Abstract
Objective
The objective of this study is to investigate the correlation between the expression levels of midkine (MDK) in the serum of patients with hepatocellular carcinoma (HCC) and various clinical features, and to evaluate the diagnostic efficacy of MDK in HCC cases that are negative for alpha-fetoprotein (AFP).
Methods
Serum samples from 330 patients were collected from electronic cases and divided into three groups: HCC, benign liver disease, and healthy people. Serum MDK levels in all three groups were detected by ELISA. Correlation analysis was conducted to evaluate the relationship between serum MDK and liver function indexes and other traditional tumor markers in the HCC group. The receiver operating characteristic curve was used to analyze the diagnostic utility of MDK in HCC and AFP-negative HCC. In addition, univariate and multivariate analyses were performed to determine the correlation between MDK level and tumor metastasis, and clinicopathological features.
Results
MDK is significantly elevated in negative HCC (P = 0.001). The serum expression level of MDK in patients with HCC was found to be positively correlated with various indicators of liver injury (P < 0.05). Notably, elevated MDK expression was significantly associated with the China liver cancer staging system (CNLC) stage (P = 0.003) and complications (P = 0.000). Furthermore, the CNLC stage significantly impacted patient survival and metastasis (P = 0.016). Specifically, we propose that high levels of MDK expression are closely linked to poor tumor prognosis.
Conclusion
The risk of tumor metastasis and the likelihood of poor prognosis in patients with HCC are significantly elevated in those exhibiting high levels of MDK. MDK could serve as a novel serum diagnostic marker for patients who are negative for AFP.
Introduction
Hepatocellular carcinoma (HCC) is a global health challenge, and is the fourth most prevalent and the second most deadly cancer in China.1,2 The tumorigenesis of HCC is a multifaceted process influenced by various risk factors, which are modulated by environmental, genetic, and immunological elements.3,4 According to statistical data, approximately 9% and 55% of HCC cases were initially diagnosed at the middle and late stages, respectively. 5 Poor progression in HCC patients is largely attributed to the fact that most cases are not diagnosed at an early stage. Delays in both diagnosis and treatment can lead to significant increases in recurrence and mortality rates. In other words, early diagnosis of HCC remains crucial for improving prognosis. In recent years, as public awareness of the severity and detrimental effects of HCC has grown, the concept of precision diagnostic medicine has gained traction. Consequently, new diagnostic methods have emerged, including novel serum markers, molecular diagnostics, and advancements in imaging techniques. 6 AFP-positive HCC (APHC) can be readily diagnosed through serum levels and characteristic imaging features of alpha-fetoprotein (AFP), a well-established biomarker for HCC. In contrast, the detection of AFP-negative HCC (ANHC) poses significant challenges due to the absence of ideal biomarkers. 7
Early diagnosis and accurate staging of liver disease are crucial in mitigating HCC-related mortality, as well as in decreasing healthcare expenditures. 8 The relationship between AFP and HCC is well-established; however, the sensitivity and specificity of AFP can be influenced by various factors, including the detection method employed, patient characteristics, and the severity of underlying liver disease. 9 In fact, a significant proportion of patients with HCC do not exhibit elevated levels of AFP, thereby diminishing the diagnostic significance of AFP. 10 Currently, numerous studies have been conducted to identify reliable biomarkers for the diagnosis of AFP-negative HCC. These potential biomarkers include des-gamma-carboxyprothrombin (DCP), alpha-fetoprotein L3(AFP-L3), microRNAs, heat shock protein 90 alpha (Hsp90α), and interleukin-41(IL41).7,10–14 Unfortunately, the data comparing DCP to AFP alone as screening tools are mixed. When AFP levels are low, AFP-L3 alone is relatively insensitive in diagnosing HCC. Research indicates that microRNAs could serve as highly effective biomarkers for both the diagnosis and prognosis of HCC. However, several issues warrant consideration. For instance, microRNAs, being part of exosomes, require multiple steps for extraction, which can be time-consuming and lack standardization in the separation process. The absence of precise definitions and specific indicators has led to confusion within this particular research field. Consequently, a lack of clear definitions and specific markers has resulted in ambiguity within this domain of study.15,16 Whether it is Hsp90α or IL-41 as emerging biomarkers, it is essential to consider the limitations regarding sensitivity and specificity when utilizing a single protein as a biomarker.14,17 Overall, the sensitivity of these markers in a screening context remains limited. Consequently, it is essential to employ a combination of various biomarkers to enhance the sensitivity, specificity, and predictive value beyond that of any single marker.18,19
Midkine (MDK), also known as neurite growth-promoting factor 2 (NEGF2), is a multifunctional secreted protein that serves as both a cytokine and a growth factor. It plays a crucial role in regulating various signaling pathways and is involved in fundamental cellular processes, including survival, proliferation, and migration. Studies have demonstrated that MDK is an effective biomarker for the early diagnosis of HCC. It not only differentiates HCC patients from healthy individuals but also distinguishes HCC patients from those with other liver diseases.20–22 Nevertheless, the diagnostic utility of MDK in identifying AFP-negative HCC remains inadequate. Considering these findings, we aimed to assess the diagnostic efficacy of MDK in patients diagnosed with AFP-negative HCC.
