Abstract
Background
M2 macrophages and regulatory T cells (Tregs) can promote tumors and development by inhibiting the anti-tumor immune response. This study investigated the effect of CD163-positive M2 macrophages and Foxp3-positive Tregs in the progression of colorectal cancer and lymph node metastasis. It also investigated the correlation between M2 macrophages and Tregs.
Methods
Postoperative tissue specimens and clinical data were collected from 197 patients with colorectal cancer who underwent initial surgical treatment in The Second Ward of Colorectal Surgery of the First Affiliated Hospital of Jinzhou Medical University from March 2020 to December 2020. Immunohistochemical methods were used to detect the expression levels of CD163 protein-labeled M2 macrophages and Foxp3 protein-labeled Tregs in colorectal cancer tissues, matched paracancer tissues, and lymph node tissues. The correlation between CD163 and Foxp3 in cancer tissues and lymph node tissues were analyzed, as well as the relationship between clinicopathological characteristics and preoperative tumor markers.
Results
M2 macrophages and Tregs were importantly positively correlated in cancer and lymph node tissues, which significantly increased in cancer and metastatic lymph node tissues. Interestingly, M2 macrophages in non-metastatic lymph nodes also increased significantly in patients with metastatic lymph nodes. In addition, both CD163 and Foxp3 were upregulated with increasing tumor node metastasis stage, depth of infiltration, and lymphatic metastasis; and both were positively correlated with carcinoembryonic antigen.
Conclusion
CD163 may be a good predictor of pre-metastatic status of colorectal cancer lymph nodes. carcinoembryonic antigen affects the distribution of M2 macrophages and Tregs in colorectal cancer. There is a certain correlation between the two types of cells. It is possible that M2 macrophages, together with suppressor Tregs cells, promote an immunosuppressive environment.
Introduction
Colorectal cancer (CRC) is one of the most common malignancies in the world and is the fourth leading cause of death among cancer patients. 1 It is a complex cancerous disease involving multiple cellular pathways starting from the initial process of tumor transformation to metastasis. 2 The tumor microenvironment consists mainly of immune cells and extracellular matrix, with specific immune cells playing an even more critical role in tumor progression. 3
Tumor-associated macrophages (TAM) are one of the major tumor-infiltrating immune cell types and can be differentiated into “classically activated” M1 macrophages and “alternative activated” M2 macrophages. 4 M1 macrophages and M2 macrophages play opposite roles in the tumor microenvironment. Increased M2/M1 ratio in CRC is closely associated with enhanced tumor cell invasion.5,6 In solid tumors, including CRC, M2 macrophages are generally associated with poorer clinical outcomes. 7 CD163 is a member of the cysteine-rich superfamily of scavenger receptors and is restricted to the monocyte/macrophage lineage. It is currently regarded as the most specific marker of M2 macrophages. 8
Regulatory T cells (Tregs) are another type of immune cell. In malignancies, Tregs promote tumor progression by suppressing effective anti-tumor immunity. 9 Tregs mostly have Foxp3 positive expression, and studies have shown that Foxp3 is the molecule that confers the suppressive activity of Tregs This transcription factor plays a key role in the immunosuppressive effects of Tregs and is closely related to the development and the performance of immunomodulatory functions of Tregs.10,11 M2 macrophages and Tregs play an important immunomodulatory role in promoting CRC. M2 macrophages can induce the formation of Tregs. 12 Studies show a correlation between increased numbers of Tregs and M2 macrophages in several types of cancer13–15; however, to the best of our knowledge, this has not been investigated in CRC progression, especially in the lymph nodes.
In this study, we analyzed the correlation and clinical significance of the number of M2 macrophages and Tregs in the paracancer, cancer, and lymph node tissues of 197 CRC patients by immunohistochemical methods, and further explored the mechanism of CRC infiltration and metastasis, thus providing new ideas for CRC diagnosis and treatment.
Materials and methods
Clinical data
Clinical data and postoperative paraffin specimens of patients undergoing radical CRC surgery at the Second Ward of the First Affiliated Hospital of Jinzhou Medical University were collected between March 2020 and December 2020. After inclusion and exclusion, 197 cases were included. The clinical data of the patients are shown in Table 1. The tissue samples of patients with CRC were divided into cancer tissue, matched paracancer tissues, and lymph node tissue. In addition, they were divided into three groups according to the presence or absence of metastatic lymph nodes. Group A included 108 cases in which one normal lymph node was randomly selected from stage I and stage II patients. Group B included 89 cases in which one pathologically positive lymph node was randomly selected from stage III and IV patients. Group C included 89 cases in which one pathologically negative node was randomly selected from stage III and IV patients. Inclusion criteria were: (a) the patient received a diagnosis of primary colorectal carcinoma; (b) they had their first surgical treatment after diagnosis at our hospital; and (c) they consented to participate in this study. The exclusion criteria were: (a) patients older than 86 years; (b) young patients with a confirmed genetic background of the disease (familial adenomatous polyposis and hereditary non-polyposis CRC); (c) the patient had received chemotherapy or radiotherapy after the diagnosis; (d) the patient received targeted immune therapy after the diagnosis; and (e) the patient had intestinal malignant tumors with multiple sites or combined with other systemic malignancies. Clinical staging was according to the American Joint Committee on Cancer (AJCC) staging criteria (8th edition). This study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Jinzhou Medical University (trial number: KYLL202090) All patients signed an informed consent form.
