Abstract
Background:
The value of pretreatment baseline 18F-fluorodeoxyglucose positron emission tomography (18F-FDG PET)/computed tomography (CT) as a prognostic factor for survival of patients with non-small-cell lung cancer (NSCLC) receiving immunotherapy remained uncertain.
Objectives:
To investigate the prognostic ability of baseline 18F-FDG PET/CT in patients with NSCLC receiving immunotherapy.
Design:
A systematic review and meta-analysis.
Data sources and methods:
We searched the PubMed, EMBASE, and Cochrane Central Register of Controlled Trials databases until May 7, 2024, and extracted data related to patient characteristics, semiquantitative parameters of 18F-FDG PET/CT, and survival. We pooled hazard ratios (HRs) to evaluate the prognostic value of the maximum standardized uptake value (SUVmax), mean standardized uptake value (SUVmean), metabolic tumor volume (MTV), and total lesion glycolysis (TLG) for overall survival (OS) and progression-free survival (PFS).
Results:
A total of 22 studies (1363 patients, average age range 30–88 years) were included. Baseline 18F-FDG PET/CT-derived MTV was significantly associated with both OS (HR: 1.124, 95% confidence interval (CI) 1.058–1.195, I2 = 81.70%) and PFS (HR: 1.069, 95% CI: 1.016–1.124, I2 = 71.80%). Other baseline 18F-FDG PET/CT-derived parameters, including SUVmax (OS: HR: 0.930, 95% CI: 0.718–1.230; PFS: HR: 0.979, 95% CI: 0.759–1.262), SUVmean (OS: HR: 0.801, 95% CI: 0.549–1.170; PFS: HR: 0.688, 95% CI: 0.464–1.020), and TLG (OS: HR: 0.999, 95% CI: 0.980–1.018; PFS: HR: 0.995, 95% CI: 0.980–1.010), were not associated with survival. Sensitivity analyses by removing one study at a time did not significantly alter the association between MTV and PFS or between MTV and OS. There was no evidence of publication bias.
Conclusion:
Pretreatment baseline 18F-FDG PET/CT-derived MTV might be a prognostic biomarker in NSCLC patients receiving immunotherapy. Further studies are needed to support routine use.
Plain language summary
Aims and Purpose of the Research We wanted to know if a type of scan called 18F-FDG PET/CT can help predict how long people with a type of lung cancer (NSCLC) will live after treatment with immunotherapy. Background of the Research This research matters because NSCLC is a common and serious type of lung cancer. Knowing how long patients might live after treatment can help doctors plan better care. Many people are affected by this disease, so finding good ways to predict survival can help a lot of patients. Methods and Research Design They reviewed and analyzed data from 22 different studies involving 1363 patients, with ages ranging from 30 to 88 years.We focused on certain measurements from the scans, like SUVmax, SUVmean, MTV, and TLG. We checked if these measurements were linked to how long patients lived and how long they lived without their cancer getting worse. Results and Importance We found that one of these measurements, the Metabolic Tumor Volume (MTV), was linked to how long the patients lived and how long they stayed free of disease after treatment. Specifically, higher MTV was associated with poorer overall survival and progression-free survival. The other measurements (SUVmax, SUVmean, and TLG) did not show a significant connection to patient survival. In conclusion, the MTV from PET/CT scans might help doctors predict the outcomes for lung cancer patients undergoing immunotherapy. However, more studies are needed to confirm these findings and to consider using this measurement regularly in clinical practice.
Keywords
Introduction
Lung cancer remains the most common cancer and the leading cause of cancer death worldwide, with an estimated 26,100 new cases and 20,500 deaths in the United States in 2021.1,2 Non-small-cell lung cancer (NSCLC) accounts for most lung cancer cases and often remains undiagnosed at advanced stages, for which treatment options are limited.3,4 For years, traditional treatments, including surgery, chemotherapy, radiation therapy, and targeted treatment, have been the standard treatment methods for NSCLC, but their effectiveness has remained suboptimal. 5
Immunotherapy has emerged as a standard treatment after chemotherapy in NSCLC patients in recent years. One promising class of immunotherapeutic agents is immune checkpoint inhibitors (ICIs), which harness the intrinsic immune response against tumor antigens by removing the brake on T-cell activation through antigen-presenting cells. 6 Randomized controlled trials have suggested that treatments with ICIs are associated with significantly longer overall survival (OS) and progression-free survival (PFS).7,8 This evidence led to the approval of three ICI drugs for patients with advanced NSCLC that target either PD-1 (pembrolizumab and nivolumab) or its ligand PD-L1 (atezolizumab). However, the overall response rates for ICIs have been reported to be as low as 14%–20% in NSCLC patients9,10; thus, early identification of patients who are likely to benefit from ICI therapies is crucial to ensure high-quality practice. Several prognostic factors are associated with poor outcomes with immunotherapy, such as the LIPI index, 11 performance status, 12 and co-mutations like KRAS/STK11. 13 Recent efforts have been made to find prognostic factors related to imaging data since they are convenient and noninvasive. However, an effective imaging-based prognostic approach has not been established.
