Abstract
Several genetic association studies have analyzed the link between the catalase (CAT) C262T variant and different cancers, but the findings remain controversial. Our research centered on establishing a comprehensive correlation between the C262T variant and different cancers. This study was conducted using RevMan 5.4 software following the PRISMA 2020 guidelines. For this meta-analysis, 53 case-control studies (18,258 cases and 47,476 controls) were chosen. The analysis revealed that three genetic models were statistically linked (P < 0.05) to overall cancer susceptibility in codominant model 2 (COD2): odds ratio (OR) = 1.16, COD3: OR = 1.21, recessive model (RM): OR = 1.20). After stratification by ethnicity, a significant link (P < 0.05) was found in Caucasians (COD2: OR = 1.18, COD3: OR = 1.17, over-dominant model (ODM): OR = 1.19) and Asians (COD3: OR = 1.49). Subgroup analyses revealed a significant correlation (P < 0.05) with blood-and-bone-marrow-related cancer, skin cancer, gastrointestinal-tract-related cancer, prostate cancer, and gynecologic cancer. Three genetic models in population-based controls (COD2: OR = 1.19, COD3: OR = 1.17, RM: OR = 1.19) and two genetic models in hospital-based controls (COD3: OR = 1.40, RM: OR = 1.24) were found to be significantly correlated (P < 0.05) with cancer. Also, three genetic models for polymerase chain reaction-restriction fragment length polymorphism (COD3: OR = 1.46; RM: OR = 1.34, ODM: OR = 0.80) and three models for MALDI-TOF + MassARRAY (COD2: OR = 1.32, RM: OR = 1.26, allele model: OR = 1.14) genotyping methods showed significant association (P < 0.05) with cancer. The meta-regression showed that the quality scores might be a source of significant heterogeneity under the COD2 model (coefficient = 0.176, P = 0.029). Trial sequential analysis also validated the adequacy of the sample size on overall findings. Our results indicate that CAT C262T variant is associated with overall cancer susceptibility, especially in Caucasians and Asians. This variant may also be associated with blood-and-bone-marrow-related, GIT-related, prostate, skin, and gynecological cancers.
Introduction
Cancer is a major worldwide public health issue at present. The prevalence of cancer and cancer-related morbidity and mortality are rising at an alarming rate. Based on the GLOBOCAN estimation, in 2020, more than 19 million people were supposed to be affected by different cancers and approximately 10 million people were supposed to die due to cancer. 1 In 2020, the most common cancer was breast cancer (2.3 million new cases) and the fifth-highest reason for global death. The second most frequent cancer and the leading cause of mortality was lung cancer, with an estimated 2.2 million cases and 1.8 million deaths in 2020. Colorectal carcinoma was the third-highest prevalent cancer and the second most common cause of death in the world. Cancer progresses for multiple reasons, including genetic, racial, environmental, lifestyle, infection, and other associated factors.1,3
Superoxide anion radicals, hydroxyl radicals, hydrogen peroxide, and singlet oxygen are examples of reactive oxygen species (ROS) that play significant roles in the regulation of cellular function. They may cause serious damage to macromolecules (DNA, protein, and lipids) at high or prolonged amounts, resulting in cancer. 4 As a result, radicals are now regarded as an important cause of malignancies. Endogenous antioxidants (e.g., superoxide dismutase (SOD), glutathione peroxidase, catalase (CAT), butyrylcholinesterase, and thioredoxin-peroxiredoxin) and non-enzymatic defenders (e.g., glutathione, vitamins A, ascorbic acid, tocopherol, and flavonoids) counteract the effects of ROS. SODs are enzymes that convert superoxide radicals into hydrogen peroxide and oxygen. 5 The CAT enzyme is an endogenous heme enzyme that converts H2O2 to H2O to neutralize ROS. 5
CAT is an essential protein commonly found in erythrocytes, liver, and kidneys. This 240 kDa tetrameric enzyme with four porphyrin heme (iron) groups is mostly found in peroxisomes. The CAT gene, which spans 34 kb and is made up of 13 exons and 12 introns, is located in the 11p13 locus. 6 Several polymorphisms in the CAT gene have been discovered, with rs1001179 (C262T) being the most widely investigated. It produces a cytosine (C) to thymine (T) alteration in the promoter region at position −262 (262 C > T). Many studies have revealed that genetic polymorphisms can affect protein expression and function, and they have shown a possible link between CAT C262T polymorphism and cancer risk. According to several studies, the CAT C262T genetic variant has been linked to prostate cancer, invasive cervical cancer, breast cancer, hepatocellular carcinoma, and endometriosis, among other cancers.7,10 However, these findings were inconsistent and inconclusive.
Based on the inconsistencies of the previously available genetic association studies, a meta-analysis is much needed to achieve a comprehensive outcome. Therefore, the goal of this study was to find a possible connection between CAT C262T polymorphism and the risk of various cancers.
