Abstract
This study aims to explore the expression of matrix metalloproteinase-9 (MMP-9) associated with both diagnostic and prognostic value in ovarian cancer by meta-analysis and bioinformatics analyses. We investigated the prognostic value of MMP-9 expression in ovarian cancer based on The Cancer Genome Atlas. Five databases were used to collect records about MMP-9 expression related to diagnostic and prognostic values in ovarian cancer from inception to June 2022. Using Stata 15.0 software, hazard ratio (HR) and odds ratio (OR) were calculated as the effect index of prognosis. We chose the pooled sensitivity, specificity, and area under the curve (AUC) to judge the diagnostic utility of MMP-9 for ovarian cancer. A total of 23 studies on prognosis, and five studies on diagnosis were entered into the meta-analysis. These suggest that high MMP-9 expression was detrimental to the overall survival of patients with ovarian cancer (HR = 1.34; 95% confidence interval (CI) 1.08∼1.66; P<0.01). High MMP-9 expression increased the risk of tumor stage (OR = 3.66; 95% CI 1.89∼7.07), but was not related to the tumor grade of ovarian cancer (P>0.05). The pooled analysis of serum MMP-9 diagnosing for ovarian cancer gave the pooled sensitivity, specificity, and AUC the values of 0.72 (95% CI 0.61∼0.81), 0.81 (95% CI 0.77∼0.85), and 0.84 (95% CI 0.81∼0.87), respectively. High MMP-9 expression can increase the tumor stage, and a correlation exists between high MMP-9 expression and poor prognosis in patients with ovarian cancer. Also, serum MMP-9 has a good diagnostic value for ovarian cancer.
Introduction
Ovarian cancer (OC) is one of the three most common malignant tumors of the female genital tract. In accordance with the latest research, approximately 300,000 cases of OC occur every year worldwide, with a mortality rate of 0.45%. 1 The histological type of epithelial ovarian cancer is complex and is most common in OC. Due to its hidden incidence and lack of indicators for early diagnosis, 70–80% of patients with OC were not diagnosed until tumor stage III or IV.2,3 Although comprehensive treatments such as tumor cell reduction surgery and chemotherapy were used in clinics, the 5-year survival rate has not been notably improved yet, and the mortality rate still ranks first among gynecological malignant tumors.4–7 Therefore, the search has been ongoing for early OC markers and valuable predictors of invasion, metastasis, and prognosis.8–10
Matrix metalloproteinases (MMPs), a family of peptidases, can degrade the basement membrane and extracellular matrix in the human body, and can destroy the histological barrier of cancer cell invasion. MMPs are widely expressed in the human body. According to its different substrates and homology of fragments, it can be divided into four categories 11 : collagenase, gelatinase, matrix lysin, and membrane matrix metalloproteinase. Under normal physiological conditions, matrix metalloproteinase-9 (MMP-9), belonging to the type of gelatinases, may cut off components of the extracellular matrix and regulate cell adhesion, participating in physiological functions such as embryonic development, wound repair, immune regulation, and tissue model remodeling. 12 While under pathological conditions, MMP-9 is involved in multifaceted syndromes such as chronic inflammation, tumor invasion and metastasis, cardiovascular remodeling, etc.13–15 Previous studies have shown that MMP-9 mainly degrades IV collagen, releasing activated cytokines and growth factors, which play an essential role in regulating cancer cell growth, invasion, metastasis, and angiogenesis. 16 A large number of studies have shown that MMP-9 is highly expressed in breast cancer, colorectal cancer, and other malignant tumors.17,18 Meanwhile, it has been found that the unregulated MMP-9 expression in hematological malignant tumors is related to the invasion, metastasis, and poor prognosis of tumors in different tissues.19,20
At present, there are many studies on the relationship between MMP-9 and the clinicopathological characteristics and prognosis of OC; however, the results are inconsistent and even contradict. Sillanpää et al. 21 reported that the low expression of MMP-9 is related to poor prognosis in OC, but not with tumor stage and grading. Alshenawy et al. 22 and Kamat et al. 23 reported that OC with a high expression of MMP-9 had poor prognoses. Hu et al. 24 showed that no correlation existed between the prognosis of OC and MMP-9 expression. The relationship between the expression of MMP-9 with the prognosis of OC is still unclear. Although Li et al. 25 conducted a comprehensive study on this, there are further high-quality studies. Hence, it was essential to carry out more stringent restrictions on the inclusion of studies, and to conduct a comprehensive analysis including the latest high-quality records. Therefore, in this study, using The Cancer Genome Atlas (TCGA) database, we obtained the hazard ratio (HR) between MMP-9 expression and overall survival (OS) in OC, and used the method of meta-analysis to explore the correlation of MMP-9 expression with clinicopathological features, plus prognostic and diagnostic values in OC to provide medical evidence for clinical targeted therapy of patients with OC.