An increasing number of studies have suggested the potential of imaging biomarkers, derived from computed tomography (CT) or 18F-fluorodeoxyglucose positron emission tomography (18F-FDG PET), to serve as objective and reproducible approaches to predict the optimal duration of immunotherapy 14 and long-term benefit in various cancers.14–16 The maximum standardized uptake value (SUVmax) is a commonly used FDG PET parameter in clinical practice and has been found to be associated with prognosis in various cancers,17–19 but its prognostic value in NSCLC is controversial.20,21 The prognostic value of metabolic tumor volume (MTV) and total lesion glycolysis (TLG) is frequently reported in many cancers,22–25 as well as in NSCLC patients undergoing surgery, chemotherapy, or radiotherapy. 26 However, some published studies hold the opposite opinion. 27 Moreover, the prognostic value of baseline 18F-FDG PET/CT in the subgroup of NSCLC patients receiving immunotherapy is still unclear.
Thus, we conducted this systematic review and meta-analysis to comprehensively evaluate the association between pretreatment baseline 18F-FDG PET/CT and survival in NSCLC patients who received immunotherapy.
Methods
Our meta-analysis was reported according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 28 A PRISMA checklist is provided in Supplemental Table 1.
Eligibility criteria
We included studies that (1) evaluated the association between baseline 18F-FDG PET/CT-derived metabolic parameters (SUVmax, SUVmean, MTV, and TLG) and OS or PFS, (2) enrolled NSCLC patients who received immunotherapy, and (3) were published in English. The exclusion criteria were as follows: (a) patients diagnosed with other malignant tumors; (b) studies in phantom or animal models; (c) case reports or small case series (⩽10 patients), reviews, poster presentations, and letters; (d) studies that did not report sufficient data to extract hazard ratios (HRs) for the association; and (e) studies that used duplicate or overlapping populations. Overlapping patient populations were defined as those from the same hospital (or, if not specified, the hospital of the corresponding author), the first author, and the study period. In cases where duplicate or overlapping patient populations were identified, data from the most informative or most recent publication were selected for inclusion in our meta-analysis. Moreover, additional research studies of possible interest were identified from the reference lists of the included articles and reviewed for eligibility.
Search strategy and study selection
A comprehensive search of the literature was conducted in PubMed, EMBASE, and the Cochrane Central Register of Controlled Trials up to May 7, 2024. We used MESH (and EMTree) terms, as well as free texts, related to the concepts of ICI therapy, NSCLC, and 18F-FDG PET, to compose the search strategy (Supplemental Table S2).
Two reviewers (M.H. and Y.Z.) independently screened titles, abstracts, and full texts for eligibility. Any disagreement was resolved through discussion.
Quality assessment and data extraction
Two reviewers (M.H. and Y.Z.) independently assessed the quality of the included studies and extracted the data. Any disagreement was resolved through discussion. The quality of each article included in the study was evaluated via the Newcastle–Ottawa Scale (NOS), 29 a systematic review tool specifically developed for assessing the quality of nonrandomized controlled trials. Articles scoring 6 points or higher on the NOS were deemed high-quality and subsequently included in the meta-analysis. The following information was extracted from each study: author, country, sample size, study design, age, sex, cancer type, stage, ICI agent, baseline 18F-FDG PET/CT-derived parameters, HRs for OS or PFS, response assessment criteria, and other endpoints reported.
Statistical analyses
We used a random-effects model to pool HRs for the evaluation of the prognostic impact of baseline 18F-FDG PET parameters on both OS and PFS because heterogeneity from sampling schemes is notable. A pooled HR greater than 1 suggested a poorer prognosis for NSCLC patients who presented higher 18F-FDG PET-derived parameters. The heterogeneity among studies was evaluated by applying Cochran’s Q-test and Higgins I-squared statistics. A p-value < 0.1 or I2 > 50% indicates significant statistical heterogeneity among studies. Sensitivity analysis was conducted by removing one study at a time. Publication bias was assessed using funnel plots, Egger’s, and Begg’s tests. p < 0.05 was used to determine statistical significance. Data from each study were analyzed via Stata version 15.0 (Stata Corp LP, College Station, TX, USA).
