Abstract
Background:
The prognostic significance of podoplanin (PDPN) in tumor cells for cancer patients’ survival remains controversial. Therefore, we performed this meta-analysis to clarify the relationship between the podoplanin-positive tumor cells and cancer prognosis.
Method:
Eligible studies were identified by searching the Pubmed and EBSCO online databases up to August 2019. Hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated to evaluate the correlation between podoplanin expression and overall survival (OS) and/or disease-free survival (DFS) and odds ratios (ORs) with 95% CIs severed as the summarized statistics for clinicopathological characteristic.
Results:
A total of 2155 patients from 21 eligible studies were included. The results revealed that high expression of podoplanin was associated with a poor survival rate in cancer patients. Further subgroup analysis stratified by tumor type showed that podoplanin-positive tumor cell infiltration had a negative prognostic effect associated with survival in esophageal cancer and oropharyngeal cancer. In addition, high expression of these cells was significantly associated with N stage, T stage, TNM stage and vascular invasion.
Conclusion:
Our study suggests the over-expression of podoplanin might be a significant prognostic indicator for patients with esophageal and oropharyngeal cancer.
Introduction
Cancer has become a heavy burden to the health care systems and is a leading cause of mortality worldwide. 1 Although diagnostic, treatment methods and supportive care have improved significantly over the past few decades, the cure rate of cancer is still relatively low. This is mainly due to the lack of screening strategies in the early stage, and the high recurrence rate of cancer. Targeted therapy early detection, and early treatment are efficient ways to prevent cancer recurrence. An increasing number of genetic targets are being used to suppress tumors. However, the sensitivity and specificity of most of the tumor markers remain controversial. Therefore, it is necessary to discover new genetic targets to prevent tumorigenesis and the development of cancer.
Podoplanin, a sialomucin-like type 1 transmembrane glycoprotein, also known as GP36, 2 GP40, 3 Aggrus, T1A, 4,5 has been found in a variety of normal tissues, including glomerular podocytes, lymphatic endothelial cells, heart, type Ⅰ alveolar cells, and skeletal muscle. 6 -8 Podoplanin plays a crucial role in cell motility, organ development, lymphangiogenesis, blood-lymph separation, platelet production, and immune response. 9,10 However, its physiological functions are still largely unknown. In pathology conditions, a growing body of evidence indicates that it plays an important role in tumorigenesis. 11 -13 There is evidence that podoplanin correlates with tumor-associated lymphangiogenesis and cancer-associated fibroblasts (CAFs). 14 -16 High expression of podoplanin has been observed in various kinds of tumor cells, such as oral cancer, esophageal cancer, lung cancer, cervical cancer, renal cancer, and cutaneous basal cell carcinoma. 17 -19 However, the function of podoplanin remains controversial in tumor cells. In most kinds of cancers, the podoplanin expression is associated with a higher carcinoma cell migration, leading to cancer metastasis, lower survival rates, and poor tumor stage. 20 -23 On the other hand, some studies suggest that podoplanin expression is a protective factor in lung and lip carcinoma. 24 -26 Thus it is an important subject that needs to be further explored.
Therefore, we performed this meta-analysis to clarify the relationship between the density of podoplanin-positive tumor cells and outcomes such as overall survival (OS), disease-free survival (DFS), and prognosis factors in cancer patients.
Materials and Methods
Searching Strategy and Registration
We searched Pubmed and EBSCO websites to obtain the density of podoplanin in solid tumors and the survival in patients up to August 2019. The search strategy was: (podoplanin OR PDPN OR GP36 OR GP40 OR Aggrus OR T1A) AND (neoplasms [Title/Abstract] OR tumor [Title/Abstract] OR cancer [Title/Abstract] OR carcinoma [Title/Abstract]). The citation lists of the studies we searched were also used to identify other eligible studies. We completed the research registration on the INPLASY website (registration number: INPLASY202160113).
Inclusion and Exclusion Criteria
The studies were included in this meta-analysis when they met the following criteria: (1) Studies were published as original research articles. (2) The density of podoplanin was evaluated in human tissues. (3) They obtained the density of podoplanin+ tumor cells by histological or pathological examinations. (4) They provided hazard ratios (HRs) or odds ratios (ORs) with 95% confidence interval (CI), or Kaplan-Meier curves of high and low podoplanin+ tumor cells density with OS, and/or DFS.