Methods
Patients and clinicopathological characteristics
All data were obtained from the electronic medical records at Guangxi International Zhuang Medical Hospital between the years 2021 and 2024. After excluding patients with other malignant tumors, serious cardiovascular or infectious diseases and lack of clinical data, we finally included 244 patients, including 163 patients with HCC and 81 with benign liver disease, where all participating patients received treatment. Finally, we collected 86 cases from healthy people as control group. Among the 163 patients diagnosed with HCC, 85 tested positive for AFP, and 78 tested negative. The cohort of patients with benign liver disease consisted primarily of individuals with fatty liver or liver abscesses. The study protocol was approved by the ethics committee of Guangxi International Zhuang Medical Hospital. Serum samples and patient medical records were collected by established medical research ethics guidelines. The information gathered includes patient demographics such as gender and age, lifestyle habits, classification according to the China HCC staging system (CNLC), family history of the disease, tumor characteristics including size and number, tumor stage, history of metastasis, as well as any complications related to the disease. All participants included in the data analysis underwent follow-up for a minimum duration of 3 years. In diagnosing HCC within this study population, an AFP level below 10 ng/mL is considered indicative of a negative result for the presence of malignancy.
Laboratory characteristics
Collect disease-related laboratory testing indicators, including quantitative test results for (HBV) virus DNA, des-gamma carboxyprothrombin (DCP), and five classic tumor markers: alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), carbohydrate antigen 199 (CA-199), carbohydrate antigen 153 (CA-153), and carbohydrate antigen 125 (CA-125). Liver function indices encompass serum total bilirubin (TBIL), direct bilirubin (DBIL), prealbumin (PA), total protein (TP), albumin (ALB), alanine aminotransferase (AST), aspartate aminotransferase (ALT), γ-glutamyltransferase (γ-GGT), alkaline phosphatase (ALP), total bile acid (TBA) and cholinesterase (CHE). Coagulation-related parameters include prothrombin time (PT) and activated partial thromboplastin time (APTT); thrombin time (TT); fibrinogen (FIB); and D-dimer. Hematological routine tests consist of white blood cells (WBC), red blood cells (RBC), hemoglobin (HGB), and platelets (PLT) among others. The laboratory instruments utilized comprise the Sysmex XN9000 automatic blood analysis assembly line, the Waffen ACL TOP 700 Las Automatic Coagulation Analyzer, and the Abbott Architect c16000 automatic biochemical analysis system.
Enzyme-linked immunosorbent assay
The serum concentration of human MDK was determined using the Human MDK enzyme-linked immunosorbent assay (ELISA) Kit. In this assay, the plate is first coated with an anti-human MDK antibody, allowing the human MDK present in the sample or standard to bind to the coated antibody during incubation. Subsequently, biotinylated anti-human MDK antibodies and horseradish peroxidase-labeled avidin are added sequentially. The biotinylated antibodies specifically bind to the human MDK that is attached to the coated antibody, forming an immune complex as avidin binds selectively to biotin. Unbound components are then washed away. Following this step, a chromogenic substrate (TMB) is introduced; under the catalytic action of horseradish peroxidase, TMB develops a blue color which subsequently turns yellow upon the addition of a stop solution. The optical density (OD) value and corresponding MDK concentration are measured at a wavelength of 450 nm using an enzyme-linked instrument. Finally, the concentration of MDK in each sample is calculated by constructing a standard curve based on these measurements.
