Abstract
Background:
A randomized controlled trial in Bangladeshi children aged 12 to 18 months with moderate acute malnutrition found that dietary supplementation with the microbiota-directed complementary food (MDCF-2) significantly improved weight gain and repaired gut microbiota compared to the ready-to-use supplementary food. However, the MDCF-2 formulation was made daily from locally available ingredients and the need for a packaged, nutritionally compliant, and organoleptically acceptable MDCF-2 prototype was essential for future large-scale clinical studies.
Objective:
The study aimed to develop and assess the acceptability of 3 alternative foil-packaged formulations of MDCF-2 in comparison to current MDCF-2.
Methods:
Of the 3 packaged formulations, the Jumpstart version was provided in 2 sachets, the other 2 formulations were provided in a retort-stable foil pouch extended by sterilization, and microbiological growth was monitored over 10 months. The acceptability study included 40 children aged 8 to 12 months living in an urban slum in Dhaka, and the organoleptic properties were assessed using a 7-point hedonic scale.
Results:
In the 100 g distributed over the 2 sessions, children consumed 82.5 ± 7.84 g (mean ± SD) of kitchen-prepared MDCF-2, 85.4 ± 7.15 g of the “Jumpstart” MDCF-2 formulation, 85.4 ± 8.70 g of the MDCF-2 with green banana powder, and 86.2 ± 4.26 g of the MDCF-2 with sweet potato formulation. The “Jumpstart” MDCF-2 and MDCF-2 with sweet potato achieved the highest overall acceptability scores on the hedonic scale; although none of the shelf-stable formulations were significantly different from the kitchen-prepared MDCF-2.
Conclusions:
Packaged, shelf-stable prototypes of MDCF-2 exhibited comparable acceptability among Bangladeshi children aged 8 to 12 months to the original freshly prepared formulation.
Plain language title
Development and Acceptability of Shelf-Stable Microbiota-Directed Complementary Foods
Plain language summary
Countries around the world are making progress in fighting malnutrition, but it may be challenging to achieve the global nutrition targets for undernourished children by 2025 at the current rate. To address this problem, we need special types of food that can help children grow, develop properly, and sustain their growth. Children with moderate acute malnutrition have imbalanced gut bacteria. By providing them with the right nutrition, we can restore the healthy bacteria in their gut using a special food called microbiota-directed complementary food (MDCF). Currently, this food is made daily in the icddr, b established field kitchen, which follows standard production measures to control the quality of MDCF preparation, but we need to create stable prototypes that can be stored and used in different settings. This study was designed to develop shelf-stable new formulations in industrial settings and check their acceptability among children with moderate acute malnutrition. The shelf life of the food was extended using a sterilization method, and its microbiological safety was monitored for a year. The actual consumption and acceptability of these foods were evaluated and all these formulations were acceptable by the children and their mother. The children consumed all 4 food formulations in substantial quantities, with consumption rates exceeding 80% for each formulation. This study showed positive results in Bangladesh, but more research is needed to see if these formulations work well in other geographies and over longer periods.
Keywords
Introduction
Nearly 45 million children under the age of 5 worldwide suffer from acute malnutrition (wasting). 1 Moderate acute malnutrition (MAM) is defined as moderate wasting (weight-for-height Z-score between <−2 to −3 compared to the World Health Organization [WHO] Child Growth Standards) and/or mid-upper-arm circumference greater or equal to 115 mm and less than 125 mm. 2 The progress made toward addressing childhood wasting, which is one of the WHO’s 6 global nutrition targets for 2025, has been somewhat slow. 1 Several studies have provided evidence that lipid-based nutritional supplements and various blended diets provide benefits in the recovery of children with MAM, though there have been relapses. 3,4
