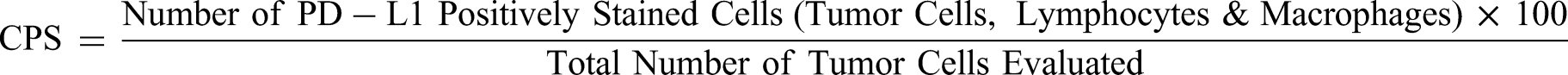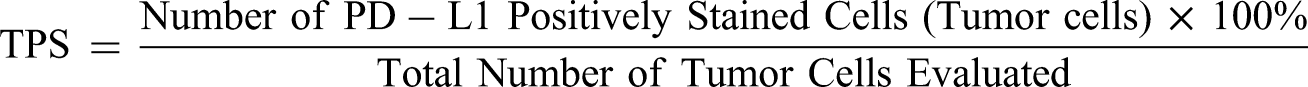Abstract
Background
Programmed death-ligand 1 (PD-L1) expression in some tumors has prognostic implications. This work aims at investigating PD-L1 expression in diffuse large B-cell lymphoma (DLBCL) and to study its association with clinicopathological variables.
Methods
The study consisted of 75 DLBCL patients who were cared for at the King Hussein Cancer Center during the period 2015–2018. The expression of PD-L1 in tumor tissue was assessed by immunohistochemistry using the anti-human PD-L1 (Clone 22C3) monoclonal antibody. The correlation between gender, age, clinical stage, pre-treatment-LDH level, tumor location, response to therapy, overall and event-free survival with PD-L1 expression was studied.
Results
Six patients were excluded from further analysis as they were in relapse at the time of tissue sampling. The tumor proportion score (TPS) was ≥1% in 16/69 (23.2%) of DLBCL cases while the combined positive score (CPS) at a cut-off of ≥20 was observed in 23/69 (33.3%) cases. No significant difference in PD-L1 expression was found between germinal center B-cell-like (GCB) and non-GCB subtypes. Similarly, no differences in PD-L1 expression (at CPS ≥20 and TPS ≥1) were found between different genders, age groups, clinical stages, tumor location, and patient response to therapy. However, base-line lactate dehydrogenase was significantly elevated in patients with PD-L1 CPS ≥20. The overall survival was not significantly different between PD-L1-positive and -negative groups. On the other hand, the median event-free survival was higher in either of the PD-L1 TPS or CPS negative groups at 107months each versus 54 months in the PD-L1 positive group of either category.
Conclusions
PD-L1 expression can predict event-free survival in DLBCL cases and therefore poor prognosis.
Keywords
Introduction
Lymphoma is a cancer that originates in the lymphatic system and appears in general as a lymphadenopathy or solid tumor. 1 It is divided into Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL). NHL ranks among the 10 most common malignancies in the Western world and accounts for 2.8% of all new cancer diagnoses globally. 2 Diffuse large B cell lymphoma (DLBCL) is the most common aggressive lymphoma subtype of NHL. 3 Geographically, the incidence of lymphoma varies secondary to a variety of genetic and environmental factors. Furthermore, the incidence rate of lymphoma increases with age. Almost half of the patients are over the age of 60 years, and around one-third are over the age of 70 years. 4
Jordan is a developing country located in the Middle East that has a population of around 10 million people, who are mainly of Arab descent. According to a recent report, NHL is the seventh most common cancer in Jordanian males and the fourth most common cancer in Jordanian females. 5 A retrospective study conducted in Jordan on 4189 patients from 2014 to 2019 found that the commonest high-grade NHL type was DLBCL, which accounted for 53% of NHL cases. 6
Patients with DLBCL are usually treated using R-CHOP regimen, namely rituximab (R) in association with prednisolone, doxorubicin, cyclophosphamide, and vincristine (CHOP). 7 According to one study, the 5-year survival rates in the first-line setting of DLBCL cases was 60%–70% while up to 50% of patients become refractory to or relapse after treatment. 8 Patients with non-germinal center B-cell-like (non-GCB) lymphoma DLBCL treated with R-CHOP have a worse prognosis compared with GCB cell lymphoma. 9 In fact, patients with poor response to R-CHOP have a 1-year overall survival. 10
The expression of programmed death-ligand 1 protein
Materials and methods
Sample collection
Seventy-five Arab, Jordanian DLBCL patients who visited the King Hussein Cancer Center (KHCC) from 2015 to 2018 were included in the study. The Institutional Review Board (IRB) approval was obtained from KHCC (IRB #21 KHCC 074). Formalin-fixed, paraffin-wax embedded blocks were retrieved. The most representative part of the blocked tissues was marked for sampling to the tissue microarray. Cases of NHL other than DLBCL were excluded. Six patients whose biopsies were taken at the time of relapse were also excluded. Data regarding the patient's age, sex, clinical stage, survival, pretreatment-LDH level, response to therapy and or progression were obtained from the Medical Records Department at KHCC.
