Abstract
Introduction
Clusterin (CLU) is a pleiotropic protein with numerous functions. It has recently attracted considerable attention owing to its association with cancer progression and metastasis. However, its role in hepatocellular carcinoma (HCC) has not been investigated. This meta-analysis is the first evaluation of the diagnostic performance of CLU in HCC.
Methods
Articles published in PubMed, EMBASE, Web of Science, Wanfang Data Knowledge Service Platform, and China Science and Technology Journal Database until January 2022 were searched. Studies that reported the usefulness of CLU for the differentiation of HCC and non-HCC (e.g., liver cirrhosis, chronic hepatitis, and other benign liver disease) patients were selected. Alpha-fetoprotein (AFP) was used as a positive control in this study. The sensitivity, specificity, diagnostic odds ratio (DOR), and area under the curve (AUC) were compared between CLU and AFP.
Results
Eight articles including 811 participants were included. The pooled sensitivity (95% confidence interval (CI)), specificity (95% CI), DOR (95% CI), and AUC (95% CI) were: 0.86 (0.78–0.91), 0.85 (0.75–0.91), 35 (13–94), and 0.92 (0.89–0.94) for CLU; 0.74 (0.67–0.81), 0.89 (0.79–0.94), 22 (8–61), and 0.87 (0.84–0.90) for AFP; 0.93 (0.88–0.96), 0.85 (0.68–0.94), 75 (21–262), and 0.95 (0.92–0.96) for CLU + AFP, respectively. Compared with AFP, CLU showed higher sensitivity, DOR, and AUC, as well as similar specificity. The combination of CLU and AFP resulted in higher sensitivity, DOR, and AUC.
Conclusions
Serum CLU is a better biomarker versus AFP for the diagnosis of HCC. The combination of CLU and AFP improved diagnostic performance.
Introduction
Liver cancer is a common malignancy worldwide; it is estimated that by the year 2025 >1 million individuals will be affected by liver cancer annually. 1 It is also one of the leading causes of cancer-related death worldwide, and the second most lethal tumor type with a 5-year survival rate of 18% in the USA and 12% in China.2,3 Hepatocellular carcinoma (HCC)—the most common form of primary liver cancer—accounts for 90% of cases. 1 It typically develops from chronic liver disease, due to hepatitis B virus infection, hepatitis C virus infection, and non-alcoholic fatty liver disease. 4 Patients diagnosed with HCC at an early stage may occasionally be cured through surgery or transplantation. However, in cases of diagnosis at an advanced stage, tumor cells may have spread either to the lymph nodes or to other organs, thereby complicating resection by surgery. Although patients with advanced disease cannot be cured, appropriate treatment and support can prolong their lives and improve their quality of life. 5 Therefore, there is an urgent need for an early and accurate diagnosis of HCC.
Numerous tests are used for the diagnosis of HCC. 6 The current methods utilized for the diagnosis of HCC in clinical practice include blood testing, ultrasound, imaging, and pathological diagnosis.7,9 Analysis of biomarkers through blood testing offers more convenient, economical, and rapid diagnosis than other examinations. Alpha-fetoprotein (AFP) is the most frequently used biomarker in this setting. 10 Elevated levels of AFP in the blood have been found in approximately 60–70% of patients with HCC. 11 Additionally, a combination of AFP and other methods has also been employed for the diagnosis of HCC. A meta-analysis concluded that the combination of AFP and ultrasound examination can also serve as a diagnostic approach. 12 However, the diagnostic performance of this combination is limited by low sensitivity and specificity. Apart from AFP, numerous other biomarkers are also used for screening, such as long non-coding RNAs, transforming growth factor β1, midkine, alpha-L-fucosidase, and des-γ-carboxyprothrombin.13,16
Clusterin (CLU) is a multifunctional secretory glycoprotein; it was discovered approximately 40 years ago in ram rete testis as a protein that causes erythrocyte clustering.17,18 Researchers revealed that this heat-stable, trypsin-sensitive protein was in charge of cell aggregates; hence, this extracellular molecule was termed “clusterin”. CLU can be found in nearly all body fluids, such as milk, plasma, and cerebrospinal fluid. 