Recent studies have shown that acute nutrition is associated with impaired development of the microbiota in early childhood (microbiota “immaturity”), with the extent of this perturbation in community structure and functional capacity greater in severe acute malnutrition (SAM) than in MAM. 5,6 Furthermore, these perturbations were not “repaired” by standard therapeutic foods. 6,7 Following preclinical studies using gnotobiotic animals colonized with microbial communities from malnourished versus healthy age-matched children established causality: microbiota from the undernourished children resulted in slower weight gain, impaired bone growth, and metabolic abnormalities in recipient animals. 7 Moreover, the growth impairment could be prevented by specific gut microbial community members found in the microbiota of healthy children who were underrepresented in the microbiota of undernourished children. 8,9
Using gnotobiotic animal models, we have screened a range of Bangladeshi complementary foods (consumed during the transition in children from exclusive milk feeding to a fully weaned state) to identify those that are able to promote the fitness and/or expressed beneficial functions of the growth-associated bacterial taxa that are underrepresented in children with MAM/SAM. 7 This initiative led to the development of a “microbiota-directed complementary food” (MDCF-2) for which efficacy was established in a 3-month randomized controlled trial in Bangladeshi children, where it produced significantly greater weight gain (improvement in weight-for-length z-score [WLZ]) than a more calorically dense “standard” ready-to-use supplementary food (RUSF). 10 This improvement in ponderal growth was associated with an increase in the abundance of the underrepresented growth-associated gut bacterial taxa, along with improvements in various plasma protein biomarkers and mediators of musculoskeletal and brain development. 7,10
To date, clinical studies with MDCF-2 have utilized a “kitchen-prepared” formulation that is produced in the icddr, b kitchen and provided daily to study participants by caregivers, through a process overseen by study staff. 11 We recently described the manufacture of scalable, foil-packaged prototypes of MDCF-2 for a pilot clinical trial to compare their efficacy to that of freshly-prepared MDCF-2 in Bangladeshi children with MAM. 12 In the current study, we assessed the shelf-life (stability) of these foil-packaged prototypes along with their organoleptic acceptability. We measured the “liking” (overall consumption in a timed feeding study) of each formulation in children and used a 7-point hedonic scale for mothers to score their color, aroma, taste, texture, and overall acceptability.
Methods
Ethical Approval
The Ethical Review Committee of the icddr, b reviewed and approved a randomized pre-proof of concept trial to compare the efficacy of the MDCF-2 prototypes in repairing the gut microbiota of 8- to 12-month-old children with MAM living in the Mirpur slum area of Dhaka, Bangladesh (PR#18079; Clinical Trials.gov ID NCT05094024). The study described here was a component of this protocol. All participants’ legal guardians provided written informed consent.
Production of Shelf-Stable MDCF-2