Patient clinical data characteristics.
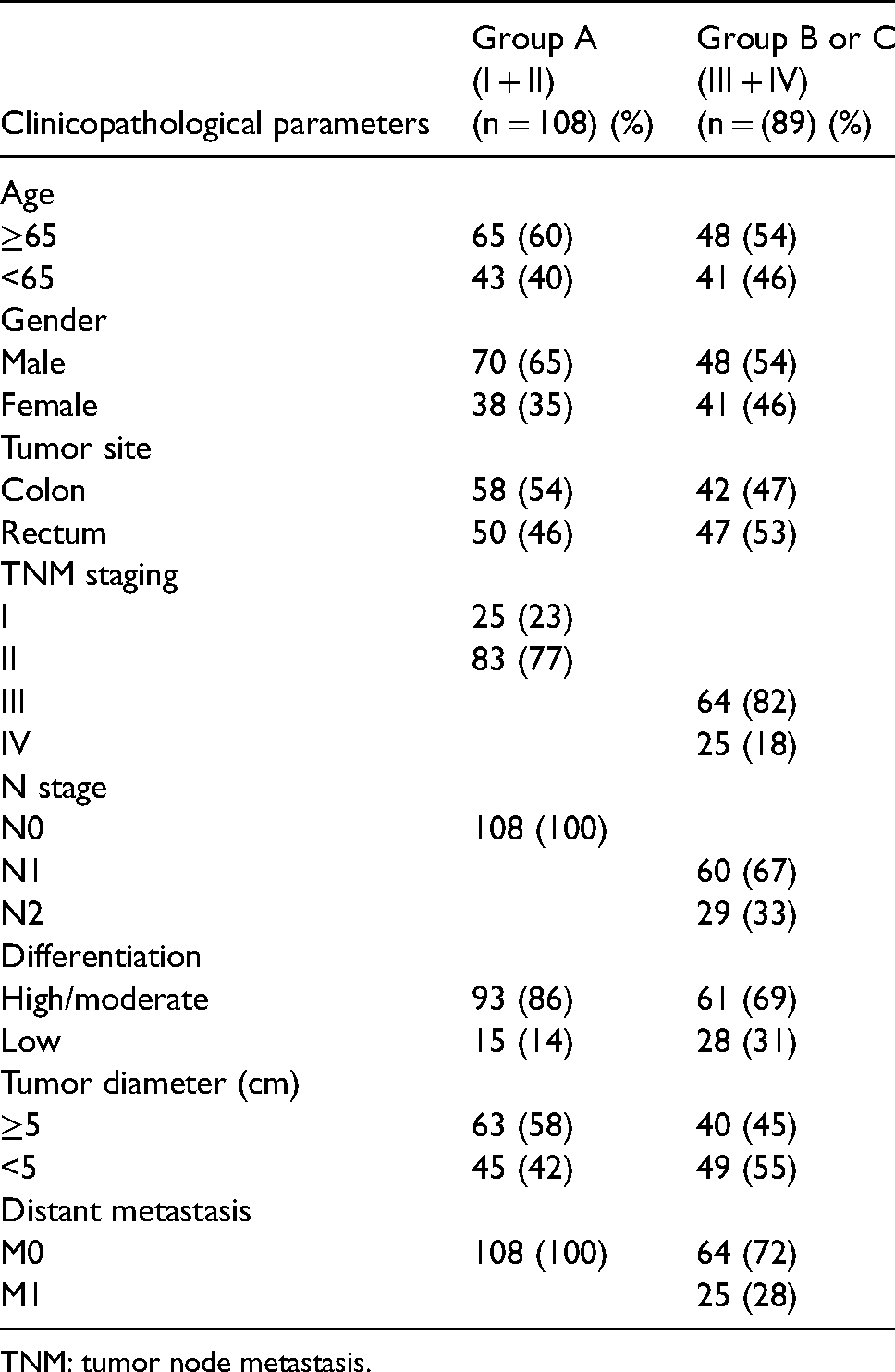
TNM: tumor node metastasis.
Immunohistochemical analysis
Using immunohistochemistry (IHC) streptomyces antibiotic protein-peroxidase linkage (SP) method: All specimens were formalin-fixed, paraffin-embedded, and cut into 4 um thick sections. Sections were placed on microscope slides, dewaxed in xylene, hydrated in a graded ethanol series, and subjected to antigen repair using ethylenediaminetetraacetic acid buffer (pH 8.0) at a sub-boiling temperature for 2.5 min. Tissue sections were incubated with endogenous peroxidase blocker for 10 min at room temperature, then closed with goat serum (KIT-9710; Maixin-Bio, Fuzhou, China) for 30 min. Sections were washed three times with phosphate buffered solution (PBS). Subsequently, anti-CD163 (1:500, ab182422, Abcam, Cambridge, UK), anti-Foxp3 (1:500, ab20034, Abcam) were added and incubated in a refrigerator at 4°C for 16 h. Biotin-labeled IgG polymer and Streptomyces anti-biotin protein-peroxidase (KIT-9710; Maixin-Bio) were added sequentially. Finally, sections were developed using the diaminobenzidine (DAB) kit (DAB-0031, Maixin-Bio) and restained with hematoxylin (G1140, Solarbio, Beijing, China).
