Abstract
Background
The purpose of this study was to construct a nomogram based on classical parameters and immunohistochemical markers to predict the recurrence of early low-risk endometrial cancer patients.
Methods
A total of 998 patients with early low-risk endometrial cancer who underwent primary surgical treatment were enrolled (668 in the training cohort, 330 in the validation cohort). Prognostic factors identified by univariate and multivariate analysis in the training cohort were used to construct the nomogram. Prediction performance of the nomogram was evaluated using the calibration curve, concordance index (C-index), and the time-dependent receiver operating characteristic curve. The cumulative incidence curve was used to describe the prognosis of patients in high-risk and low-risk groups divided by the optimal risk threshold of the model.
Results
In the training cohort, grade (P = 0.040), estrogen receptor (P < 0.001), progesterone receptor (P = 0.001), P53 (P = 0.004), and Ki67 (P = 0.002) were identified as independent risk factors of recurrence of early low-risk endometrial cancer, and were used to establish the nomogram. The calibration curve showed that the fitting degree of the model was good. The C-indexes of training and validation cohorts were 0.862 and 0. 827, respectively. Based on the optimal risk threshold of the nomogram, patients were split into a high-risk group and a low-risk group. The cumulative incidence curves showed that the prognosis of the high-risk group was far worse than that of the low-risk group (P < 0.001).
Conclusion
This nomogram, with a combination of classical parameters and immunohistochemical markers, can effectively predict recurrence in early low-risk endometrial cancer patients.
Keywords
Introduction
Endometrial cancer (EC) is the sixth most common diagnosed carcinoma in women. 1 Due to the symptoms of abnormal or post-menopausal bleeding, early detection can be attained by almost 75% of patients with EC. 2 In contemporary series, early low-risk EC is defined as the age of patients <60 years old, stage IA, endometrioid histology, histologic grade 1 or 2, without lymphovascular space invasion (LVSI).3,4 The largest group among EC patients is those with early low-risk EC; it has been reported that patients with stage IA accounted 77.1% of the total population.5,6 Currently, surgery is the main therapy for early low-risk EC in clinical practice, and follow-up after surgery is usually recommended for patients according to most guidelines.3,4 However, a proportion of early low-risk EC may still progress, resulting in the development of recurrence and even death from relapse. The 5-year survival rate of early-stage EC has been reported to be as high as 92%, but 5–10% of patients still relapse. 7 Ignatov et al. 5 showed a recurrence rate of 6% in low-risk EC patients. Although the proportion is low, these patients are still a huge group due to the large base of early low-risk patients. Thus, there is a critical need to explore the risk factors related to the recurrence of early low-risk EC in order to screen patients with the potential high risk of recurrence from early low-risk patients, and to take the necessary preventive measures and prognostic management for them.
A nomogram is a commonly used tool to estimate prognosis by integrating important pathological and clinical characteristics in oncology.8–10 Currently, most models for the prediction of EC recurrence have been developed based on classical clinicopathological parameters. 11 For instance, Ouldamer et al. 12 have developed a nomogram to predict the prognosis of stage I∼III EC patients based on age, histologic type, stage, grade, surgical nodal staging, and LVSI. However, classical parameters appear to be insufficient to accurately predict EC prognosis; nomograms for predicting the recurrence of EC could be refined by adding other useful predictors.
Presently, immunohistochemical markers have been applied in clinical practice for a variety of cancers. In luminal breast cancer, the threshold of Ki67 was reported as an important prognostic indicator. 13 Furthermore, the scoring system including estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2) and Ki67 developed by Abubakar et al. 14 can be used as an aid to the prognosis management of luminal breast cancer. In EC, ER, PR, P53, and Ki67 have been frequently used as prognostic parameters. 15 For example, a retrospective study identified the combined ratio of ([P53 + Ki67] / [ER + PR]) as a predictable marker of lymph node metastasis in endometrioid-type EC. 16 However, models that include the molecular markers to predict early low-risk EC recurrence are rare. Therefore, we aimed to construct a nomogram based on classical parameters and immunohistochemical markers to predict the recurrence of early low-risk EC.
Methods
Study population
From October 2013 to May 2018, the clinicopathological data of EC patients who underwent primary surgical treatment were extracted from the First Affiliated Hospital of Chongqing Medical University and the Second Affiliated Hospital of Chongqing Medical University in China. This study was approved by the Ethics Committee of Chongqing Medical University. The inclusion criteria were: (a) patients who received initial surgical treatment and were confirmed as EC by postoperative pathology; (b) patients were diagnosed as stage IA EC; and (c) patients with integral case records: age, body mass index (BMI), the results of preoperative CA125 examination, tumor characteristics (histologic type, histologic grade, LVSI), immunohistochemical makers including ER, PR, P53, and Ki67. Exclusion criteria were: (a) age of patients ≥60 years old; (b) patients with non-endometrial EC; (c) patients with LVSI; (d) patients with histologic grade 3; (e) patients who did not receive the standard surgery; (f) patients without complete medical records; (g) patients with irregular follow-up; and (h) patients with other malignancies. Patients from the First Affiliated Hospital of Chongqing Medical University were defined as the training cohort (n = 668), while patients from the Second Affiliated Hospital of Chongqing Medical University were assigned as the validation cohort (n = 330) (Supplementary Figure 1).