18 It modulates the cell–cell and/or cell-matrix interactions, and performs a variety of functions (e.g., lipoprotein transport, survival/apoptosis regulation, inhibition of complement-mediated cell lysis, and tumorigenesis).19,20 Recently, several studies have reported the potential oncogenic role of CLU in the development/progression of several types of cancer. Bertuzzi et al. 21 showed that detection of plasma CLU might improve the identification rate of people at risk of colorectal cancer. Shabayek et al. 22 reported that combining of angiogenin and CLU could achieve an excellent performance in the diagnosis of bladder cancer. Panico et al. 23 found the prognostic role of CLU in resected adenocarcinomas of the lung. The expression of CLU is higher in prostate, renal cell, lung, and hepatocellular cancer.24,28 However, due to the low number of cases analyzed, these studies yielded inconsistent results. Moreover, the diagnostic performance of CLU in HCC has not been elucidated. In this meta-analysis, we conducted the first systematic analysis of the currently available relevant literature. The objective was to evaluate the diagnostic potential of CLU for the detection of HCC and to provide insight into the clinical application of this approach.
Materials and methods
This meta-analysis was performed according to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 29
Literature search
We searched mainstream databases: PubMed, Embase, Web of Science, Wanfang Data Knowledge Service Platform, and China Science and Technology Journal Database for relevant research studies published until January 2022. There were no restrictions set regarding the language and year of publication. The following keywords and their synonyms were searched in multiple combinations: “liver”, “neoplasm”, “carcinoma”, “hepatocellular”, “clusterin”, “CLU”, “diagnosis”, “SGP-2”, “SP40-40”, “TRPM-2”, “ApoJ”, and “diagnostic marker”. The article titles and abstracts were screened, and irrelevant articles were excluded.
Inclusion and exclusion criteria
The inclusion criteria were: (a) patients with confirmed HCC; (b) non-HCC participants, including patients with benign liver disease, patients with liver cirrhosis, or healthy individuals (control group); (c) measurement of serum levels of CLU performed prior to treatment; (d) availability of sufficient data for the extraction or calculation of true positive (TP), false positive (FP), true negative (TN), false negative (FN) to generate the 2 × 2 contingency tables. The exclusion criteria were: (a) lack of relevant data (i.e., 2 × 2 contingency tables could not be found or calculated from the article); (b) case reports; (c) incomplete and unpublished research reports; (d) studies performed on cell lines or experimental animal models; and (e) studies investigating other diseases.
Data extraction
First, information was extracted from each publication (i.e., first author's name, date of publication, countries, sample type, number of participants, sex, age, control type, detection methods, and cutoff values used for defined positives of CLU). Second, diagnostic performance data were extracted, including TP, FP, TN, FN, specificity, sensitivity, negative likelihood ratio (NLR), and positive likelihood ratio (PLR). The Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) tool was used to assess the quality of included studies from two main aspects: the risk of bias and applicability of primary diagnostic accuracy studies. 30 Answers to questions regarding the risk of bias were “yes,” “no,” or “unclear.” Items concerning the applicability assessment were labeled as “high,” “low,” or “unclear.”
Statistical analysis
The 2 × 2 contingency tables were used to calculated the sensitivity, specificity, PLR, NLR, and diagnostic odds ratio (DOR) with the corresponding 95% confidence interval (CI). The PLR denotes the ratio of TP to FP, while NLR represents the ratio of FN to TN. Higher PLR and lower NLR values were indicative of better diagnostic performance. DOR is the ratio of PLR to NLR, reflecting the degree of association between CLU and HCC. Values >1 denoted better diagnostic performance of CLU. Additionally, summary receiver operating characteristics (SROC) curves were constructed using data from the contingency tables. Furthermore, the area under the curve (AUC) for each SROC curve was calculated for the assessment of diagnostic performance. The data were analyzed using Stata Version 15.1, and data pooling was performed using a random-effects model due to the expected heterogeneity. Significant heterogeneity was indicated by an I2 >50% and a P-value of 0.05. Meta-regression was utilized to test whether non-threshold effects were the cause of heterogeneity. A P-value <0.05 denoted statistically significant differences. We also performed a series of predetermined subgroup analyses based on the number of participants, country, and type of control. Deeks’ funnel plot was performed to check for publication bias.