Results
Eligible studies and characteristics
The initial search identified 406 relevant records, and according to the titles and abstracts, 318 records were excluded. After full-text screening, 22 studies enrolling 1363 patients were included in this systematic review, and all studies were pooled in the meta-analysis. The PRISMA flowchart of literature selection is illustrated in Figure 1. Twenty-two studies reported OS, and 19 reported PFS. We included both parameters derived from primary lesions20,30–35 and metastatic lesions27,36–39 in the analysis. Most studies were retrospectively designed (17, 77%), focused on the East Asian population (18, 82%), enrolled patients solely in advanced stages (18, 82%), and collected metabolic parameters before treatment (22, 100%). Detailed information on the basic characteristics of the patients is shown in Table 1.

Flow chart of selection studies and specific reasons for exclusion.
The basic characteristics of the included studies.
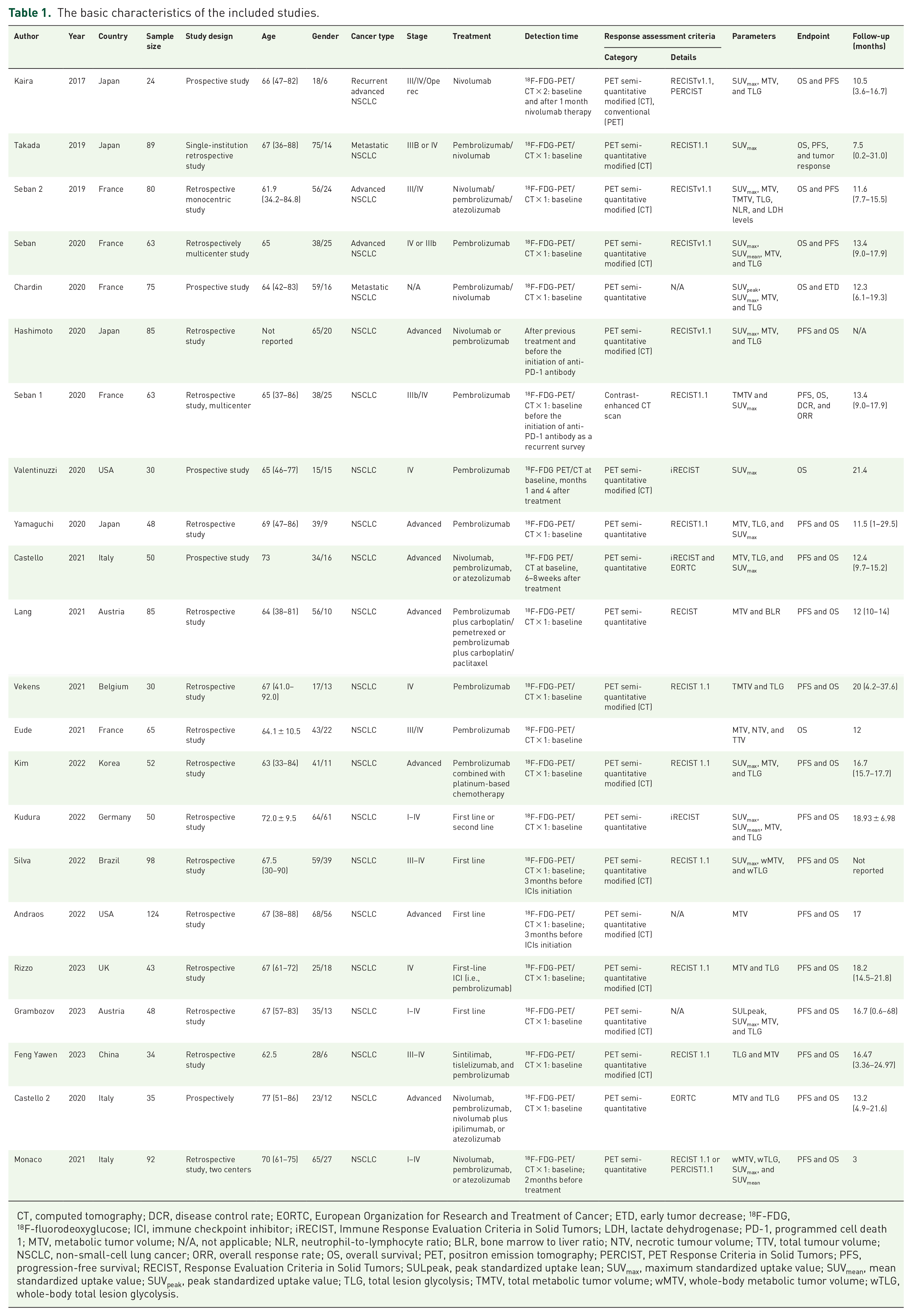
CT, computed tomography; DCR, disease control rate; EORTC, European Organization for Research and Treatment of Cancer; ETD, early tumor decrease; 18F-FDG, 18F-fluorodeoxyglucose; ICI, immune checkpoint inhibitor; iRECIST, Immune Response Evaluation Criteria in Solid Tumors; LDH, lactate dehydrogenase; PD-1, programmed cell death 1; MTV, metabolic tumor volume; N/A, not applicable; NLR, neutrophil-to-lymphocyte ratio; BLR, bone marrow to liver ratio; NTV, necrotic tumour volume; TTV, total tumour volume; NSCLC, non-small-cell lung cancer; ORR, overall response rate; OS, overall survival; PET, positron emission tomography; PERCIST, PET Response Criteria in Solid Tumors; PFS, progression-free