Materials & methods
Methods of literature search
We followed the preferred reporting items for systematic reviews and meta-analyses (PRISMA) 2020 principles for our meta-analysis. 11 From October 15, 2021 to December 07, 2021, the articles were carefully reviewed in electronic literature resources such as PubMed, ScienceDirect, the Wiley Online Library, BMC, and the Cochrane Library. Duplicate studies were eliminated using EndNote × 7.0 software. The following terms—“catalase”, “CAT”, “C262T”, “rs1001179”, “polymorphism”, “variant”, “cancer”, “tumor”, “malignancy”—were used in searches, either alone or in combination. In this meta-analysis, no language or country restrictions were applied, and only online literature was considered for evaluation. We looked over the reference lists of all of the papers we selected to see if any studies were missing.
Study eligibility
For studies to be included, the following criteria were implemented: (a) human-sample-based case-control or cohort studies; (b) cancer was assessed as the major outcome of the study; (c) studies analyzed CAT C262T polymorphism and various cancers; and (d) the publications provided enough data to assess odds ratio (OR) and 95% confidence interval (CI) values.
The criteria for exclusion were: (a) reviews, expert opinions, letters to the editor, and case reports; (b) duplicate or overlapped research; (c) articles that examined other CAT polymorphisms; and (d) animal studies.
Data extraction
The data were collected individually by two authors (MAB and SJ), followed by the above-mentioned criteria. They individually performed a literature review and evaluation, and entered data into an Excel spreadsheet. If a disagreement appeared during the inquiry, it was resolved through discussion by other investigators (MAA and MSI). The first author's name, publication year, country of publication, ethnicity, cancer types, age of cases and controls, cancer stages, genotyping methods, type of control, number of patients and controls, genotypes, and P-value from HWE were selected for data collection from each study. To select publications for the meta-analysis, the Rayyan QCRI was applied. 12
Methodological quality assessment
To assess the quality of the included studies, the Newcastle–Ottawa Scale (NOS) was applied. 13 The Jadad scale was used to verify data integrity in randomized controlled trials, if necessary. 14
Statistical analysis, heterogeneity, and publication bias