Judgment of staining results
CD163 + staining was expressed as the presence of tan or yellow particles in the cell membrane or cytoplasm of M2 macrophages. The results were determined by two pathologists using a semi-quantitative scoring method under a double-blind method of independent film review. (Semi-quantitative score = cell membrane staining intensity score × cell membrane staining ratio score.) The staining intensity was divided into four levels: 0 (negative), 1 (weak), 2 (moderate), 3 (strong). The percentage of positive cells was 0 (0%–10%), 1 (11%–25%), 2 (26%–40%), 3 (41%–75%), and 4 (76%–100%). Semi-quantitative scores > 4 are positive cells, and < 4 are negative cells. Five randomly selected high magnification fields (400×) from stained pathology sections were counted and the mean calculated.
Foxp3 + staining was expressed as the presence of tan or yellow particles in the nucleus of Tregs. Five high magnification fields (400×) were randomly selected from the stained pathological sections. Image J was applied to determine the positive cells, which were then confirmed by two pathologists under double-blind methodological conditions. For pathology sections stained with the Anti-Foxp3 monoclonal antibody, the mean number of positive cells was calculated for the five high magnification fields, and this mean was the final score for this section.
Statistical analysis
The SPSS version 26.0 software program and GraphPad Prism 8 were used to analyze the data. Measurement data were expressed as means ± SD. The independent sample t-test was used to compare differences in M2 macrophages and Tregs in cancer tissues with different clinicopathological parameters, as well as differences within the M2 macrophage and Tregs groups. Spearman correlation analysis and linear regression analysis were used to analyze the correlation between the two variables. A value of P < 0.05 was considered significant.
Results
Immunohistochemical results of CD163 and Foxp3 in different tissues
CD163 is mainly characterized by the presence of yellow or brownish-yellow medium granules on the membrane of M2 macrophages and weak staining in the cell matrix (Figure 1(a) and (b)). Foxp3 is mainly characterized by the presence of brownish-yellow granules on the nuclei of Treg cells, distributed throughout the tissues of CRC patients (Figure 1(c) and (d)). Cancer tissue stained significantly more than paracancerous tissue (P < 0.05) (Figure 1(a) and (c)), and metastatic lymph node tissue stained significantly more than non-metastatic tissue (P < 0.05), (Figure 1(b) and (d)).
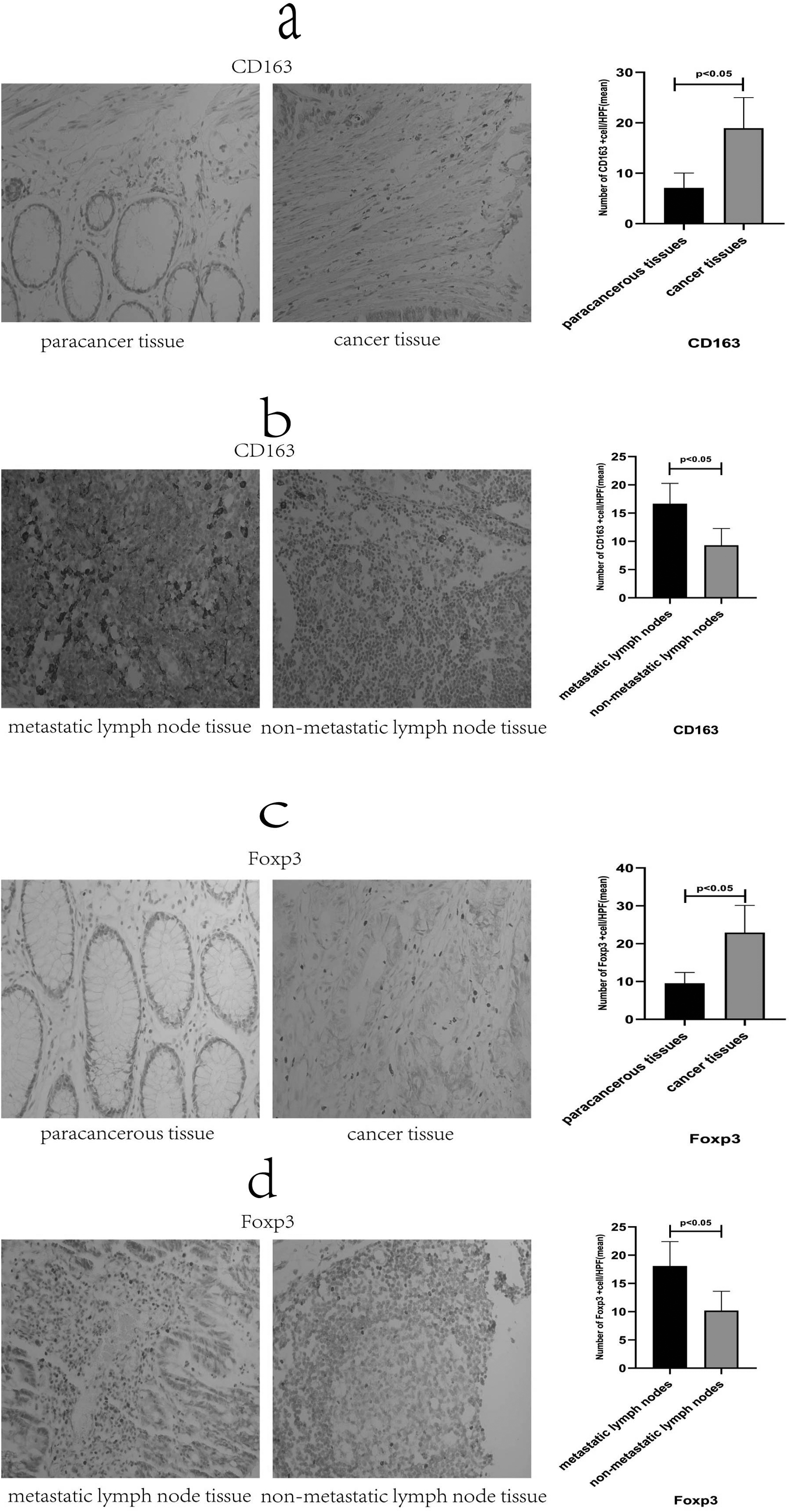
Immunohistochemical results of CD163 and Foxp3 in different tissues. As shown in the figure: representative pictures of different tissues by IHC experiments (400× field of view). (a) CD163 staining and number in paraneoplastic and cancerous tissues; (b) CD163 staining and number in metastatic and non-metastatic lymph nodes; (c) Foxp3 staining and number in paraneoplastic and cancerous tissues; and (d) Foxp3 staining and number in metastatic and non-metastatic lymph nodes.