The studies were excluded when they met the following criteria: (1) They were published as letters, case reports, reviews and conference abstracts, and in non-English languages. (2) Studies did not provide enough information to extract the data. (3) Studies detected podoplanin density not in tumor cells, or metastatic tissues. (4) Duplicate publications.
Data Extraction
All the information for this meta-analysis was extracted independently by 2 authors. The data we extracted were as follows: first author’s name, publication year, number of patients, median age, gender, time of follow-up, tumor type, assessment method applied to quantify podoplanin+ tumor cells, and cut-off values of podoplanin expression levels. OS, DFS, and clinic pathological information including Tumor, Lymph Node, Metastasis (TNM) stage, lymphatic invasion were extracted from the text, tables, or Kaplan-Meier curves.
If the included studies showed the HRs and 95%CI in text or tables, we extracted it directly. Otherwise, we calculated the HRs and 95%CI from Kaplan-Meier curves via Engauge Digitizer version 4.1.
Quality Assessment
The quality of included studies was assessed by The Newcastle-Ottawa Scale (NOS). Two independent authors provided the NOS score and achieved consensus. A score ≥ 6 denoted a high quality.
Statistical Analysis
Statistical analysis was performed using STATA-MP software (Version 15.0, College Station, Texas, USA). Statistical heterogeneity was assessed with the Q-test or I 2 statistic were pooled based on the random-effect model in the presence of significant heterogeneity (I 2 >50%). Otherwise, the fixed-effect model was applied. Sensitivity analysis, Begg funnel plot, and Egger test were performed to assess the influence of individual study and the publication bias. All tests were 2-sided, and P < 0.05 was considered statistically significant.
Results
Search Results and Study Characteristics