Treatment and follow-up
Patients received at least total hysterectomy + bilateral salpingo-oophorectomy. According to the National Comprehensive Cancer Network (NCCN) guideline, observation was preferred for early low-risk EC patients after surgery. 3 Patients received regular follow-up: every 3 months for the first 2 years, semiannually for the following 3 years, and then annually. Each visit included physical examination and necessary auxiliary checks such as the examination of CA125, the cytology of the vaginal stump, and imaging examinations. 17 The follow-ups were started from the day of surgery, and continued until June 2021.
Histopathology and immunohistochemistry
All postoperative specimens from each patient were processed in the pathology department with the same standard. Cancerous parts were confirmed by H&E staining. The histologic grade, primary tumor diameter, and LVSI (negative/positive) were firstly judged by the junior doctors in the pathology department, and then re-examined by the experienced pathologists. Immunohistochemical staining of the expressions of ER, PR, P53, and Ki67 were carried out using the automated immunostainer (Leica Bond-Max, Milton Keynes, UK). ER and PR immunohistochemical staining were performed using anti-ER (clone 1D5, 1:50) and anti-PR (clone PgR636, 1:500). For Ki67 and P53 assessment, anti-Ki67 (clone MIB-1, 1:300) and anti-P53 (clone DO-7, 1:200) were used.
The assessment of ER, PR, Ki67, and P53 staining was performed by two senior pathologists independently under double-blind conditions. Results were shown as the percentage of positive tumor cells. It was considered concordant when the results of the two senior pathologists differed ≤10%. However, if the difference was >10%, it was re-evaluated by an unblinded method until agreement was reached. Finally, the average of the proportion evaluated by the two senior pathologists was identified as the final result. 18
Definition of molecular markers
The clinically accepted threshold of CA125 in EC was 35U/mL.19,20 According to Ferrandina et al., 15 when >5% of tumor cells expressed ER ( + ) or PR ( + ), ER and PR were classified as positive, respectively. When >40% of tumor cells expressed Ki-67 ( + ), Ki-67 was defined as high expression.16,21 According to Köbel et al., 22 the complete absence and overexpression were both regarded as P53 abnormal, whereas the wild-type pattern was considered P53 normal. (Supplementary Table 1).
Recurrence
If the lesion was verified by examinations (physical examination, histological findings, or imaging examination), recurrence was considered. 23 Recurrence could be either local or distant: vaginal or intrapelvic recurrence were considered local relapse; upper para-aortic lymph node metastasis, or abdominal metastasis, or metastasis to other organs were considered distant recurrence. 24 The crude cumulative recurrence incidence (CCRI) was the cumulative probability of tumor recurrence. The crude cumulative mortality (CCM) was the cumulative probability of death. 25
Statistical analysis
The differences between the training cohort and the validation cohort were compared using the χ2 test or Fisher's exact test for categorical variables; the student's t-test or rank sum test was used for continuous data. If P values were <0.05, the differences were considered statistically significant. Due to the competing risks in this study, the univariate and multivariate analysis were conducted using the Fine–Gray competing risk model to analyze the association of each factor with early low-risk EC recurrence. Factors with P < 0.05 in the univariate analysis were entered into the multivariate analysis, and the hazard ratio of each factor was calculated. Independent risk factors (P < 0.05) identified from the multivariate analysis were included in the nomogram. The risk threshold of the nomogram model was represented by calculating the optimal threshold of the 3-year CCRI. Then the optimal threshold of the 3-year CCRI rate calculated by the nomogram was determined by the receiver operating characteristic (ROC) curve and the Youden index (Youden index = sensitivity + specificity −1). 26 Based on the optimal cut-off value, patients in two cohorts were split into high-risk and low-risk groups. The distributions of CCRI and CCM were calculated by the cumulative incidence curve, and were compared by the Fine–Gray test. The calibration curve was used to assess the predictive ability of the nomogram. The predictive performance of the nomogram was evaluated by concordance index (C-index). 27 The value of the C-index ranged from 0.5 to 1.0. When the C-index was 0.5, the nomogram had no discriminating ability. A C-index equal to 1 demonstrated a perfect ability to divide patients into different prognosis groups. The time-dependent ROC curves were established to show the discrimination of the nomograms for 1-, 3-, and 5-year CCRI. 10 Statistical analyses were performed by using SPSS software (version 25.0, IBM statistics, Chicago, IL, USA) and R software (version 3.6.1, http://www.r-project.org) (Supplementary R Code).
Results
Patient characteristics
A total of 998 patients with early low-risk EC were included in the study, which involved 668 in the training cohort and 330 in the validation cohort (Supplementary Figure 1). Clinical features of patients in two cohorts are provided in Table 1. There was no significant difference between the training and validation cohorts. In the training cohort, 62 (9.3%) patients relapsed, 32 (4.8%) patients died, of which 30 (4.5%) died due to recurrence. In the validation cohort, 31 patients (9.4%) developed recurrence, 22 patients (6.7%) died, of which 18 (5.5%) died due to recurrence. The features of relapsed patients in the two cohorts are presented in Supplementary Table 2.