The Review Manager 5.4 (RevMan 5.4) software was applied to investigate the link between CAT C262T polymorphism and various malignancies. To assess the significance of the link, seven distinct genotypic models were used: codominant model 1 (COD1), COD2, COD3, dominant model (DM), over-dominant model (OD), recessive model (RM), and allele model (AM). We employed the χ2-based I2-statistic and the Q-test to assess potential heterogeneity. If the P-value was lower than 0.1, the Q results indicated significant heterogeneity, and a random-effects model was applied. In other cases (P > 0.1), the fixed-effects model was applied. Sensitivity assessments were carried out by deleting chosen studies one at a time in order to classify any substantial risk differences across studies for each parameter. The funnel plot asymmetry, Begg–Mazumdar rank correlation, and Egger's test were applied to evaluate the publication biases. The threshold for statistically significant publication bias was P ≤ 0.05. In addition, various subgroups were implemented in our investigation, including ethnicity, cancer types, genotyping methods, and type of control sources (hospital or population).
The meta-regression analysis was also performed to evaluate the impact of the quality scores of studies on overall findings or to identify the source heterogeneity. In addition, trial sequential analysis (TSA) was applied to test whether the estimated sample sizes are adequate enough to generate a statistically significant result.
Results
Characteristics and quality assessment of eligible studies
There were 321 entries identified in five databases during the primary search. A total of 185 documents were eliminated due to redundancy; 25 articles were excluded for being discursive studies; 21 studies were eliminated after reading the manuscript title and full abstract; 15 were removed from the rest 90 studies for different arguments and reasons. Finally, based on the inclusion and elimination characteristics, 53 complete studies7,9,15,58 with 18,258 cancer patients and 47,476 controls, were included for the assessment in this meta-analysis (Figure 1 and Table 1).
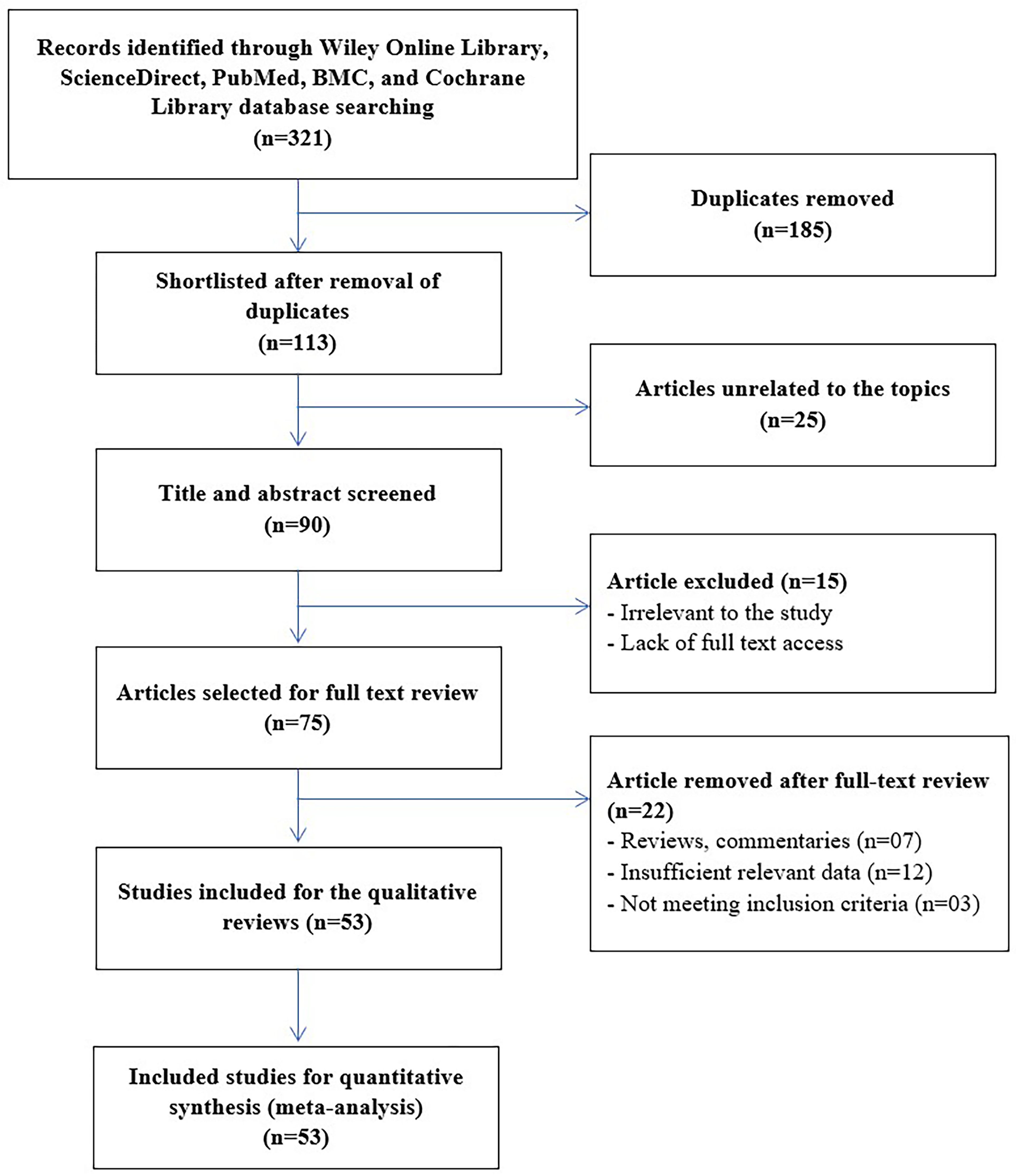
Study selection process according to PRISMA guidelines.
Characteristics of the selected studies for detecting the connection of CAT C262T (rs1001179) polymorphism with cancer.
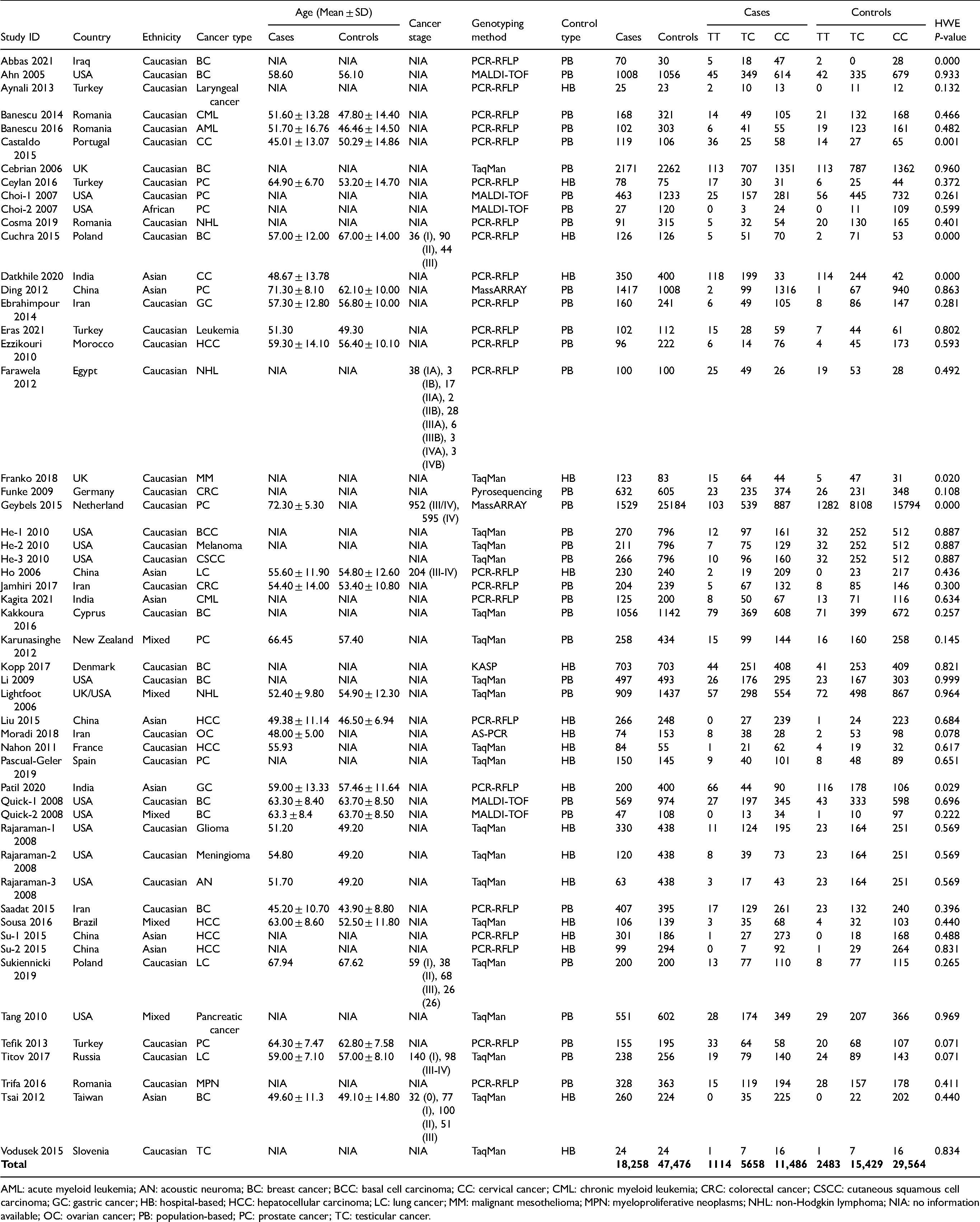
AML: acute myeloid leukemia; AN: acoustic neuroma; BC: breast cancer; BCC: basal cell carcinoma; CC: cervical cancer; CML: chronic myeloid leukemia; CRC: colorectal cancer; CSCC: cutaneous squamous cell carcinoma; GC: gastric cancer; HB: hospital-based; HCC: hepatocellular carcinoma; LC: lung cancer; MM: malignant mesothelioma; MPN: myeloproliferative neoplasms; NHL: non-Hodgkin lymphoma; NIA: no information available; OC: ovarian cancer; PB: population-based; PC: prostate cancer; TC: testicular cancer.