Statistical analysis
The Kolmogorov–Smirnov test is employed to assess the normality of data. Continuous variables in laboratory parameters are described using either the median (M) and interquartile range (IQR) or the mean (M) and SD. For demographic characteristics, absolute values and percentages are utilized to represent categorical variables. When comparing three groups of data, analysis of variance (ANOVA) is applied for pairwise comparisons of continuous variables that conform to a normal distribution, while the Kruskal–Wallis H test is used for analyzing other types of variables. To evaluate statistical differences between the two groups, appropriate methods such as the Student's t-test or Mann–Whitney U test are employed for quantitative data comparison. The chi-square test is utilized for gender comparisons among the studied groups. Spearman correlation coefficients are calculated to examine relationships between two variables. Both univariate and multivariate analyses are conducted to determine correlations between MDK levels and tumor metastasis as well as clinicopathological characteristics. The receiver operating characteristic (ROC) curve along with the area under the ROC curve (AUC) is generated to assess the diagnostic efficiency of MDK. Statistical analyses and graphical processing of all data were performed using SPSS 25, GraphPad Prism 5, and Origin Statistical Software for Windows. The P-value of less than 0.05 (two-tailed) was considered statistically significant.
Results
MDK is significantly elevated in the serum of AFP-negative HCC patients
A total of 330 patients were included in this study, comprising 163 patients with hepatocellular carcinoma, 81 with benign liver disease, and 86 healthy controls. The clinical laboratory parameters for each study group are presented in Table 1. As indicated in Table 1, serum MDK expression was significantly elevated in HCC patients compared to those with benign liver disease and healthy controls (P = 0.000). We further stratified the HCC cohort and discovered that serum MDK levels differed significantly between AFP-negative and AFP-positive patients (P = 0.001), with MDK concentrations being notably higher in AFP-positive HCC patients than in their AFP-negative counterparts (Table 2). Given the potential association of MDK with various tumor expression