Characteristics of the training (N = 668) and the validation (N = 330) cohorts.
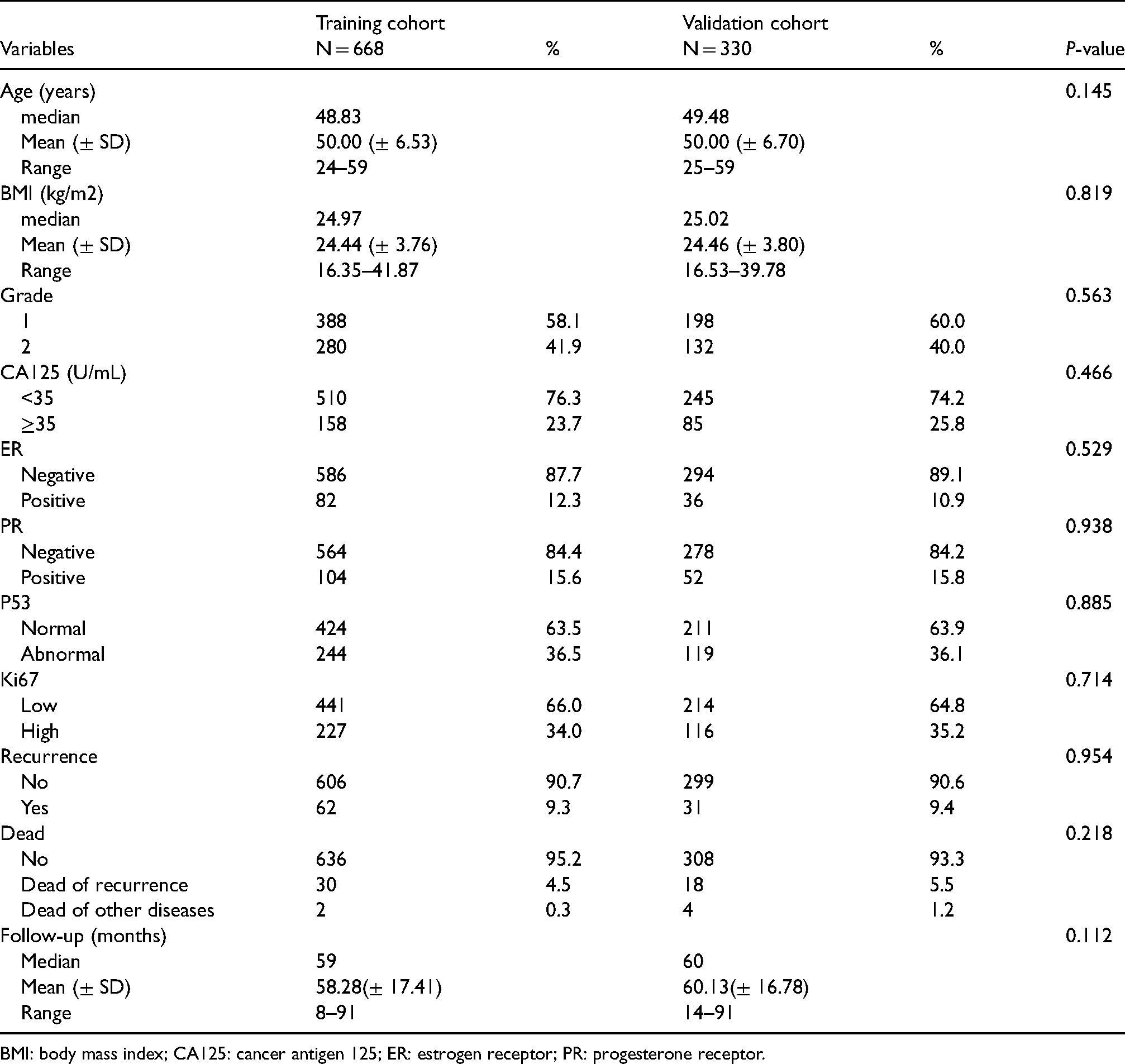
BMI: body mass index; CA125: cancer antigen 125; ER: estrogen receptor; PR: progesterone receptor.
Univariate and multivariate analysis
The univariate analysis indicated that age, grade, CA125, ER, PR, P53, and Ki67 were related to recurrence of early low-risk EC. In the multivariate analysis, grade (P = 0.040), ER (P <0.001), PR (P <0.001), P53 (P = 0.004), and Ki67 (P = 0.002) were independent prognostic factors of recurrence of early low-risk EC (Table 2).
Selected variables in the training cohort for predicting low-risk EC recurrence by univariate and multivariate analysis.