The detailed flow diagram of the screening process is presented in Figure 1. A total of 8828 records were initially identified after searching on Pubmed and EBSCO. We then excluded duplicates and irrelevant studies after screening the titles and abstracts. Finally, we determined that 21 studies, including 2155 patients with solid tumors, met our inclusion criteria of this meta-analysis. 24 -44 The quality of included studies was assessed by NOS, and the score ranged from 6 to 8. Characteristics of the included studies are shown in Table 1.
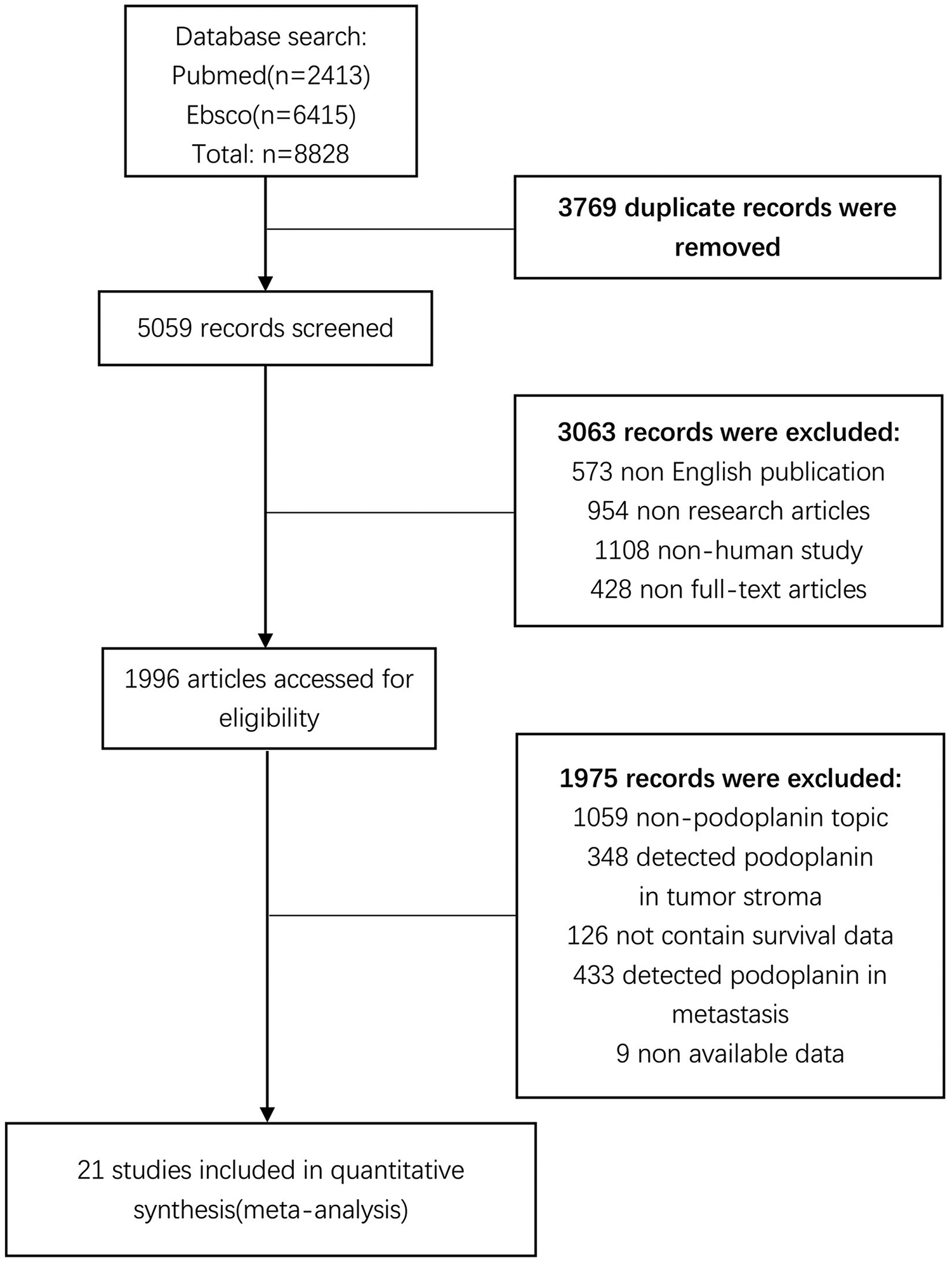
Flow chart of the data selection process. Finally, a total of 2155 patients from 21 eligible studies were included.
Characteristics of the Datasets Included in the Meta-Analysis of Association Between Podoplanin Expression and Solid Tumor.

Abbreviations: ESCC, esophageal squamous cell carcinoma; LSCC, lip squamous cell carcinomas; SCCHN, squamous cell carcinoma of the head and neck; OSCC, oral squamous cell carcinoma; CSCC, cutaneous squamous cell carcinoma; ccRCC, clear cell renal cell carcinoma; IRS, immunoreactive score; IOD score, integrated optical density score; OS, overall survival; DFS, disease-free survival; PFS, progression-free survival; NR, not reported.
Meta-Analysis Results
Overall survival (OS)
We combined extracted data into meta-analyses with STATA. The results have showed that podoplanin+ tumor cells were significantly associated with decreased OS (HR 1.739 95%CI 1.164 to 2.600, P = 0.007) in cancer patients (Figure 2).

Forest plot describing HR of the association between podoplanin+ tumor cells and OS in tumors. Podoplanin+ tumor cells were significantly associated with decreased OS (HR 1.739, 95%CI 1.164 to 2.600, P = 0.007) in cancer patients.
The stratified analyses were implemented based on cancer type. As shown in Figure 3, pooled data indicated that podoplanin+ tumor cells were significantly associated with poor OS in esophageal carcinoma (HR 1.99, 95%CI 1.274 to 3.115, P = 0.003) and oropharyngeal carcinoma (HR 2.097, 95%CI 1.172 to 3.754, P = 0.013) with no heterogeneity detected (I2 = 0%, P = 0.574).

Stratified analyses describing HRs of the association between podoplanin+ tumor cells and OS. Podoplanin+ tumor cells were significantly associated with poor OS in esophageal carcinoma (HR 1.99, 95%CI 1.274 to 3.115, P = 0.003) and oropharyngeal carcinoma (HR 2.097, 95%CI 1.172 to 3.754, P = 0.013).
