Abstract
Background
Many types of cancer exhibit high nuclear factor erythroid 2-related factor 2 (NRF2), which is effective in resisting drugs and radiation. However, the role of NRF2 gene expression in predicting the prognosis of esophageal squamous cell carcinoma (ESCC) remains unclear.
Methods
The association between NRF2, heme oxygenase-1 (HO-1), baculovirus IAP repeat 5 (BIRC5), P53 gene expression levels and their relationship to immune-infiltrating cells were assessed using the Cancer Genome Atlas dataset, the Human Protein Atlas and the TISDB database. The expression of NRF2, HO-1, BIRC5, and TP53 in 118 ESCC patients was detected by immunohistochemistry, and the relationship between their expression level and clinicopathological parameters and prognosis was analyzed.
Results
In ESCC, NRF2 overexpression was significantly associated with Han ethnicity, lymph node metastasis, and distant metastasis. HO-1 overexpression was significantly associated with differentiation, advanced clinical staging, lymph node metastasis, nerve invasion, and distant metastasis. BIRC5 overexpression was significantly associated with Han ethnicity and lymph node metastasis. TP53 overexpression was significantly associated with Han ethnicity and T staging. The NRF2/HO-1 axis expression was positively correlated with BIRC5 and TP53. Kaplan–Meier and multivariate Cox regression analysis showed that NRF2, BIRC5, and TP53 genes co-expression was an independent prognostic risk factor. TISIDB dataset analysis showed that immune-infiltrating cells were significantly negatively correlated with NRF2 and BIRC5.
Conclusion
NRF2, BIRC5, and TP53 axis gene expressions are predictors of poor prognosis for ESCC. The overexpression of the NRF2/HO-1/BIRC5 axis may not be related to immune-infiltrating cells.
Introduction
The two predominant types of esophageal cancer are squamous cell carcinomas(ESCC) and adenocarcinomas, with the former accounting for around 90% of cases. 1 Although the exact causes of ESCC remain unknown, increasing evidence suggests that highly constitutive activation of NRF2 is associated with an increased risk of various human tumors. 2 Nuclear factor-erythroid-2-related factor 2, nuclear factor erythroid-derived 2-like 2 ((NRF2), nuclear factor erythroid-derived 2-like 2 gene (NFE2L2)) as a sensor of oxidative or electrophilic stress induces nuclear translocation of transcription factors, which regulate the expression of antioxidant and detoxification genes such as HO-1 by combining with antioxidant/electrophilic response element (ARE). Abnormally active NRF2 promotes the occurrence and development of various types of tumors, including hepatocellular carcinoma, 3 pancreatic tumorigenesis, 4 and head and neck squamous cell carcinoma. 5 Knockdown of NRF2 small interfering RNA (siRNA) or inhibition of NRF2 activity by some chemicals makes cancer cells prone to apoptosis. 6 Cell apoptosis inhibition serves as the hallmark of tumor development and progression. 7 BIRC5 (Baculoviral IAP Repeat Containing 5), a unique member of the inhibitor of apoptosis (IAP) family, may promote cell proliferation and cancer progression by inhibiting apoptosis-related signaling pathways. Dysregulation of BIRC5 has been demonstrated to be a key signature of a variety of cancers, such as renal cell carcinoma, glioma, melanoma, and endometrial cancers.8,9 High levels of BIRC5 expression have also been associated with cancer progression, chemotherapy resistance, and a poor prognosis.10–12 An earlier study found that nuclear BIRC5 expression was related to the grade and tumor node metastasis (TNM) stage of oral squamous cell carcinoma. 13 Some studies suggested that TP53 and BIRC5 played a role in the AURORA kinase signaling pathway and thus were likely to be involved in pancreatic carcinogenesis.14,15 Frequent mutations of the TP53 gene in 96% of high-grade serous ovarian cancer tumors could lead to the de-repression of oncogenic DREAM target genes, such as BIRC5. Overexpression of BIRC5 in cells sensitive to p53-dependent cell death markedly inhibits apoptosis induced by ultraviolet light. In addition, NRF2 can increase the nuclear localization of recruitment on ARE sequence by combining with mutant TP53, and inhibit the transcriptional activation of gene HO-1, thus reducing the survival and metastasis ability of breast cancer cells. 16 Also in breast cancer cells, it is worth noting that in another study, BIRC5 acts as a downstream regulator of NRF2-mediated survival-promoting phenotype. 17 In type 2 papillary renal cell carcinoma, over-expressed NRF2 inhibits reactive oxygen species (ROS) levels by up-regulating the transcription of gene HO-1 carrying antioxidant response elements, thereby activating the transcription of BIRC5 and inhibiting the death of tumor cells. The NRF2/ BIRC5 pathway is overexpressed in endometrial tumor tissue, which may be related to progesterone resistance. During this period, TP53 may also play a reasonable role, but at present, it is not significantly associated with the occurrence of progesterone treatment response. On the other hand, this study also proves that BIRC5 is one of the key downstream regulators.18,19 With these understandings, we hypothesized that the anti-apoptosis of tumor cells driven by NRF2/HO-1 may be mediated by BIRC5 regulation, to maintain the survival ability of esophageal squamous cell carcinoma and maintain proliferation.