Materials and methods
The relationship between MMP-9 and prognosis of patients with OC in TCGA database
The TCGA biolinks software package of R 4.0.3 was used to download the gene expression data of OC in TCGA database (https://portal.gdc.cancer.gov/), with the corresponding clinical information captured. The merge function in R software was applied to integrate the MMP-9 expression data with the clinical information of OC. The survival of OC was analyzed according to the MMP-9 expression. OS is defined as the time from the beginning of the operation to death or the last follow-up. With the median as the cut-off value, two groups including MMP-9 high and low expressions were generated. The survival curve was drawn. Meanwhile, HR, as well as 95% confidence interval (CI), was utilized to analyze the MMP-9 expression on the OS of OC.
Document retrieval strategy
This meta-analysis was performed following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. 26 The databases (PubMed, Excerpta Medica (Embase), Web of Science, China National Knowledge Infrastructure (CNKI) and China Wanfang) were accessed for studies on the association between MMP-9 expression and clinicopathological features, plus prognostic and diagnostic values of OC from inception to June 2022. The retrieval strategy was as follows: (“Ovarian cancer” OR “Ovarian neoplasm” OR “Ovarian tumor” OR “Ovarian carcinoma”, OR “Epithelial ovarian cancer”) AND (“MMP-9” OR “Matrix metalloproteinase-9”) AND (“Prognosis” OR “prognostic value” OR “Survival” OR “Pathological features” OR “diagnosis” OR “diagnostic value”). There were no restrictions on language. The literature was searched independently by two researchers and finally cross-checked.
Inclusion and exclusion criteria
Inclusion criteria
The inclusion criteria were: (a) patients pathologically diagnosed with OC; (b) the study population was divided into the MMP-9 high expression and low expression groups for prognosis analysis (diagnostic value of serum MMP-9 in OC); (c) the HR and its 95% CI were reported in the analysis of prognosis, or log-rank X2, survival curve and P-value were presented; in the studies related to clinicopathological features, the number of cases of high and low expression of MMP-9 in OC stage and grade were given; and (d) in the analysis of diagnostic value, the record directly or indirectly provided true positive, false positive, false negative, and true negative.
Exclusion criteria
The exclusion criteria were: (a) studies that did not focus on humans; (b) articles with higher influencing factors and complete data were selected when the same author or the same medical center had duplicate data; and (c) insufficient data.
Data extraction
Two researchers independently scanned the titles and abstracts of the articles to confirm whether they should get the full texts or not. If the article did not meet the inclusion criteria, it would be excluded. Whenever the two researchers encountered differences, they resolved them through discussion. The following data were extracted from the studies: (a) basic information: first author, year of publication, country, number of patients and controls, ages of cases, tumor stage, treatment measures of prognosis, detection method, cut-off value; (b) prognostic value: follow-up time, HR and 95% CI; (c) tumor stage and grade: odds ratio (OR) and 95% CI; and (d) diagnosis: sample source, sensitivity, and specificity.
Quality evaluation
According to the Newcastle–Ottawa scale (NOS), 27 a strict quality evaluation of all included studies of prognosis was conducted. The highest score was 9; articles with 6 scores or more were defined as high-quality studies and were entered into this meta-analysis. When there was a disagreement, an agreement was reached through cross-checking and discussion.
The risk bias of the studies on diagnostic value was assessed using the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2). 28 The total score was 11. When the score was greater than 7, the study was considered high quality and was included in this meta-analysis.
Statistical analysis
The data analysis was performed using Stata 15.0 software. The relationship between MMP-9 expression and prognosis of OC was evaluated by HR and its 95% CI. If the HR of the OS rate was not provided in the original text, Engauge Digitizer software would be utilized to extract the survival curve data, and then the HR was calculated. The relationship between MMP-9 expression and the stage and grade of OC was expressed by OR and 95% CI. We adopted I2 statistics and Q test to assess the heterogeneity. In the analysis of prognosis, if I2 ≥ 50%, or P ≤ 0.05, heterogeneity was significant; thus we chose a random-effects model (REM); otherwise, a fixed-effects model was adopted. For diagnostic tests, the pooled diagnostic indexes from sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), and diagnostic odds ratio (DOR) to area under the curve (AUC) of summary receiver operating characteristic (SROC) were investigated adopting a bivariate mixed-effects model. When the sample size was greater than seven, publication bias was detected. The publication bias was explored by whether the funnel plot was symmetrical or not; the funnel plot was performed by the filling and trimming method. Also, Egger's Test was utilized to assess publication bias. The ethnicity, detection method, and tumor stage were analyzed to find the sources of heterogeneity regarding the prognostic analysis. The survival curve was created by R 4.0.3 software.
Results
MMP-9 associated with OS of OC
A total of 254 patients with OC selected from TCGA database were identified in our prognostic analysis. The average age was 60.0 years old (SD: 11.4). The white population accounted for 85.96%. The tumor stage was between I and IV (two cases did not report a tumor stage), with a median survival time of 31.7 months.
Survival analysis demonstrated no correlation of MMP-9 expression with OS of OC (Figure 1(a)) (HR = 0.86; 95% CI 0.58∼1.26; P > 0.05). Stratified analysis according to tumor stage displayed that, for tumor stage IC-II, III, and IV, the expression difference of MMP-9 was not correlated with OS of OC (all P > 0.05).