In this meta-analysis, 38 studies of Caucasians, 9 of Asians, and 6 of mixed and other populations were analyzed. The included studies were also categorized based on cancer as: 5 studies were blood-and-bone-marrow-related cancer (chronic myeloid leukemia + acute myeloid leukemia + leukemia + myeloproliferative neoplasms), 3 lung, 11 breast, 3 non-Hodgkin lymphoma, 4 gastrointestinal tract (GIT)-related (gastric cancer + colorectal cancer), 8 prostate, 4 head and neck cancer and central nervous system cancer (glioma + meningioma + acoustic neuroma + laryngeal cancer), 3 skin (basal cell carcinoma + melanoma + cutaneous squamous cell carcinoma), 3 gynecological (cervical cancer + ovarian cancer), 6 hepatocellular, and 3 other cancers (pancreatic cancer + testicular cancer + multiple myeloma). A total of seven different genotyping methods (polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP), MALDI-TOF, TaqMan, MassARRAY, pyrosequencing, kompetitive allele-specific PCR (KASP), and allele-specific-PCR (AS-PCR)) were used for the identification of polymorphisms in patients and controls. The sample sizes ranged from 24 to 2,171 in cases and from 23 to 25,184 in controls. The general properties of all studies are listed in Table 1.
According to the quality assessment tool (NOS), the maximum studies included had a score of more than 5 (high quality). Only one study was of moderate quality (score 5; Table S1).
Meta-analysis on overall cancer and ethnicity
Our findings demonstrated that the CAT C262T SNP was significantly linked with higher cancer risk in three genotype models: COD2 (CC vs. TT: OR = 1.16, CI = 1.03–1.32, P = 0.018), COD3 (CC vs. TC: OR = 1.21, CI = 1.11–1.33, P = 1.82 × 10−5), and RM (CC vs. TT + TC: OR = 1.20, CI = 1.10–1.31, P = 1.95 × 10−5) (Table 2, Figure 2). The connection between C262T variant and cancer risk in the Caucasian population was evaluated in 38 case-control studies with a number of 13,112 cancer patients and 41,436 controls. As shown in Table 2, COD2 (OR = 1.18, CI = 1.03–1.38, P = 0.027), COD3 (OR = 1.17, CI = 1.06–1.29, P = 0.003), and the ODM (OR = 1.19, CI = 1.04–1.36, P = 0.011) showed a significant link (P > 0.05) with cancer. In Asians, nine articles with 3,248 patients and 3,200 controls were assessed. Only one genetic model, COD3 (OR = 1.49, CI = 1.17−1.91, P = 0.002), showed a strong association with cancer. No statistically significant relationship was observed in any genetic model in the mixed and other populations, as shown in Table 2 and Figure S1.

Meta-analysis for detecting the connection of CAT C262T (rs1001179) polymorphism with overall cancer and ethnicity.