Quick-Ray Manual Tissue Microarrayer (TMA) was used to generate 60 equivalent cores, 2 mm in size. Afterwards, a 2 mm tissue punch was used to extract cylindrical tissue cores from the selected patient paraffin donor block using the evaluated hematoxylin and eosin (H&E) sections as a guide to punch the tissue. After the tissue was punched out, it was re-embedded into the prepared recipient mold at defined array coordinates. Then, the wax mold was placed in an incubator for 1 hour at 65–70°C with an embedding cassette placed on it. Two 4 μm sections were created for each block: the first was stained with H&E and was used to evaluate appropriateness of the tumor cores; the second was placed on a charged slide (Thermo Scientific, USA) and was further processed for immunohistochemistry (IHC) staining.
IHC staining of tumor tissues with PD-L1 antibody
IHC was performed using the Benchmark-Ultra machine, Ventana Amplification kit and Optiview DAB IHC detection kit (Ventana Medical Systems, Tucson, AZ, USA). The protocol used involves deparaffinization (at 72°C) of the TMA sections, placing sections in xylene then hydrating them with descending concentrations of ethanol. On-board antigen retrieval was performed by incubation with Ventana cell conditioning reagent (PH = 8) at 100°C for 48 min. The endogenous biotin is blocked prior to the addition of PD-L1 primary antibody using Ventana Endogenous Biotin Blocking kit for 4 min. Anti-human PD-L1 monoclonal mouse antibody Clone 22C3 (Dako, Agilent, Carpentaria, CA, USA) was used (1:40 dilution) and incubated for 60 min at 36°C. After washing, the ultraView Universal HRP Multimer (which is an HRP enzyme that is directly conjugated to the secondary antibody) was added and incubated for 8 min at room temperature. After developing the slides and applying chromogen (DAB), the slides were counterstained with hematoxylin (Harris Hematoxylin, Roche, USA). Finally, the slides were dehydrated, mounted with DPX, and cover slipped as in routing processing. PD-L1 IHC 22C3 PharmDx control slides (Dako, Agilent Carpentaria, CA, USA) were used to check the validity of the IHC staining run. Each control slide contained two pelleted cell lines: NCI-H226 with moderate PD-L1 protein expression and MCF-7 with negative PD-L1 protein expression.
Interpretation of results
`IHC evaluation for each section was performed by an experienced pathologist (MAS) using the manual exact count method. The entire core of each case was evaluated at 20× magnification with at least 100 tumor cells present per case using an Olympus microscope (Japan). Assessment was done in two ways:
Combined positive score (CPS) which is defined as:
Specimens were considered PD-L1-positive at TPS ≥ 1% and CPS ≥ 20.
Statistical analysis
Patients’ demographic and clinical data were presented using descriptive statistics. Online-version of Fisher's exact test was used to find out if a significant difference exists between PD-L1-positive and ----negative groups. A P-value of ≤0.05 was considered statistically significant. Survival analysis was carried out using the Kaplan–Meier test utilizing SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). The Kaplan–Meier analysis was performed to study the correlation between PD-L1 expression and OS; the interval of time that individuals diagnosed with DLBCL have been alive since the date of diagnosis and between PD-L1 expression and EFS; and the interval of time that patients are free from events including relapse of the lymphoma or death.
Results
This study included 69 patients diagnosed with DLBCL from 2015 to 2018 in KHCC. Participants’ age ranged from 9–96 years (mean = 48 ± 19.2 years), including 40 (58%) males and 29 (42%) females. Forty-five (65.2%) patients in this cohort showed complete remission (CR) while 24 (34.8%) had no or partial response to therapy. Sixty (87%) patients had a known clinical stage, namely: 22 (31.8%) were at an early stage (I and II), 38 (55%) were at a late stage (III and IV), while 9 (12%) had no available data regarding the stage (Tables 1 and 2).
Correlation of PD-L1 (CPS: cutoff ≥20) positive score with clinicopathological variables.