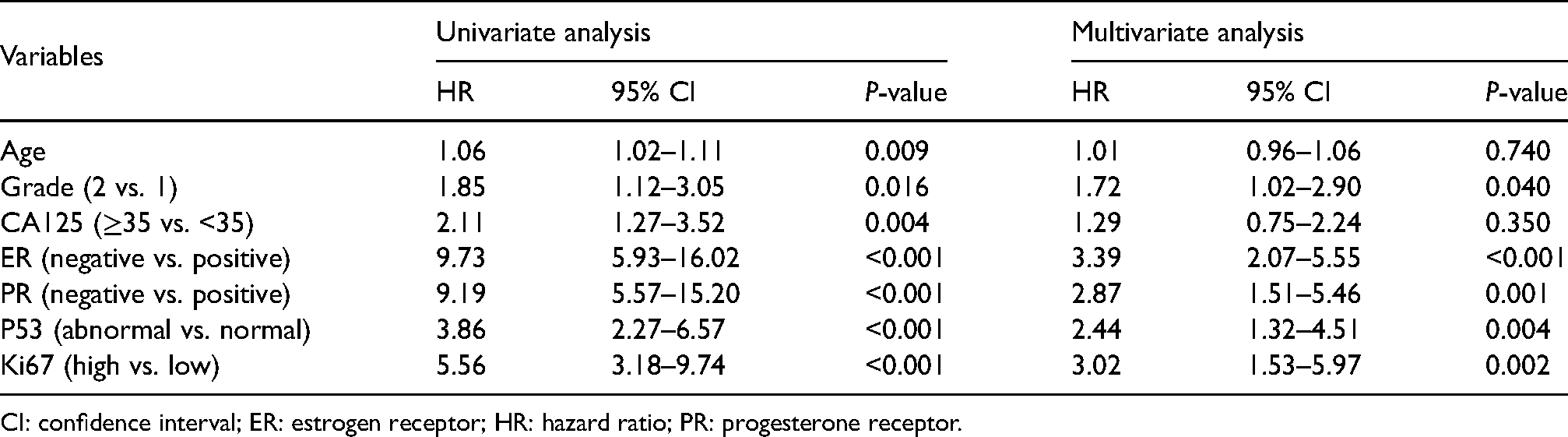
CI: confidence interval; ER: estrogen receptor; HR: hazard ratio; PR: progesterone receptor.
Nomogram model establishment and validation
Based on the independent prognostic factors, a nomogram was constructed. For patients with early low-risk EC, this nomogram could be used to predict the 1-, 3-, and 5-year CCRI rates (Figure 1). The results of the calibration curves did not show substantial bias in predicting 1-year, 3-year, 5-year CCRI probability in the training (Figure 2(a) to (c)) and validation cohorts (Figure 2(d) to (f)). The C-index in the training set was 0.862 (95% confidence interval (CI) 0.790, 0.934), and was 0.827 (95% CI 0.725, 0.929) in the validation set (Supplementary Table 3). The time-dependent ROC curves also indicated the good discriminative abilities of this nomogram. In the