Results
Study selection and characteristics
The search yielded 69, 93, 45, 58, and 5 publications from PubMed, Embase, Web of Science, Wanfang Data Knowledge Service Platform, and China Science and Technology Journal Database, respectively. After excluding duplicate documents, 166 potentially relevant publications were found. Following a preliminary screening of titles and abstracts, 15 publications satisfied the inclusion criteria. After evaluating the full text content, analyzing the article content and data integrity according to the exclusion criteria, and excluding relevant literature, 8 studies were finally included in this meta-analysis (Supplementary Figure 1).31,38 The studies were conducted in Egypt and China. The attributes of the studies are listed in Table 1, and the raw data of the contingency table are listed in Supplementary Table 1. These studies also evaluated the diagnostic performance of AFP; however, only four studies analyzed the combined diagnostic potential of CLU and AFP. These studies involved 518 patients with HCC and 293 control participants (223 patients with liver cirrhosis, 60 with chronic hepatitis, and 10 healthy individuals). For the quantification of the expression levels of CLU and AFP, all researchers used enzyme-linked immunosorbent assay. All studies reported the diagnostic performance of AFP in HCC, and four studies investigated the diagnostic accuracy of the combination of CLU and AFP in this setting.
Main characteristics of included studies.
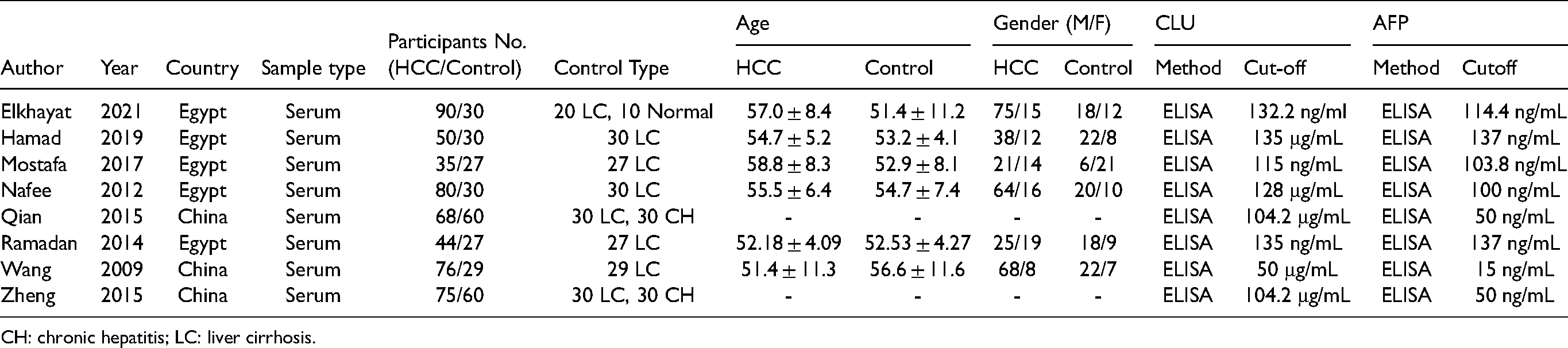
CH: chronic hepatitis; LC: liver cirrhosis.
Quality evaluation
The results of the QUADAS-2 analysis are illustrated in Supplementary Figure 2. In the “risk of bias” part, the major concerns were “patient selection” and “index test.” In the “patient selection” section, the high risk of bias was due to the case-control design, regardless of random or consecutive sample collection. Most of the included studies were case-control studies, in which patients with confirmed HCC instead of suspected patients were enrolled. For the “index test” section, studies were evaluated as “high risk” or “unclear” due to inexact reporting or unmentioned pre-specified threshold for diagnosis. The risk of bias in the other two sections was low. Most of the studies showed low risk for the “applicability concerns” domains. Therefore, there were no important concerns for the applicability.