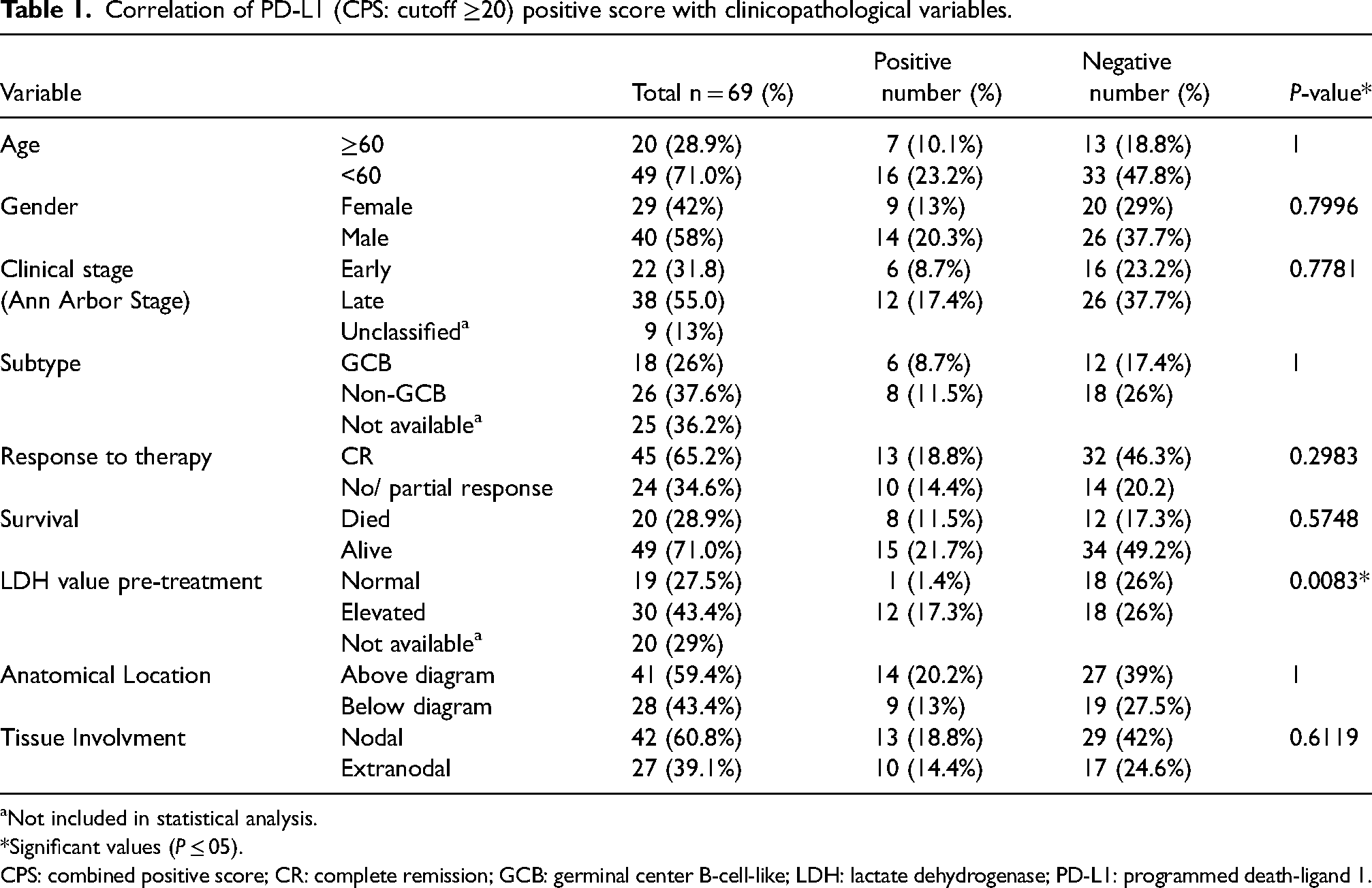
Not included in statistical analysis.
*Significant values (P ≤ 05).
CPS: combined positive score; CR: complete remission; GCB: germinal center B-cell-like; LDH: lactate dehydrogenase; PD-L1: programmed death-ligand 1.
Correlations of PD-L1 (TPS cutoff: ≥1) with clinicopathological variables.