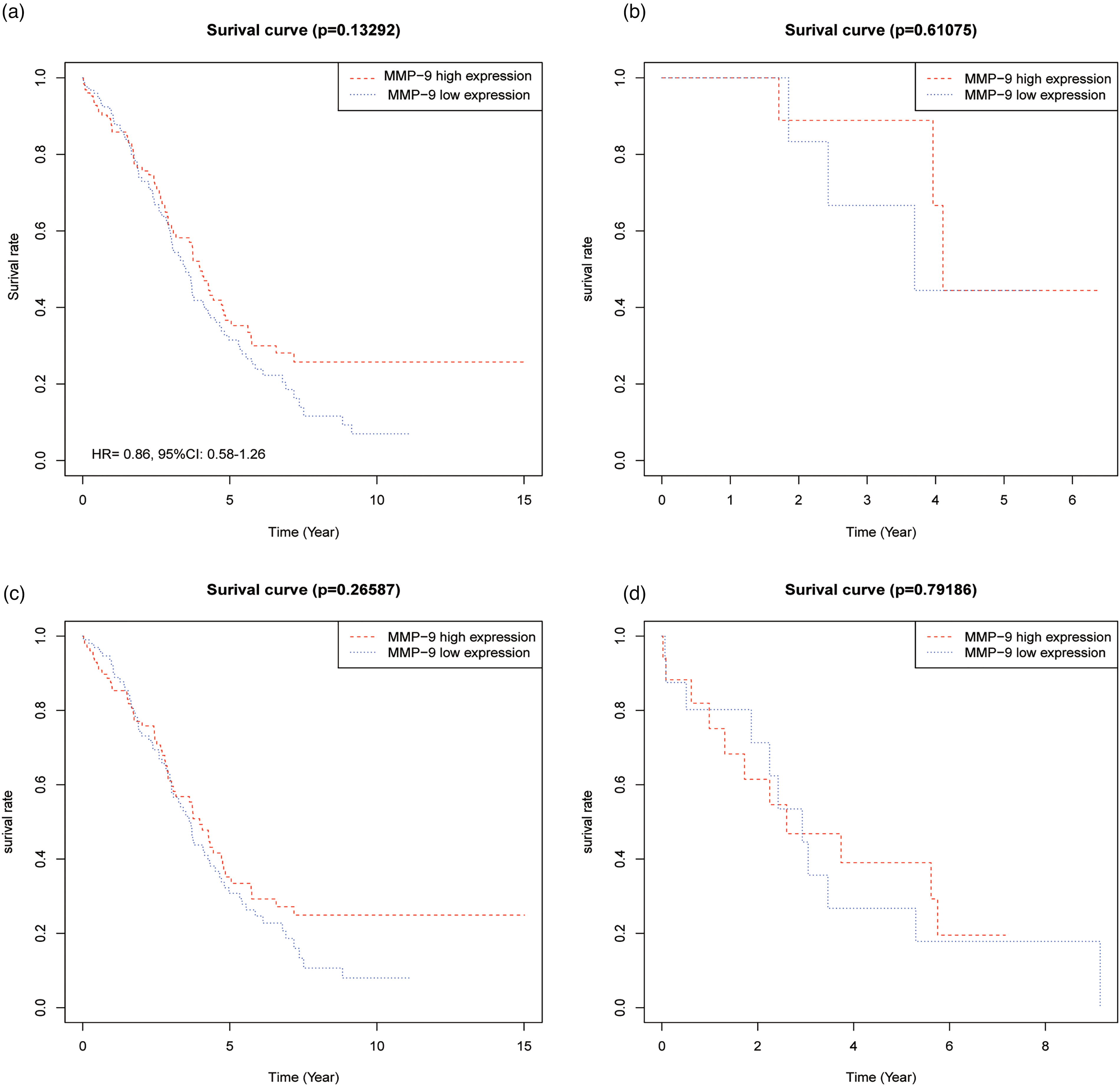
Relationship between MMP-9 expression and overall survival of ovarian cancer in TCGA database. (a) Tumor stage I–IV (n = 254); (b) tumor stage IC–II (n = 18); (c) tumor stage IC–III (n = 201); (d) tumor stage IV (n = 33).
Records of literature retrieval
A total of 22 published papers on prognosis21–24,29–46 and 5 papers on diagnosis47–51 were identified in this meta-analysis (Supplemental Figure 1). For prognosis, the basic information and NOS scores of the identified records are demonstrated in Table 1. The treatment of OC in the prognostic analysis is provided in Supplemental Table 1. Together with the prognostic data obtained in this study, 23 records were eventually entered into our meta-analysis. The total number of patients was 2444. As shown in Table 1, the NOS scores of all the identified records were not less than 6.
Basic characteristics of the included studies about prognosis.