Diagnostic performance
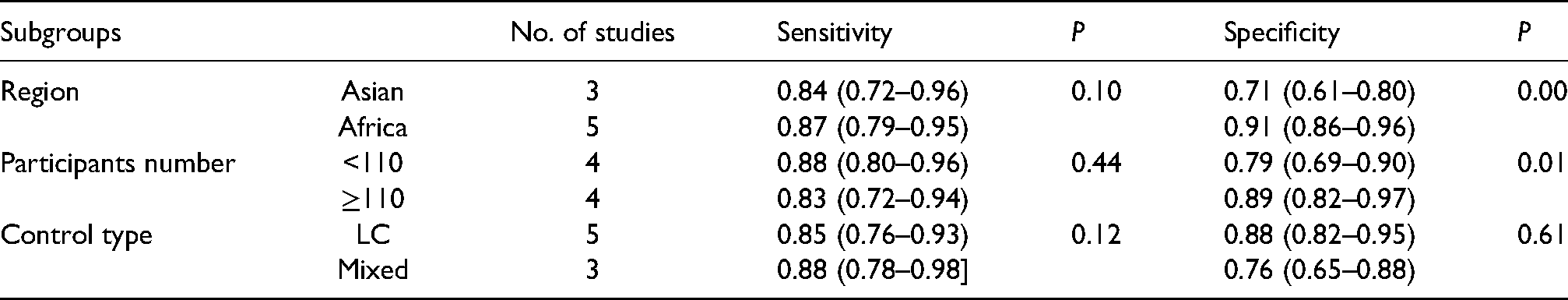
The random-effects model was suitable for data analysis due to substantial heterogeneity (All I2 >50, P = 0.00). In general, the sensitivity and specificity of CLU for the diagnosis of HCC in this study ranged from 0.70 to 0.96 (median: 0.86) and 0.67–0.97 (median: 0.88), respectively (Figure 1(a)). For AFP, these values were 0.59–0.90 (median: 0.75) and 0.75–1.00 (median: 0.86), respectively (Figure 1(b)). The pooled sensitivity and specificity of CLU for the detection of HCC were 0.84 (95% CI: 0.77–0.90) and 0.90 (95% CI: 0.81–0.95), respectively (Figure 1(a)). However, the pooled sensitivity and specificity of AFP were only 0.74 (95% CI: 0.67–0.81) and 0.89 (95% CI: 0.79–0.94), respectively (Figure 1(b)). The pooled PLR, NLR, and DOR for CLU were 5.8 (95% CI: 3.2–10.2), 0.17 (95% CI: 0.10–0.28), and 35 (95% CI: 13–94), respectively. For AFP, the pooled PLR, NLR, and DOR were 6.5 (95% CI: 3.2–13.1), 0.29 (95% CI: 0.21–0.41), and 22 (95% CI: 8–61), respectively (Supplementary Table 2). As shown in the SROC curves, the AUC values for CLU and AFP were 0.93 (95% CI: 0.90–0.95) and 0.87 (95% CI: 0.84–0.90), respectively (Figure 2). Collectively, the results suggested that CLU exhibited a relatively better performance to that of AFP in distinguishing HCC from non-HCC. The threshold effect test (Spearman's correlation coefficient: 0.59, P = 0.34, for CLU; Spearman's correlation coefficient: 1.00, P = 1.00, for AFP) suggested that the cutoff value was not the source of heterogeneity. According to the sensitivity analysis, the pooled estimates were stable and were not influenced by a single study (Figure 3). Subgroup analyses of CLU studies were additionally performed, and the summarized results are presented in Table 2. Furthermore, the Deeks’ symmetric funnel plot did not indicate publication bias in the selected studies (P = 0.94) (Supplementary Figure 3).