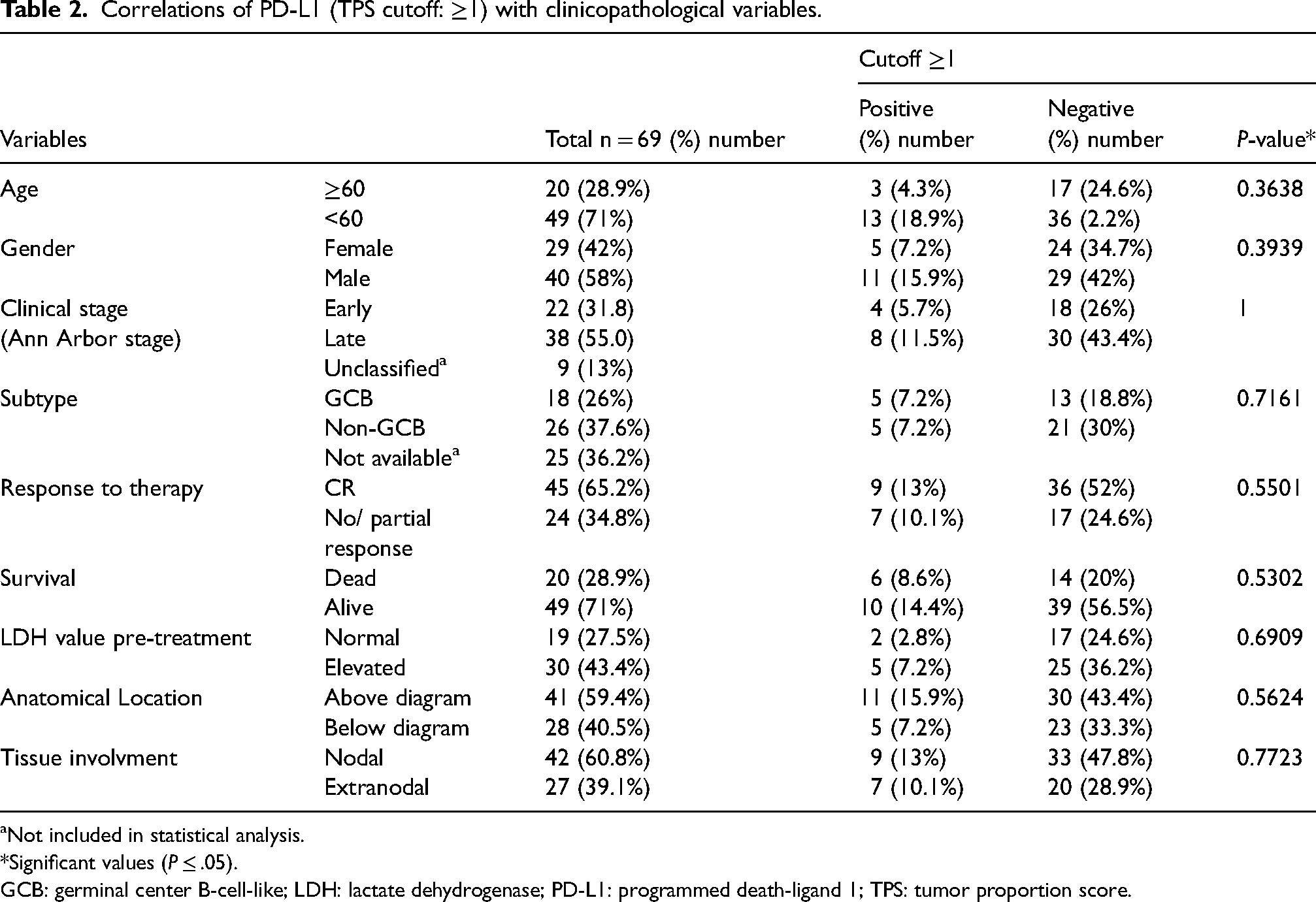
Not included in statistical analysis.
*Significant values (P ≤ .05).
GCB: germinal center B-cell-like; LDH: lactate dehydrogenase; PD-L1: programmed death-ligand 1; TPS: tumor proportion score.
Microscopic examination showed that PD-L1 expression was mostly membranous in the tumor cells, and both cytoplasmic and membranous in nontumorous mononuclear inflammatory cells. The population's expression pattern was diverse as shown in Figure 1. Only 16 (23.2%) specimens showed positive results in tumor cells (TPS score, cutoff ≥1). On the other hand, 23 (33.3%) showed positive results in tumor microenvironment staining immune cells including (lymphocyte and macrophage) plus the tumor cells (CPS) at threshold ≥20. There was no statistically significant difference between PD-L1-positive and -negative groups for most of the studied clinicopathological variables based on both CPS and TPS scores (Tables 1 and 2). However, pre-treatment lactate dehydrogenase (LDH) levels were significantly higher in patients with a CPS ≥20.