However, there was no significant association between podoplanin+ tumor cells and OS in lung cancer (HR 0.697, 95%CI 0.324 to 1.499, P = 0.356) and head and neck carcinoma (HR 5.171, 95%CI 0.567 to 47.179, P = 0.145).
Disease-free survival (DFS)
When we combined the DFS data into the meta-analysis, no significant relevance was observed in podoplanin+ tumor cells with DFS (HR 1.496, 95%CI 0.961 to 2.329, P = 0.074) (Figure 4).

Forest plot describing HR of the association between podoplanin+ tumor cells and DFS in tumors. There was no significant relevance in podoplanin+ tumor cell with DFS (HR 1.496, 95%CI 0.961 to 2.329, P = 0.074).
In stratified analysis by tumor type, as shown in Figure 5, podoplanin+ tumor cells were significantly associated with poor DFS in esophageal carcinoma (HR 1.943, 95%CI 1.357 to 2.783, P < 0.001). And, conversely, elevated expression of podoplanin in tumors was not significantly associated with DFS in oropharyngeal carcinoma (HR 0.933, 95%CI 0.132 to 6.610, P = 0.944) and in lung carcinoma (HR 0.803, 95%CI 0.292 to 2.209, P = 0.672).
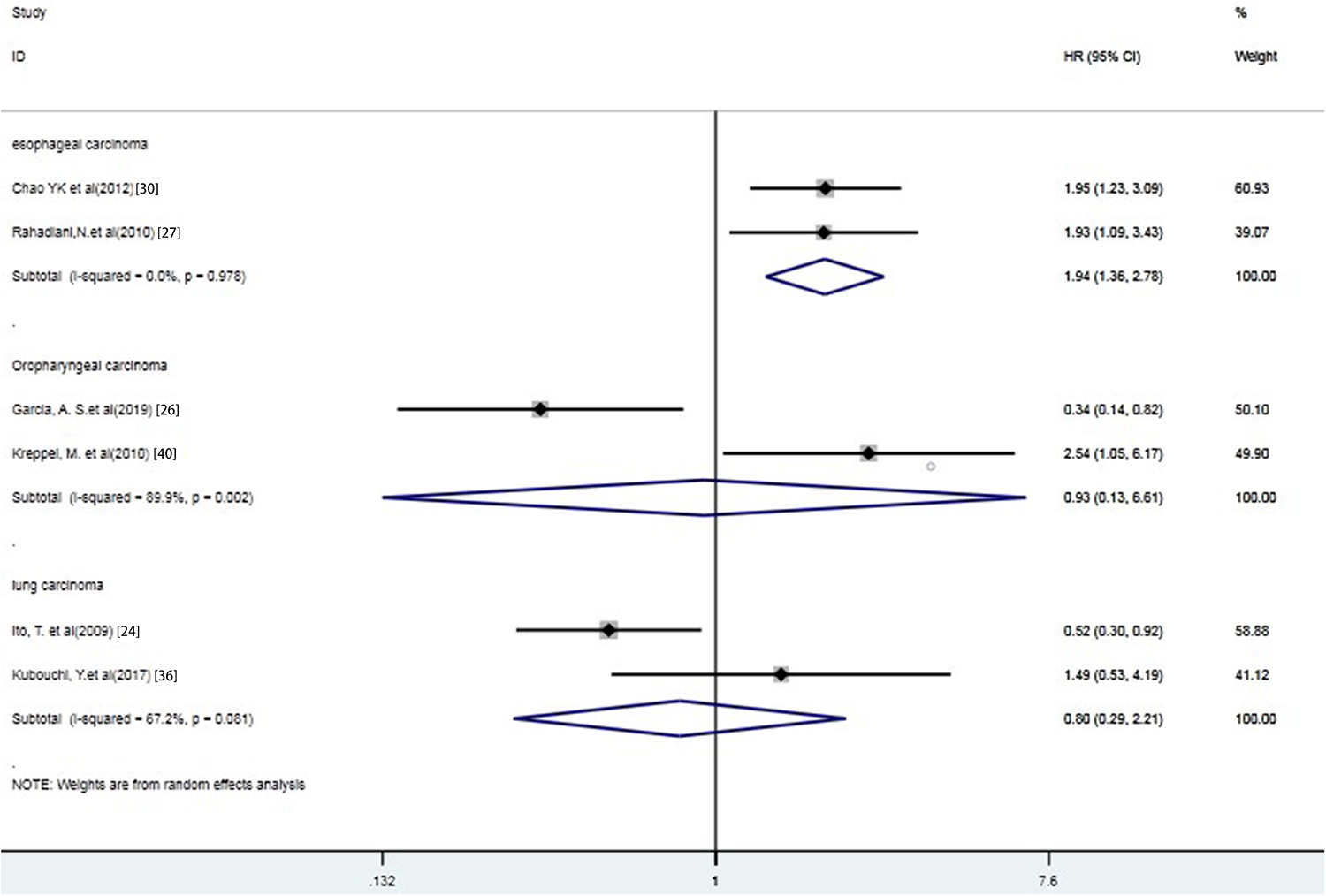
Stratified analyses describing HRs of the association between podoplanin+ tumor cells and DFS. Podoplanin+ tumor cells were significantly associated with poor DFS in esophageal carcinoma (HR 1.943, 95%CI 1.357 to 2.783, P < 0.001).
