Abstract
Background:
Estrogen receptors (ER) and progesterone receptors (PR) have important prognostic value in endometrial cancer, but there is no recognized positive immunohistochemical threshold for predicting the recurrence of early low-risk endometrial cancer. The purpose of this study was to clarify the optimal positive thresholds of the immunohistochemical parameters the ER and PR in early low-risk endometrial cancer.
Methods:
A total of 332 patients with stage IA endometrial cancer were enrolled from the First Affiliated Hospital of Chongqing Medical University and People’s Hospital of Sha ping ba District in Chongqing between January 2013 and December 2018. First, univariate and multivariate Cox regression analyses were used to analyze the correlation between various clinical factors and the prognosis of early low-risk endometrial cancer. Then, the receiver operating characteristic curve (ROC curve) and Youden index were used to determine the positive thresholds of ER and PR.
Results:
The positive thresholds of ER and PR for predicting the recurrence of early low-risk endometrial cancer were 12% and 8%, respectively. Multivariate analysis showed that ER (P = 0.004), PR (P = 0.026), and p53 (P = 0.021) were risk factors for the prognosis of patients with early low-risk endometrial cancer. The recurrence-free survival and the overall survival in the low ER group and PR group were much lower than those in the high ER group and PR group (P < 0.001 of all).
Conclusions:
ER and PR positive thresholds of 12% and 8%, respectively, are the most suitable for predicting the recurrence of early low-risk endometrial cancer.
Keywords
Introduction
Endometrial carcinoma (EC) is one of the most common gynecological malignancies among women in developed countries, and its morbidity and mortality are increasing yearly, 1 showing a younger trend. Endometrial cancer is a malignant tumor derived from the epithelium of the endometrium. This cancer primarily manifests as abnormal vaginal bleeding, which is easy to detect early. Among EC cases, 70% to 80% are type I estrogen-dependent endometrioid adenocarcinomas, 2 and the 5-year overall survival rate exceeds 80%. 3 The European Society of Medical Oncology (ESMO) guidelines 4 define early low-risk endometrial cancer as stage IA, G1, or G2 endometrioid adenocarcinoma. 5 Studies have reported that 20% of early low-risk patients have relapsed, 6 and recurrence remains one of the most common causes of death in patients with early low-risk endometrial cancer. 7
At present, the treatment for patients with endometrial cancer is determined by clinicopathological factors, including stage, tumor grade, histological type, and lymphatic vascular space invasion (LVSI). 8 However, these factors cannot accurately assess the risk of recurrence. Clinicians need accurate markers to predict the outcome of endometrial cancer patients and implement personalized management.6,9 Estrogen receptor (ER) and progesterone receptor (PR) are currently the most common biomarkers used in clinical practice.10,11 Although the significance of ER and PR cutoff values in the prognosis of endometrial cancer was reported,12,13 the effects on early low-risk endometrial cancer were not emphasized. Therefore, this study aimed to clarify the clinical value of ER and PR as prognostic indicators for patients with early low-risk endometrial cancer and to identify an optimal cutoff value of ER and PR related to the prognosis of these patients.
Materials and Methods
Research objects
The retrospective study was approved by the Ethics Committee of Chongqing Medical University (Ethics approval number: 2020-192) and conducted in accordance with the Declaration of Helsinki. The archived data of the patients with endometrial cancer who underwent radical surgical treatment between January 2013 and December 2018 were retrieved from the First Affiliated Hospital of Chongqing Medical University and People’s Hospital of Sha ping ba District in Chongqing. Inclusion criteria: (1) patients were diagnosed with stage IA endometrial cancer according to the 2009 International Union of Obstetrics and Gynecology (FIGO) guidelines and 14 (2) patients had complete medical records, including age, body mass index (BMI), comorbidities (hypertension or diabetes), detailed surgical records, postoperative pathological results (including tumor histological type and grade, depth of muscle invasion, depth of cervical stromal invasion, etc.), immunohistochemical indices (CA125, ER, PR), postoperative adjuvant treatment methods and whether the treatment is adequate. Exclusion criteria: the patients had one of the following high-risk factors:3,15 age ⩾ 60 years, postoperative pathology showed nonendometrioid adenocarcinoma or mixed histological types, or high-grade endometrioid adenocarcinoma or accompanied by LVSI (lymphatic vascular infiltration). Patients who were finally included in the study according to the above inclusion and exclusion criteria were defined as ultralow-risk endometrial cancer patients.