factors, we analyzed the correlation between serum MDK levels and conventional tumor markers. Our findings revealed a positive correlation between MDK and AFP, CA-199, CA-125, as well as DCP (P < 0.05, Supplemental Figure 1(a)). Among the assessed indices, the correlation between serum MDK and CA-199 in HCC patients was found to be the most significant (P < 0.05, R = 0.589, Supplemental Figure 1(b)).
The clinical feature of the studied groups.
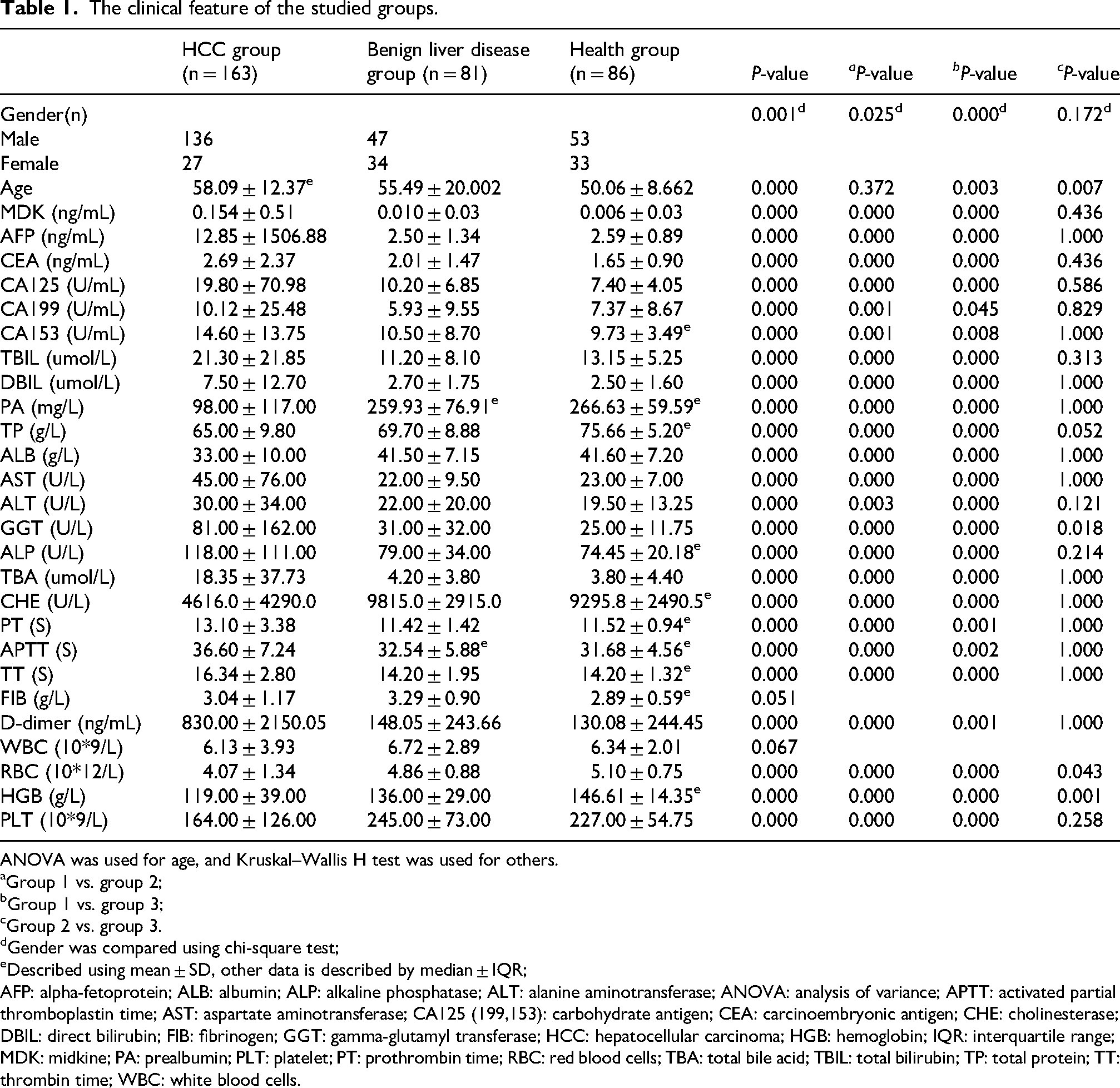
ANOVA was used for age, and Kruskal–Wallis H test was used for others.
aGroup 1 vs. group 2;
bGroup 1 vs. group 3;
cGroup 2 vs. group 3.
dGender was compared using chi-square test;
eDescribed using mean ± SD, other data is described by median ± IQR;
AFP: alpha-fetoprotein; ALB: albumin; ALP: alkaline phosphatase; ALT: alanine aminotransferase; ANOVA: analysis of variance; APTT: activated partial thromboplastin time; AST: aspartate aminotransferase; CA125 (199,153): carbohydrate antigen; CEA: carcinoembryonic antigen; CHE: cholinesterase; DBIL: direct bilirubin; FIB: fibrinogen; GGT: gamma-glutamyl transferase; HCC: hepatocellular carcinoma; HGB: hemoglobin; IQR: interquartile range; MDK: midkine; PA: prealbumin; PLT: platelet; PT: prothrombin time; RBC: red blood cells; TBA: total bile acid; TBIL: total bilirubin; TP: total protein; TT: thrombin time; WBC: white blood cells.
Comparison of main laboratory parameters between AFP-positive hepatocellular carcinoma group and AFP-negative hepatocellular carcinoma group.

aDescribed using mean ± SD, other data is described by median ± IQR.
bIndependent sample T test. Mann–Whitney U test for others.
AFP: alpha-fetoprotein; ALB: albumin; ALP: alkaline phosphatase; ALT: alanine aminotransferase; ANOVA: analysis of variance; APTT: activated partial thromboplastin time; AST: aspartate aminotransferase; CA125 (199,153): carbohydrate antigen; CEA: carcinoembryonic antigen; CHE: cholinesterase; DBIL: direct bilirubin; DCP: des-gamma carboxyprothrombin; FIB: fibrinogen; GGT: gamma-glutamyl transferase; HBV: hepatitis B virus; HCC: hepatocellular carcinoma; HGB: hemoglobin; IQR: interquartile range; M: mean; MDK: midkine; PA: prealbumin; PLT: platelet; PT: prothrombin time; RBC: red blood cells; TBA: total bile acid; TBIL: total bilirubin; TP: total protein; TT: thrombin time; WBC: white blood cells.