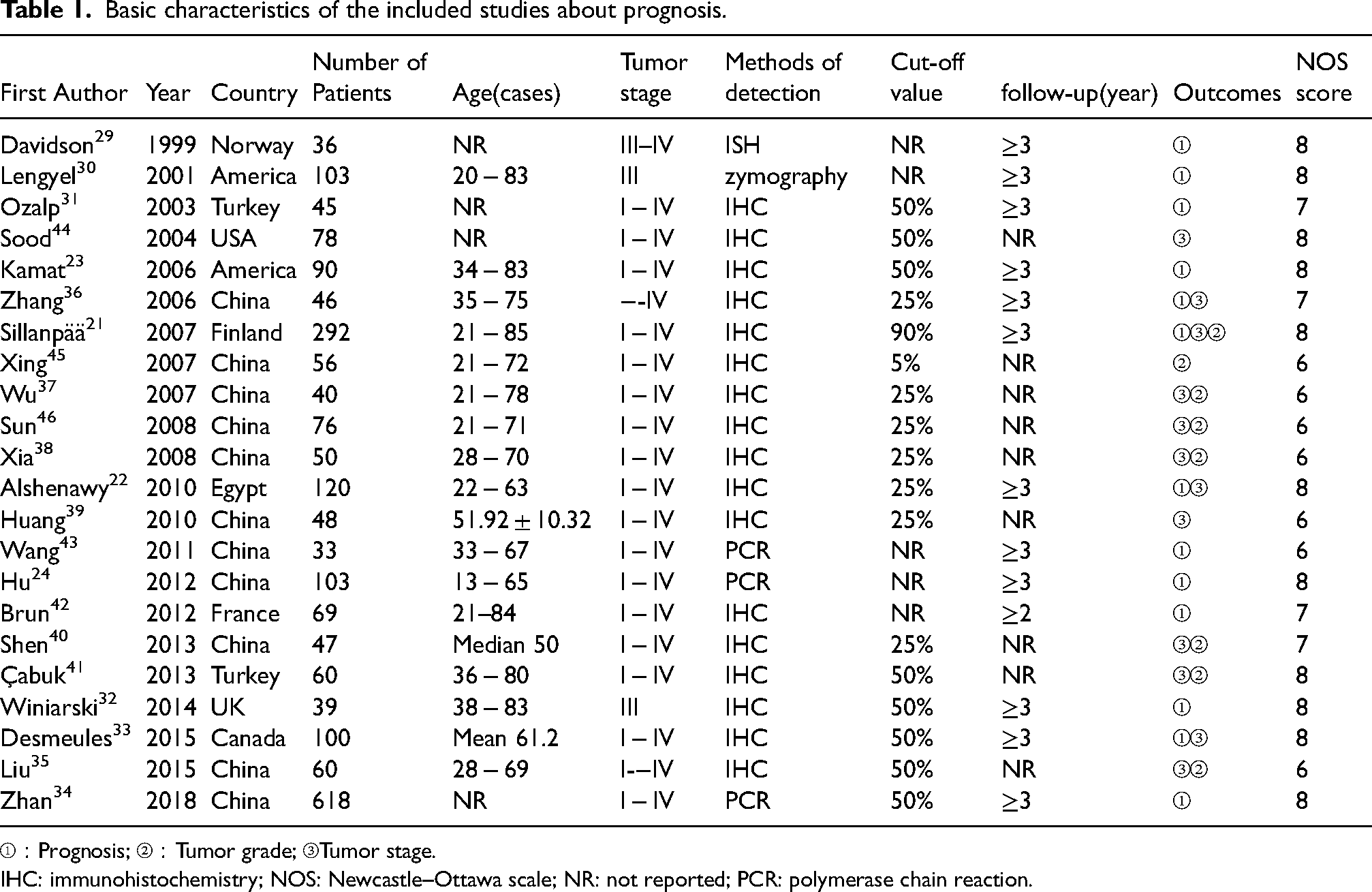
①:Prognosis; ②:Tumor grade; ③Tumor stage.
IHC: immunohistochemistry; NOS: Newcastle–Ottawa scale; NR: not reported; PCR: polymerase chain reaction.
For diagnosis, the basic information and QUADAS-2 scores of the five studies are provided in Table 2. The number of patients with OC was 616, and the number of controls was 733. The NOS scores were all more than 7.
Basic characteristics of five included diagnostic studies.

ELISA: enzyme-linked immunosorbent assay; N: number; Sen: sensitivity; NR: not reported; QUADAS-2: Quality Assessment of Diagnostic Accuracy Studies-2; Spe: specificity.
Results of the pooled analysis
MMP-9 expression for diagnosing OC
Heterogeneity was significant in the meta-analyses of sensitivity, specificity, NLR, and DOR, except for low heterogeneity in PLR analysis (Supplemental Table 2). The bivariate mixed-effects model demonstrated that the pooled sensitivity, specificity, PLR, NLR, and AUC of SROC (Figure 2) were 0.72 (95% CI 0.61, 0.81), 0.81 (95% CI 0.77, 0.85), 3.85 (95% CI 3.25, 4.56), 0.34 (95% CI 0.25, 0.48), and 0.84 (95% CI 0.81, 0.87), respectively. Therefore, serum MMP-9 has a good diagnostic value for OC.