survival; RECIST, Response Evaluation Criteria in Solid Tumors; SULpeak, peak standardized uptake lean; SUVmax, maximum standardized uptake value; SUVmean, mean standardized uptake value; SUVpeak, peak standardized uptake value; TLG, total lesion glycolysis; TMTV, total metabolic tumor volume; wMTV, whole-body metabolic tumor volume; wTLG, whole-body total lesion glycolysis.
Quality assessment of included studies
The included studies were of acceptable quality. All studies were deemed of high quality in terms of representativeness of the exposed cohort, selection of the nonexposed cohort, ascertainment of exposure, control for important factors, additional factors, and assessment of outcomes. Five (23%) studies did not have adequate follow-up of cohorts, 3 (14%) did not follow-up long enough for outcomes to occur, and 3 (14%) did not ensure that outcomes were not present at the start of the study. The details are shown in Table 2.
Non-RCT quality assessment.
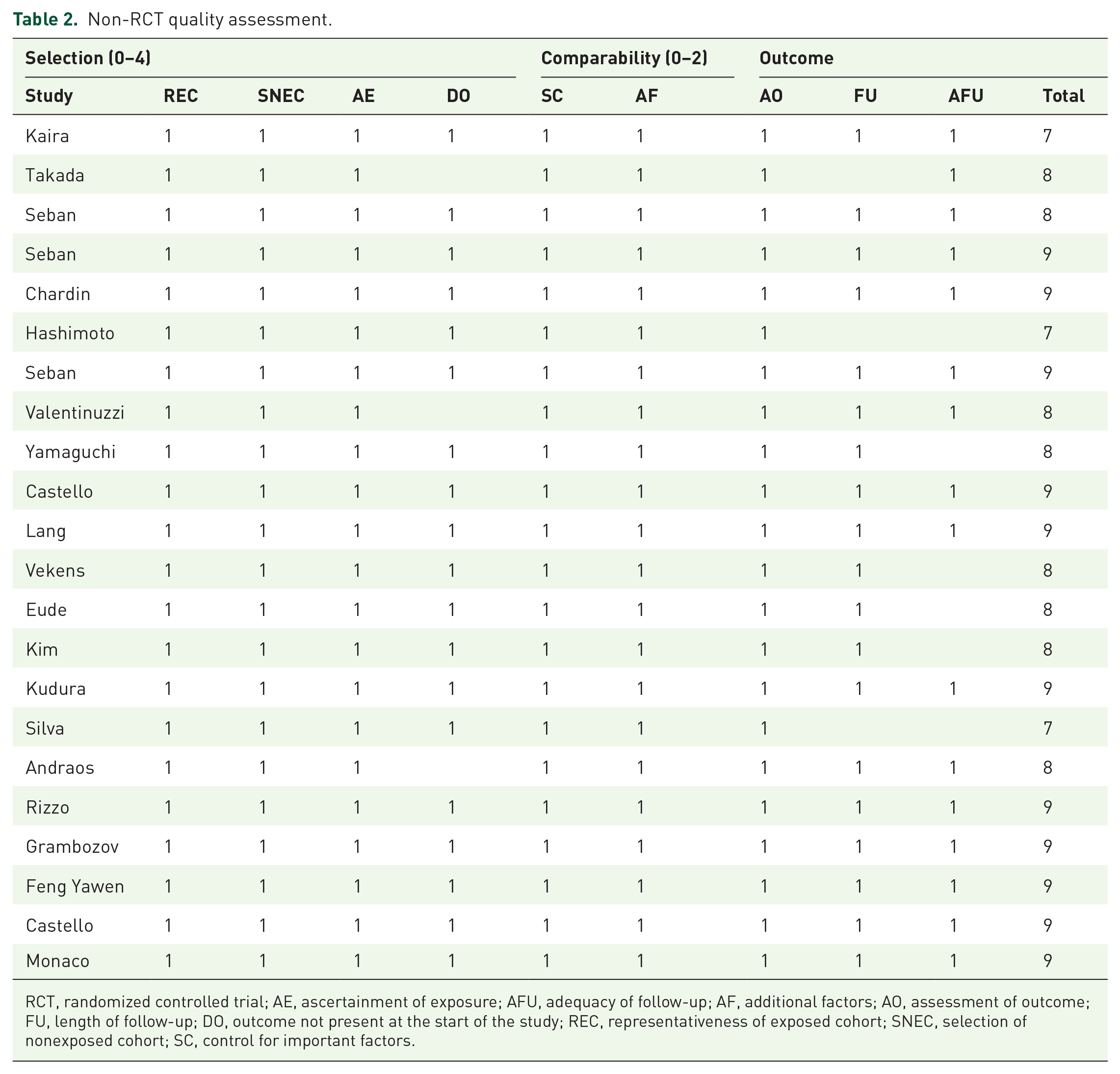
RCT, randomized controlled trial; AE, ascertainment of exposure; AFU, adequacy of follow-up; AF, additional factors; AO, assessment of outcome; FU, length of follow-up; DO, outcome not present at the start of the study; REC, representativeness of exposed cohort; SNEC, selection of nonexposed cohort; SC, control for important factors.
Summary of outcomes
Associations of baseline 18F-FDG PET/CT-derived parameters with OS in NSCLC patients receiving immunotherapy (Table 3).
The pooled HR of SUVmax, SUVmean, MTV, and TLG on PFS and OS.

CI, confidence interval; HR, hazard ratio; MTV, metabolic tumor volume; OS, overall survival; PFS, progression-free survival; SUVmax, maximum standardized uptake value; SUVmean, mean standardized uptake value; TLG, total lesion glycolysis.