Association of Podoplanin Expression With Prognosis Factors
Moreover, we carried out meta-analyses with respect to prognostic characteristics. We found that high expression of podoplanin was significantly associated with poor N stage (N1,2 vs N0, OR = 3.652, 95%CI 2.366 to 5.636, P < 0.001), T stage (T3-4 vs T1-2, OR = 2.72, 95%CI 1.992 to 3.714, P < 0.001), TNM stage (III-IV vs I-II, OR = 3.083, 95%CI 2.141 to 4.439, P < 0.001) and vascular invasion (yes vs no, OR = 3.448, 95%CI 1.338 to 8.888, P = 0.010).
However, high podoplanin expression was not significantly related to age (≥60 vs <60, OR = 1.23, 95%CI 0.729 to 2.074, P = 0.438), gender (male vs female, OR = 1.113, 95%CI 0.768 to 1.611, P = 0.961), tumor size (≥3 vs <3cm, OR = 0.847, 95%CI 0.595 to 1.205, P = 0.355), Lymph node status (yes vs no, OR = 1.826, 95%CI 0.881 to 3.784, P = 0.105), lymph vascular invasion (present vs absent, OR = 2.300, 95%CI 0.906 to 5.842, P = 0.080) and tumor differentiation (well vs poor, OR = 1.353, 95%CI 0.673 to 2.721, P = 0.396).
Sensitivity Analysis
Sensitivity analysis indicated that each included study had no influence on the overall HR for OS or DFS (Figure 6).
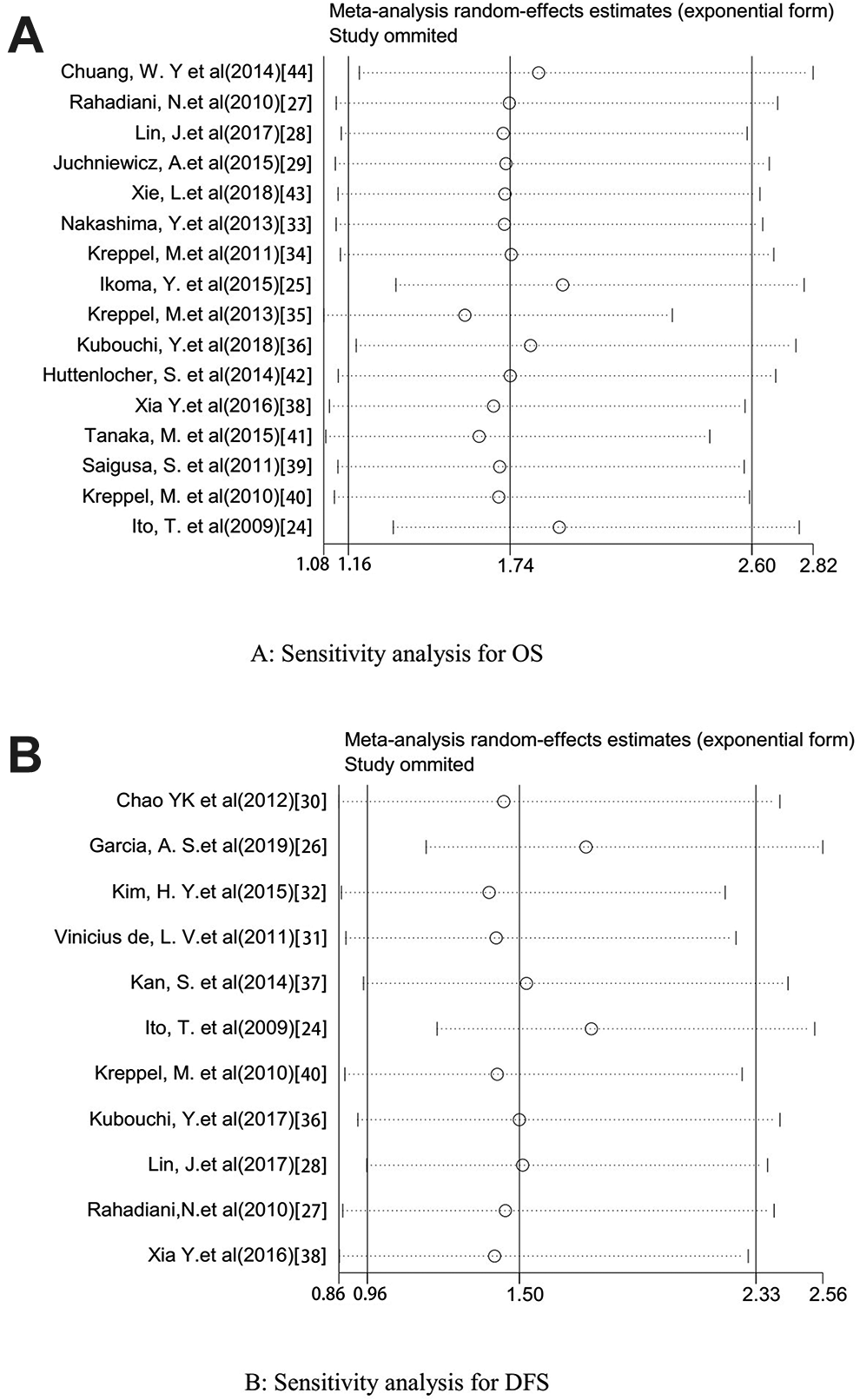
Plots describing the influence of individual studies on the overall HRs for OS (A) and DFS (B) in tumor patients. The results shown that each included study had no influence on the overall HR for OS or DFS.
Publication Bias
The shape of the funnel plot and Egger’s test did not reveal any publication bias existing between podoplanin-positive tumor cells and OS (P = 0.063) or DFS (P = 0.533).
Discussion