training cohort, the area under the curve (AUC) values of the nomogram to predict 1-, 3-, and 5-year CCRI were 0.913 (95% CI 0.868, 0.959), 0.878 (95% CI 0.833, 0.923), and 0.872 (95% CI 0.826, 0.919), respectively (Supplementary Figure 2(a)). In the validation cohort, the AUC values of the nomogram to predict 1-, 3-, and 5-year CCRI were 0.917 (95% CI 0.879, 0.956), 0.860 (95% CI 0.793, 0.928), and 0.845 (95% CI 0.774, 0.917), respectively (Supplementary Figure 2(b)).
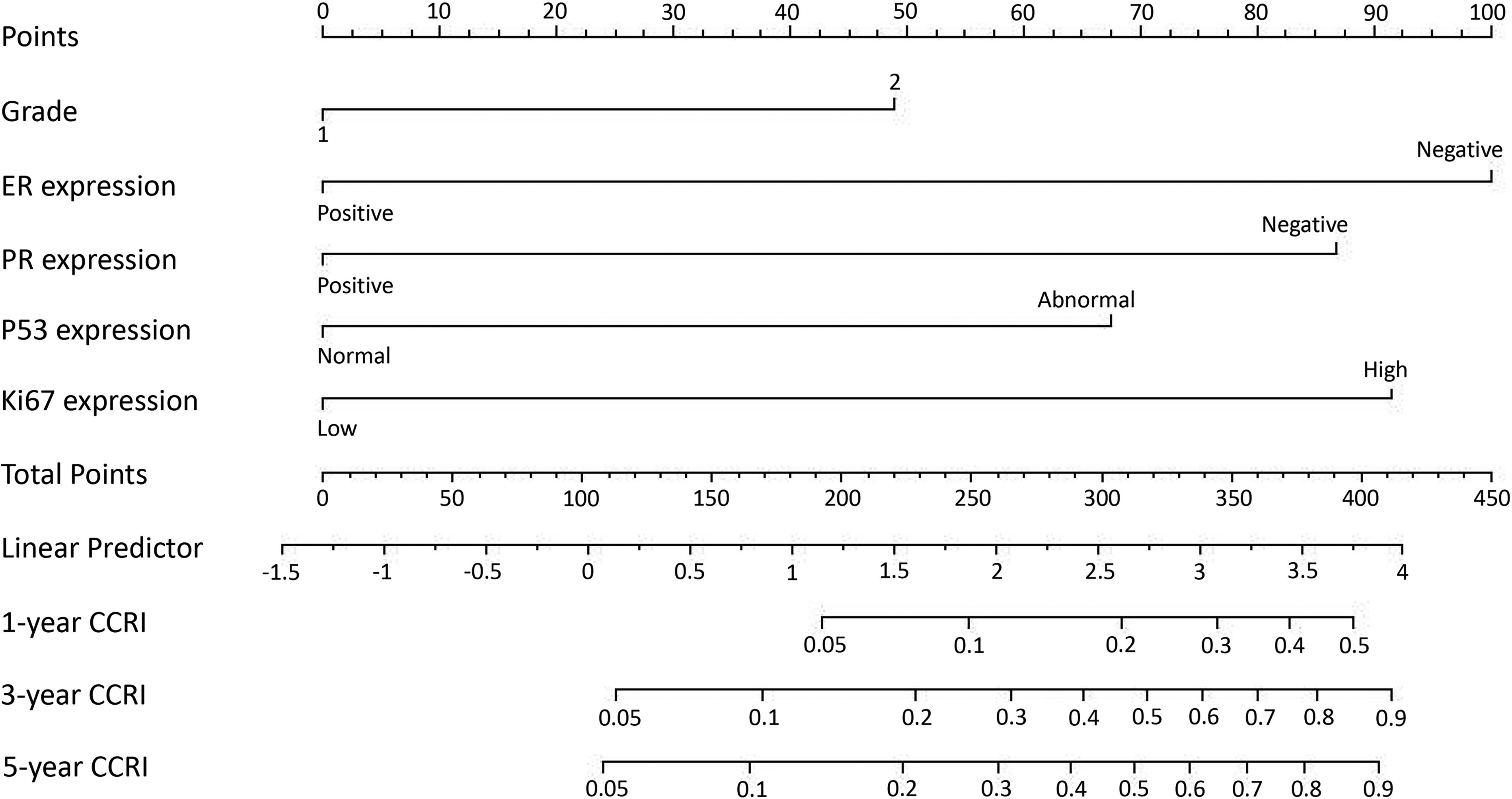
Nomogram model for predicting the 1-, 3-, and 5-year CCRI of EC patients.
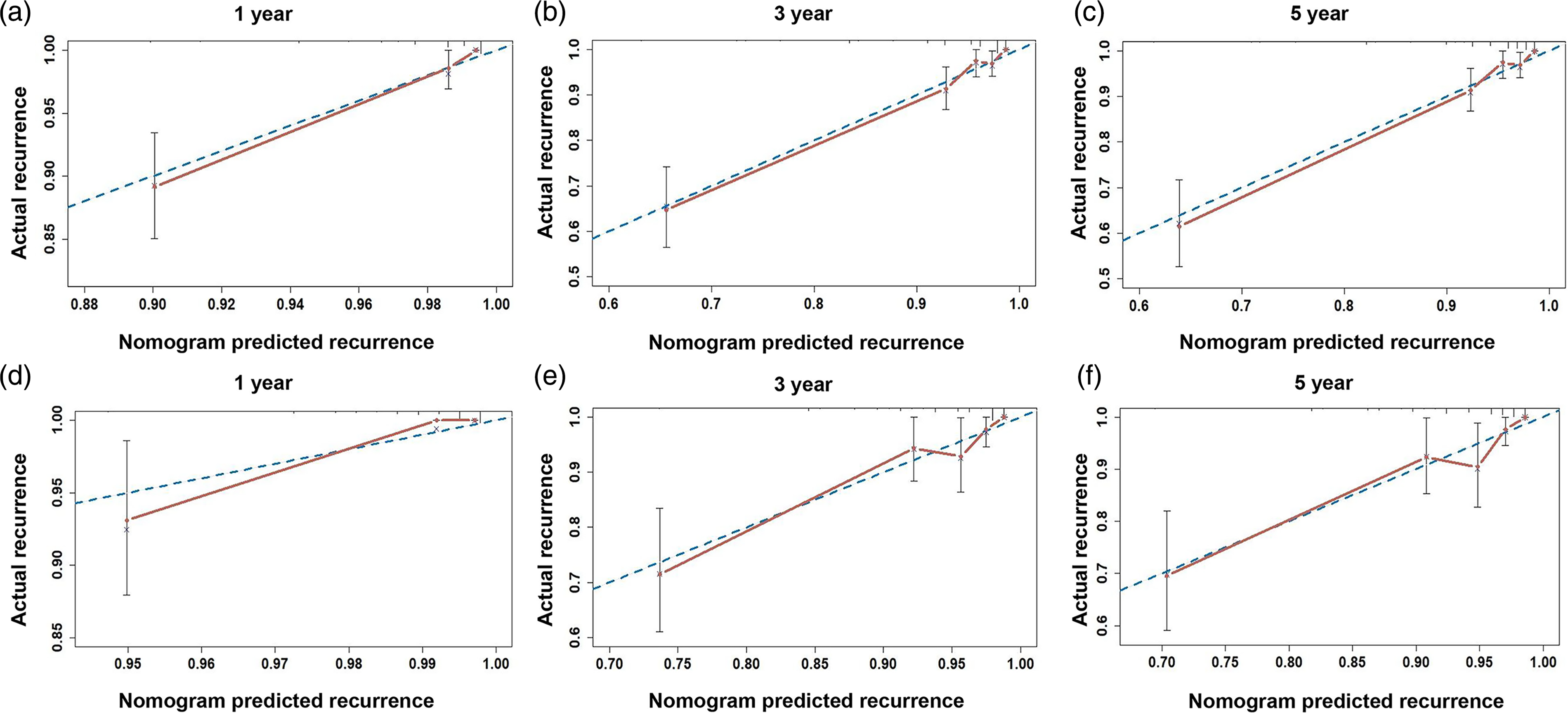
The calibration curves of the nomogram in the training cohort (a) to (c); and validation cohort (d) to (e). Notes: The blue dotted line is the preference line; the red solid line is the prediction curve given by the nomogram.