SUVmax and SUVmean
Thirteen studies investigated the association between the baseline SUVmax and OS in NSCLC patients receiving immunotherapy. The pooled results revealed that the SUVmax was not significantly associated with OS (HR: 0.930; 95% confidence interval (CI): 0.718–1.230, p = 0.579; I2 = 53.70%, Figure 2(a)). When removing one study at a time, the pooled HR ranged from 0.859 to 1.006, with the lower bound of the 95% CI ranging from 0.673 to 0.797 and the upper bound of 95% CI ranging from 1.097 to 1.140 (Supplemental Figure S1(A)).

Forest plot of a meta-analysis of the prognostic role of SUVmax (a) and SUVmean (b) on overall survival.
Six studies investigated the association between the baseline SUVmean and OS and found no significant association between the SUVmean and OS (HR: 0.801; 95% CI: 0.549–1.170, p = 0.251; I2 = 1.10%, Figure 2(b)). When removing one study at a time, the pooled HR ranged from 0.706 to 0.884, with the lower bound of the 95% CI ranging from 0.456 to 0.594 and the upper bound of the 95% CI ranging from 1.094 to 1.324 (Supplemental Figure S1(B)).
MTV and TLG
In all, 19 studies reported an association between the baseline MTV and OS in NSCLC patients receiving immunotherapy. The pooled HR showed that patients with higher MTV had significantly poorer OS than those with lower MTV, despite high heterogeneity (HR: 1.124; 95% CI: 1.058–1.195, p = 0.001; I2 = 81.70%, Figure 3(a)). When removing one study at a time (Supplemental Figure S2(A)), the pooled HR ranged from 1.105 to 1.296, with the lower bound of the 95% CI ranging from 1.060 to 1.153 and the upper bound of the 95% CI ranging from 1.170 to 1.456.