In recent years, the correlation between podoplanin expression and tumor prognosis has been explored in several studies, both in tumor stroma and tumor cells. However, the effect of podoplanin in tumor cells remains controversial. To further clarify the prognosis value of podoplanin in tumor cells, we performed this meta-analysis according to the Preferred Reporting Project (PRISMA) guideline. 45
The present study involved 2155 patients from 21 eligible studies. Data from these studies revealed that high expression of podoplanin was associated with a poor survival rate in cancer patients. To determine the prognostic value of podoplanin in different cancers, we conducted stratified analyses by tumor types. The results showed that podoplanin-positive tumor cell infiltration had a negative prognostic effect associated with survival in esophageal cancer and oropharyngeal cancer. However, no relationship between podoplanin expression and survival was observed in other tumor-type patients. The results show a difference between OS and DFS. The reason for this discrepancy may be due to the limited number of eligible studies including DFS data, in which changes of some included studies could influence the pooled data. In addition, high expression of podoplanin in tumor cells was significantly associated with N stage, T stage, TNM stage and vascular invasion. No significant correlations were found with age, gender, tumor size, lymph node status, and tumor differentiation. These results provide meaningful evidence that the expression of podoplanin in tumor cells is a negative prognostic marker in esophageal cancer and oropharyngeal carcinoma. For such patients with high podoplanin expression in esophageal and oropharyngeal tumor cells, close follow-up is necessary. And targeting podoplanin is a new direction for cancer drug development.