Patterns of PD-L1 expression in DLBCL. PD-L1 expression was scored using CPS and TPS on 20X fields as follows: (a) Negative expression. (b) Positive control. (c) TPS = 2%, CPS = 10. (d) TPS = 20%, CPS = 30. (e) TPS = 20%, CPS = 50. (f) TPS = 2%, CPS = 60. (g) TPS = less than 1%, CPS = 50. (h) TPS = 90%, CPS = 100. A positive reaction appears as a brown color at the sites of PD-L1 expression, while a negative reaction appears blue in color (hematoxylin stained).
The range of follow-up of patients was 10 months to 12 years (mean = 29.48 months, median = 26 months). No significant difference between PD-L1-positive and -negative patients for OS and EFS was found based on TPS and CPS (Figure 2). However, a trend in favor of PD-L1 negative patients is evident in both EFS and OS for both TPS and CPS, which is more evident in the TPS where the median EFS is 107 months versus 54 months in favor of PD-L1-negative patients (Figure 2).

Association of PD-L1 expression with survival. (a) OS of PD-L1 CPS-positive and -negative patients at cutoff ≥20. (b) OS of PD-L1 TPS- positive and -negative patients at cutoff ≥1%. (c) EFS of PD-L1 CPS-positive and -negative patients at cutoff ≥20, (d) EFS of PD-L1 TPS-positive and -negative patients at cutoff ≥1%.
Discussion
The present investigation illustrated that PD-L1 expression in Jordanian cases of DLBCL was 23.2% in tumor cells (TPS ≥ 1%) while 33.3% of specimens were positive based on a CPS score of ≥20. In comparison, Xing et al. 16 performed a study on 86 American DLBCL patients, which showed that 16% of cases were considered PD-L1-positive (TPS) at cutoff ≥30%. In another study conducted by Kiyasu et al. 17 on 1253 DLBCL Japanese patients, PD-L1-positive and micoenvironment PD-L1- (mPD-L1) positive cases were 11% and 15.3%, respectively, at 30% threshold. A higher rate of PD-L1 expression was reported in Greece using 27 of newly diagnosed DLBCL cases. Of the malignant cells, 25.9% were positive at cutoff 5, while 48.1% were positive in both malignant and non-malignant cells. 18 In an Egyptian study, a higher PD-L1 expression was present (38%) in DLBCL patients. 19 It should be noted that different studies used different cutoff values for CPS and TPS. 16 In fact, the choice of cutoff value depends on several factors including the type of antibody used, the staining platform, and the IHC staining protocol.20,21 To the best of our knowledge, our study is the only one that has assessed PD-L1 using the CPS algorithm and the specified cutoffs of 1% and 20 for TPS and CPS respectively. Future studies are warranted to standardize cutoffs for a given antibody and staining protocol.
DLBCL is classified into GCB and non-GCB types according to cell origin. 22 In this study, no significant difference in the expression of PD-L1 TPS was found between GCB (7.2% positive PD-L1) and non-GCB types (7.2% positive PD-L1). Comparing the results of this study with a previous study conducted on 83 American patients, 70% of PD-L1 negative cases were GCB type and 30% were non-GCB type with a significant difference between the two subtypes. 16 In another study conducted on Japanese DLBCL untreated patients, 4.7% (22 out of 461) GCB cases were positive for PD-L1 in tumor cells compared to 13.8% (110 out of 792) cases in the non-GCB type. 17
Aggressive NHL, including DLBCL, has been staged according to the Ann Arbor staging system, which was originally designed for HL. 23 In this study, patients were divided according to diagnostic stage into two groups: early stages (I, II) and late stages, which included stage III and stage IV. Only 5.7% of early-stage cases showed positive results of PD-L1 expression (TPS) while 11.5% of those with late stages were positive. The CPS expression on the other hand was presnt in 8.7% and 17.4%, respectively. Compared with a previous study, none of the cases diagnosed at an early stage showed positive PD-L1 expression while 100% showed positive PD-L1 expression at a late stage. 16 In a Japanese study, 71% of patients showed positive PD-L1 expression in a late stage. 17 In a study conducted in China on 204 DLBCL patients, 42% of specimens showed positive PD-L1 results at early stages and 58% showed positive PD-L1 results at a late stage. 24 Similarly in another study, out of 27 cases, 33.3% showed PD-L1 positive results at an early diagnostic stage, and 44.4% were positive in a late stage with no significant difference between early and late stages. 18
Despite the aggressive nature of the DLBCL, the current standard of care, R-CHOP chemotherapy, can cure up to 50%–70% of patients. 25 In this study, 65% of patients had a CR after treatment while 35% showed no or partial response to therapy. No significant difference was found between CR and no or partial response in PD-L1 expression based on both TPS and CPS PD-L1 scores. Only 13% of patients who had a CR had PD-L1 positive expression for TPS while 10.1. % of patients with no or partial response showed positive PD-L1 TPS expression. Looking at this from a different angle 56.3% of the patients with positive TPS had CR in contrast to 67.9% of those with negative TPS. In a previous study, it was found that 71% of positive PD-L1 expression cases had CR or partial response while 75% of negative PD-L1 expression cases had a similar response with no significant difference. 16 In another study, 64% and 72% of PD-L1-positive and PD-L1-negative patients had CR, respectively, with no significant difference. 17 In a third study, 66% of cases with either positive or negative PD- L1 expression had CR. 18 Based on these findings, despite the lack of unified PD-L1 assessment, response to therapy appears independent of PD-L1 expression.
