various assays.19–23 In 2017 and 2018, the results of the Blueprint (BP) Programmed Death-Ligand 1 (PD-L1) Immunohistochemistry Comparability Project, which assesses the feasibility of harmonizing the clinical use of five independently developed commercial PD-L1 IHC assays (22C3, 28-8, SP142, SP263, and 73-10), found a compatibility of three antibodies (22C3, 28-8, and SP263) for determining the TPS of primary lung cancer cells.19,20 These results were confirmed by different trials, some of which also showed 22C3 to be the most sensitive PD-L1 IHC assay for tumor cell expression.21–26 Besides comparing the commercial PD-L1 IHC assays, D-L1, Tsimafeyeu et al. 27 detected the PD-L1 protein expression by polymerase chain reaction (PCR) RNA and compared the result with the PD-L1 IHC assays. The PCR RNA expression analysis was not equivalent to IHC, but had some potential for the identification of PD-L1-negative tumors. Overall, these findings demonstrate that the interchangeability of the commercial different PL-L1 IHC assays—22C3, 28-8, and SP142—may be acceptable. However, another challenge is the interobserver agreement of PD-L1 staining that was also evaluated in different trials. Overall, the concordance in the interobserver agreement of TPS was found to be substantial to near prefect.26,28,29
Impact of chemotherapy, immunotherapy, and targeted therapy on PD-L1 expression
PD-L1 expression on tumor cells may vary due to chemotherapy or immunotherapy. Clinical trials described a downregulation of the PD-L1 expression of tumor cells after a cisplatin/gemcitabine combination, paclitaxel-based regimen, or tyrosine kinase inhibitor (TKI)-based therapy.30,31 Fujimoto et al. 32 evaluated the PD-L1 protein expression pre- and post-chemoradiation therapy in 45 patients with NSCLC. They found a significant decrease in the percentage of tumor cells with PD-L1 expression. Moreover, they investigated the association between changes in PD-L1 expression and survival time. The median overall survival of patients with decreased, unchanged, or increased PD-L1 expression was 85, 93, and 15 months, respectively, demonstrating the impact of the PD-L1 alteration on survival. A similar study that evaluated the impact of chemoradiation therapy on PD-L1 levels was published by Choe et al. 33 In more than half of the 43 patients who underwent neoadjuvant concurrent chemoradiation therapy, an up- or down-regulation of PD-L1 expression was found. Patients without PD-L1 expression following the treatment had a survival benefit.
Takahashi et al. 34 reported a remarkable alteration of PD-L1 expression after immune checkpoint therapy in two patients with NSCLC. The transbronchial biopsies in these two patients showed a high PD-L1 expression and the patients were treated by immunotherapy. However, the patients experienced disease progression and passed away. The autopsies revealed a considerable decrease of the PD-L1 expression, so the authors hypothesized that the decrease was associated with little effect of the immunotherapy and thus contrary to the results reported by Fujimoto et al.32 and Choe et al.33 It must be considered that the biopsy techniques varied; thus, the biopsy technique and/or tumor heterogeneity may have significant impact. Furthermore, the number of patients was too small for concluding a valid statement. Frank et al. 35 investigated the PD-L1 change in 46 lung cancer patients after chemotherapy or immunotherapy. PD-L1 TPS change was observed in 33% of patients, whereby they observed both: a change from PD-L1 positive to negative and vice-versa. A significantly higher chance of PD-L1 TPS change was observed in chemotherapy-treated patients.
Another study, which compared the PD-L1 expression of tumor cells of biopsies and surgical specimens of patients who underwent chemotherapy and/or immunotherapy in-between, revealed contrary results. 7 The authors found a statistically significant correlation of the PD-L1 status prior to and following neoadjuvant treatments indicating that chemotherapy and/or immunotherapy did not have a relevant impact on the PD-L1 status. Isomoto et al. 36 described the PD-L1 change after a targeted treatment in patients with epidermal growth factor receptor (EGFR)-mutated NSCLC. Prior to the targeted treatment with EGFR-TKI 14% of patients presented a PD-L1 expression level of ≥50%. Rebiopsy, which was performed due to disease progression, revealed a PD-L1 expression of ≥50% in 28% of patients indicating a significant increase. Median progression-free survival for subsequent treatment with ICI was longer for patients with a high PD-L1 expression than for those with a low expression after EGFR-TKI.
However, further trials are needed that evaluate the consistency of the PD-L1 expression around chemotherapy, immunotherapy, and/or targeted treatment for patients with lung cancer.
Conclusion
PD-L1 expression on tumor cells predicts the efficacy of immunotherapy in patients with NSCLC. However, the interpretation of the PD-L1 expression is complicated by intra-tumoral and inter-tumoral heterogeneity, the impact of the biopsy techniques, and the use of different PD-L1 IHC assays. To minimize these limitations associated with PD-L1 assessment, larger specimens and multiple biopsies from multiple points within the tumor and its metastases are recommended. Nevertheless, so far, PD-L1 is a useful biomarker for predicting the efficacy of immunotherapy, but efforts are being made to find a complementary biomarker in conjunction with the PD-L1 status to improve predictive capability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