Our findings regarding the adverse effects of podoplanin-positive tumor cells are consistent with those of prior studies. 11 The following reasons could possibly be responsible for the findings presented in this study. The extracellular region of podoplanin can bind to the C-type lectin-like receptor-2 (CLEC-2) receptor on platelets to induce platelet aggregation. 46,47 This interaction can promote tumor progression by releasing more cytokines like TGF-β and platelet-derived growth factor (PDGF) from the platelets and facilitate tumor-related thrombosis. 48 Some studies have shown that podoplanin localizes to the invadopodia and the invasive front of the tumor to facilitate tumor cell invasion and promote the progression of various types of cancer. 49 Podoplanin expression has been shown to enhance matrix metalloproteinase (MMP) expression, which degrades the extracellular matrix (ECM) during tumor invasion. 20 In addition, epithelial-mesenchymal transition (EMT) is a phenotypic change that manifests as epithelial cells lose polarity and epithelium markers such as E-cadherin, and obtaining migration factors such as snail and vimentin, a characteristic of fibroblasts. 50 This transition indicates a significant increase in tumor aggressiveness and metastasis. Several studies have revealed that podoplanin plays an important role in the procession of EMT. High expression of podoplanin promotes EMT of tumor cells by releasing TGF-β from platelets. 51 Podoplanin can also promote vimentin expression in tumor cells, suggesting that it is involved in vimentin-associated EMT. 28
In addition, the effect of podoplanin in tumor stroma was also worthy of study. Cremasco et al showed that podoplanin+ CAFs exhibited immunosuppressive potential by releasing nitric oxide and then inhibited the proliferation of activated T cells. 52 Suzuki et al showed that podoplanin+ CAFs exhibited higher expression of TGFβ1 and were associated with CD204+ tumor-associated macrophages infiltration in stage-I lung squamous cell carcinoma. 13 Dai et al showed that the upregulation of podoplanin in CAFs increase tumor migration, proliferation, metastasis and malignancy. 53 Neri et al demonstrated that podoplanin+ CAFs displayed a higher RhoA activity and treatment with a ROCK inhibitor canceled the increased invasion ability of podoplanin+ CAFs and subsequently decreased the invasion and metastasis ability of tumor cells. 54 Hu et al performed a meta-analysis and found that podoplanin+ CAFs infiltration had a negative prognostic effect associated with survival in breast cancer, lung cancer, pancreatic cancer and cholangiocarcinoma. 55 These findings all suggest that podoplanin+ CAFs were associated with the immunosuppressive tumor microenvironment and tumor malignancy. Combined with our study, it is necessary to conduct a comprehensive systematic evaluation on the effect of podoplanin both in tumor and tumor stroma.
In this study, we made a comprehensive effort to present the clinical value of podoplanin expression in various cancer. However, several limitations should be considered when assessing the results from these analyses. First, non-English papers were excluded from this meta-analysis. Moreover, studies with negative results and low-quality may not have been published, which could lead to publication bias. However, there was no publication bias assessed by funnel plot and Egger’s test in included studies. Second, some trials with negative results were missing relevant survival data, despite great efforts to contact the corresponding authors. Finally, podoplanin expression level was assessed via different concentrations of the podoplanin antibody, pathologists, and cut-off values, which may have led to some of the heterogeneity. Despite these limitations, the sensitivity analysis and Egger’s test showed that these meta-analysis results were credible.
Conclusion
In conclusion, our study suggests that the over-expression of podoplanin might be a significant prognostic indicator for patients with esophageal and oropharyngeal cancer, implicating that it is a potential therapeutic target to improve survival in these cancer patients. However, more multi-center clinical investigations with larger sample sizes should be conducted to confirm these findings.
Footnotes
Authors’ Note
Jx Zhang conceived of the study and participated in its design. Xh Wang and Xy Wang extracted data, performed the statistical analysis and drafted the manuscript; Sb Liu, Qq Wang and V. C participated in the statistical analysis; Tt Li, Qq Wang, Jb Wang, Y Chen, Cm Ni and V. C participated in the language editing of the manuscript. All authors read and approved the final manuscript. Data will be shared by the corresponding author upon reasonable request. All studies included in our meta-analysis had been approved by their institutes’ Ethical Committee.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the National Nature Science Foundation of China (NSFC-81870534).