Treatment and follow-up
Patients received standard surgical treatment, including at least total hysterectomy with bilateral salpingectomy with or without pelvic ± abdominal para-aortic lymphadenectomy. 7 Given that patients in the ultralow-risk group were included, according to international guidelines, these patients were not supplemented with adjuvant therapy after surgery. Postoperative follow-up was achieved through outpatient clinics and by telephone every 3 months for the first 2 years, every 6 months for the following 3 years, and once yearly thereafter. The follow-up included a physical examination and related imaging examinations. 7
Histopathology and immune tissue analysis
All specimens were fixed with formalin solution within 2 hours of isolation and then sent to the pathological laboratory of Chongqing Medical University for further processing. All specimens were processed in the pathology department with the same standards in accordance with pathological section staining and immunohistochemistry-related procedures. 16
Immunohistochemistry (IHC) was used to stain ER and PR with an automatic immunestainer (Leica Bond-Max, Milton Keynes, UK). All specimens were embedded in paraffin after being fixed in 10% formalin solution. Representative parts of the paraffin specimens were marked according to HE staining. The core of endometrial tissue (1 mm diameter) with a high percentage of tumors (>70%) was selected and transferred to the receptor block. Each sample was made into a tissue chip wax block and placed 2 mm apart. The blocks were sliced and transferred to a glass slide to generate a tissue chip. The thickness of the tissue chip sections was 4 μm. The sections were usually deparaffinized with xylene in water, hydrated with gradient alcohol, and subject to antigen retrieval with 0.01 mol/L citrate buffer (pH 6.0) sodium. To reduce nonspecific background staining caused by endogenous peroxidase, the slices were incubated in 3% hydrogen peroxide at room temperature for 30 minute. The tissue specimens were blocked with 5% bovine serum albumin (BSA) blocking solution at room temperature for 30 minute and incubated with rabbit anti-human polyclonal ER (clone 1D5, 1:50), PR (clone PgR636, 1:500), and P53 antibodies at 37℃ for 1 hour. The samples were incubated for 30 minute at room temperature using an anti-rabbit secondary antibody. Then, diaminobenzidine (DAB) was developed at room temperature, and the reaction time was controlled under a microscope. The changes in tissue staining were observed under a microscope to assess the degree of positivity.
The histological type, grade, size of the lesion, depth of muscular layer infiltration, and the results of immunohistochemical analysis of the tumor were preliminarily judged by the primary pathologist and then independently evaluated by two experienced senior pathologists. The percentage of positively stained ER and PR tumor cells was recorded (0%-100%). 17 If the positive ratio of tumor cells was ⩽ 10%, it was considered consistent. If the ratio was > 10%, the result was re-evaluated (unblinded), and a consensus was reached. The final proportion of positive tumor cells was the average value evaluated by two pathologists. According to the P53 reading criteria, P53 is defined as either normal expression (wild-type expression) or abnormal expression (including complete deletion or overexpression). 18
Recurrence
Recurrence was confirmed by physical examination, histological examination, and/or imaging examination (including CT, MRI, ultrasound, bone imaging, FDG-PET, or specific X-ray examination). 7 Based on location, recurrence was divided into 7 vaginal stump recurrence, pelvic, and abdominal lymph node recurrence (pelvic or para-aortic lymph nodes), pelvic local recurrence, peritoneal metastasis, and distant metastasis. Recurrence-free survival (RFS) was the time interval between the date of surgery and the date of recurrence (confirmed by histology or imaging). 19 Overall survival (OS) was defined as the time interval between the date of surgery and the date of death of the patient. 7
Statistical analysis
SPASS 26.0 statistical software was used to process the data. Enumeration data were expressed as a percentage, and comparisons between groups were implemented by chi-square test. Normal measurement data (such as age and BMI) were expressed as the mean ± standard deviation (X ± S), and t test was used to compare between groups. Nonnormal measurement data (such as ER, PR, and CA125) were represented by median and interquartile range (Q1, Q3), and rank sum test was used for comparison between groups. Univariate and multivariate analyses of prognostic factors were implemented using the Cox regression model. The positive thresholds (cutoff values) of ER and PR were determined by the ROC curve and Youden index (Youden index = sensitivity + specificity - 1).17,20 Finally, patients were divided into 2 groups according to the optimal cutoff value of the ER-positive percentage: patients with an ER-positive percentage less than the cutoff value comprised the low ER-positive proportion group, and those with values greater than or equal to the cutoff comprised the high ER-positive proportion group. Similarly, according to the optimal PR positive percentage, the patients were divided into 2 groups: patients with a PR positive percentage less than the cutoff value comprised the low PR positive ratio group, and those with a value greater than or equal to the cutoff comprised the high PR positive ratio group. The Kaplan–Meier method was used to calculate the survival rate. The differences between groups were analyzed using the log-rank test, and the statistical significance level was α = 0.05.