HO-1, heme oxygenase 1 gene (HMOX1)) as a downstream target gene for NRF2, plays a crucial role in anti-oxidation, anti-inflammation, anti-apoptosis, and cyto-protection. 20 HO-1 was reported to play a part in the pathogenesis and progression of cancers.21,22 Mesenchymal stem cells (MSC) increase the expression of HO-1, which directly controls the anti-apoptotic function of MSC by promoting mitochondrial biogenesis. 23 Pharmacological induction of HO-1 increases TP53 expression in human breast cancer cells. 24 However, the correlation between NRF2/HO-1 and the expression of the BIRC5 and TP53 proteins, and their relationships with ESCC outcome remains largely unknown. In the present study, we measured the expressions of NRF2/HO-1, BIRC5, and TP53 in ESCC samples to assess their correlations with key clinical parameters and ESCC outcomes.
Tumors have the ability to maintain immunosuppressive microenvironment, which contains a variety of infiltrating immune cells, and different immune microenvironments have different effects on the response of tumor patients to immunotherapy. A variety of factors, including strict control of Nrf2 activity, cell type and specific tumor microenvironment, regulate the transition between Nrf2's carcinogenic and anti-cancer effects. In the early stages of tumors, Nrf2 activation has anti-cancer effects, while in advanced stages, constitutive expression of Nrf2 can cause cancer by protecting cancer cells from damage caused by oxidative stress.25,26 In tumors that continuously activated NRF2, the infiltration of dendritic cells and CD4+ and CD8+ T cells was significantly reduced, suggesting that NRF2 pathway activation favored the creation of an immune evasive tumor microenvironment. 27 Activation of HO-1 impairs anti-tumor CD8+ T cell responses in the tumor microenvironment by regulating STAT1/3 and NFκB. 28 In addition to this, positive HO-1 immunoreactivity was detected in tumor-associated macrophages of cancerous tissue, suggesting that HO-1 may affect cancer progression by modulating the tumor microenvironment. 29 Long-term survivors in the BIRC5-positive group may be associated with an anti-BIRC5 immune response. 30 An increase in the copy number of BIRC5 somatic cells affects the level of infiltration of various immune cells, thereby reducing survival time in patients with low-grade gliomas. 31 Reconstitution of functional TP53 into head and neck squamous cell carcinoma (HNSCC) cells could alter the tumor immune microenvironment and either directly enhance anti-tumor immunity or improve responses to immune-activating therapies. 32 In the study, using online database analysis, to observe the correlation between gene expression levels in esophageal cancer and the enrichment of immune cells infiltrating esophageal cancer.
Materials and methods
Bioinformatic analysis
The Cancer Genome Atlas (TCGA) gene expression array database (https://www.oncomine.org/) is an online cancer microarray database. UCSC XENA (https://xenabrowser.net/datapages/) RNAseq data in TPM format for TCGA and GTEx through the Toil process. 33 Extraction of normal tissue data from ESCC (esophageal cancer) and GTEx of TCGAs were also performed. The mRNA expression levels of NRF2, HO-1, BIRC5 and TP53 were especially compared between clinical ESCC samples and normal controls. The Human Protein Atlas (http://www.proteinatlas.org/) is a publicly available database with millions of high-resolution images showing the spatial distribution of proteins in normal human tissues and different cancers. 34 In our study, this database was used for immunohistochemical staining profiles of NRF2, HO-1, BIRC4, and TP53 in head and neck squamous cell carcinoma are presented. TISIDB (cis.hku.hk/TISIDB/), a web portal for tumor and immune cell interactions developed by the Zhang laboratory in 2019, was used to evaluate the associations between the expressions of NRF2, HO-1, BIRC5 and various types of lymphocytes. 35 All the data were from the PubMed database and TCGA.