The composition and preparation of the kitchen-prepared MDCF-2 formulation has been previously described. 10,11 In this study, kitchen-prepared MDCF-2 was compared with 2 industrial prototypes and another kitchen-prepared prototype that comprised 2 separate sachets, called “Jumpstart” MDCF-2. The formulations that are prepared in icddr, b field kitchen not in an industrial setting are referred to as kitchen-prepared MDCF-2. Two formulations were made in the kitchen; one named kitchen-prepared MDCF-2, and another named Jumpstart MDCF-2. The “Jumpstart” formulation is comprised of individually packaged MDCF-2 components in a separate sachet of dry and liquid ingredients. These ingredients are prepared in the field kitchen and have a shelf life of around 7 days. The nutritional composition of the formulations is shown in Table 1.
Nutritional Composition of the MDCF-2 Prototypes.
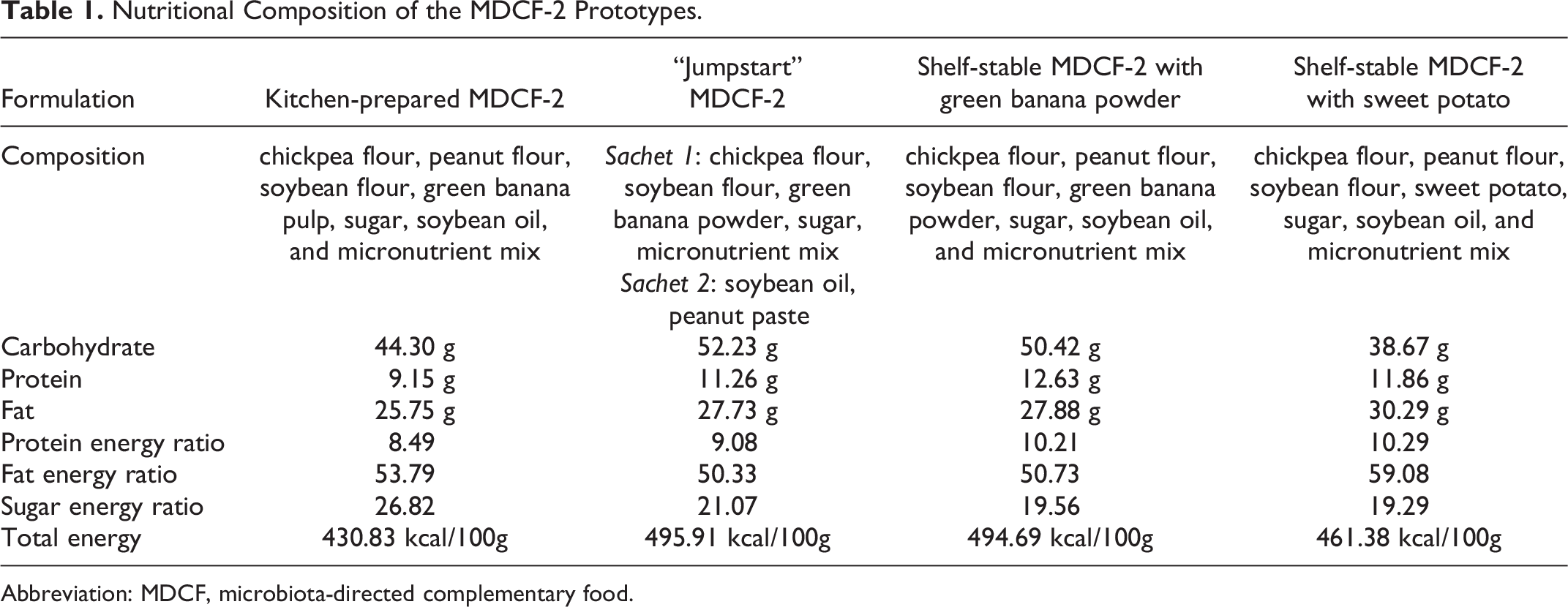
Abbreviation: MDCF, microbiota-directed complementary food.
The industrial prototype production was conducted in collaboration with a large food processing company in Bangladesh. A dedicated Food Processing Laboratory that adhered to British Retail Consortium and Hazard Analysis Critical Control Point compliance standards was established, ensuring that the processing of raw ingredients and packaging of the final product were performed under highly controlled conditions. Initially, chickpeas, soybeans, and green bananas were roasted at 120°C to 130°C for 8-10 minutes without burning. After cooling, burnt particles were removed, and the roasted ingredients were finely ground using an electric grinder. Additionally, the sugar was ground into fine powder. Peanut paste was prepared by dry roasting peanuts at 140°C for 30 minutes, followed by grinding in a high-speed electric blender. For the MDCF-2 with sweet potato, sweet potatoes were washed and boiled for 20 minutes, cooled, peeled, and blended. The required amounts of soybean oil and a vitamin-mineral pre-mix for each formulation were measured and subsequently, all ingredients were combined in an electric mixer to produce MDCF-2 paste in batches. The paste was then filled into trilaminar retort-stable aluminum foil pouches (manufacturer: XI’AN Lucky Star Co. Ltd.) using a semi-automatic filling machine and sealed using an Impulse sealing machine. After packaging and sealing, sterilization was performed in batches using a retort machine (121°C at 0.208 Mpa pressure for 1 hour).
The “Jumpstart” MDCF-2 (Table 1) was prepared in a dedicated kitchen established at the study field office at Mirpur area of Dhaka City. This prototype was prepared, as it is simple to mix and administer to the child. In addition, green banana pulp has been substituted with powder in this prototype to ensure microbiological safety, as the water in boiled green banana can be a source of contamination. Trained kitchen staff processed all raw ingredients following standard operating procedures (SOPs). Once all ingredients had been prepared, 2 separate aluminum foil pouches were filled; one containing the dry ingredients and the other containing peanut and soybean oil. Each pouch was appropriately labeled, sealed, and stored at room temperature for a maximum of 5 days between feeding sessions. The kitchen’s adherence to established SOPs ensured uniformity in the product’s preparation and packaging.
Determination of Quality and Stability of MDCF-2 Prototypes
Storage stability tests
Storage stability tests were conducted to assess how well MDCF-2 retained organoleptic and microbiological acceptability over time under various storage conditions. The purpose of these tests is to evaluate the product’s shelf life, potential changes in its sensory attributes, and any risks associated with extended storage. MDCF-2 with green banana powder and sweet potato were stored in room temperature to represent typical storage scenarios. The storage duration ranged from 1-10 months, with regular testing intervals at months 1, 2, 3, 6, and 10. Chemical stability was assessed by (moisture, pH, and water activity). In addition, sensory evaluation was performed at 5 time points (months 1, 2, 3, 6, and 10) in a blinded fashion by a trained panel of icddr, b personnel to assess any changes in the product’s taste, texture, color, and overall acceptability.