Results
Clinical characteristics
From January 2013 to December 2018, a total of 1,021 patients with stage IA endometrial cancer received standard surgical treatment at the First Affiliated Hospital of Chongqing Medical University and People’s Hospital of Sha Ping Ba District in Chongqing, of which 332 patients met the inclusion criteria. In total, 689 patients were excluded. Among these patients, 45 patients had incomplete case data, 31 patients had no regular follow-up after surgery, 23 patients did not undergo standard surgery due to the desire to preserve fertility, and 575 patients had one or more of the following high-risk factors: age ⩾ 60, nonuterine endometrioid carcinoma, high-grade endometrioid carcinoma, and LVSI (lymphatic vascular infiltration) (Figure 1).

Flow chart of patient admission.
Table 1 summarizes the clinicopathological characteristics of the 332 patients in this study. There were 185 patients (55.7%) with BMI ⩾ 24 kg/m2. There were 79 patients (23.8%) with CA125 values greater than 35 U/ml, and 40 patients (12%) were diagnosed with diabetes before the operation. Grades 1 and 2 tumors accounted for 57.8% (192 patients) and 42.2% (140 patients), respectively. The distribution range of ER was 0% to 95% (median 90%); the distribution range of PR was 0% to 95% (median 90%). The normal expression of p53 was 64.5% (n = 214), the abnormal expression was 35.5% (n = 118). A total of 31 patients (9.3%) relapsed, of which 16 patients (4.8%) died. The median recurrence-free survival was 45.5 (0.73) months, and the mean standard deviation was 46.58 ± 16.42 months.
Clinical data characteristics of all patients (n = 332).

BMI: body mass index; ER: estrogen receptor; PR: progesterone receptor; SD: standard deviation.
Clinical characteristics of relapsed patients
In total, 31 relapsed patients were included in this study (Table 2). The follow-up time of relapsed patients was 34.49 ± 16.682 (range: 0%-60%) months, and the median follow-up was 31 months. The relapse-free survival was 17.90 ± 10.678 (range: 0%-44%) months, and the median was 15 months. The recurrence sites included the vagina, cavum pelvis, lymph gland, and peritoneum.
Comparison of clinical parameters in patients who relapsed (n = 31).

SD: standard deviation.
Univariate and multivariate analysis
A univariate Cox regression model was used to analyze clinicopathological factors affecting the prognosis of early low-risk endometrial cancer, and factors with a P value greater than 0.05 were excluded from the multivariate analysis, including age (Wald = 3.264, P = 0.071), BMI (Wald = 2.031, P = 0.154), and pathological grade (Wald = 2.244, P = 0.134). Factors with P values less than 0.05, including ER (Wald = 32.360, P = 0.000), PR (Wald = 24.896, P = 0.000), CA125 (Wald = 4.280, P = 0.039), and p53 (Wald = 8.716, P = 0.003) were related to prognosis in multivariate Cox regression analysis. The results of multivariate analysis showed that ER (Wald = 8.134, P = 0.004), PR (Wald = 4.939, P = 0.026), and P53 (Wald = 5.296, P = 0.021) were risk factors for the prognosis of patients with early low-risk endometrial cancer (Table 3).
Univariate and multivariate analyses for predicting the recurrence factors of endometrial cancer.

BMI: body mass index; ER: estrogen receptor; PR: progesterone receptor; HR: hazard Ratio; CI: confidence interval.