Specimens and tissue microarrays
Archived formalin fixed paraffin embedded tumor tissue was obtained from 118 patients(62 Han and 56 Kazak) diagnosed with primary ESCC between January 2013 and December 2018 at the First Affiliated Hospital of Xinjiang Medical University (Xinjiang Uygur Autonomous Region, China). The harvested samples were made into tissue microarrays. Tissue cores with a diameter of 1.5 mm were extracted from each donor block with each tissue microarray containing 60 cases. The inclusion criteria were as follows: primary ESCC of the Han and Kazakh populations. The exclusion criteria were radiotherapy or chemotherapy before surgery, secondary esophagal malignancies, and patient ethnicities other than Han and Kazakh. The following information was recorded for each patient: age, sex, ethnicity, tumor location, tumor size, degree of differentiation, and TNM staging, lymph node status, vascular invasion, nerve invasion, postoperative radio-chemotherapy, and progression of the disease. The use of human tissues was approved by the local institutional review board. All patients signed the informed consent forms.
Immunohistochemical staining
Tissue microarray wax blocks were sliced into 4-micrometer-thick serial sections for immunohistochemical staining. Tissue chips were placed in the 71CC slicer for 40 min, followed by section deparaffinization using xylenes and graded ethanol. After dewaxing, the tissue chips were immersed in a boiling EDTA repair solution (pH = 8.0) for 15 min. The primary antibodies used for immunohistochemical staining included NRF2 (ab62352, abcam, 1:100), HO-1 (af5393, 1:200), BIRC5 (ab76424, abcam, 1:500), and TP53 (Dako, 1:100) The DAB chromogenic agent was used for visualization. Representative mages were taken while the tissue microarray was being examined under an optical microscope
Expression scoring
Each stained section was independently evaluated under 200× magnification by two authors. Immunoreactivities of NRF2, HO-1, BIRC5, and TP53 were all semi-quantitatively assessed by calculating the proportion of positively stained cells multiplied by the staining intensity. The staining intensity was evaluated as follows: 1 = no staining or just faintly stained, 2 = intensely stained. The percentage of positively-stained tumor cells was rated as follows: 1 = 0–25%), 2 = 26%–74%) and 3 = 75%–100%. A sum score of ≥3 was judged positive, while a value of <3 was deemed negative.
Statistical analyses
All statistical analyses were carried out using the SPSS 25.0 program. The Student's t-test or one-way analysis of variance was employed. For categorical variables, the X2 test or Fisher's exact test was utilized. For correlation analysis, a Spearman rank correlation test was applied. The mean ± standard deviation was utilized to show the data. All significance tests were two-sided. Overall survival (OS) time was defined as the period from operation to death or final follow-up as the research endpoint. The OS rate of all patients was determined using Kaplan–Meier analysis and then a log-rank test to establish statistical significance. For univariate and multivariate analyses, the Cox proportional hazards models were applied. A two-sided P < 0.05 denoted statistical significance.
Results
The expression levels of NRF2/HO-1, BIRC5, and TP53 in pan-cancer, especially esophageal carcinoma
To investigate the RNA expression levels of NRF2, HO-1, BIRC5, and TP53 in patients with pan-cancer and their corresponding normal samples, we first analyzed 33 types of tumors in the TCGA database that encompassed 10,538 samples. The expressions of NRF2, HO-1, BIRC5, and TP53 were significantly higher in the tumor group than in the control group (Figure 1(a) to (d)), notably in esophageal carcinoma. The expression of NRF2(P < 0.001), HO-1(P = 0.0013), BIRC5(P < 0.001), and TP53(P < 0.001) in esophageal carcinoma was moderately higher than those in the normal tissue (Figure 1(e) to (h)). As a result, we examined the expression patterns and cell localization of NRF2, HO-1, BIRC5, and TP53 in immunohistochemistry (IHC) pictures of HNSCC (Human Protein Atlas (HPA)). NRF2, BIRC5, and TP53 were all moderate to strongly stained in tumor tissue, while HO-1 was unstained or weakly stained (Figure 1(i) to (l)).

Expression of NRF2/NFE2L2, HO-1/HMOX1, BIRC5, and TP53 in pan-cancer and esophageal carcinoma. (a) to (d) Box plot showing the expression level of NRF2, HO-1, BIRC5, and TP53 in different cancer types compared to adjacent normal tissues determined based on data from The Cancer Genome Atlas (TCGA). (e) to (h) Histogram showing the expression of NRF2, HO-1, BIRC5, and TP53 in esophageal carcinoma and normal tissues(TCGA). (i) to (l) The image shows four proteins expressed in squamous cell carcinoma of the head and neck from The Human Protein Atlas(HPA). The significance computed by the Wilcoxon rank sum test was annotated by the number of stars. Magnification is 40×.