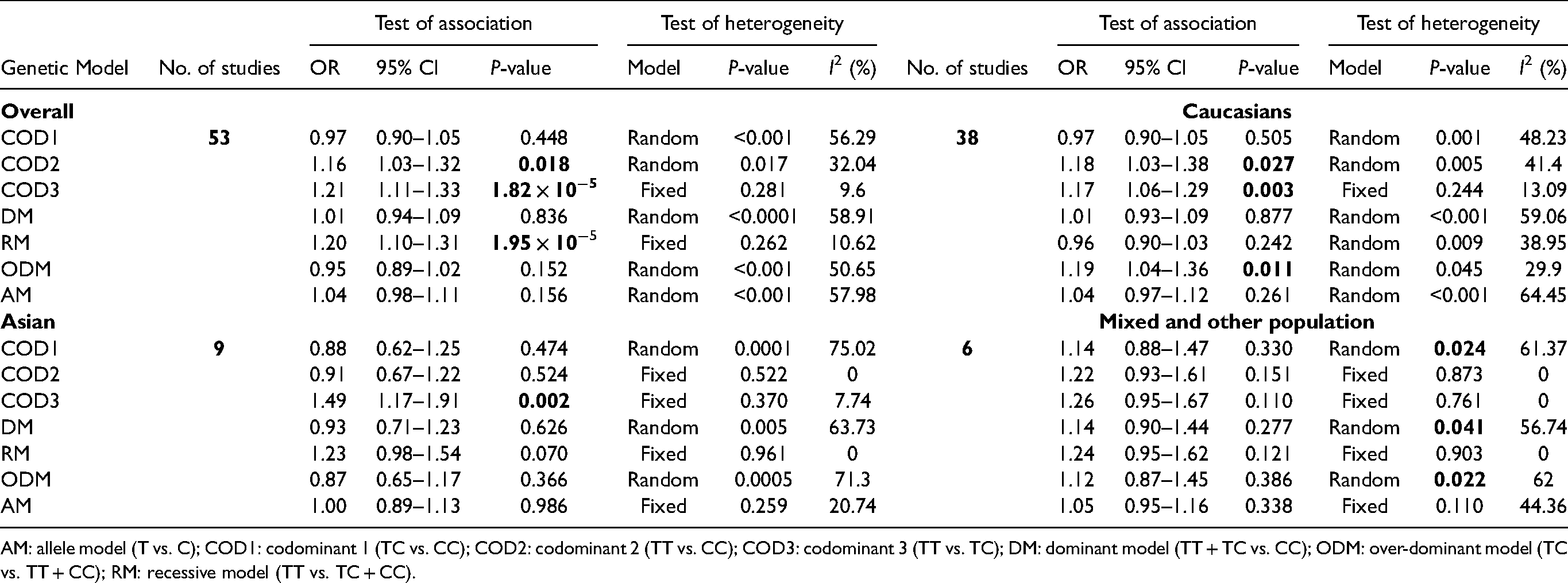
AM: allele model (T vs. C); COD1: codominant 1 (TC vs. CC); COD2: codominant 2 (TT vs. CC); COD3: codominant 3 (TT vs. TC); DM: dominant model (TT + TC vs. CC); ODM: over-dominant model (TC vs. TT + CC); RM: recessive model (TT vs. TC + CC).
Meta-analysis based on cancer types
Three genetic models, including COD1 (OR = 0.77, CI = 0.64−0.93, P = 0.008), DM (OR = 0.80, CI = 0.67−0.95, P = 0.013), and ODM (OR = 0.78, CI = 0.65−0.94, P = 0.009) showed a significant association between C262T SNP and blood-and-bone-marrow-related cancer. Only one genetic model, ODM (OR = 0.86, CI = 0.76−0.98, P = 0.023), showed a significant link between C262T and GIT-related cancer. The C26 T variant was also linked with the risk of prostate cancer in all genotypic models: COD1 (OR = 1.13, CI = 1.04−1.24, P = 0.005), COD2 (OR = 1.51, CI = 1.27−1.80, P = 3.15 × 10−6), COD3 (OR = 1.30, CI = 1.08–1.55, P = 0.005), RM (OR = 1.43, CI = 1.21−1.70, P = 3.85 × 10−5), ODM (OR = 1.09, CI = 1.00−1.19, P = 0.048), and AM (OR = 1.21, CI = 1.03–1.43, P = 0.021), except DM. Three genetic models, including COD1 (OR = 1.21, CI = 1.02−1.44, P = 0.033), DM (OR = 1.19, CI = 1.00−1.41, P = 0.044), and ODM (OR = 1.21, CI = 1.02−1.45, P = 0.032) revealed a substantial link with the risk of skin carcinoma. Our findings also revealed that the rs1001179 variant is significantly linked with gynecological malignancy in four genetic models: COD2 (OR = 2.94, CI = 1.06−8.16, P = 0.039), DM (OR = 1.73, CI = 1.01−2.97, P = 0.045), RM (OR = 2.51, CI = 1.01−6.19, P = 0.047), and AM (OR = 1.73, CI = 1.04–2.88, P = 0.036) (Table 3, Figure S2).
Meta-analysis for detecting the connection of CAT C262T (rs1001179) polymorphism with different cancer subtypes.