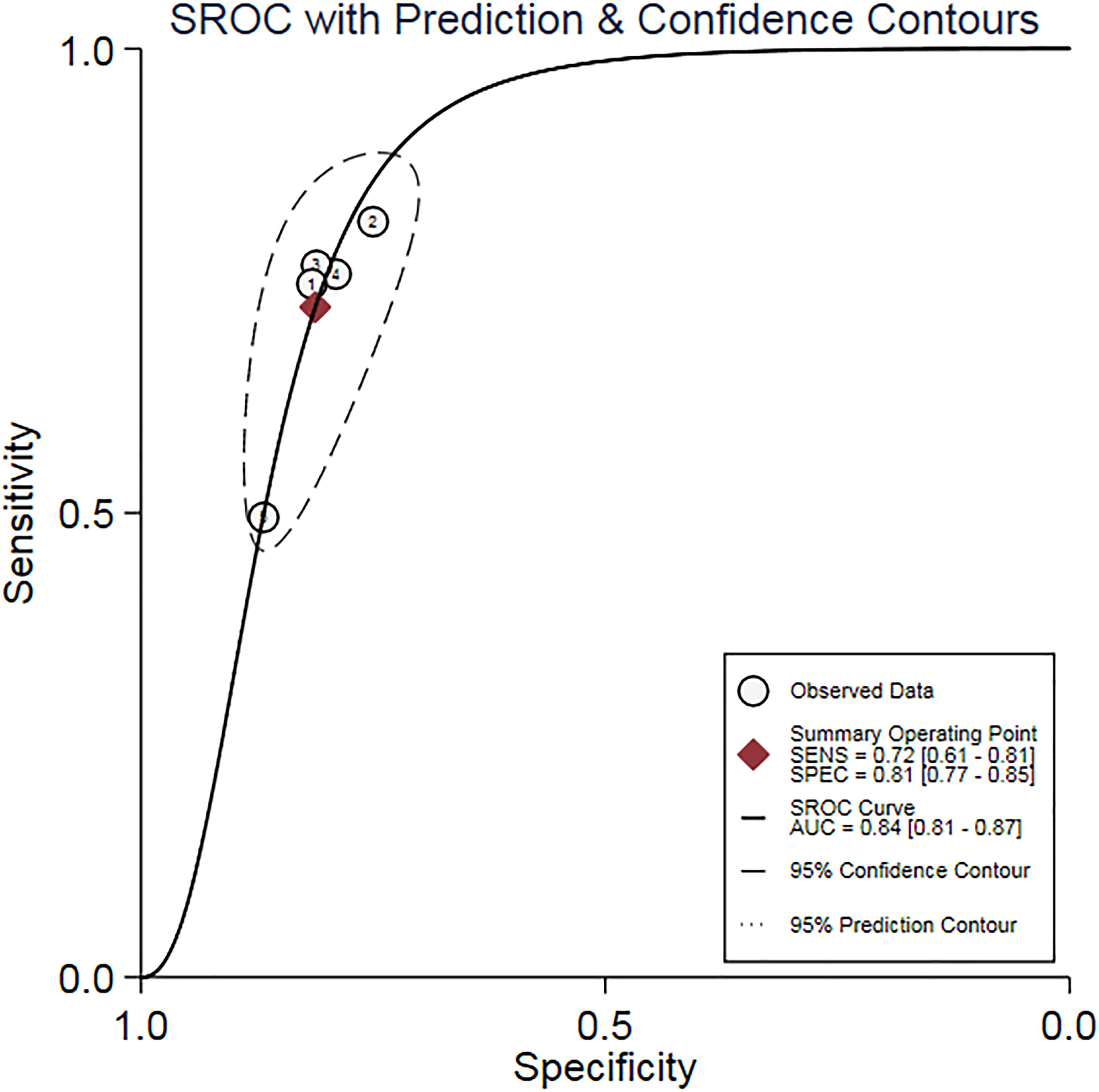
SROC curve of MMP-9 in the diagnosis of ovarian cancer.
MMP-9 expression influencing prognosis of OC
A total of 14 studies, 13 from the published papers,21–24,29–34,36,42,43 and 1 from our study, were entered into the prognostic analysis. As the heterogeneity was high (I2 = 81.6%), a statistic model of REM was applied for data analysis (Supplemental Table 3). The forest plots of prognostic value and clinicopathological features are shown in Figure 3(a) to (c). The results showed that the high expression of MMP-9 significantly increased the risk of death in OC compared to the low MMP-9 expression, with HR = 1.34 (95% CI 1.08∼1.66; P < 0.01) (Figure 3(a)). The subgroup analysis based on ethnicity displayed a noteworthy difference in China (HR = 2.39; 95% CI 1.30∼4.42), but not in the Caucasian population (HR = 1.31; 95% CI 0.99∼1.73). The subgroup analysis according to the detection method also gave a statistical significance (HR = 1.46; 95% CI 1.05∼2.02) in the people tested with immunohistochemistry (IHC). However, the heterogeneity did not decrease remarkably. Funnel plots of prognostic value and clinicopathological features are shown in Supplemental Figure 2(a) to (c). No obvious publication bias occurred because of the symmetrical funnel plot (Supplemental Figure 3(a)), as well as the P-value of Egger's Test of more than 0.05.