Increased NRF2/HO-1 and BIRC5 protein expressions in primary ESCC
A summary of patient clinicopathological characteristics is displayed in Supplemental Table 1. These patients were followed up with a median time of 34 months(2–108 months). Approximately two-thirds of patients were ≥60 years of age at the time of surgery and 27.1% were males. By using IHC, the levels of NRF2/HO-1, and BIRC5 protein expression in tissues around the tumor as well as ESCC were evaluated (Figure 2). As shown in Supplemental Table 2, the protein expression of NRF2(P = 0.022), HO-1(p < 0.001) and BIRC5(p < 0.001) in ESCC were significantly higher than those in normal tissues.
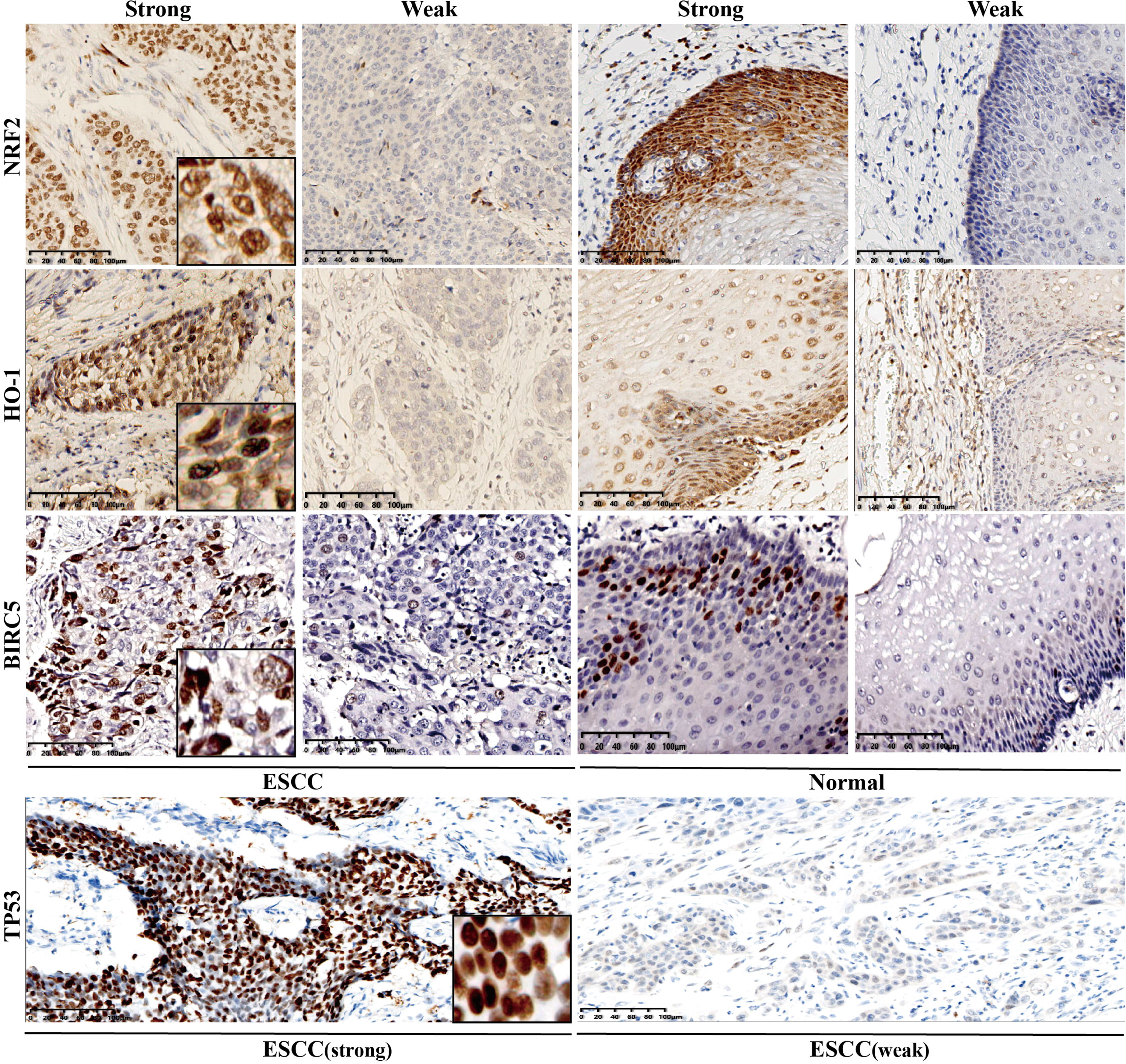
Representative immunohistochemistry staining images were shown for the expressions of NRF2 (first line), HO-1 (second line), BIRC5 (third line), and TP53 (last line) in esophageal squamous cell carcinoma (ESCC) and normal esophageal epithelium tissues. First and third columns, positive/strong expression at high magnification (brown); second and last columns, negative/weak expression at high magnification (lightly brown or no strain). The first two columns are ESCC tissues; the last columns, are normal adjacent esophageal tissues. The final two columns are normal neighboring esophagus tissues; the first two columns are ESCC tissues.