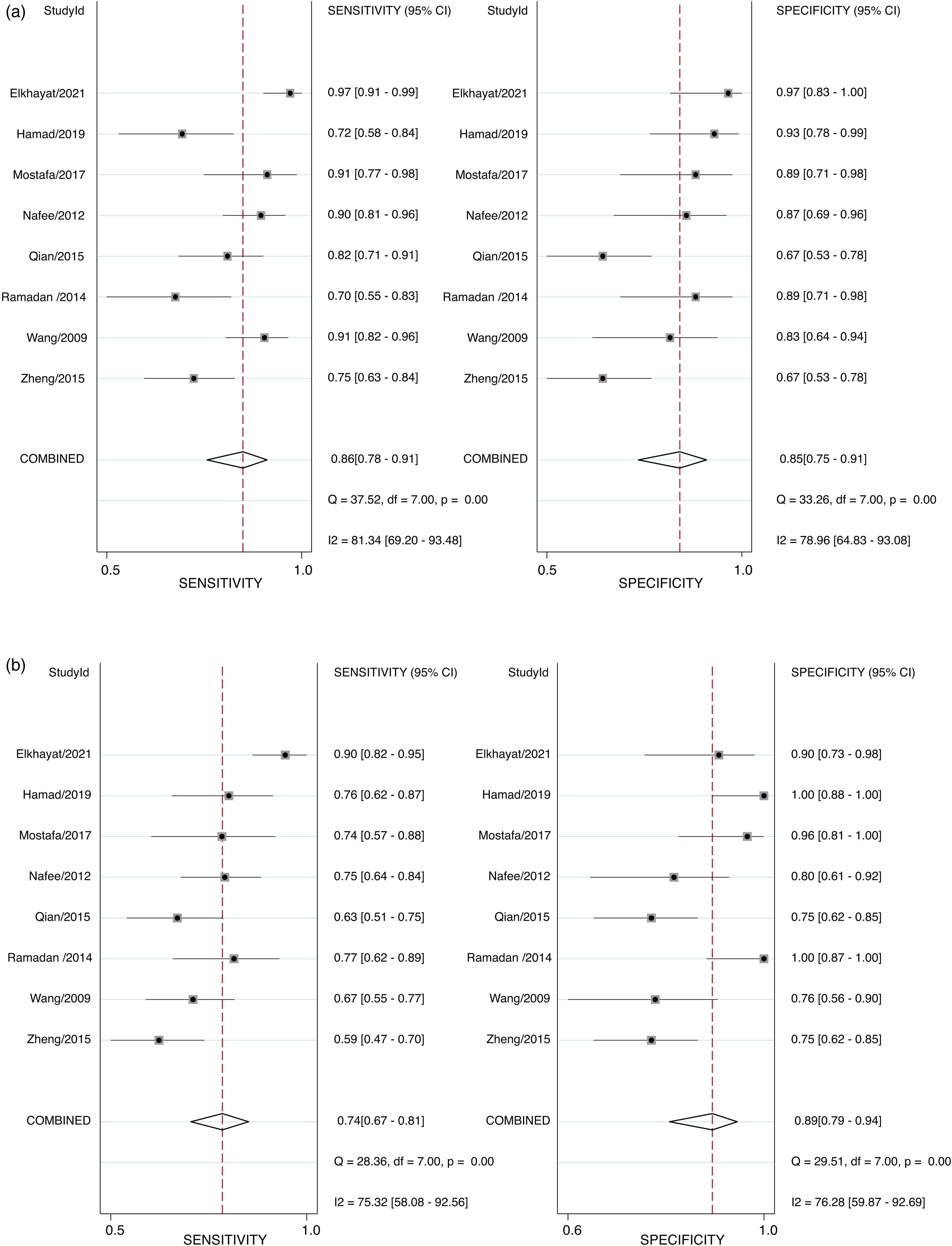
Forest plots for the sensitivity and specificity of CLU (a) and AFP (b) in the diagnosis of HCC.