Additionally, we sought to determine the relationship between MDK levels and serum liver function indices to explore its association with liver injury markers. Interestingly, we observed that MDK exhibited positive correlations with TBIL, DBIL, AST, ALT, γ-GGT, ALP, and TBA while showing negative correlations with PA, ALB, and CHE (P < 0.05, Supplemental Figure 1(c)). Notably among these indices was the significant correlation between serum MDK levels and TBIL in HCC patients (P < 0.05, R = 0.485, Supplemental Figure 1(d)).
MDK expression suggests malignant progression of HCC
To further evaluate the diagnostic performance of MDK, we analyzed the correlation between MDK expression and various clinicopathological features in patients with HCC. Our findings indicate that there are significant differences in MDK levels across different CNLC stages (Table 3). Specifically, MDK levels were markedly higher in patients classified as stage D compared to those in stages A and B (Supplemental Figure 2(a)). Furthermore, we observed a relationship between MDK expression and the occurrence of complications in HCC patients. The serum MDK levels were elevated in patients experiencing complications such as ascites, splenomegaly, and portal hypertension when compared to those without such complications (Table 3; Supplemental Figure 2(b)). This suggests that an increase in serum MDK expression is associated with a deterioration of the patient's condition. To investigate the relationship between clinicopathological features and survival or recurrence in patients with HCC, we conducted a comprehensive analysis of various factors including CNLC stage, cirrhosis, tumor number, tumor size, complications, HBV infection, and detrimental lifestyle habits. Our findings indicate that the CNLC stage significantly influences patient survival and recurrence (Table 4). Specifically, patients classified as CNLC stages C and D exhibit a higher likelihood of tumor recurrence and poorer prognosis compared to those in stages A and B. In conjunction with previous research findings, our study reveals that elevated expression levels of MDK are closely associated with advanced CNLC stages. We propose that high MDK expression is intricately linked to unfavorable tumor prognoses. Furthermore, while liver cirrhosis, tumor number, tumor size complications, HBV infection, and adverse lifestyle choices were examined in relation to patient outcomes, these factors appear to have minimal impact on the prognosis of individuals diagnosed with HCC.
The relationships between MDK expression in HCC patients and clinical pathological features.
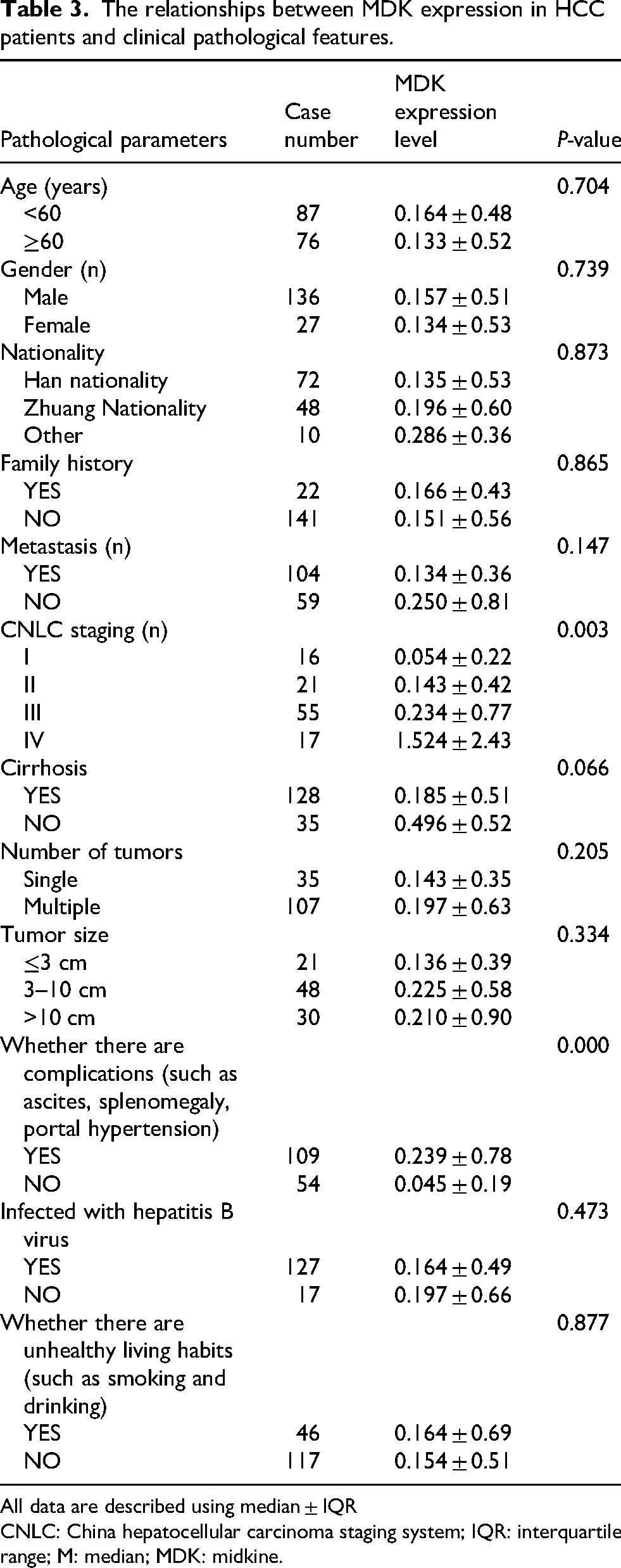
All data are described using median ± IQR
CNLC: China hepatocellular carcinoma staging system; IQR: interquartile range; M: median; MDK: midkine.