Positive thresholds of ER and PR
Univariate and multivariate analyses demonstrated that ER and PR positivity were risk factors for recurrence, as confirmed by the ROC curve and Youden index (Youden index = sensitivity + specificity -1). The optimal cutoff value of ER for predicting the recurrence of early low-risk endometrial cancer was 12% (area under the curve = 0.794; sensitivity was 64.5%; specificity was 89.7%) (Figure 2A), and the optimal cutoff value of PR was 8% (area under the curve = 0.725; sensitivity was 58.1%; specificity was 88.7%) (Figure 2B).

Receiver operating characteristic curves of ER and PR.
Comparison of clinicopathological parameters between the high ER group and the low ER group
According to the positive threshold of ER, patients with ER < 12% were defined as the low ER-positive proportion group, and those with ER ⩾ 12% were defined as the high ER-positive proportion group. When the ER cutoff value was set to 12%, the high ER-positive ratio (⩾12%) was significantly related to the following factors: PR expression (x2 = 77.431, P = 0.000), recurrence (x2 = 63.542, P = 0.000), and death (x2 = 36.853, P = 0.000) (Table 4).
Comparison of clinicopathological parameters among patients with high and low ER and PR positive proportions (n = 332).

ER: estrogen receptor; PR: progesterone receptor.
The 3-year recurrence-free survival (RFS) rates of patients in the low ER-positive proportion group and the high ER-positive proportion group were 62% (95% CI = 48.5%-75.5%) and 95.8% (95% CI = 93.3%-98.3%), respectively. The 5-year RFS rates were 58.9% (95% CI = 44.8%-73%) and 95.8% (95% CI = 93.3%-98.3%), respectively, indicating a significant difference (x2 = 72.389, P < 0.001) (Figure 3A). The 3-year overall survival (OS) rates of patients in the low ER-positive proportion group and the high ER-positive proportion group were 78.9% (95% CI = 67.1%-0.7%) and 98.5% (95% CI = 82.8%~100%), respectively, whereas the 5-year OS rates were 76.1% (95% CI = 63.6%~88.6%) and 98% (95% CI = 80.3%~100%), respectively (x2 = 38.6, P < 0.001) (Figure 3B).
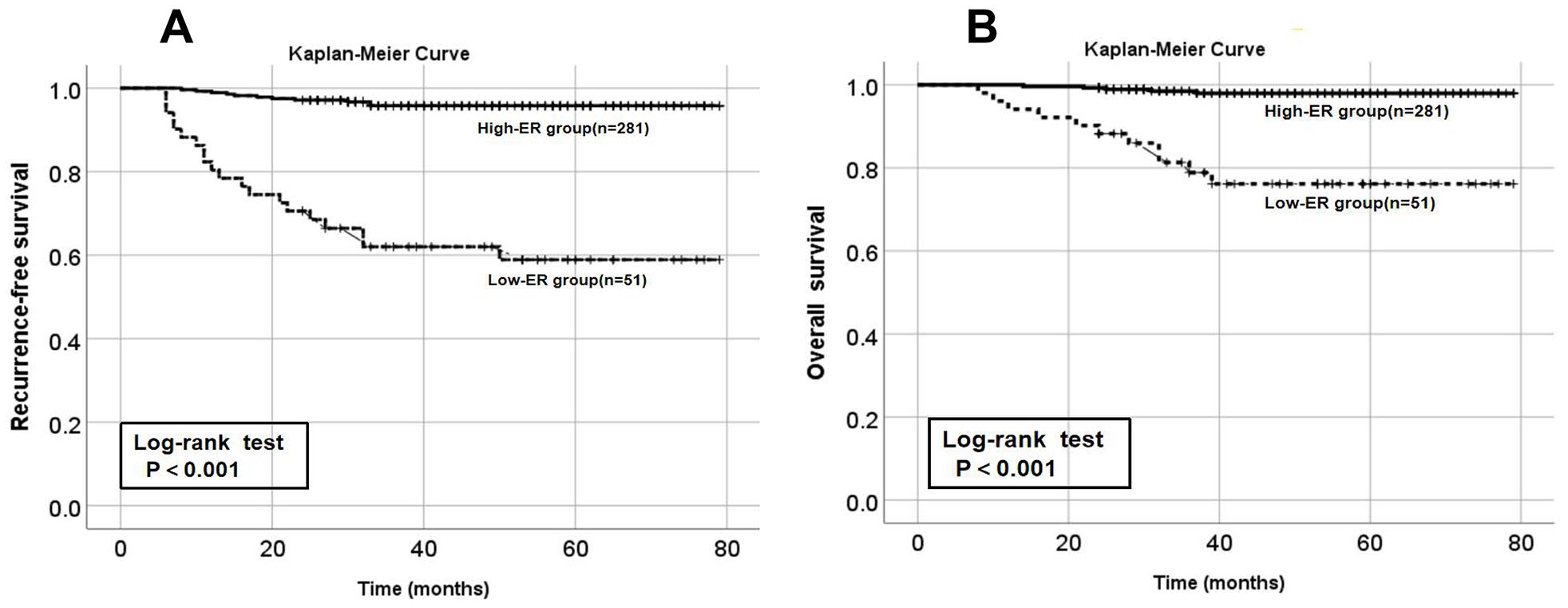
Kaplan–Meier survival curves of the high ER group and the low ER group.