Forest plot of a meta-analysis of the prognostic role of MTV (a) and TLG (b) on overall survival.
In all, 14 studies investigated the association between baseline TLG and OS and reported no association between TLG and OS (HR: 0.999; 95% CI: 0.980–1.018, p = 0.883; I2 = 84.10%, Figure 3(b)). When removing one study at a time, the pooled HR ranged from 0.997 to 1.004, with the lower bound of the 95% CI ranging from 0.967 to 0.981 and the upper bound of the 95% CI ranging from 1.042 to 1.050 (Supplemental Figure S2(B)).
Associations of baseline 18F-FDG PET/CT-derived parameters with PFS in NSCLC patients receiving immunotherapy.
SUVmax and SUVmean
In all, 11 studies examined the association between the baseline SUVmax and PFS in NSCLC patients receiving immunotherapy. The pooled HR revealed no association between the SUVmax and PFS (HR: 0.979; 95% CI: 0.759–1.262, p = 0.868; I2 = 62.1%, Figure 4(a)). When removing one study at a time, the pooled HR ranged from 0.913 to 1.040, with the lower bound of the 95% CI ranging from 0.702 to 0.809 and the upper bound of the 95% CI ranging from 1.157 to 1.336 (Supplemental Figure S3(A)).

Forest plot of a meta-analysis of the prognostic role of SUVmax (a) and SUVmean (b) on progression-free survival.
Five studies investigated the association between the SUVmean and PFS in NSCLC patients receiving immunotherapy. The pooled HR showed that the SUVmean was not significantly associated with PFS (HR: 0.688; 95% CI: 0.464–1.020, p = 0.062; I2 = 26.60%, Figure 4(b)). When removing one study at a time, the pooled HR ranged from 0.624 to 0.791, with the lower bound of the 95% CI ranging from 0.388 to 0.500 and the upper bound of the 95% CI ranging from 0.875 to 1.253 (Supplemental Figure S3(B)).
MTV and TLG
In all, 17 studies reported an association between MTV and PFS in NSCLC patients receiving immunotherapy. The pooled HR showed that patients with higher MTV were significantly associated with poorer PFS compared to those with lower MTV, despite moderate heterogeneity (HR: 1.069; 95% CI: 1.016–1.124, p = 0.010; I2 = 71.80%, Figure 5(a)). When removing one study at a time, the pooled HR ranged from 1.053 to 1.132, with the lower bound of the 95% CI ranging from 1.008 to 1.040 and the upper bound of the 95% CI ranging from 1.010 to 1.235 (Supplemental Figure S4(A)).

Forest plot of a meta-analysis of the prognostic role of MTV (a) and TLG (b) on progression-free survival.
In all, 13 studies investigated the association between TLG and PFS outcomes and reported no association between TLG and PFS (HR: 0.995; 95% CI: 0.980–1.010, p = 0.543; I2 = 87.10%, Figure 5(b)). When removing one study at a time, the pooled HR ranged from 0.992 to 0.998, with the lower bound of the 95% CI ranging from 0.970 to 0.982 and the upper bound of the 95% CI ranging from 1.009 to 1.021 (Supplemental Figure S4(B)).
Publication bias
The present study employed funnel plots, Egger’s test, and Begg’s test to assess publication bias in studies pertaining to OS and PFS, with Egger’s p-value of 0.060 and Begg’s p-value of 0.951 for OS, and Egger’s p-value of 0.195 and Begg’s p-value of 0.640 for PFS. The analysis indicated no significant publication bias for either OS (Figure 6(a)) or PFS (Figure 6(b)).