The relationship between the expression of NRF2, HO-1, BIRC5 and TP53 and clinical pathological parameters
As shown in Supplemental Table 3, a strong NRF2 expression in primary ESCC tumors was significantly correlated with Han ethnicity (P = 0.008), lymph node metastasis (P = 0.042), and distant metastasis (P = 0.032). A higher expression of HO-1 in primary ESCC was significantly correlated with poor differentiation (P = 0.041), higher AJCC stage (P = 0.021), lymph node metastasis (P = 0.029), nerve invasion (P = 0.018) and distant metastasis (P = 0.042). Significant correlations between higherBIRC5 high expression and Han ethnicity (P = 0.018), mph node metastasis (P = 0.037) were observed. Higher TP53 expression was significantly associated with Han ethnicity (P < 0.001) and full thickness invasion (P = 0.046).
NRF2/HO-1 overexpression is linked to BIRC5 and TP53 expression
As presented in Table 1, baseline expression levels were significantly correlated between all four proteins. A significant positive correlation was found between NRF2 and HO-1 expression (r = 0.52, P = 0.008), NRF2 and BIRC5 expression (r = 0.357, P < 0.001), NRF2 and BIRC5 expression (r = 0.357, P < 0.001), NRF2 and BIRC5 expression (r = 0.269, P = 0.003). Remarkably, the co-expression of the proteins NRF2/HO-1 was shown to have significant associations with TP53 (r = 0.306, P = 0.001) and BIRC5 (r = 0.284, P = 0.002), respectively.
The correlations between different gene expressions.

Co-expression refers to the co-expression of NRF2 and HO-1 proteins.
*Correlation is significant at the 0.01 level (2-tailed). The values in the chart are R-values.
**Correlation is significant at the 0.05 level (2-tailed).
BIRC5: baculovirus IAP repeat 5; HO-1: heme oxygenase-1; NRF2: nuclear factor erythroid 2-related factor 2; TP53: tumor protein P53.
Higher NRF2/HO-1 co-expression correlates with decreased OS
Among 118 ESCC patients, 30 were younger than 60 years old, of which 4 were NRF2/HO-1 positive and 26 were arbitrary negative, with a positive rate of 13.3%. There were 55 Han patients, of which 17 were NRF2/HO-1 positive, 38 were random negative, and the positive rate was 30.9%. There were 69 patients with tumors infiltrating into the whole layer, including 14 patients with positive NRF2/HO-1 expression and 55 with negative NRF2/HO-1 expression, with a positive rate of 20.3%. There were 17 patients in American Joint Committee on Cancer (AJCC) stage IV, of which 7 were positive for NRF2/HO-1 and 10 were negative at random, with a positive rate of 41.2%. There were 19 patients in AJCC stage III, including 2 with positive NRF2/HO-1 co-expression and 17 with negative NRF2/HO-1, with a positive rate of 10.5%. There were 16 patients with vascular infiltration, of which 5 were positive for NRF2/HO-1 co-expression and 11 were negative at random, with a positive rate of 31.3%. There were 19 patients with nerve invasion, of which 5 were positive for NRF2/HO-1, 14 were negative at random, and the positive rate was 26.3%. Kaplan–Meier survival analysis (Figure 3) showed that patient OS with NRF2/HO-1 co-expression was significantly shorter than those with either negative NRF2 or HO-1, concerning patients under the age of 60 (P = 0.043), Han ethnicity (P = 0.046), patients with tumor full layer invasion (P = 0.012), AJCC stage III (P = 0.010), AJCC stage IV (P = 0.026), vascular invasion (P = 0.001), and nerve invasion (P = 0.025). In addition, the analysis results showed that the progression-free survival (PFS) in patients with NRF2/HO-1 co-expression was worse than that of any negative patients, respectively in females, <60 years old, Han ethnicity, the tumor was located in the middle of the thoracic segment of the esophagus, tumor size between 1.5 cm and 2.5 cm, moderate differentiation, full-layer invasion, AJCC stage III, lymph node metastasis, vascular invasion, neural invasion, and patients who received radio-chemotherapy. The outcomes are displayed in Figure 4.