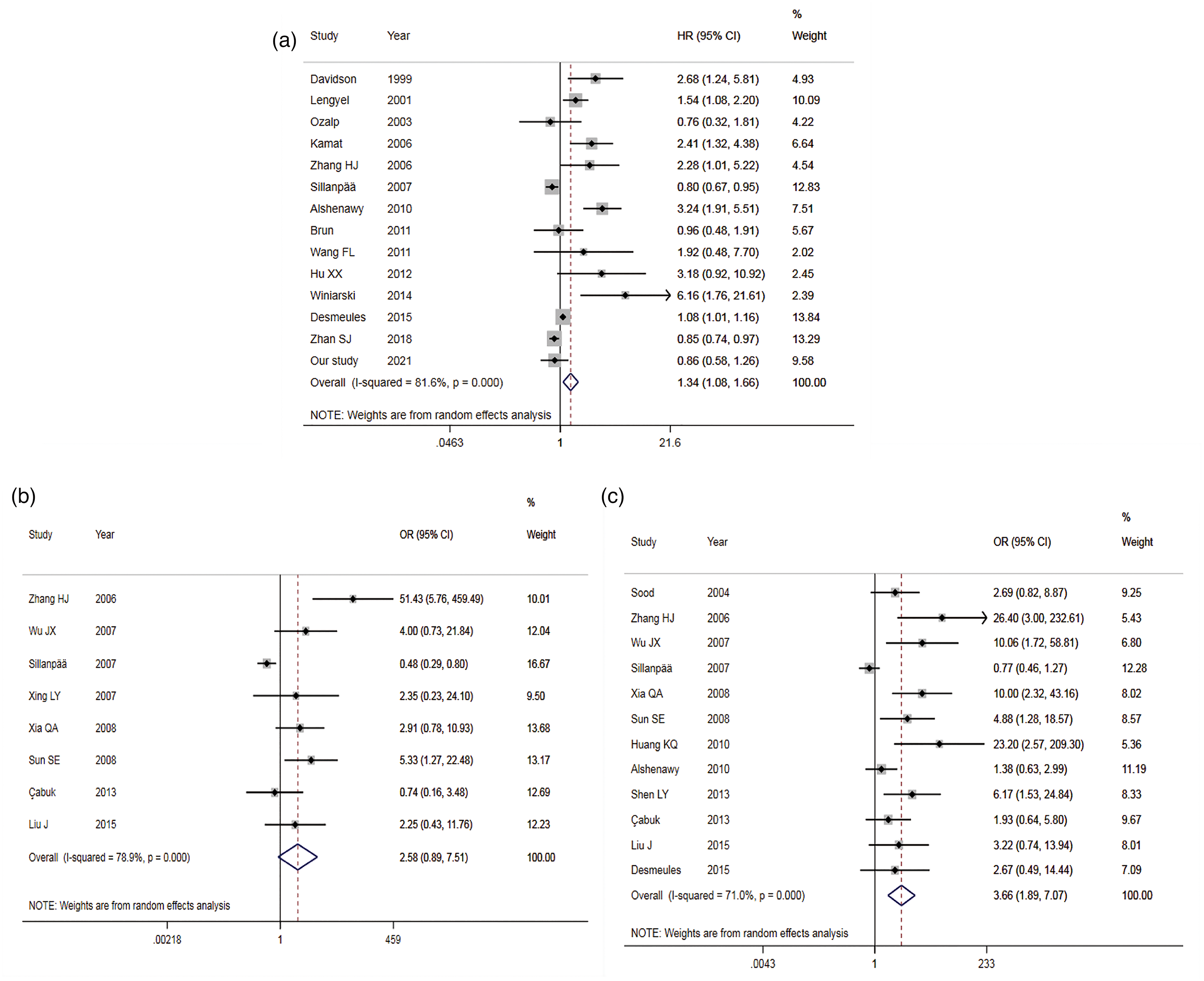
Forest plots of MMP-9 expression and prognosis, pathological features of ovarian cancer ((a) MMP-9 and prognosis; (b) MMP-9 and tumor grade; (c) MMP-9 and tumor staging).
In terms of the effect of tumor stage on prognosis, four studies22,29,30,32 were entered into the meta-analysis of advanced OC (tumor stage III + IV). The results showed that the heterogeneity across the studies decreased significantly (I2 = 54.0%; P > 0.05), with HR = 1.86 (95% CI 1.25∼2.77), which indicated that the prognosis of OC with high MMP-9 expression was not good, especially in patients with advanced OC. However, the Egger regression test gave a P-value less than 0.05 (Supplemental Table 3), suggesting a certain publication bias in the pooled analysis of advanced OC.
MMP-9 expression associated with tumor differentiation in OC
A total of eight studies21,35,37,38,40,41,45,46 were included, covering 156 poorly differentiated patients and 517 moderately and highly differentiated patients. There was statistical heterogeneity across the studies (I2 = 78.9%), so a REM was selected for analysis (Supplemental Table 3). The combined results indicated a statistically significant difference between the two groups, with the middle and high differentiation of OC as the control, OR = 1.10 (95% CI 0.54∼2.24, P = 0.803) (Figure 3(b)). This suggested there was no correlation between the expression of MMP-9 and the OC differentiation degree. The symmetrical funnel plot (Supplemental Figure 2(b)), as well as Egger's Test showing P > 0.05, displayed no significant publication bias.