Optimal threshold of the nomogram model
Since the recurrence time of EC patients is mostly concentrated within 3 years after surgery, we calculated the optimal threshold of the 3-year CCRI of patients, which was used to represent the risk threshold of the model. The optimal threshold of the 3-year CCRI was determined to be 0.11 (AUC = 0.878; sensitivity = 77.4%; specificity = 84.5%) by the ROC curve and the Youden index (Supplementary Figure 3). The AUC of the nomogram constructed based on the five predictors was larger than that of each single predictor (Supplementary Figure 3 and Supplementary Table 4). Patients in two cohorts were all split into two groups according to the optimal threshold: the low-risk group (3-year CCRI rate ≤0.11), and the high-risk group (3-year CCRI rate >0.11). In the training and validation cohorts, the cumulative incidence curves indicated that patients in the high-risk group had higher CCRI and CCM than patients in the low-risk group (Figure 3, Supplementary Table 5).
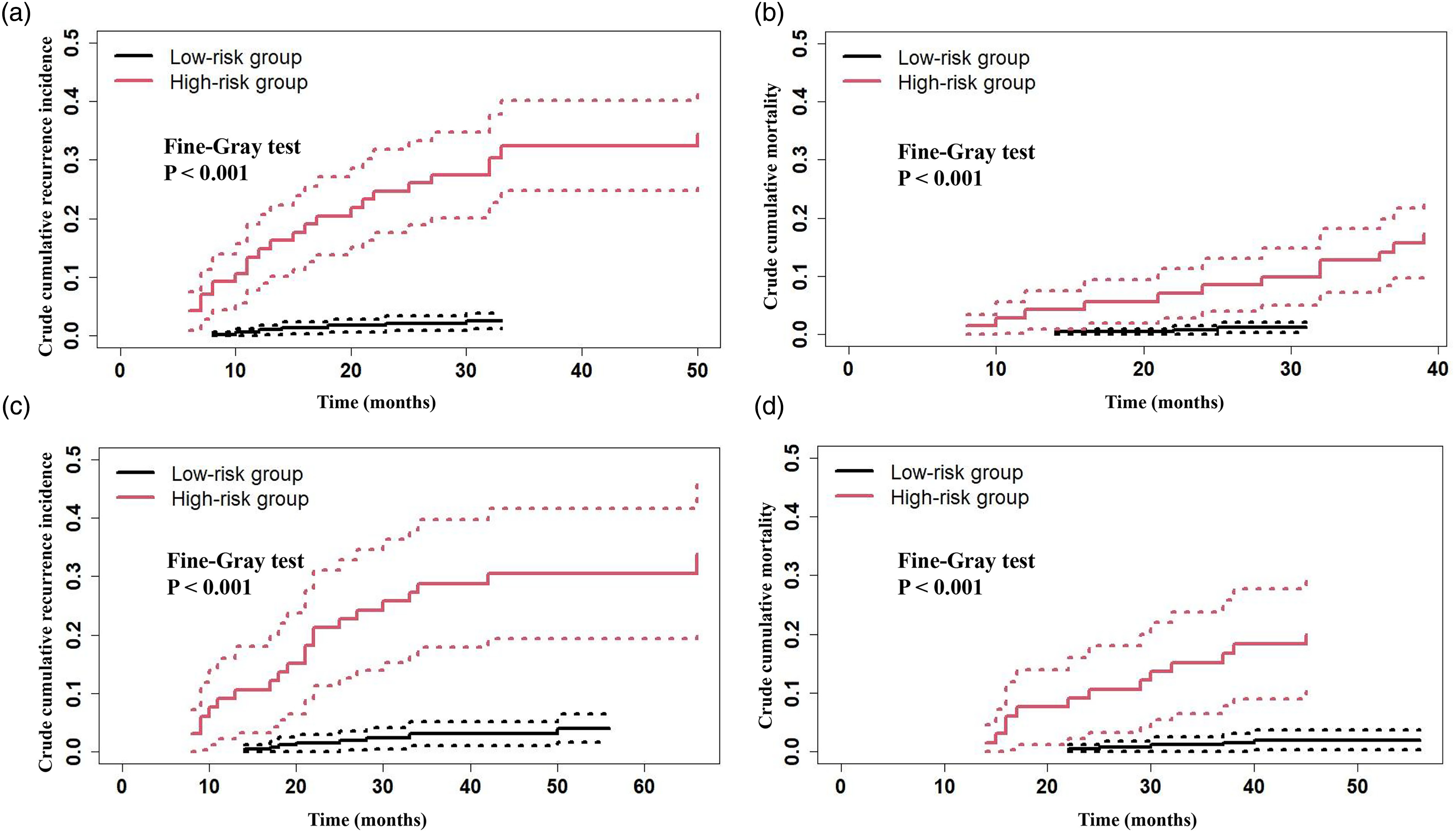
The cumulative incidence curves of low- and high-risk group in two cohorts. Description: (a) the cumulative incidence curve of EC recurrence and (b) the cumulative incidence curve of EC death in the training cohort; (c) the cumulative incidence curve of EC recurrence; and (d) the cumulative incidence curve of EC death in the validation cohort.