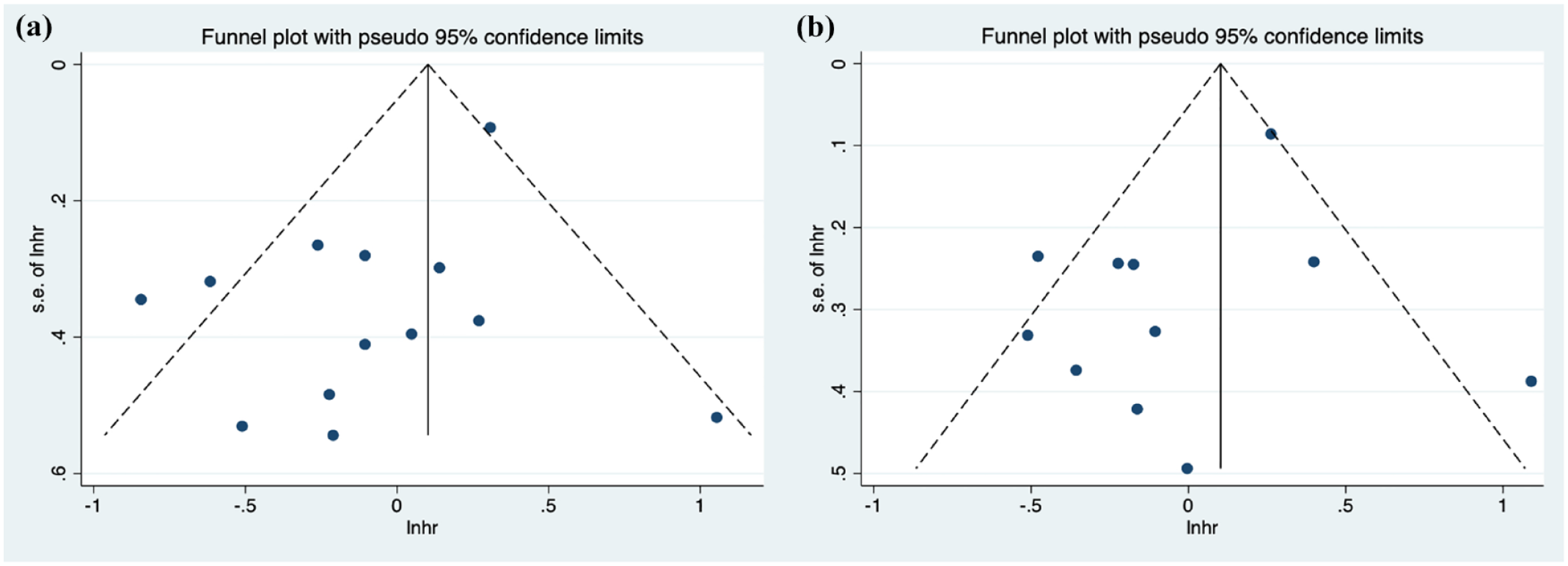
The funnel plot of the prognostic role of SUVmax on overall survival (a) and progression-free survival (b).
Discussion
Main findings
This is the largest meta-analysis to date to examine the prognostic value of pretreatment baseline 18F-FDG PET/CT-derived parameters in NSCLC patients undergoing immunotherapy. We found that high MTV is weakly associated with both poor PFS and poor OS (with borderline HRs and high heterogeneity), while other parameters, including the SUVmax, SUVmean, and TLG, were not predictive of survival outcomes.
The SUV is a widely used parameter in PET/CT interpretation, and its prognostic value in NSCLC has been assessed in many studies. 40 In our meta-analysis, both SUVmax and SUVmean were found to be nonpredictive for survival. This finding is inconsistent with a published meta-analysis, 41 in which a significant association was found between the baseline SUVmean and PFS. This inconsistency might be explained by the application of random-effects versus fixed-effects models when pooling data, which has been frequently observed in previous practice of meta-analyses. 42 We adopted a random-effects model given the clinical heterogeneity introduced during sampling and its stability was proven by sensitivity analysis. In addition, the previous meta-analysis pooled both univariate and multivariate HRs from the same population in one analysis, which might be another source of bias. In addition, the authors also acknowledged that the presence of pseudoprogression during immunotherapy would lead to inaccurate assessment of PFS, and, consequently, an unreliable association with the SUVmean. The limited prognostic value of SUVmax in NSCLC patients might be explained by the partial-volume effect and dependence of the SUVmax on tumor size and T stage. 43 In addition, the SUV only reflects the metabolic activity of lesions and does not account for the overall volume of the tumor, which is more relevant to prognosis, particularly in those with intratumoral heterogeneity.44,45
The MTV is a volume parameter that can be measured quantitatively and can reflect the metabolic volume of lesions in a certain anatomical location. Although we found a statistically significant association between MTV and both OS and PFS, we have to admit that the HRs were borderline with notable heterogeneity. Thus, the application of MTV in clinical practice shall be considered with caution. The prognostic value of MTV has been reported by published studies, 41 but a few studies challenged this finding.27,38,46 These inconsistencies might also be explained by heterogeneous patient characteristics, including a wide age range, different clinical stage and disease subtypes, and different software and the different ways used for the definition of the threshold of MTV. 47
The mechanisms underlying the predictive value of MTV are unclear. The prognostic value of MTV in oncology might be explained by its ability to quantify both the extent and metabolic activity of tumors. Specifically, a larger MTV reflects a greater number of tumor cells, which correlates with advanced disease stages and increased tissue invasion. An elevated MTV also indicates increased glucose uptake and metabolism and is a trait characteristic of aggressive tumors that demand substantial energy for rapid growth. Furthermore, larger tumors are prone to hypoxic regions, which contribute to resistance to certain therapies and promote more aggressive behavior.48–50