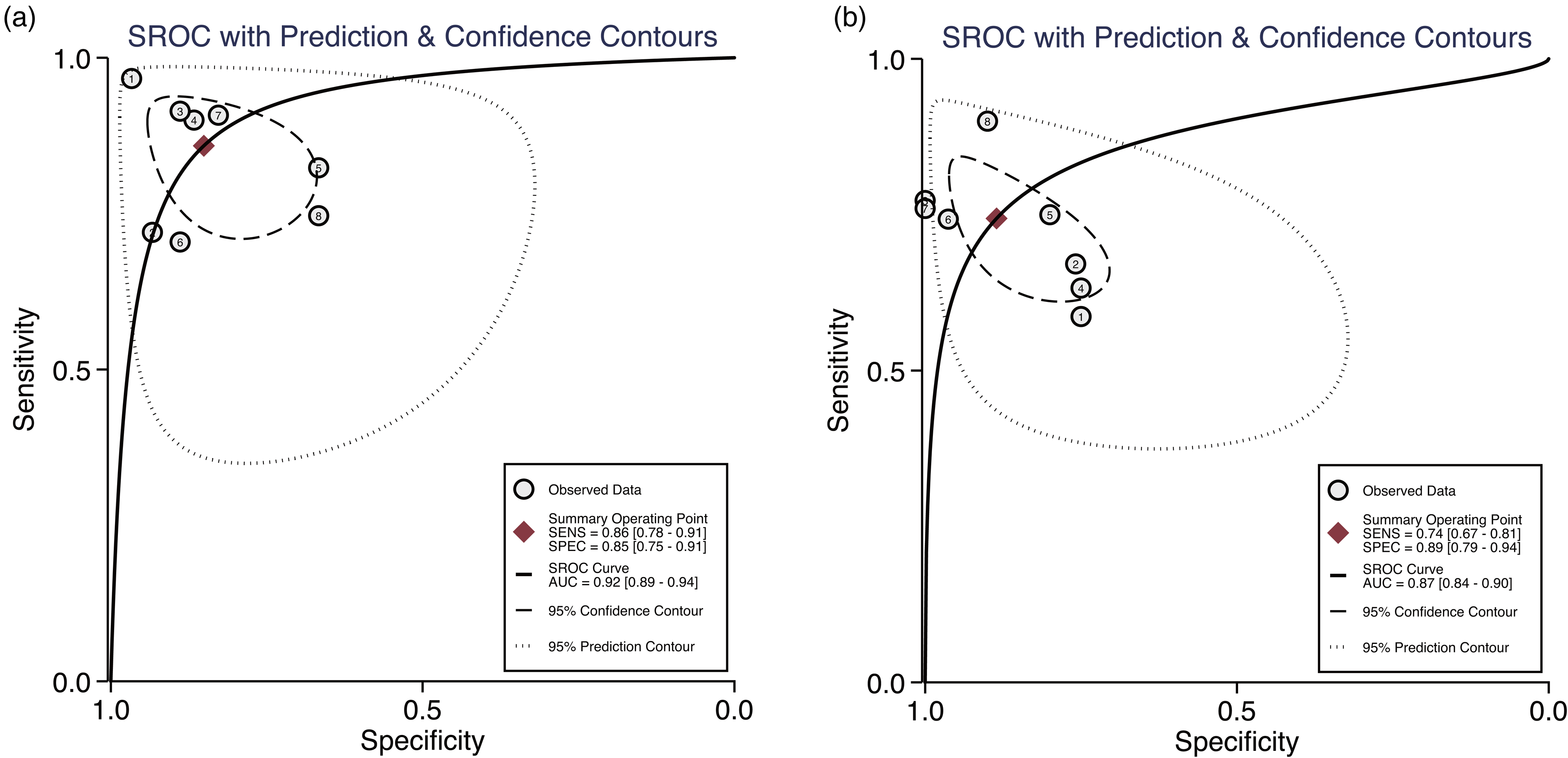
Summary receiving operator characteristics (SROC) curves for CLU in the diagnosis of HCC. Included studies are arranged in alphabetical order (A–Z) by the first letter of the first author.