MMP-9 expression related to tumor stage in OC
A total of 12 articles21,22,33,35–41,44,46 were included, including 588 patients with tumor stage III + IV and 324 patients with the tumor stage I + II. The heterogeneity across the studies (I2 = 71.0%) was significant, so a REM was adopted (Supplemental Table 3). Comparing tumor stage III + IV with I + II (Figure 3(c)), OR was 3.66 (95% CI 1.89∼7.07; P < 0.05). The funnel plot being symmetrical (Supplemental Figure 2(c)), combined with Egger's Test (P < 0.05), indicated partial publication bias. This suggested that the overexpression of MMP-9 could promote tumor staging in patients with OC.
Sensitivity analysis
In order to explore whether the conclusion is robust or not, a sensitivity analysis was conducted. After excluding each study, a meta-analysis was re-conducted to verify if there was any difference between the current and previous conclusions. The results showed that in the meta-analysis of the expression of MMP-9 influencing the prognosis of OC (Supplemental Figure 3(a)), the conclusion changed when one study was omitted. 22 In the meta-analysis of the MMP-9 expression related to tumor differentiation of OC (Supplemental Figure 3(b)), the conclusion changed after the removal of one study. 21 In the meta-analysis of the MMP-9 expression associated with tumor stage of OC (Supplemental Figure 3(c)), after excluding any record, the conclusion did not alter. Therefore, the conclusion of tumor staging is relatively robust, while the conclusions of prognosis and tumor differentiation still need to be carefully explained.
Discussion
MMP-9 played an essential role in the occurrence and development of OC. The lncRNA-microRNA-mRNA axis can modulate the progression of OC. 52 Lin et al. 53 reported an alteration of MMP-9 expression in the process of AOC4P inhibiting epithelial OC metastasis via regulating epithelial–mesenchymal transformation. LncRNA TP73-AS1 could promote the proliferation and metastasis of OC cells by regulating MMP-9. 54 Pomegranate, ellagic acid, and luteolin could inhibit the metastasis of OC via down-regulating MMP-2 and MMP-9. 55 Ghasemi et al. 56 believed that leptin induces the invasion of OC cells through the overexpression of MMP7, MMP-9, and upA. In the past few decades, many candidate biomarkers have been identified to diagnose hyperserous ovarian cancer (HGSOC), but most of them do not have enough sensitivity or specificity for clinical application. MMP-9 may be a new HGSOC biomarker with satisfactory specificity. 57 Suri et al., 58 in a study of OC transgenic mice, found that in ovarian tumors of obese and non-obese mice, celecoxib treatment resulted in reduced proliferation, increased apoptosis, and abated expression of COX-2 and MMP-9 proteins compared with the control group.
The meta-analysis showed that the OC patients with high MMP-9 expression compared with low MMP-9 expression had a poor prognosis (HR = 1.34). As a great deal of heterogeneity was found, there were probably other factors influencing the outcomes. The subgroup analysis indicated a correlation of MMP-9 expression with the prognosis of OC in China, but not in the Caucasian population. However, the heterogeneity in the Caucasian population did not significantly decrease. The IHC subgroup demonstrated a significant difference with the heterogeneity still high. This suggested that the main sources of heterogeneity might not come from ethnicity and detection methods. In the pooled analysis of patients with advanced OC (tumor stage III + IV), the heterogeneity decreased significantly, showing one of the main sources of heterogeneity resulting from tumor staging. Notably, high expression of MMP-9 in advanced OC was detrimental to the OS of OC patients, but the Egger regression test showed certain publication bias, so caution should still be taken regarding this conclusion. The funnel plot and Egger's Test demonstrated no obvious publication bias in the pooled analysis of OS. The sensitivity analysis suggested a certain degree of instability regarding the result that MMP-9 expression affected the prognosis of OC, which should be treated carefully.
The meta-analysis revealed that the overexpression of MMP-9 could aggravate the tumor stage, but no such correlation was found between MMP-9 expression and tumor grade in OC. High MMP-9 expression promoted tumor staging of OC (tumor stage III + IV vs. I + II: OR = 3.66; P < 0.05). Although the analysis in TCGA database showed that MMP-9 expression did not affect the prognosis of OC in different tumor stages, this meta-analysis clearly showed that MMP-9 expression influenced the prognosis of advanced OC. Moreover, our review indicated that MMP-9 expression significantly influenced different tumor stages of OC. Although the sensitivity analysis verified the findings obtained on the tumor stage, the Egger regression showed certain publication bias, so further studies are needed to verify this correlation in the future. No publication bias was found on tumor differentiation, but clinicians should be cautious regarding correlation because of instability in the sensitivity analysis. Li et al. 25 showed that MMP-9 overexpression affected the poor prognosis of OC, promoting tumor stage and grade; the conclusions on prognosis and tumor stage were roughly consistent with our study. However, our review displayed that MMP-9 expression did not affect the tumor grade of OC. The reason why such results occurred might be that the latest high-quality studies were chosen by more stringent inclusion criteria. More importantly, we incorporated the data of MMP-9 expression and prognosis of OC patients in TCGA database into our meta-analysis.