LDH is a component of the International Prognostic Index, which is a good predictor of survival in NHL. 26 LDH value above the normal level indicates a dismal prognosis, while extremely high levels are significantly worse. 27 In this study, an abnormally elevated pre-treatment LDH level was significantly associated with a positive CPS at a cutoff of CPS ≥20 (p = .008) confirming the poor prognostic effect of PD-L1 expression. On the other hand, Hu et al. 24 found no significant differences between elevated and normal LDH value in PD-L1 expression. However, another study found that 80% of positive PD-L1 patients showed an elevated pretreatment LDH value that was not statistically significant. 16 Kiyasu et al. 17 had similar results in which 76% of PD-L1-positive patients had elevated LDH values but with no significant difference between PD-L1-positive and -negative patients.
DLBCL disease generally affects elderly people. Since Jordanian society is a young society, many DLBCL patients in our study were young. Patients over 60 years represented only 29% of the cohort in this study. In fact, age is considered a significant prognostic factor in DLBCL that has a direct impact on treatment options, chemotherapeutic feasibility, and tolerability. 28 In our study, 10.1% of patients were over the age of sixty and had positive results of CPS while only 4.3% of the same patients had PD-L1-positive TPS results with no significant difference between the two age groups. In another study, 76% of patients were above 60 years and had PD-L1-positive expresion. 17 Similar results were obtained by others with no significant differences in PD-L1 expression between the two age groups. 24
In this study, no significant differences were found between the two genders in terms of PD-L1 expression. Previous studies also had similar results with no significant difference.16–18,24
The IPI is the most widely used model for predicting DLBCL survival; it contains five criteria to determine four risk groups: age, serum lactate dehydrogenase level, Eastern Cooperative Oncology Group performance status, stage, and number of extranodal locations. 29 In this study, 32% of patients died of disease during the follow-up period while 68% are still alive. Only 30% of patients who died showed a positive TPS score for PD-L1 expression while 20% of patients who were alive showed positive PD-L1 expression with no significant difference in PD-L1 expression for both TPS and CPS. The results of our study showed a trend towards correlation between PD-L1 expression and OS and EFS based on CPS or TPS. In the study of Xing et al., 16 no significant difference in OS between the mPD-L1-positive and mPD-L1-negative subgroups was found, while a statistically significant difference in progression-free survival (PFS) and OS in the PD-L1-positive group was present. In another study, patients with PD-L1 expression in tumor cells had significantly lower 5-year OS and 5-year PFS rate compared with patients without PD-L1 expression in tumor cells. The 5-year OS and PFS rates were similar in patients with and without PD-L1 expression in the tumor microenvironment. 24 Our finding, in terms of median survival advantage of PD-L1-negative patients, is similar in terms of prognostic implications to the previously cited studies. Some other studies showed that PD-L1 expression had no prognostic implications in DLBCL. 30 Again it seems that methods of assessment might have played a role in these differences.
Conclusion
The current study sheds light on several points: (a) It evaluates the frequency of PD-L1 expression (TPS and CPS) in DLBCL Jordanian Arabs. (b) PD-L1 expression was significantly associated with elevated pre-treatment LDH level but not with age, gender, stage, or DLBCL subtype. (c) The median EFS was higher in PD-L1 negative subjects. Therefore, EFS may have a prognostic value for patients suffering from DLBCL.
Footnotes
Acknowledgments
This work was published with the support of Al-Ahliyya Amman University, Amman, Jordan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.