The relationship between the expression of M2 macrophages and Tregs and the clinical characteristics of tumors
To clarify the infiltration characteristics of M2 macrophages and Tregs cells within CRC, we performed a statistical analysis of the mean number of M2 macrophages and Tregs in 197 CRC tissues with clinicopathological parameters (Table 2). The mean number of M2 macrophages and Tregs increased with increasing tumor stage and depth of infiltration (P < 0.05). In addition, in the case of lymph node metastasis, vascular tumor thrombus, nerve invasion, and distant metastasis, the more significant the increase in the mean number of M2 macrophages and Tregs expression (P < 0.001). However, in patient age, gender, tumor diameter, tumor site, and degree of differentiation, there were no significant differences between the two types of cells (P > 0.05).
Clinicopathological characteristics of 197 patients with nodal cancer.
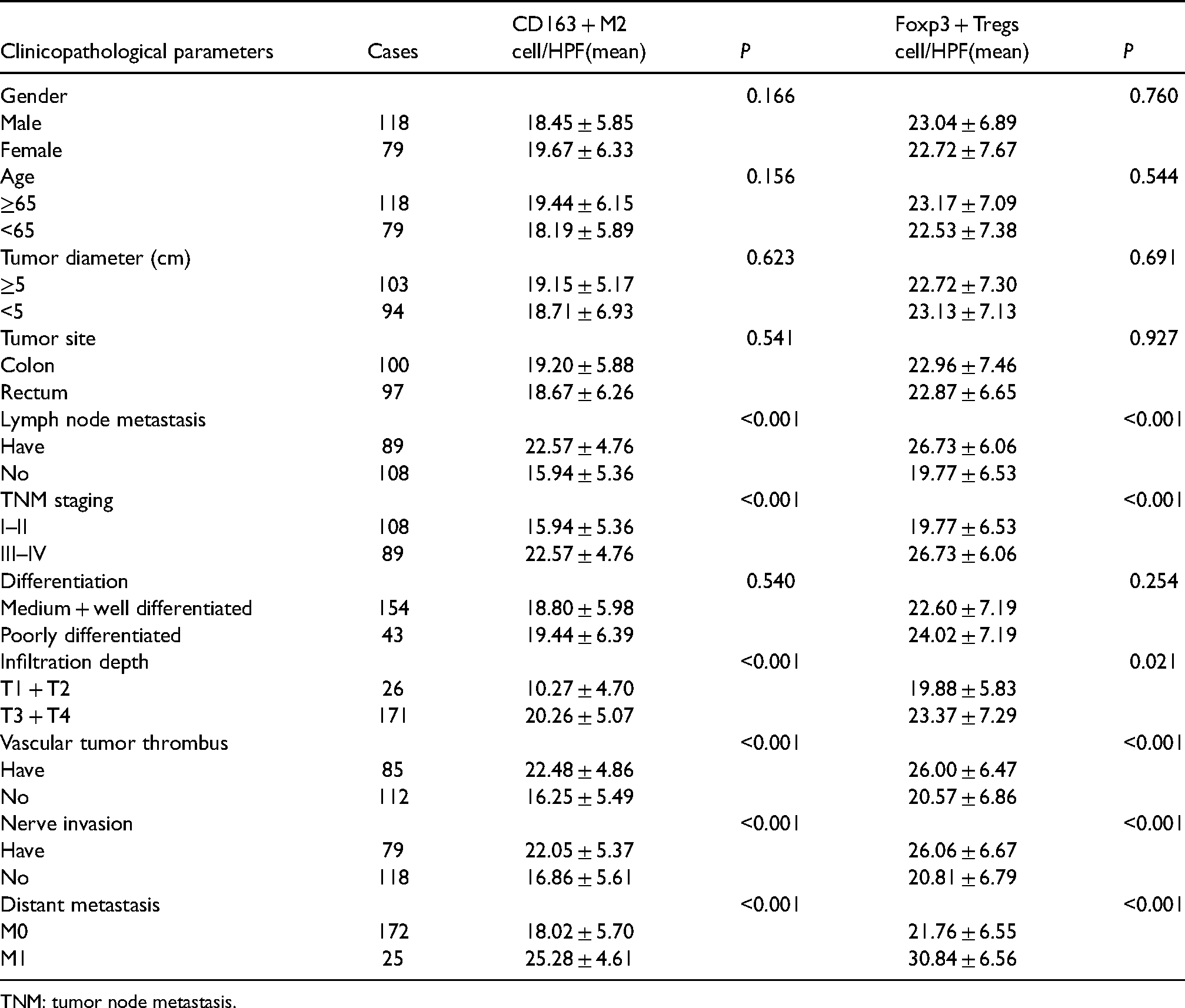
TNM: tumor node metastasis.