TLG combines the volume and metabolic activity of lesions and is calculated by multiplying the MTV by the SUVmean, providing an overall assessment of the tumor’s metabolic burden of the tumor. TLG is not prognostic according to our pooled results, which is, however, inconsistent with a published meta-analysis. 41 This inconsistency might be explained by our update of the literature search, which resulted in the identification of additional new published studies.19,46,51–55 Moreover, we included a broader patient population by setting no limitation on the stage of disease. However, a subgroup analysis based on cancer stage was not applicable because many studies enrolling patients with broad stages did not provide data on the early-stage group (i.e., stages I–II). In addition, the published meta-analysis revealed high heterogeneity in the associations between TLG and both OS (83.7%) and PFS (86.8%). Thus, we believe that conclusions concerning the prognostic value of TLG cannot be drawn given the current evidence.
Clinical implications
18F-FDG PET/CT is a widely used in practice to evaluate advanced NSCLC patients before immunotherapy. Our findings suggest that PET/CT-derived parameters, such as MTV, may hold potential prognostic value and aid in treatment planning for patients with advanced NSCLC. However, given the high cost and limited accessibility of PET/CT, more evidence, particularly cost-effectiveness evidence, is warranted to support the routine use of this modality in advanced NSCLC patients prior to immunotherapy in daily practice.
Strengths and limitations
The strengths of our study include the inclusion of the largest number of studies to examine the efficacy of 18F-FDG PET/CT in predicting survival outcomes in patients with NSCLC receiving immunotherapy, encompassing nearly all relevant parameters of 18F-FDG PET/CT.
This study also has limitations. First, high heterogeneity was observed for the majority of outcomes, particularly between MTV and survival. We performed subgroup analysis and confirmed the stability of results. However, our findings should still be interpreted with caution. Second, the incorporation of other imaging modalities or biomarkers might provide a more comprehensive assessment of immunotherapy response since 18F-FDG PET/CT may not capture all aspects of tumor biology or treatment response. However, we did not include studies of combined imaging biomarkers from FDG PET/CT and other modalities due to insufficient data. Third, we did not assess the impact of tumor stage on outcomes since data on early-stage groups were not extractable from most studies.
Conclusion
In summary, this study suggested the prognostic value of pretreatment FDG-derived parameters in the prediction of survival in NSCLC patients receiving immunotherapy. Particularly, a high MTV might predict poorer PFS and OS. However, future prospective studies with larger sample sizes are warranted to support the value of 18F-FDG PET/CT in the prognosis of NSCLC patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241293364 – Supplemental material for The role of baseline 18F-FDG PET/CT for survival prognosis in NSCLC patients undergoing immunotherapy: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241293364 for The role of baseline 18F-FDG PET/CT for survival prognosis in NSCLC patients undergoing immunotherapy: a systematic review and meta-analysis by Mingxing Huang, Yuheng Zou, Weichen Wang, Qianrui Li and Rong Tian in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241293364 – Supplemental material for The role of baseline 18F-FDG PET/CT for survival prognosis in NSCLC patients undergoing immunotherapy: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359241293364 for The role of baseline 18F-FDG PET/CT for survival prognosis in NSCLC patients undergoing immunotherapy: a systematic review and meta-analysis by Mingxing Huang, Yuheng Zou, Weichen Wang, Qianrui Li and Rong Tian in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359241293364 – Supplemental material for The role of baseline 18F-FDG PET/CT for survival prognosis in NSCLC patients undergoing immunotherapy: a systematic review and meta-analysis
Supplemental material, sj-docx-3-tam-10.1177_17588359241293364 for The role of baseline 18F-FDG PET/CT for survival prognosis in NSCLC patients undergoing immunotherapy: a systematic review and meta-analysis by Mingxing Huang, Yuheng Zou, Weichen Wang, Qianrui Li and Rong Tian in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