Comparison of clinicopathological parameters between the high PR group and the low PR group
According to the positive threshold of PR, patients with PR < 8% were defined as the low PR-positive proportion group, whereas those with PR ⩾ 8% were defined as the high PR-positive proportion group. When the PR cutoff value was set to 8%, the high PR positive ratio (⩾ 8%) was significantly related to the following: factors: age (x2 = -2.967, P = 0.015), ER expression (x2 = 77.431, P = 0.000), recurrence (x2=46.539, P = 0.000) and death (x2=35.865, P = 0.00) (Table 4).
The 3-year RFS rates in the low PR-positive proportion group and the high PR-positive proportion group were 64.6% (95% CI = 51%~77.9%) and 95.3% (95% CI = 92.7%~97.8%), respectively, whereas the 5-year RFS rates were 64.6% (95% CI = 51%~77.9%) and 94.6% (95% CI = 91.7%~97.5%) (x2 = 54.482, P = 0.000), respectively (Figure 4A). The 3-year OS rates of the patients in the low PR-positive proportion group and the high PR-positive proportion group were 78.2% (95% CI = 66%-90.3%) and 98.5% (95% CI = 82.8%-100%), respectively, whereas the 5-year OS rates were 75% (95% CI = 61.7%~88.3%) and 98% (95% CI = 80.4%~100%), respectively (x2 = 50.234, P = 0.000) (Figure 4B).

Kaplan–Meier survival curves of the high PR group and the low PR group.
The prognostic value of ER and PR combined with P53
Considering that P53 expression is closely related to the prognosis of endometrial cancer, the above multivariate analysis also showed that P53 is an independent influencing factor of recurrence. Therefore, we used the ROC curve to compare the prognostic value of ER, PR and P53 and found that the prognostic values of ER and PR combined with P53 (AUC = 0.829, 95% CI = 74.7%-91.1%) were superior to each single predictor, including ER (AUC = 0.739, 95% CI = 63.1%-84.7%), PR (AUC = 0.729, 95% CI = 62.1%-83.6%), and P53 (AUC = 0.642, 95% CI = 53.8%-74.6%) (Figure 5).

The prognostic value of ER and PR combined with P53.
Discussion
Many researchers have used ER- and PR-positive thresholds to assess the prognosis and recurrence 3,21 -23 of tumors.12,13 Some studies have used ER- and PR-positive thresholds to predict the prognosis and recurrence of patients with stage I-III endometrial cancer. It is believed that patients below the positive thresholds have a poor prognosis and a high risk of recurrence. 3 In addition, the literature has shown that the lack of ER and PR expression in early low-risk endometrial cancer is not only an independent predictor of adverse outcomes 12 but can also predict the risk of lymph node metastasis 24 and recurrence. 14 However, there is currently no accepted standard for ER- and PR-positive thresholds in early low-risk endometrial cancer.