Discussion
Although the overall prognosis of patients with early low-risk EC is relatively good, a subset of patients will still relapse due to insufficient attention of the clinicians and patients themselves. In recent years, the molecular biological behavior of cancer has been very important and worthy of attention. The four subgroups, defined by The Cancer Genome Atlas (TCGA) as POLE-mutant, microsatellite-instable (MSI), p53-abnormal, and p53-wild-type, 28 were reported to be related to the prognosis of EC, in which patients in the p53-abnormal subgroup have the worst prognosis.29,30 However, routine gene sequencing and molecular profiling for each patient are expensive, which limits their use in assessing the prognosis of EC in many settings. It is well known that immunohistochemistry remains as an important component of postoperative histopathological survey. Studies have shown that immunohistochemistry may be an alternative method for gene sequencing in some cases due to its simplicity, rapidity, and low cost. 31 Therefore, it is warranted to establish an effective model integrating immunohistochemical markers to predict the prognosis of early low-risk EC.
In this study, the commonly used clinical immunohistochemical markers: Ki67, ER, PR, and P53 were analyzed and included in the model. The subgroup with the worst prognosis in TCGA molecular classification (p53-abnormal) was represented by P53 immunohistochemistry. This model can be used to predict the risk of postoperative recurrence in each early low-risk EC patient. For example, an early low-risk EC patient with histologic grade 2 (48 points), with ER expression “negative” (100 points) and PR expression “positive” (0 points), with P53 expression “normal” (0 points), and with Ki67 expression “high” (92 points) has a sum-point equal to 240, which corresponds to a 3-year CCRI is 24%. Based on the multiple predictors in the nomogram, the risk of recurrence can be expressed in a quantitative form. Furthermore, the internal and external validation of the calibration curve confirmed the high predictive accuracy and consistency of the model.
Currently, adjuvant treatment (chemotherapy and/or radiotherapy) is the main treatment method for EC after surgery. However, follow-up after surgery is usually recommended for early low-risk EC according to guidelines. 3 Unfortunately, a fraction of patients with early low-risk EC relapsed during postoperative follow-up. 5 Hence, 998 patients with early low-risk EC were finally included in this study. The results showed that 9.3% of patients relapsed in the training cohort, and 9.4% patients developed recurrence in the validation cohort, which is similar to other studies. Therefore, a nomogram of patients with early low-risk EC was established, and a subgroup analysis was performed. The optimal risk threshold of the nomogram was determined to divide the patients into a low-risk group and a high-risk group. It was found that the patients in the high-risk group had a poor prognosis compared with those in the low-risk group. This suggested that there were still many early low-risk EC patients who may experience recurrence according to the current guidelines, indicating that these patients may be under-treated. Based on this model, early low-risk EC patients can be stratified in more detail, and appropriate prognostic management could be performed. For example, for the high-risk group of patients identified by this model, even if they are early low-risk patients according to the guidelines, they may also need certain adjuvant therapy, closer follow-up or multiple treatments. Of course, postoperative adjuvant treatment for early low-risk patients is still controversial, and prospective studies are still needed. The model proposed by this study may be helpful for screening patients in prospective studies.
Recent studies have shown that the application of molecular-based classification in EC is reproducible and promising.28,32 The TCGA molecular classification for EC has gained prominence for its direct clinical and prognostic significance. 33 This was mentioned at the 2016 ESMO-ESGO-ESTRO consensus conference. 4 The ESGO-ESTRO-ESP guidelines encouraged the determination of the molecular classification in all EC patients. 34 A previous study showed that EC patients with the POLE mutant had a good prognosis; patients with MSI-H and copy-number-low had a medium prognosis; and patients with copy-number-low had poor outcomes. 28 Unfortunately, due to resource constraints, such as the demand for fresh frozen tissue, the high cost, the long turn-around time, and complex methodologies, TCGA classification is not suitable for routine practice at present. Thus, finding other markers that could precisely reflect the TCGA molecular subtype is a practical way to overcome this obstacle. Compared with the TCGA molecular subtype, our model is more accessible, costs less, and can be performed rapidly, which may assist the transition to future molecular subtyping.