The meta-analysis of serum MMP-9 in OC showed an AUC of 0.84 with moderate diagnostic sensitivity and specificity. Also, the difference in DOR is significant. However, the PLR of 3.85 was not high, and the NLR of 0.34 was not low. A meta-analysis conducted by Liao et al. 59 illustrated that the diagnostic sensitivity, specificity, and the AUC of serum CA-125 for diagnosing OC in China were 0.75, 0.80, and 0.839, respectively. This is comparable to the diagnostic performance of serum MMP-9 for diagnosing OC in our study. Therefore, MMP-9 is expected to be a potential biomarker combined with other diagnostic measures to diagnose OC in clinical practice.
Certainly, this study had several limitations:
The included studies had significant heterogeneity. When doing subgroup analysis, such as studying population race and testing methods, heterogeneity still existed. In the separate analysis of tumor staging, the heterogeneity reduced significantly, but it was only limited to patients with advanced OC. For early disease stage patients we could not perform further research due to the limitations of the data.
The methods used to evaluate the expression of MMP-9 were different. Although most of our studies regarding prognosis used IHC, they did not use the same first antibody, antibody manufacturers, batch numbers, etc.
Publication bias occurred in terms of tumor stage.
Some instability happened in the sensitivity analysis regarding prognosis, as well as tumor differentiation.
As different literature defined different criteria for the high and low expression of MMP-9, this may have led to heterogeneity.
Only five records were enrolled in the diagnostic meta-analysis, all being from China, which might have a certain influence on the extrapolation of the conclusion.
A total of 11 studies on the prognosis of OC by MMP-9 were conducted in China, while there was a lack of studies from other Asian countries.
Despite the above limitations, this is the first time that the meta-analysis method was used to find that serum MMP-9 has a good diagnostic performance for OC. Moreover, this study is also a more in-depth study on the prognostic value of MMP-9 in OC. All these aspects make MMP-9 a hopeful diagnostic and prognostic marker of OC, which has crucial clinical significance.
In summary, our meta-analysis suggested that high expression of MMP-9 could promote tumor staging in OC, is closely related to the poor prognosis of OC, but is not associated with tumor grade. In addition, serum MMP-9 has good diagnostic utility for OC. However, given the limitations of this meta-analysis, more research is warranted to confirm the findings concluded in our study.
Supplemental Material
sj-tif-1-jbm-10.1177_03936155221140421 - Supplemental material for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis
Supplemental material, sj-tif-1-jbm-10.1177_03936155221140421 for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis by Changyu Liu, Ying Shen and Qiyan Tan in The International Journal of Biological Markers
Supplemental Material
sj-tif-2-jbm-10.1177_03936155221140421 - Supplemental material for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis
Supplemental material, sj-tif-2-jbm-10.1177_03936155221140421 for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis by Changyu Liu, Ying Shen and Qiyan Tan in The International Journal of Biological Markers
Supplemental Material
sj-tif-3-jbm-10.1177_03936155221140421 - Supplemental material for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis
Supplemental material, sj-tif-3-jbm-10.1177_03936155221140421 for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis by Changyu Liu, Ying Shen and Qiyan Tan in The International Journal of Biological Markers
Supplemental Material
sj-doc-4-jbm-10.1177_03936155221140421 - Supplemental material for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis
Supplemental material, sj-doc-4-jbm-10.1177_03936155221140421 for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis by Changyu Liu, Ying Shen and Qiyan Tan in The International Journal of Biological Markers
Supplemental Material
sj-doc-5-jbm-10.1177_03936155221140421 - Supplemental material for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis
Supplemental material, sj-doc-5-jbm-10.1177_03936155221140421 for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis by Changyu Liu, Ying Shen and Qiyan Tan in The International Journal of Biological Markers
Supplemental Material
sj-doc-6-jbm-10.1177_03936155221140421 - Supplemental material for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis
Supplemental material, sj-doc-6-jbm-10.1177_03936155221140421 for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis by Changyu Liu, Ying Shen and Qiyan Tan in The International Journal of Biological Markers
Supplemental Material
sj-doc-7-jbm-10.1177_03936155221140421 - Supplemental material for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis
Supplemental material, sj-doc-7-jbm-10.1177_03936155221140421 for Diagnostic and prognostic values of MMP-9 expression in ovarian cancer: A study based on bioinformatics analysis and meta-analysis by Changyu Liu, Ying Shen and Qiyan Tan in The International Journal of Biological Markers
Footnotes
Acknowledgments
Our project is supported by Hainan Province Clinical Medical Center.
Author's contributions
LCY, SY, TQY: Critical revision of the manuscript; LCY, SY, TQY: Substantial contribution to the conception and design of the work, manuscript drafting; LCY, SY, TQY: Acquisition, analysis, and interpretation of the data; LCY, SY, TQY: Revising the manuscript critically, and final approval of the version to be published. All authors have read and approved the final manuscript. LCY and SY equally contributed to this work.
Ethics approval and consent to participate
Ethical approval was not needed because this is a bioinformatics analysis and meta-analysis.
Consent for publication
Not applicable.
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