In this study, we included patients in the ultralow-risk group. According to international guidelines (such as NCCN guidelines), these patients are suggested to undergo follow-up after surgery without the need for supplementary adjuvant therapy. However, we found that these patients in the ultralow-risk groups still had recurrence, and the recurrence rate increased yearly.25 -28 Therefore, it is urgent to identify new prognostic indicators for the prognostic management of these patients. It is also important to assess whether ER and PR can be used as prognostic indicators for patients with ultralow-risk endometrial cancer. We used univariate analysis to reveal that the loss of ER and PR, the increase in CA125 expression and P53 expression were risk factors for the recurrence of early low-risk endometrial cancer. However, other clinicopathological factors, such as age, BMI and grade, were not significantly related to the recurrence of early low-risk endometrial cancer patients. Furthermore, multivariate analysis found that ER, PR and P53 were risk factors for the prognosis of patients with early low-risk endometrial cancer. Then, the ROC curve and Youden index showed that the positive threshold of ER was 12% and the positive threshold of PR was 8%. The patients were divided into groups according to the positive thresholds of ER and PR to compare clinicopathological factors, recurrence-free survival and overall survival between the groups. The results showed that the prognosis of patients with early low-risk endometrial cancer was strongly related to ER and PR and less affected by age and grade. In addition, the analysis showed that patients in the low ER and PR groups exhibited more recurrences and deaths than those in the ER and PR high-positive ratio groups. Finally, the results also showed that the recurrence-free survival rate and overall survival rate of patients in the high ER- and PR-positive groups were much higher than those in the low ER- and PR-positive groups, and the difference was statistically significant. For patients with ER and PR positivity ratios below the positivity threshold, even for patients with early-low-risk endometrial cancer, follow-up alone according to current guidelines may no longer be appropriate and may require appropriate adjuvant radiotherapy and chemotherapy. In addition, more frequent follow-up or adjuvant radiotherapy and chemotherapy are needed. 29 At present, the choice of adjuvant therapy for endometrial cancer patients mainly depends on classic clinicopathological factors, 15 such as age, LVSI, tumor volume, depth of invasion, cervical stromal invasion or endocervical gland invasion.30,31 For early low-risk endometrial cancer patients, ER and PR may be strong and effective indicators for postoperative management. For example, the 2021 NCCN Guidelines noted that ER detection is recommended for recurrent endometrioid cancer. Therefore, the positive thresholds of the immunohistochemical markers ER and PR are expected to provide guidance for the postoperative adjuvant treatment of patients with early low-risk endometrial cancer. Moreover, our study demonstrated that combining P53 expression status with the prognosis of endometrial cancer can further optimize the prognostic value of a single index, providing a theoretical basis for the application of molecular indices in early-stage low-risk endometrial cancer in the future.
In univariate analysis, age and pathological grade were suggested as “recurrence risk factors for prognosis” (HR > 1). However, the P values of age and pathological grade were greater than 0.05, which indicates that these factors were not significant. However, this finding does not suggest that they are prognostic indicators in endometrial cancer, as they have been shown to have important prognostic value in endometrial cancer in other studies. 32 Multivariate analysis found that CA125 was not a significant risk factor for recurrence in the multivariate analysis (P = 0.099), but it was suggested as a “recurrence risk factor” (HR > 1). Other studies have reported that CA125 is related to prognostic factors, such as clinical stage, depth of myometrial invasion, and lymph node metastasis, in endometrial cancer. 33 It can be inferred that CA125 has a greater significance for the prognosis of endometrial cancer patients. Finally, the greatest limitation of our study is that this is a two center retrospective study, and the patient cohort structure has a certain bias. For example, the recurrence rate of our patient cohort is greater than that of other similar studies (9.3% vs. 2.5%), 34 so the conclusion of this study still needs to be verified by multicenter studies and prospective studies.
Conclusions
In summary, in this study, the positive thresholds of ER and PR revealed the clinical value of these biomarkers in the prognosis of patients with early low-risk endometrial cancer, and this information can be instructive for the administration of adjuvant treatment in these patients.
Footnotes
Acknowledgements
The authors are grateful to all patients who participated in this study.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
NL: Obtaining and analyzing data; PJ: Data analysis and interpretation; YT: Important content revision; YH and JZ: Sample collection and preparation; WK and SJ: Visualization; YJW: Obtaining data; XRZ: Obtaining data; QNX: Obtaining data; RY: Final revision of manuscript.
Availability of Data and Material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The Ethics Committee of Chongqing Medical University approved this study (Ethics approval number: 2020-192). All patients provided their informed consent before starting the treatment and gave consent to have their data published. As it was a retrospective clinical study, all the patients were contacted by telephone to obtain verbal informed consent, and the study was approved by the ethics committee. All patient data were anonymized or maintained with confidentiality.