Different expressions of M2 macrophages and Tregs in different tissues
We observed (Figure 2(a) and (b)) the mean number of M2 macrophages and Tregs in cancer tissues (18.94 ± 6.06) and (22.91 ± 7.20), significantly higher than the paracancerous tissues (7.08 ± 2.97) and (9.55 ± 2.82), (P < 0.05); In cancer tissues, the mean number of M2 macrophages at stage III + IV (22.57 ± 4.76) was significantly higher than at stage I + II (15.94 ± 5.36) (Figure 2(c)), (P < 0.05). Meanwhile, the mean number of Tregs at stage III + IV (26.73 ± 6.07) was also significantly higher than at stage I + II (19.77 ± 6.53) (Figure 2(d)), (P < 0.05). To further investigate the progression of M2 macrophages and Tregs in CRC patients. We examined their lymph nodes and counted them. It was found that the mean number of M2 macrophages and Tregs in group A (8.82 ± 3.28) and (9.99 ± 3.70) were significantly lower than those in group B (16.65 ± 3.60) and (18.07 ± 4.33), (P < 0.05) (Figure 2(e) and (f)). However, it is not clear whether the number of M2 macrophages and Tregs in the lymph nodes of patients with stage I + II is different from the number of M2 macrophages and Tregs in the non-metastatic lymph nodes of patients with stage III + IV. Therefore, we counted the M2 macrophages in group A and C, and found (Figure 2(g)) that the mean number of M2 macrophages in group A (8.82 ± 3.28) was lower than that in group C (9.89 ± 2.47), (P < 0.05). (Figure 2(h)) There was no difference in the mean number of Tregs (9.99 ± 3.70) and (10.42 ± 3.06) between group A and group C, respectively, (P > 0.05).
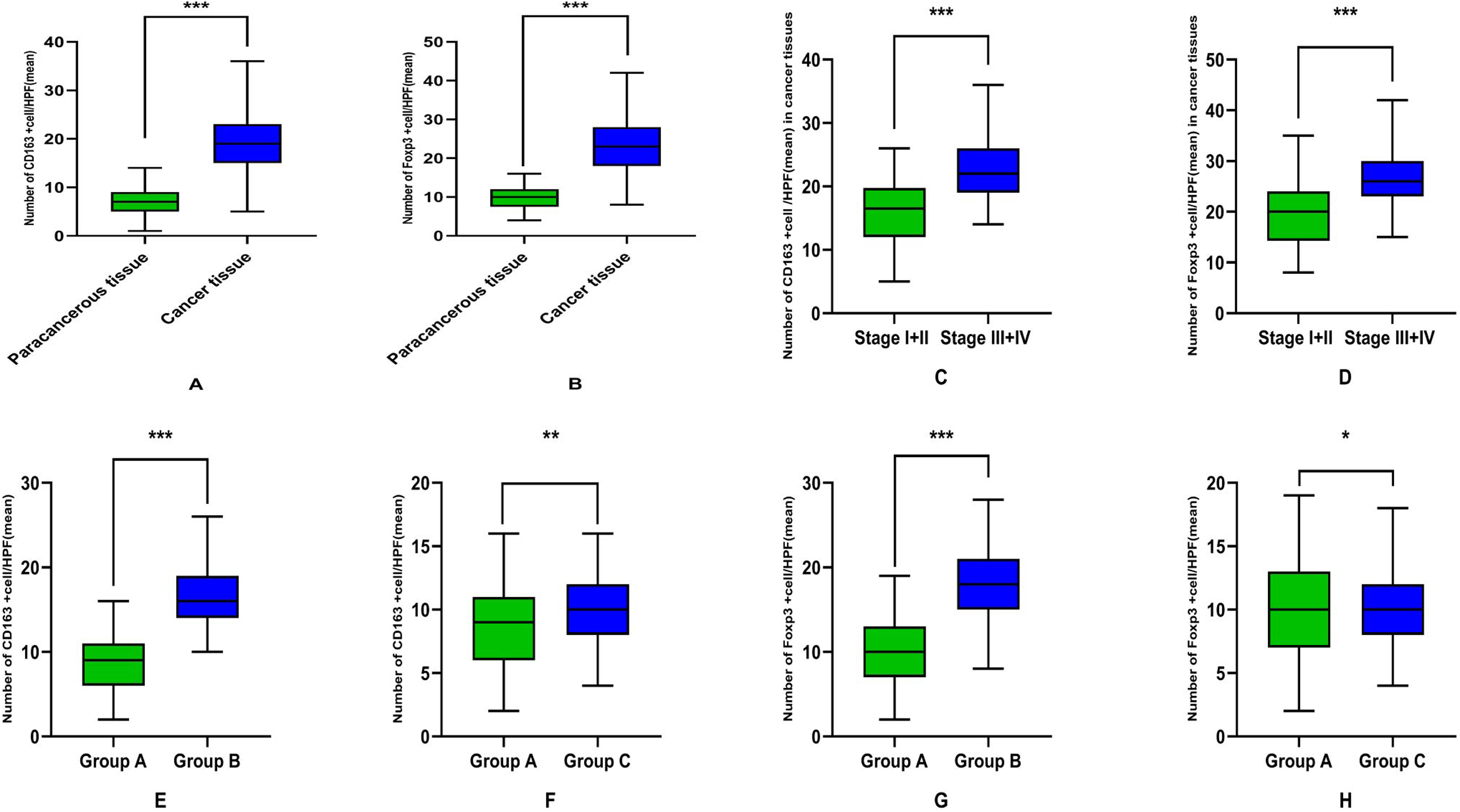