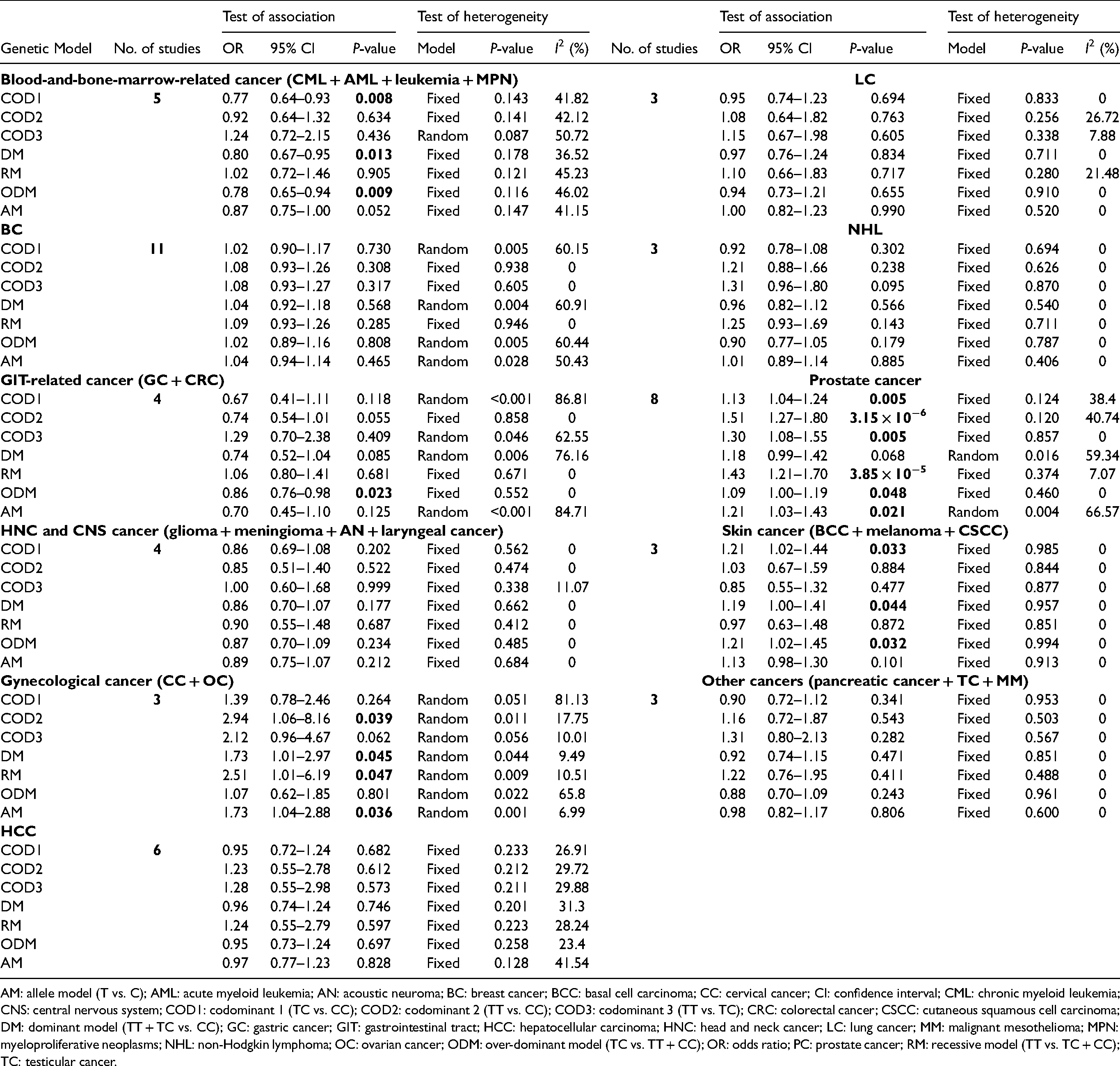
AM: allele model (T vs. C); AML: acute myeloid leukemia; AN: acoustic neuroma; BC: breast cancer; BCC: basal cell carcinoma; CC: cervical cancer; CI: confidence interval; CML: chronic myeloid leukemia; CNS: central nervous system; COD1: codominant 1 (TC vs. CC); COD2: codominant 2 (TT vs. CC); COD3: codominant 3 (TT vs. TC); CRC: colorectal cancer; CSCC: cutaneous squamous cell carcinoma; DM: dominant model (TT + TC vs. CC); GC: gastric cancer; GIT: gastrointestinal tract; HCC: hepatocellular carcinoma; HNC: head and neck cancer; LC: lung cancer; MM: malignant mesothelioma; MPN: myeloproliferative neoplasms; NHL: non-Hodgkin lymphoma; OC: ovarian cancer; ODM: over-dominant model (TC vs. TT + CC); OR: odds ratio; PC: prostate cancer; RM: recessive model (TT vs. TC + CC); TC: testicular cancer.
Meta-analysis based on control sources
The connection between the CAT C262T variant with cancer, depending on population-based control sources, was evaluated in 33 studies with 14,546 cancer patients and 42,644 controls. Three genetic models showed a link between the C262T SNP and the risk of cancer in COD2 (OR = 1.19, CI = 1.08−1.32, P = 0.0003), COD3 (OR = 1.17, CI = 1.06−1.29, P = 0.003), and RM (OR = 1.19, CI = 1.08−1.31, P = 0.0004). In contrast, an analysis of hospital-based control sources was evaluated in 20 studies with 3,712 cancer patients and 4,832 controls, which revealed that two genetic models, COD3 (OR = 1.40, CI = 1.16−1.70, P = 0.0005) and RM (OR = 1.24, CI = 1.04−1.48, P = 0.018), were correlated with the risk of cancer (Table S2).