In the TISIDB database, the Spearman's rank correlation coefficient was used to examine the relationship between the immune infiltrate and the expression of the proteins NRF2/NFE2L2 (column 1), HO-1/HMOX1 (column 2), and BIRC5 (column 3). Each row represents the same type of immune infiltrating cells. (a) type 1 helper cell (Th1); (b) type 17 helper cell (Th17); (c) immature B cell (Imm-B); (d) CD56dim natural killer cell (CD56 dim); (e) myeloid-derived suppressor cell (MDSC); (f) eosinophil; (g) monocyte.
All variables on univariate Cox regression analysis with P ≤ 0.10 were submitted to multivariate Cox regression analysis to uncover independent prognostic factors. In the univariate analysis, the risk variables for OS were revealed to be gender (P = 0.011), higher NRF2 expression (P = 0.031), and higher BIRC5 expression (P = 0.014). The results of the multivariate Cox regression analysis revealed that gender (hazard ratio (HR) 4.561, 95% confidence interval (CI) 1.411, 14.75) and NRF2 expression (HR 0.424, 95% CI 0.183, 0.983) were independent predictive factors for OS. The results are shown in Supplemental Table 4. Gender (P = 0.024), lymph node metastasis (P = 0.048), vascular invasion (P = 0.011), nerve invasion (P = 0.032), NRF2 high expression (P = 0.004), HO-1 high expression (P = 0.012), and BIRC5 high expression (P = 0.018) were found to be risk variables for PFS in the univariate analysis. Multivariate analysis (Supplemental Table 5) showed that PFS was predicted by gender(HR 3.863, 95% CI 1.175, 12.695, P = 0.026). As shown in Table 2, univariate survival analysis for OS showed that BIRC5, TP53, NRF2, and HO-1 were co-expressed (P = 0.024), BIRC5, TP53, and NRF2 were co-expressed (P = 0.004). The effect on OS of ESCC patients was statistically significant, but the co-expression of BIRC5 and TP53 (P = 0.082) did not affect OS. However, further multivariate analysis showed that BIRC5, TP53, and NRF2 co-expression (P = 0.01) had significant effects on OS . Univariate survival analysis for PFS showed that BIRC5, TP53, NRF2, and HO-1 were co-expressed (P = 0.012); BIRC5, TP53, and NRF2 were co-expressed (P = 0.003); and BIRC5 and TP53 were co-expressed (P = 0.037). Likewise, the multivariate analysis showed that BIRC5, TP53, and NRF2 co-expression (P = 0.007) was associated with PFS. These results suggested that inhibiting the expression of both BIRC5, TP53, and NRF2 proteins at the same time is beneficial to prolong the OS and PFS in ESCC.
Effects of co-expression of BIRC5, TP53, NRF2, and HO-1 on the prognosis of ESCC.

BIRC5: baculovirus IAP repeat 5; CI: confidence interval; HO-1: heme oxygenase-1; NRF2: nuclear factor erythroid 2-related factor 2; OS: overall survival; PFS: progression-free survival; TP53: tumor protein P53.