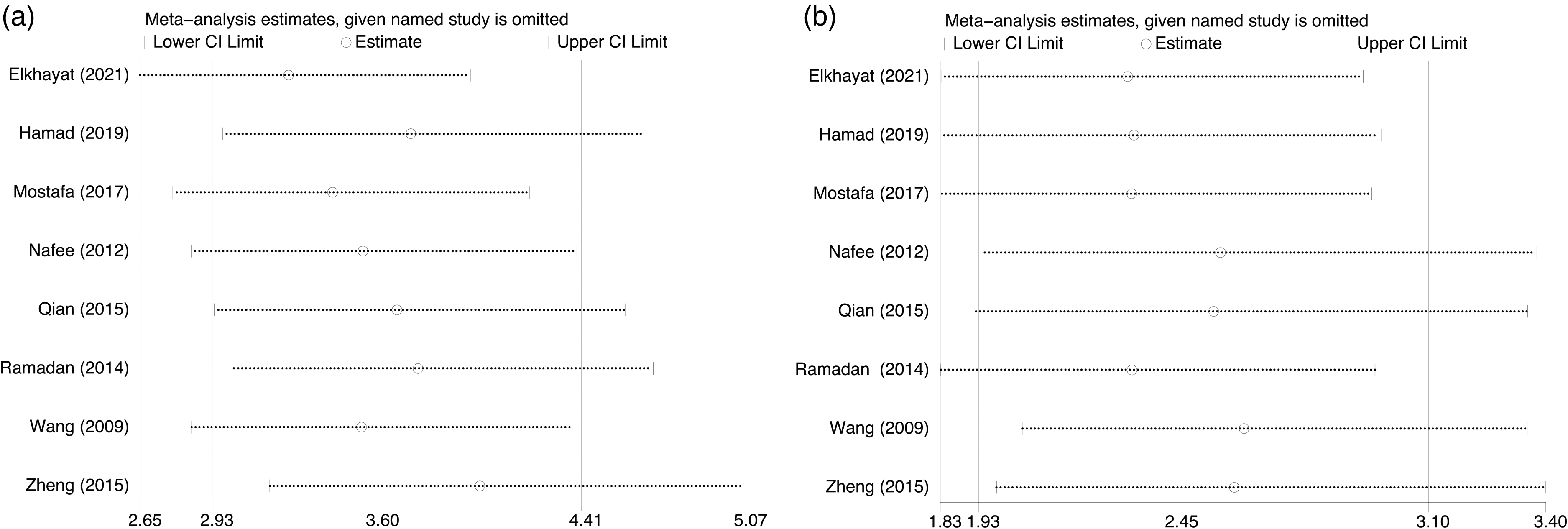
Sensitivity analysis plots for CLU (a) and AFP (b).
Results of subgroup and meta-regression analysis.
Discussion
HCC is a major cause of cancer-related mortality in some regions of the world, such as South Asia and North Africa. This is consistent with the country distribution observed in this study. 39 Apart from AFP, there are several potential serum biomarkers. We compared the diagnostic performance of CLU and AFP in this study. CLU is a glycoprotein that is highly conserved and present in various tissues. CLU has been linked to a variety of physiological processes, including cholesterol transport, cell adhesion, cell–cell/cell–substratum interactions, tissue remodeling, promotion or inhibition of apoptosis, and immune system regulation. 40 It is also found in exosomes and contributes to the survival of cancer cells in distant locations, which is required for tumor metastasis. Therefore, CLU may play key roles in carcinogenesis and cancer invasion. Studies have shown that CLU is overexpressed in several metastatic cancer cells, including bladder cancer, colon cancer, HCC, and renal cell carcinoma.24–27 However, the use of CLU in HCC is currently limited. Although previous reports have highlighted the potential of serum CLU in predicting disease recurrence in patients with HCC, its stand-alone application has not been recommended by international guidelines. Of note, the diagnostic significance of CLU in HCC has not been determined. In the present study, we conducted a literature review for the potential application of serum CLU in the detection of HCC.