Multivariate Cox hazard analysis of clinical features for poor prognosis for HCC.
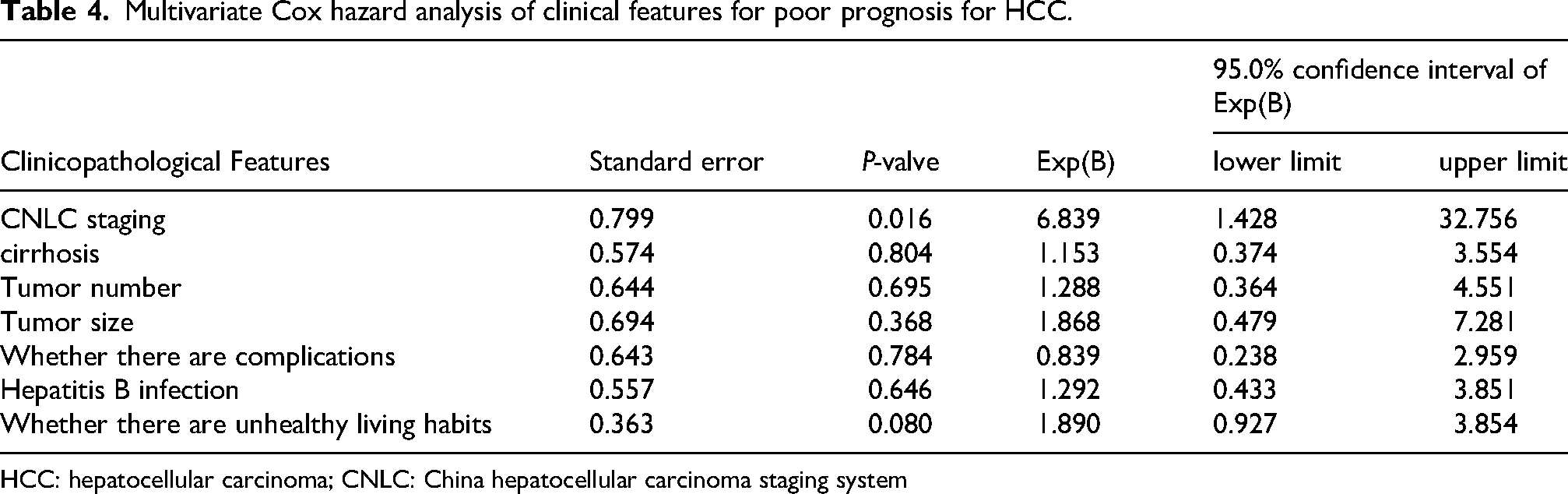
HCC: hepatocellular carcinoma; CNLC: China hepatocellular carcinoma staging system
MDK can be used as a novel serodiagnostic marker for HCC
As previously described, we observed a significant overexpression of MDK in 163 patients with HCC (Table 1). Notably, within this cohort, the expression levels of MDK were significantly elevated in AFP-positive HCC patients compared to those who were AFP-negative (Table 2). This finding suggests that MDK may serve as a potential diagnostic biomarker. To evaluate the diagnostic efficacy of MDK, we conducted a ROC curve analysis. The results indicated that the AUC for MDK in diagnosing HCC was 0.8029. When the cutoff value is 0.06 ng/mL, the sensitivity is 66% and the specificity is 87% (Table 5, Supplemental Figure 3(a)). It should be emphasized that the optimal critical value of MDK in AFP-negative HCC patients is 0.12 ng/mL, AUC is 0.7304, sensitivity is 54%, and specificity is 95% (Table 5, Supplemental Figure 3(b)). In conclusion, our findings suggest that MDK is a promising sensitive serum marker for the diagnosis of HCC and warrants further investigation and application.