Relationships between NRF2, HO-1, BIRC5 expression and infiltrating immune cells
We evaluated whether NRF2/NFE2L2, HO-1/HMOX1 and BIRC5 were also involved in the tumor-immune interactions in esophageal carcinoma patients using TISIDB, an algorithm developed to analyze the abundance of tumor-infiltrating immune cells comprehensively and flexibly. According to the findings, NRF2/NFE2L2 and BIRC5 expression levels were negatively correlated with infiltrating type 1 T helper cell (r = −0.187, P = 0.011, and r = −0.343, P < 0.001, respectively); type 17 T helper cell (r = −0.281, P < 0.001, and r = −0.422, P < 0.001, respectively); immature B cell (r = −0.188, P = 0.01, and r = −0.433, P < 0.001, respectively); CD56dim natural killer cell (r = −0.227, P = 0.002, and r = −0.222, P = 0.002, respectively); myeloid-derived suppressor cell (r = −0.159, P = 0.031, and r = −0.273, P < 0.001, respectively); eosinophil (r = −0.214, P = 0.003, and r = −0.52, P < 0.001, respectively); and monocyte (r = −0.402, P < 0.001, and r = −0.31, P < 0.001, respectively) (Figure 5(a) to (g)). HO-1 expression levels were positively correlated with infiltrating levels of type 1 T helper cell (r = 0.204, P = 0.005); type 17 T helper cell (r = 0.192, P = 0.009); immature B cell (r = 0.185, P = 0.012); CD56dim natural killer cell (r = 0.307, P < 0.001); myeloid-derived suppressor cell (r = 0.314, P < 0.001); eosinophil (r = 0.281, P < 0.001); and monocyte (r = 0.246, P < 0.001) (Figure 5(a) to (g)). These results suggest that Nrf2 and BIRC5 may play important synergistic roles in immune invasion and exhaustion in esophageal cancer, while HO-1 does the opposite.
Discussion
One of the most prevalent and deadly malignancies in the world is esophageal cancer, particularly ESCC. The defense program of oxidative stress is a new way of thinking about anti-tumor in the tumor microenvironment. In addition to conventional treatment strategies, the development of therapies targeting oxidative stress defense represents an attractive strategy to enhance the sensitivity of tumor cells to existing therapies. 36 To a certain extent, this treatment can help alleviate the situation of high mortality in the middle and late stages of esophageal cancer. Therefore, the analysis of biomarkers in ESCC has practical appeal. As many studies have reported, these biomarkers are involved in the development, angiogenesis, and invasion of ESCC.37–39
As a result, using data from the TCGA and HPA databases, we comprehensively investigated the expression of NRF2/HO-1, BIRC5, and TP53 in patients with esophageal carcinoma. We discovered that the expression levels of the aforementioned four transcription factors were substantially higher than those of the normal samples. We also noted that the NRF2, HO-1, BIRC5, and TP53 were expressed in the neucleus using the HPA database. Similar conclusions were also observed in 118 ESCC patients. Additionally, we performed a Spearman's rank correlation analysis on the expressions of NRF2, HO-1, BIRC5, and TP53 in our data based on the results of the published research. The findings shown in Table 1 revealed that they were statistically significant and associated, and the findings from the STRING database supported these findings (Supplemental Figure 2).
The prognosis of the NRF2/HO-1 co-expression group is significantly worse than that of other groups. The majority of patients with worse outcome are under 60 years old, of Han descent, with tumor infiltration into the whole esophagus, vascular infiltration, and AJCC stage III–IV. However, only the overexpression of HO-1 was not connected to the poor prognosis of OS. Additionally, although the prognosis of co-over-expression NRF2 and HO-1 was similar to that of other groups, the prognosis of NRF2 and BIRC5 co-overexpression was significantly worse than that of other groups (Supplemental Figure 2). The co-expression of NRF2, HO-1, and BIRC5 on prognosis in patients with TP53 overexpression was examined in light of the impact of TP53 mutation in malignancies; however, the difference in findings was not statistically significant (Supplemental Figure 3). Although TP53 mutation is thought to play a role in the tumorigenicity of NRF2/HO-1 in many types of tumors, this study discovered that the co-expression of BIRC5 induced by NRF2 may result in a poor prognosis in patients with ESCC, even though TP53 mutation may not be a component. However, this finding needs to be confirmed through further experimental research. Gender, lymph node metastasis, vascular metastasis, NRF2, HO-1, and BIRC5 were all shown to be significantly linked to both PFS and OS in the univariate COX analysis. A multi-factorial analysis was performed to examine the independent prognostic variables. Female patients were independent prognostic factors for shorter PFS and OS in a multivariate analysis after adjustment for possible confounders, whereas patients with overexpression of NRF2 were independent prognostic factors for poor OS.