Of note, there are still several limits. First, this nomogram was performed by retrospective analysis, which requires further efforts to collect prospective data for validation. Second, the relationship between CA125 and EC recurrence was not significant for the multivariate analysis; this would need further investigations about multivariate prognostic models of tumor recurrence, supported by an adequate number of observed events. Some studies have shown that CA125 is an important prognostic indicator of ovarian cancer, and CA125 is also closely related to the prognosis of EC.20,35 Certainly, studies have shown that other molecular markers such as HE4, L1CAM, PTEN, and LHCG-R are related to the prognosis of EC, so it is necessary to include more molecular markers to predict the recurrence of EC.31,36–40 In particular, recent studies have reported that TCGA molecular classification has an important prognostic value in EC. 41 However, only the subgroup with the worst prognosis in TCGA molecular classification (p53-abnormal) was included in this study, which may affect the accuracy of the model to a certain extent. Therefore, it is necessary to incorporate more TCGA molecular classifiers to gradually improve the model, and to provide more accurate and individualized treatment for early low-risk EC. Moreover, it is worth mentioning that there is a lack of consensus on the use of labeling index for Ki67, ER, PR, and P53. The main issue in Ki67, ER, PR, and P53 detecting is that different laboratories and observers are using different antibody clones, scoring methods, and platforms. Hence, the detection of Ki67, ER, PR, and P53 needs to be standardized and optimized. Finally, there is no comparison with other predictive models in this study because there seems to be no more models for predicting the recurrence of early low-risk EC. This suggests that we need to pay more attention to low-risk EC patients, and build more available models for predicting the recurrence of low-risk EC.
Conclusions
A prognostic nomogram model was established to predict 1-, 3-, and 5-year CCRI in early low-risk EC patients. This has been helpful for clinicians in individualizing the treatment for patients with early low-risk EC.
Supplemental Material
sj-jpg-1-jbm-10.1177_03936155221132292 - Supplemental material for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer
Supplemental material, sj-jpg-1-jbm-10.1177_03936155221132292 for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer by Wei Kong, Yuan Tu, Peng Jiang, Yuzhen Huang, Jingni Zhang, Shan Jiang, Ning Li and Rui Yuan in The International Journal of Biological Markers
Supplemental Material
sj-jpg-2-jbm-10.1177_03936155221132292 - Supplemental material for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer
Supplemental material, sj-jpg-2-jbm-10.1177_03936155221132292 for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer by Wei Kong, Yuan Tu, Peng Jiang, Yuzhen Huang, Jingni Zhang, Shan Jiang, Ning Li and Rui Yuan in The International Journal of Biological Markers
Supplemental Material
sj-jpg-3-jbm-10.1177_03936155221132292 - Supplemental material for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer
Supplemental material, sj-jpg-3-jbm-10.1177_03936155221132292 for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer by Wei Kong, Yuan Tu, Peng Jiang, Yuzhen Huang, Jingni Zhang, Shan Jiang, Ning Li and Rui Yuan in The International Journal of Biological Markers
Supplemental Material
sj-doc-4-jbm-10.1177_03936155221132292 - Supplemental material for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer
Supplemental material, sj-doc-4-jbm-10.1177_03936155221132292 for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer by Wei Kong, Yuan Tu, Peng Jiang, Yuzhen Huang, Jingni Zhang, Shan Jiang, Ning Li and Rui Yuan in The International Journal of Biological Markers
Supplemental Material
sj-docx-5-jbm-10.1177_03936155221132292 - Supplemental material for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer
Supplemental material, sj-docx-5-jbm-10.1177_03936155221132292 for Development and validation of a nomogram involving immunohistochemical markers for prediction of recurrence in early low-risk endometrial cancer by Wei Kong, Yuan Tu, Peng Jiang, Yuzhen Huang, Jingni Zhang, Shan Jiang, Ning Li and Rui Yuan in The International Journal of Biological Markers
Footnotes
Author contributions
Wei Kong: conceptualization, formal analysis, investigation, methodology, software, supervision, validation, writing - original draft, and writing - review and editing. Yuan Tu: conceptualization, formal analysis, investigation, methodology, validation, writing (original draft, revision, and editing). Peng Jiang: investigation, data curation, and validation. Yuzhen Huang: investigation, data curation, and validation. Jingni Zhang: investigation, data curation, and validation. Shan Jiang: investigation, data curation, and validation. Ning Li: investigation, data curation, and validation. Rui Yuan: project administration, conceptualization, methodology, writing (revision and editing). Wei Kong and Yuan Tu contributed equally to the study and were co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