ROC curve analysis to determine the diagnostic value of MDK for HCC.

AFP: alpha-fetoprotein; AUC: area under the curve; HCC: hepatocellular carcinoma; LCL: MDK: midkine; ROC: receiver operating characteristic; LCL: Lower Control Limit; UCL: Upper Control Limit).
Discussion
HCC represents a significant global health challenge, with projections indicating over 1 million new cases by the year 2025. 23 Common risk factors for HCC include viral infections, particularly chronic liver diseases (CLD) such as HBV virus infection and liver cirrhosis, as well as detrimental lifestyle choices like alcoholism. Due to the nonspecific nature of its symptoms, the early diagnosis of HCC is often challenging. Furthermore, the heterogeneity of HCC at both molecular and histological levels adds complexity to its diagnosis and treatment. Unfortunately, the overall burden on patients with advanced HCC remains substantial, and the survival rate continues to be alarmingly low.1,24 Although significant efforts have been made in the areas of prevention, early detection, and treatment advancements, the sensitivity and specificity of traditional diagnostic methods such as ultrasound and AFP for the early diagnosis of HCC remain insufficient for accurate diagnosis.25,26 Therefore, there is an urgent necessity for innovative and dependable biomarkers to complement AFP, with the aim of enhancing the diagnostic accuracy of HCC. Various factors underscore the necessity for large-scale identification of novel noninvasive biomarkers aimed at facilitating the early diagnosis of HCC, positioning this area as a significant field of research.
It has been reported that MDK, a developmentally regulated gene product, has been shown to promote cell growth, survival, differentiation, migration, and angiogenesis. Studies have shown that it is a key factor associated with recurrent, invasive, and metastatic phenotypes in most malignant tumors.27,28 However, there remains a diversity of opinions—some even contradictory—regarding the diagnostic utility of utilizing MDK as a serum marker for HCC.29–31 Therefore, the clinical application of MDK in the diagnosis of HCC requires long-term studies and follow-up investigations to further validate its efficacy.
Our study found that MDK is highly expressed in HCC patients. Many studies have demonstrated that MDK plays a significant role in the malignant progression of HCC. This participation can be attributed to its regulation of various signaling pathways, including the activation and modulation of the PI3 K/Akt pathway as well as the integrin α6/Src/Shc signaling pathway, thereby promoting pathological angiogenesis. Furthermore, MDK interacts with other factors such as granule protein precursors, negative regulators of Wnt-β-catenin-TGF signal transduction, and promoters of extracellular matrix degeneration, all contributing to the progression of HCC.21,27,32–34 Importantly, we observed an increase in serum MDK levels among a significant proportion of AFP-negative patients. This finding suggests that MDK could serve as a valuable diagnostic marker for this subset of patients. In addition, we conducted an analysis of the correlation between MDK and AFP in all serum samples from patients with HCC. Our findings revealed a correlation between these two markers; however, the correlation coefficient was relatively low (R = 0.25). This contrasts with the conclusions drawn by Zhu et al., 35 who posited that there is no significant correlation between MDK and AFP. The discrepancy may be attributed to differences in sample size and heterogeneity among the collected samples. The lack of a strong relationship between AFP and MDK suggests that MDK may reflects tumor biology through different pathways. Nevertheless, both studies agree that MDK plays a distinctive role in differentiating AFP-negative HCC from various control groups. In addition, we evaluated MDK’s capacity to differentiate HCC patients using the ROC curve analysis. The AUC was found to be 0.8029, indicating that the diagnostic accuracy can be considered good. Furthermore, MDK serves as a relatively sensitive marker for AFP-negative HCC, with an AUC of 0.7304. Other studies have also provided a high evaluation of the test efficiency of MDK. For instance, several studies have highlighted that the sensitivity of MDK protein surpasses that of AFP; however, its specificity is relatively low.22,34,36 Our results contrast with previous findings. Our research shows that the specificity of MDK in diagnosing HCC is obviously higher than the sensitivity. MDK is reputed to be a superior marker for diagnosing HCC, demonstrating a remarkable specificity of 95% in AFP-negative cases. However, its limited sensitivity underscores the necessity for complementary diagnostic tools. For example, the introduction of highly sensitive IL-41 as an auxiliary diagnostic index in the diagnosis of AFP-negative HCC can significantly improve the accuracy of diagnosis. 14 In a word, MDK has been proven to have great diagnostic value in the detection of AFP-negative HCC. Further research should be done to supplement this discovery.