Meta-analysis based on genotyping methods
Subgroup analysis depending on the genotyping methods (Table S3) showed that three genetic association models, including COD3, RM, and ODM were significantly associated with cancer risk for PCR-RFLP from 23 studies (OR = 1.46, CI = 1.15−1.86, P = 0.002, OR = 1.34, CI = 1.08−1.67, P = 0.009, and OR = 0.80, CI = 0.70−0.93, P = 0.003, respectively) and three genetic association models—COD2, RM, and AM—were significantly associated with cancer risk for MALDI-TOF + MassARRAY from 7 studies (OR = 1.32, CI = 1.11−1.56, P = 0.001, OR = 1.26, CI = 1.07−1.49, P = 0.005, and OR = 1.14, CI = 1.07−1.21, P = 5.68 × 10−5, respectively). No statistically significant correlation was found for TaqMan (20 studies) and other genotyping methods (3 studies).
Heterogeneity and publication bias analysis
From the analysis, we identified that different genetic models showed significant heterogeneity (P < 0.1) in which random-effects models were implemented (Table 2, Table 3, Table S2, Table S3). In our meta-analysis, we used both Begg's funnel plot and Egger's test to investigate the publication bias, which revealed that there was no bias (Table S4). A similar result is shown in the funnel plots of Figure S3.
Meta-regression and sensitivity analysis
Based on the quality scores of studies as a source of heterogeneity, we performed a meta-regression as shown in Table S5. The analysis showed that the quality scores might be a source of significant heterogeneity under the COD2 genetic model (coefficient = 0.176, CI = 0.018–0.335, P = 0.029). During the sensitivity study, individual papers in the meta-analysis were removed one by one to see whether individual data changed the pooled ORs; our results were found to be consistent among all genetic models (Figure S4).
TSA
We performed a TSA for CAT C262T in the allele models for the overall study, Caucasian, Asian, and mixed and other populations as depicted in Figure S5(a) to (d) with a sample size of 26,251, 30,517, 7,403, and 6,367 subjects, respectively. As per the analysis, the cumulative Z-curve (indicated by the blue line) exceeded the futility boundaries (indicated by the red line) in the overall, Caucasians, and Asians, and the total sample were firm enough to validate our results. Therefore, our findings were sufficient, and no further analysis was required for validation.
Discussion
Cancer is the most severe public health problem in the present world and the second-highest cause of mortality in this century. In this meta-analysis, we looked at 53 case-control studies with 65,734 samples (18,258 cases and 47,476 controls) from 18 countries using seven genetic models and found that there is a link between the CAT C262T variant and the risk of various cancers.
External variables, such as ROS or ionizing radiation, build up over time and may impact DNA expression in hematopoietic stem cells by causing double strand breaks. The CAT gene has many SNPs, including rs7943316, rs769214, and rs1001179.59,60 In the regulatory regions of the CAT gene, a C → T mutation at nucleotide −262 in the 5′ non-coding region is a common polymorphism (C262T/ rs1001179). This variant within the promoter region has been demonstrated to alter transcription factor binding. As a result, the CAT gene's transcription and expression are altered. CAT C262T polymorphism is linked not only to cancer, but also to many other disorders.61,63 In the present meta-analysis, three genotype models, namely COD2 (OR = 1.16, P = 0.018), COD3 (OR = 1.21, P = 1.82 × 10−5), and RM (OR = 1.20, P = 1.95 × 10−5) showed a statistically significant association with cancer. Also, a significant association was found between the C262T variant and cancer in three genotypic models of the Caucasian population (COD2: OR = 1.18, P = 0.027; COD3: OR = 1.17, P = 0.003; ODM: OR = 1.19, P = 0.011) and one genotypic model of the Asian population (COD3: OR = 1.49, P = 0.002).
Significant amount of oxygen radicals trigger the destruction of cellular structures and functions, as well as the death of a cell. As a result, ROS-induced DNA damage in cells can cause aberrant genetic changes in cells, which can contribute to cancer initiation and progression. The CAT gene is essential in the neutralization of reactive species and is involved in antioxidant defense systems. A study in Romania demonstrated that the CAT C262T polymorphism is correlated with chronic myeloid leukemia (OR = 0.59, P = 0.01) 41 ; however, another study found no significant association with the CAT −21A/T (P = 0.037) polymorphism with the risk of CML 45 and leukemia. 44 In our meta-analysis, we found significant associations between the CAT C262T polymorphism and blood-and-bone-marrow-related cancer in three genetic models (COD1: OR = 0.77, P = 0.008; DM: OR = 0.80, P = 0.013; ODM: OR = 0.78, P = 0.009) and skin cancer in three genetic models (COD1: OR = 1.21, P = 0.033; DM: OR = 1.19, P = 0.044; ODM: OR = 1.21, P = 0.032). Previous research also revealed a link between the C262T variant and a higher risk of skin cancer (CT + TT vs. CC: OR = 1.19, P = 0.04; CT vs. CC: OR = 1.21, P = 0.03). 23 Several studies have found no link between the C262T variant and the incidence of gastric 36 and colorectal cancer.37,49 In our meta-analysis, we found that only one genetic model (ODM: OR = 0.86, P = 0.023) was significantly connected with GIT-related cancer. Populations from Europe, Australia, New Zealand, and North America have the greatest rates of colon cancer, with Hungary and Norway ranked top for men and women, respectively. 1
According to Liu et al. 2016, CAT gene polymorphisms are related to an elevated risk of prostate cancer. 64 The TT genotype of C262T showed a considerably higher prostate cancer risk than the CC genotype. When compared to the CC genotype, the TT genotype exhibited 1.94- and 3.83-fold higher susceptibility to disease and malignancy, respectively. 31 A significant association was also reported by another study conducted in New Zealand. 25 In our meta-analysis, we found that six genetic models of C262T (COD1: OR = 1.13, P = 0.005; COD2: OR = 1.51, P = 3.15 × 10−6; COD3: OR = 1.30, P = 0.005; RM: OR = 1.43, P = 3.85 × 10−5; ODM: OR = 1.09, P = 0.048; AM: OR = 1.21, P = 0.021) have a significant connection with prostate cancer. According to a study conducted in Portugal, the C262T polymorphism showed a higher risk of cervical cancer (OR = 3.03, P = 0.003). 9 In our meta-analysis, we found that four genetic models (COD2: OR = 2.94, P = 0.039; DM: OR = 1.73, P = 0.045; RM: OR = 2.51, P = 0.047; AM: OR = 1.73, P = 0.036) have a significant connection with gynecologic cancer.