This meta-analysis included data from eight studies. Notably, 77.6% of patients with HCC were males. This finding is consistent with the results reported by El-Zayadi et al., 41 showing that males are more likely to suffer from HCC than females. The authors proposed that estrogens and androgens could modulate hepatocellular carcinogenesis and males are at a higher risk. In all included studies, the serum levels of CLU were higher in the HCC group versus the liver cirrhosis group, implying a role in carcinogenesis. Approximately 37.5% (3/8) of the included studies were performed in Asian countries, while others were conducted in Egypt. This is consistent with epidemiological trends related to geographic locations, which show that Eastern and South-Eastern Asia, as well as almost all of sub-Saharan Africa, are characterized by higher HCC incidences and mortality rates. 39 In this analysis, we used AFP as a positive control for comparison with CLU in the diagnosis of HCC. We found that the sensitivity, DOR, and AUC of CLU were higher than those of AFP. These findings indicated that the diagnostic value of CLU was better to that of AFP. The combination of CLU and AFP has been evaluated in four studies, demonstrating improvement in diagnostic accuracy for HCC. Many of the articles (4/8) included in the present analysis did not evaluate the combined use of biomarkers; thus, further research on the combined use of biomarkers for the diagnosis of HCC is warranted.
To our knowledge, this is the first meta-analysis of the diagnostic accuracy of CLU in patients with HCC. We conducted the present systematic review in accordance with the PRISMA guidelines to ensure the internal validity of our conclusions. To avoid selection bias, a thorough search of online public databases without language restrictions was conducted. QUADAS-2, an accurate tool for diagnostic meta-analyses, was used by two investigators (G.G. and X.L.) to independently extract data and assess the quality of studies. A random-effects model was used to synthesize the raw data.
Limitations
First, despite a thorough search, only eight eligible studies were found. Quality assessment revealed high or unclear bias risk for these studies. The major reasons responsible for these risks were suboptimal study designs, unmentioned pre-specified threshold for diagnosis, and the lack of suspected patients. Second, only four studies had sufficient data for the evaluation of the diagnostic accuracy of combined CLU and AFP. Therefore, conducting a comparative study of the combined and individual diagnostic accuracies is difficult. The absence of AFP studies in the included literature, as well as the use of different cutoff values for AFP, may also weaken the robustness of our findings. Third, the complexity of the control group would weaken the accuracy of the specificity values. The enrollment of healthy individuals promotes specificity. However, participants with benign liver disease and HCC are required to distinguish patients. The difference in the expression levels of CLU between these two groups is relatively smaller than that noted between healthy individuals and patients with HCC. Fourth, due to the limited publications, the populations included in the present study were in two groups (Chinese and Egyptian). There is limited evidence for this study to investigate the diagnostic value of CLU for other populations. Also, there is insufficient data to evaluate whether geographic location and ethnicity influence the diagnostic accuracy of CLU. Therefore, additional or follow-on studies are encouraged for further confirmation.
Conclusion
This meta-analysis demonstrated that CLU has a higher diagnostic accuracy for HCC versus AFP. It is also recommended to combine CLU with other diagnostic biomarkers (e.g., AFP) for the diagnosis of HCC. Additional well-designed and larger studies are required to investigate the differential expression of CLU in blood samples obtained from patients with different degrees of liver cirrhosis, and to determine its cutoff value for patients with cirrhotic and non-cirrhotic liver cancer. Further investigation is warranted for a comprehensive evaluation of combined CLU and AFP as a screening option for the clinical investigation of HCC.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155221101206 - Supplemental material for Diagnostic performance of clusterin in hepatocellular carcinoma: A meta-analysis
Supplemental material, sj-docx-1-jbm-10.1177_03936155221101206 for Diagnostic performance of clusterin in hepatocellular carcinoma: A meta-analysis by Ge Gao and Xuke Luan in The International Journal of Biological Markers
Footnotes
Author contributions
Ge Gao and Xuke Luan contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