The protein levels of NRF2, BIRC5, and TP53 were all associated with certain racial or ethnic groupings in the correlation analysis of clinicopathological parameters. Our further analysis found that the co-expression of NRF2, HO-1, BIRC5, and TP53 proteins had an impact on the OS rate and PFS rate of ESCC. A multivariate Cox analysis showed that NRF2, BIRC5, and TP53 may be independent prognostic factors for patients with ESCC. This once again fully proves that NRF2, HO-1, and BIRC5 are involved in the occurrence and development of ESCC, and have an impact on the survival and prognosis of patients. Therefore, we conclude that simultaneously inhibiting the expression of NRF2 and BIRC5 proteins is beneficial to prolong the OS of ESCC patients, and NRF2 and BIRC5 proteins can be used as molecular markers of poor prognosis in patients with ESCC. Recent research has demonstrated that BIRC5 is one of the genes that is downregulated in lung tissue when NRF2 -/- mice are exposed to cigarette smoke, indicating that NRF2 may directly interact with the trans gene expression of BIRC5. 17 It is generally known that BIRC5 is a significant gene involved in the study of the TP53 pathway. Thus, by directly up-regulating BIRC5, NRF2 may adjust the P53 pathway and sustain the capacity of ESCC to proliferate.
ESCC is associated with an imbalance of T helper cell subsets, and a substantial infiltration of T helper cell 1 is associated with a good prognosis. A positive prognosis for individuals with ESCC can be predicted by the mast cells that express IL-17A in the intrinsic muscle layer.40,41 The relationship between immature B cells and the prognosis of ESCC has not yet been investigated. Nevertheless, the infiltration of immature B cells in the high-risk group was dramatically decreased in HNSCC. 42 The cytotoxicity of a Neem leaf preparation to malignant cells can be increased by activating natural killer (NK) cells.
Responders had a statistically significant increase in CD56dim NK cells as compared to non-responders. 43 Patients with high numbers of myeloid-derived inhibitory cells had a higher OS among ESCC patients receiving postoperative chemotherapy. 44 Higher levels of eosinophil infiltration were linked to both improved OS and PFS. 45 Monocyte count decline suggests worsening OS and DFS for ESCCs receiving radical resection. 46 In this study, we discovered that the clinicopathological characteristic of positive lymph node metastasis was linked with the levels of the proteins NRF2, HO-1, and BIRC5. The expression of NRF2 and BIRC5 was shown to be inversely linked with the amount of immunological infiltration (including type 1 helper cell, type 17 helper cell, immature B cell, CD56dim NK cell, myeloid-derived suppressor cell, eosinophil, and monocyte), but HO-1 was entirely the opposite. This was confirmed using the TISIDB database. Given that NRF2 and BIRC5 are positively connected with the expression of the HO-1 protein in Spearman's correlation analysis, we may anticipate that HO-1 affects immune cell infiltration differently in various tumor types (SCC and adenocarcinoma). The cases in the TISIDB database contain both SCC and adenocarcinoma, but the data in this analysis are all SCC. On the other hand, the role of HO-1 in various types of tumors has been under discussion, and there is no irrefutable conclusion on promoting or inhibiting lymph node metastasis, and this study shows that the positive effect of HO-1 on lymph node metastasis may not be mainly reflected in the abundance of immune cell infiltration.
Conclusions
In conclusion, NRF2, HO-1, BIRC5, and TP53 are commonly expressed in ESCC and have significant prognostic value. NRF2, BIRC5, and TP53 co-expression are independent risk factors for ESCC. High expression of the NRF2/HO-1 axis in ESCC is associated with advanced tumor and vascular invasion. The NRF2/HO-1 axis may promote lymph node metastasis in tumors by regulating the expression of BIRC5, but this metastatic mechanism may not be related to immune cell infiltration such as T helper cells, immature B cells, CD56dim NK cells, myeloid-derived suppressor cells, eosinophils, and monocytes. Further studies are needed to elucidate the specific mechanism by which NRF2/HO-1/BIRC5 affect lymph node metastasis in ESCC. Our results suggest that NRF2 may be a key transcription factor that activates downstream target genes and the biological behavior of ESCC, and is a potential therapeutic target for the treatment of ESCC.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231176571 - Supplemental material for NRF2/HO-1 axis, BIRC5, and TP53 expression in ESCC and its correlation with clinical pathological characteristics and prognosis
Supplemental material, sj-docx-1-jbm-10.1177_03936155231176571 for NRF2/HO-1 axis, BIRC5, and TP53 expression in ESCC and its correlation with clinical pathological characteristics and prognosis by Yongmei Gao, Li Wan, Mengyan Li, Bo Wang and Yuqing Ma in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The State Key Laboratory of Pathogenesis, Prevention and Treatment of High Incidence in Central Asia (No. SKL-HIDCA-2020-4) and Xinjiang Medical University Graduate Innovation and Entrepreneurship Project (grant_number CXCY2022010).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