Different expressions of M2 macrophages and Tregs in different tissues. (a) and (b) M2 macrophages and Tregs were significantly higher in cancer tissue than in paracancerous tissue, with statistically significant differences (both ***P < 0.001). In cancer tissues (c) and (d), M2 macrophages and Tregs in stage I + II were significantly lower than those in stage III + IV, and the difference was statistically significant (both ***P < 0.001). (e) and (f) M2 macrophages and Tregs in group A were significantly lower than those in group B, and the differences were statistically significant (both***P < 0.001). In the non-metastatic lymph node tissue (g), the mean number of M2 macrophages in group A was lower than that in group C, and the difference was statistically significant (**P < 0.05). (h) There was no significant difference in Tregs between group A and group C, and the difference was not statistically significant (*P > 0.05).
Correlation analysis of M2 macrophages and Tregs in cancer tissues and lymph nodes
By comparative analysis (Figure 3) we found that M2 macrophages and Tregs were abundantly distributed in cancerous tissues and lymph nodes. In order to verify whether there is a certain correlation between the two in the development of CRC and lymph node metastasis, we used Spearman correlation analysis to find that: Figure 3(a) and (b) M2 macrophages and Tregs are positively correlated in CRC and lymph nodes (r = 0.269, P < 0.001; r = 0.541, P < 0.001).
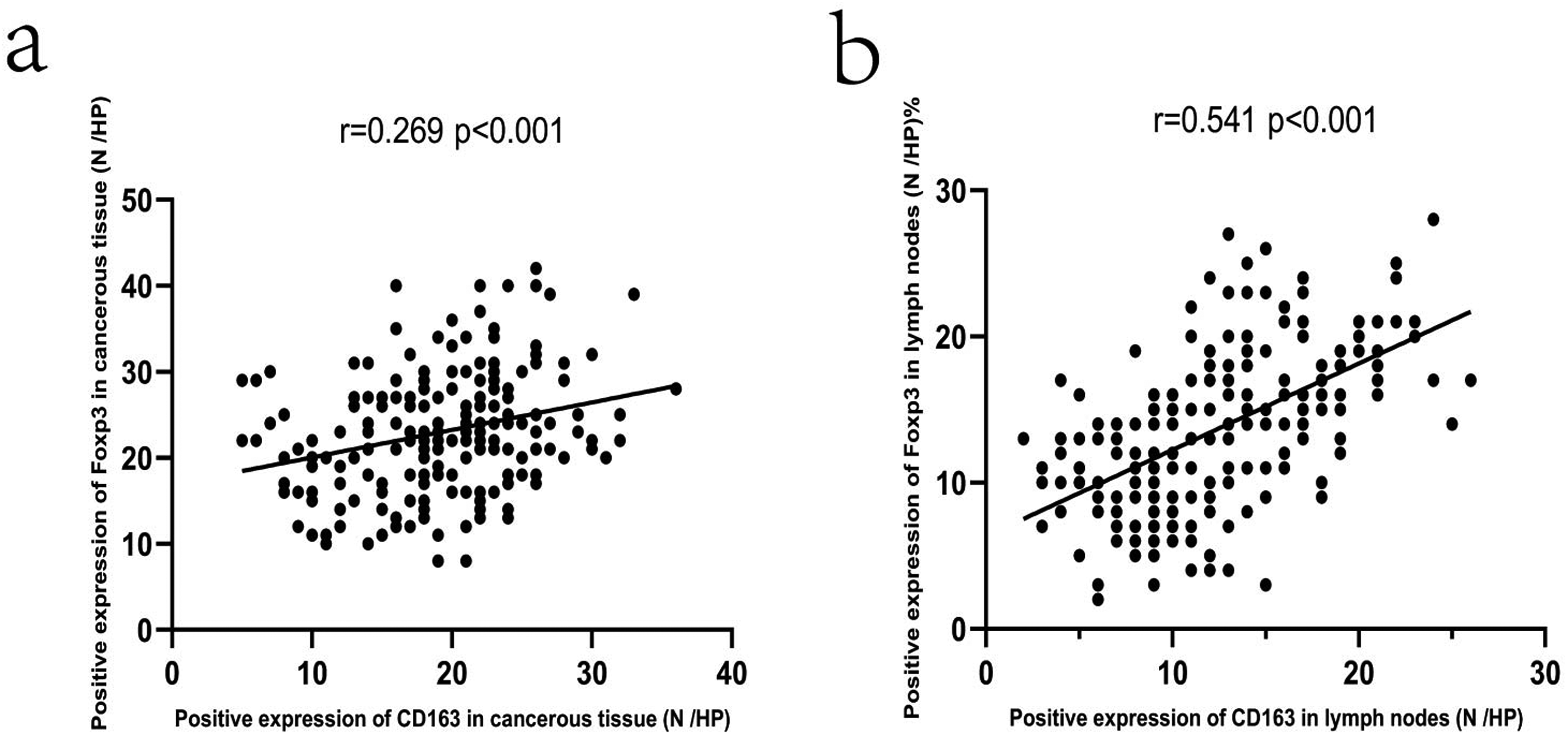
(a) Correlation of M2 macrophages and Tregs in cancer tissue (r = 0.269, P < 0.001); (b) correlation of M2 macrophages and Tregs in lymph node tissue (r = 0.541, P < 0.001).