Given the high incidence of HCC associated with chronic hepatitis in China, we tried to further investigate the relationship between MDK levels and serum liver function indices. Interestingly, we found that MDK is closely associated with the liver function indices of patients with HCC. It is important to note that the expression levels of serum MDK in patients with HCC exhibit a positive correlation with various indices indicative of liver injury, such as DBIL, ALT, and AST. This suggests that MDK may be associated with the progression of liver injury. Equally important, it has been proved that MDK can predict the poor prognosis of HCC patients. Tumor metastasis and poor prognosis are more likely to occur in patients with high expression of MDK in CNLC phase C and D. Like other promising biomarkers, such as AFP, AFP-L3, and DCP, MDK may play a role in various cell signal transduction pathways and mediate the proliferation of tumor cells. Sun et al. 27 demonstrated that HCC patients with elevated levels of MDK exhibit a higher number of circulating tumor cells and a significantly increased recurrence rate. Increasing evidence emphasizes that MDK plays an important role in cancer-related activities such as proliferation, migration, and angiogenesis in different types of solid tumors, including HCC. 37 However, the molecular mechanisms underlying the overexpression of MDK in promoting the malignant progression of HCC require further investigation to provide additional insights.
In recent years, there has been an increasing focus on the clinical application of serum biomarkers as predictors of tumor recurrence and for post-treatment monitoring. This growing interest is primarily attributed to their noninvasive nature, simplicity, and repeatability in assessment. In this study, we demonstrate that the detection of MDK expression is beneficial for diagnosing HCC, particularly in patients with AFP-negative HCC. Furthermore, MDK expression can serve as a predictor for tumor metastasis and mortality. These findings suggest that in clinical practice, patients exhibiting elevated serum MDK levels may warrant more frequent follow-up and early intervention treatment strategies. Our research presents several advantages, including a diverse array of research cohorts. This allows us to conduct subgroup analyses across different cohorts and to examine various patient characteristics or disease stages, thereby enhancing our understanding of the correlation between MDK and specific clinical parameters in greater detail. Furthermore, we have analyzed the relationship between serum MDK expression and clinicopathological parameters in patients with HCC for the first time. This analysis provides valuable insights into the potential role of MDK in both the diagnosis and prognosis of HCC. However, it is important to acknowledge that the reliability of our findings is limited by the small number of available studies. It will be crucial to recruit additional samples from multiple platforms to verify the accuracy and generalizability of our results. Moreover, the combination of MDK with AFP or other biomarkers still lacks long-term follow-up and prognostic data, warranting further investigation in future studies.
Conclusion
In summary, MDK is closely associated with the progression of HCC and serves as a potential predictor of poor prognosis and malignant advancement in HCC. Furthermore, MDK may pave the way for novel diagnostic approaches for patients with AFP-negative HCC.
Footnotes
Ethical disclosure
The authors indicate that they have obtained the approval of the appropriate institutional review committee.
Author contributions
L. Lu and Q. Peng drafted the overall design scheme of this paper. C. Zhu and L. Yi completed the data collection and analysis. This article was drafted by L. Lu, reviewed and edited by S. Li, M. Fan and J. Lin. All the authors agreed to publish the final version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Guangxi Natural Science Foundation , Youth Project of Guangxi International Zhuang Medicine Hospital, Discipline Project of Guangxi International Zhuang Medicine Hospital and Research Fund of Guangxi International Zhuang Medicine Hospital .(grant number NO. 2024JJA140801, NO. [2022]203, NO. [2021]33, NO. RCYJ202201)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