Different studies revealed that genetic variations in the CAT gene were not associated with lung cancer, 24 breast cancer,26,29 acoustic neuroma, 28 malignant mesothelioma, 17 hepatocellular carcinoma, 20 and non-Hodgkin lymphoma.21,33 However, we did not find any significant risk of the CAT C262T polymorphism and lung cancer, non-Hodgkin lymphoma, head and neck cancer, central nervous system cancer, breast cancer, and hepatocellular carcinoma. Since the late 1970s, the case-fatality percentages of hepatocellular carcinoma have dropped in many Eastern and South-East Asian nations, including China, Taiwan, the Republic of Korea, and the Philippines. 65 In our meta-analysis, we also found that three genetic models (COD2: OR = 1.19, P = 0.0003; COD3: OR = 1.17, P = 0.003; RM: OR = 1.19, P = 0.0004) in population-based controls and two genetic models (COD3: OR = 1.40, P = 0.0005; RM: OR = 1.24, P = 0.018) in hospital-based controls have a statistically significant connection with cancer. In addition, three genetic models for PCR-RFLP (COD3: OR = 1.46, P = 0.002; RM: OR = 1.34, P = 0.009; ODM: OR = 0.80, P = 0.003) and three models for MALDI-TOF + MassARRAY (COD2: OR = 1.32, P = 0.001; RM: OR = 1.26, P = 0.005; AM: OR = 1.14, P = 5.68 × 10−5) genotyping methods showed a significant association with cancer progression.
The consistency and repeatability of our results across all of the genetic models confirmed their validity. We used heterogeneity analysis, publication bias assessment, meta-regression, and sensitivity analyses to conduct our meta-analysis. Also, the TSA results suggest that no additional studies are required to validate our findings. Despite the fact that we approached this meta-analysis with caution, there are several drawbacks that must be noted. First, only published case-control studies were included. Second, the total number of studies included in the analysis is rather limited (53), despite a large number of patients (cases + controls = 49,027). Third, we stratified studies only based on ethnicity, cancer type, genotyping methods, and source of control—no other characteristics. With the exception of these limitations, the included case-control publications are of excellent quality, the analysis of the data is robust, and the findings of this study are acceptable.
Conclusion
In summary, our findings support the hypothesis that the CAT C262T polymorphism is significantly associated with overall cancer, especially in Caucasians and Asians. This variant may also be associated with other cancers such as blood-and-bone-marrow-related, GIT-related, prostate, skin, and gynecological cancers. To confirm the risk identified in the current statistical analysis, large-scale investigations should be undertaken in the future.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155221104128 - Supplemental material for Catalase C262T genetic variation and cancer susceptibility: A comprehensive meta-analysis with meta-regression and trial sequential analysis
Supplemental material, sj-docx-1-jbm-10.1177_03936155221104128 for Catalase C262T genetic variation and cancer susceptibility: A comprehensive meta-analysis with meta-regression and trial sequential analysis by Md Abdul Barek, Sarah Jafrin, Md. Abdul Aziz and Mohammad Safiqul Islam in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155221104128 - Supplemental material for Catalase C262T genetic variation and cancer susceptibility: A comprehensive meta-analysis with meta-regression and trial sequential analysis
Supplemental material, sj-docx-2-jbm-10.1177_03936155221104128 for Catalase C262T genetic variation and cancer susceptibility: A comprehensive meta-analysis with meta-regression and trial sequential analysis by Md Abdul Barek, Sarah Jafrin, Md. Abdul Aziz and Mohammad Safiqul Islam in The International Journal of Biological Markers
Footnotes
Acknowledgements
Not applicable.
Ethical approval
Not applicable.
This work received no funding.
Data availability
All data related to the manuscript were added in the manuscript main file, figures, tables and supplementary materials. The corresponding author will provide additional information on a valid request if required.
Author contributions
The literature search and data collection were performed by Md Abdul Barek and Sarah Jafrin. Mohammad Safiqul Islam performed the statistical analyses for this meta-analysis. The initial draft of the manuscript was co-written by Md Abdul Barek and Md. Abdul Aziz. Mohammad Safiqul Islam reworked the manuscript, critically updated it for important intellectual substance, and made substantial contributions to conceptualization and design. The paper was read and approved for submission by all the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