Correlation analysis of the number of M2 macrophages and Tregs with tumor markers in CRC
According to Supplementary Table 3, Spearman correlation analysis was used to determine the relationship between the mean number of M2 macrophages and Tregs and preoperative carcinoembryonic antigen (CEA), CA199, and CA724 levels. It was found that the mean number of M2 macrophages and Tregs were found to be significantly and positively correlated with preoperative CEA levels (both P < 0.001).
Discussion
Immune cells play a crucial role in the development of tumors in the tumor microenvironment. 16 In this study, IHC staining was performed on paracancerous tissue, cancerous tissue, and its surrounding lymph node tissue of 197 patients with CRC. The number of M2 macrophages and Tregs was found to be significantly higher in cancer tissue than in paracancerous tissue. In lymph node tissue, increased numbers of M2 macrophages and Tregs infiltrated into the metastatic lymph nodes compared to the non-metastatic lymph nodes. Also, M2 macrophages infiltrated into non-metastatic lymph nodes in patients who also exhibited metastatic lymph nodes. These observations indicate that M2 macrophages were closely associated with lymph node metastasis in colorectal carcinoma. Notably, our study found a positive correlation between M2 macrophages and Tregs in cancer tissues and lymph node tissues.
M2 macrophages are a kind of activated macrophages induced by Th2 cytokines interleukin (IL)-4, IL-10, and IL-13. It promotes tumor angiogenesis and leads to tumor progression by suppressing T-cell-mediated anti-tumor immune responses.17,18 The rapid growth of tumor tissue leads to increased metabolism and uneven vascularization around it. The lack of blood vessels in the tumor area causes tumor hypoxia. Hypoxia causes tumor cells to release factors such as HIF-1α and HIF-2α, which can increase the expression of micro RNAs (miRNAs) through the PI3K/AKT/mTOR pathway, and promote the polarization of M2 cells.19–21 Therefore, M2 macrophages are significantly increased in tumor tissue. Lian et al. 22 also noted that colon cancer cells secrete epithelial growth factor (EGF), which can bind to the epithelial growth factor receptor (EGFR) on monocytes, activate the smad-PI3K-Akt-MTOR pathway, and promote the differentiation of monocytes into M2 macrophages. We also found higher mean numbers of M2 macrophages in tumor tissue in the presence of more severe vascular cancer thrombosis, nerve invasion, and distant metastases. The increase in the mean number of M2 macrophages was closely associated with the tumor node metastasis (TNM) stage, lymph node metastasis and depth of infiltration of the tumor. It indicates that M2 macrophages are involved in the formation of the immunosuppressive microenvironment in cancer tissues, which may be one of the important factors leading to the occurrence of CRC. The EGF secreted by M2 macrophages activates the EGFR on tumor cells, which in turn up-regulates the vascular (V)EGF/VEGFR signals in surrounding tumor cells to support tumor cell proliferation and migration. 23 Our study found that in lymph node tissue, the mean number of M2 macrophages increased significantly not only in metastatic lymph node tissue, but also in non-metastatic lymph nodes in patients with metastatic lymph nodes. It suggests that in patients with metastatic lymph nodes, there is more M2 macrophage infiltration in the part where tumor metastasis has not occurred. It shows that the microenvironment of the lymph node has changed before metastasis, and M2 macrophages are involved in it. This is the same result as our previous study. 24 Therefore, we speculate that M2 macrophages play an important role in the process of lymph node metastasis in CRC, which indicates a poor prognosis. Tacconi et al. 25 stated that VEGF-C promotes proliferation and expansion of lymphatic vessels, thereby increasing the pathway of tumor metastasis to lymph nodes. Therefore, M2 macrophages may change the tumor microenvironment and promote the lymph node metastasis of CRC.24,26
Tregs are a subset of T lymphocytes with immunosuppressive effects, which can induce tumor immune escape by inhibiting effector T cells, and induce tumor growth and immune tolerance. 27 To investigate the changes in Tregs during CRC progression, we examined the changes in Tregs in paracancerous, cancerous, and lymph node tissues. CRC was divided into early (stage I + II) and late (stage III + IV) stages for analysis. It was found that Tregs in cancer tissues were significantly higher than those in paracancerous tissues, and significantly higher in late stages than in early stages, which is consistent with previous studies. 28 We analyzed tumor cells and microenvironmental macrophages to produce chemokine CCL22, which can bind to the CCR4 receptor highly expressed on the surface of FOXP3 + Tregs. This helped to recruit Tregs to tumor tissues.29,30 In addition, we analyzed the correlation between Tregs and clinicopathological characteristics. It was found that Tregs in CRC are closely related to TNM staging, lymph node metastasis, and distant metastasis. TNM stage and lymph node metastasis are important indicators of the prognosis and survival time of tumor patients. Thus, our results suggest that Tregs infiltration is associated with CRC metastasis and poor prognosis.
Our research found that M2 macrophages and Tregs are closely related in tumor tissue and lymph node tissue. M2 macrophages can produce immunosuppressive cytokines and chemokines. These cytokines and chemokines are involved in recruiting lymphocytes and stimulating their development into Tregs.29,31 In addition, Tregs produce high levels of IL-10, IL-32, and TGF-β, which further inhibit the anti-tumor inflammatory response and stimulate M2 macrophages to increase the production of cytokines and chemokines, thereby being able to recruit additional Tregs. 32 Studies have pointed out that M2 macrophages and Tregs have synergistic effects in promoting proliferation, tumor angiogenesis and metastasis in ovarian cancer. 33 Sun et al. 15 studied 65 patients with laryngeal squamous cell carcinoma (LSCC) and found that Tregs and M2 macrophages were positively correlated with each other in LSCC, and demonstrated that the two formed a positive feedback loop. In addition, we also preliminarily confirmed that M2 macrophages may induce the production of regulatory T cells by activating the TGF-β/Smad signaling pathway. 34 Therefore, we speculate that M2 macrophages in CRC may have the ability to induce the formation of Tregs and increase the expression of Tregs in tumor tissues. The interaction between the two may change the tumor microenvironment and promote the development of CRC and lymph node metastasis.
The specific mechanism of action of M2 macrophages and Tregs in CRC, especially the molecular mechanism of interaction in local lymph nodes, will be the next focus of our group's research. In addition, studies have pointed out that CD163 can be used as a potential prognostic biomarker for CRC patients, and Foxp3 can directly affect the prognosis of CRC patients.35,36 To further verify the impact of both on the monitoring and prognosis of CRC patients, our study analyzed the preoperative CEA, CA199, and CA724 levels in correlation with the number of M2 macrophages and Tregs. It was found that the level of CEA was significantly positively correlated with the mean number of M2 macrophages and Tregs. This is consistent with our previous findings. 24 We know that CEA plays an important role in monitoring the recurrence and metastasis of CRC. In addition, studies have shown that IL-10 stimulated by CEA can activate the JAK1/STAT3 signaling pathway to differentiate macrophages into M2 macrophages.37,38 At the same time, TGF-β combined with CEA in malignant tumors is a key regulatory signal of Tregs and one of the main functional cytokines secreted by Tregs.39,40 It is evident that CEA has an important effect on the distribution of M2 macrophages and Tregs in colon cancer.
In summary, the mechanisms leading to CRC progression and lymph node metastasis are very complex. The presence of M2 macrophages and Tregs in CRC patients is an important indicator of CRC progression and lymph node metastasis. Our results provide some insights for M2 macrophages and Tregs in the progression of CRC and its lymph node metastasis. CD163 may be a good predictor of pre-metastatic status of CRC lymph nodes. CEA affects the distribution of M2 macrophages and Tregs in CRC. There is a certain correlation between the two. The relationship between M2 macrophages and Tregs may affect the progression of CRC and lymph node metastasis. This could be a potential future target for immunotherapy and could provide important clinical value for CRC diagnosis, treatment, and prognosis.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155221132572 - Supplemental material for A study on the correlation between M2 macrophages and regulatory T cells in the progression of colorectal cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155221132572 for A study on the correlation between M2 macrophages and regulatory T cells in the progression of colorectal cancer by Yanlei Chen, Yu Gao, Xueqian Ma, Yanping Wang, Jinhao Liu, Chunyu Yang, Yue Wang, Cuifen Bao, Xiaoyu Song, Yang Feng, Yan Sun and Shifeng Qiao in The International Journal of Biological Markers
Footnotes
Acknowledgments
The authors would like to thank the laboratory of Jinzhou Medical University for providing technical guidance
Authors’ contributions
YLC, YG, XQM, CYY, and YW performed the experiments. YLC, YPW, JHL, and CFB contributed to the statistical analysis and drafted the manuscript. YLC, XYS, YF, and YS participated in collecting the data, and contributed to the statistical analysis and manuscript writing. SFQ conceived the current study and helped revise the manuscript. All authors read and approved the final manuscript.
Ethics approval
The study was conducted in accordance with the Ethics Committee of the First Affiliated Hospital of Jinzhou Medical University (KYLL202090).
Availability of data and materials
The analyzed data sets generated during the study are available from the corresponding author on reasonable request. Inquiries for data access may be sent to the following e-mail address: shifengqiao2020@163.com.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