Microbiological tests
Microbiological food safety tests were performed on samples of the 2 industrially produced foil-packaged formulations by an internationally recognized external testing facility (SGS, Bangladesh, and India). Microbiological tests (total plate count, total coliform, Enterobacteriaceae, enumeration of Bacillus, Staphylococcus aureus, yeasts, molds, Salmonella, Shigella, aflatoxin, Escherichia coli, and Cronobacter sakazakii) were performed prior to the acceptability trial.
Acceptability Trial
The primary outcome of the acceptability trial was to assess the acceptability of 2 industrially produced formulations and the “Jumpstart” MDCF-2 in comparison with the kitchen-prepared MDCF-2 by measuring the amount of each formulation that was consumed by children in a timed feeding session. The secondary outcome was the mothers’ opinion on the food’s organoleptic properties (color, taste, aroma, texture, and overall acceptability) using a 7-point hedonic scale.
Children aged 8-12 months with MAM, whose WLZ score <−2 to −3 and without bilateral pedal oedema were enrolled from an urban slum settlement located in the Mirpur area of Dhaka city, Bangladesh. We excluded participants with any acute or chronic illness, such as tuberculosis, any congenital/acquired disorder, known cases of trisomy-21, cerebral palsy, or a history of soy, peanut, or milk protein allergy. 12 Mother’s expressed an interest in testing new food formulations and provided informed consent for the study. Mothers were advised not to feed their children for 2 hours prior to the feeding session. Children were randomized to receive 1 of the 4 formulations (10 children/formulation). Two separate feeding sessions were performed for each child on a single day. To ensure that feeding was not coerced, the feeding session was observed directly by a field research assistant. If children turned their heads away, closed their mouths, gritted their teeth, spit out the food, wailed, became agitated, or refused to swallow the food provided, we interpreted that as a refusal to eat. The amount consumed was determined by deducting the weight of the leftover food from the 50 g that was initially provided. The amount of food given to each child was calculated with the goal of providing 50% of their daily energy requirements (a total of 100 g split over 2 sessions of 50 g each, providing 450 kcal energy). If a child with a body weight of <4 kg consumed an amount of 11.5 g, likewise at least 23 g was consumed if she/he had a body weight of 4-9.9 kg within 45 minutes, the child was considered to have passed the appetite test. 13 -15 Any adverse reactions, such as nausea, vomiting, or fever were also documented.
For mothers, we provided the food after her child’s feeding session was completed to avoid influencing the child’s feeding behaviour. Mothers were asked to rate the food formulation on different attributes (color, taste, aroma, texture, and overall acceptability) using a 7-point hedonic scale (1 = dislike extremely, 2 = dislike moderately, 3 = dislike slightly, 4 = neither dislike nor like, 5 = like slightly, 6 = like moderately, 7 = like extremely). 16
Data Analysis
Data related to socioeconomic status, household characteristics, and demographic information of the children are presented as means with standard deviation (SD), frequencies, or percentages. Additionally, we calculated the mean (SD) of total consumption in grams and consumption percentages for each arm. One-way analysis of variance (ANOVA) was used to determine the statistical significance of differences in consumption and acceptability between the formulations tested in the human study.
Results
Microbiological and Chemical Stability Assessment of the Industrial Foil-Packaged Prototypes
Microbiological and chemical stability evaluations were conducted on two industrially produced foil-packaged formulations after 1, 2, 3, 6, and 10 months after production; formulations were stored at 35°C to 39°C (Supplementary Table 1). The assessments encompassed a range of parameters including total plate count, total coliform, yeast count, mold count, and Salmonella count. Total plate count, yeast count, mold count, and Salmonella count were <10 cfu/gm across all time points for both formulations. Moisture content, pH, and water activity were also determined at the same time points and remained stable (Supplementary Table 1).
Acceptability Trial
Forty mother-child pairs were recruited in the acceptability trial, and all of them completed the trial. The sociodemographic characteristics of the participants in the acceptability trial are shown in Table 2. The WLZ was −2.28 ± 0.23 (mean ± SD) and age 10.25 ± 1.46 months. Twenty of the children (50%) were male and all participants were breastfeeding and consuming complementary foods during the study.
Sociodemographic Information of the Study Participants.
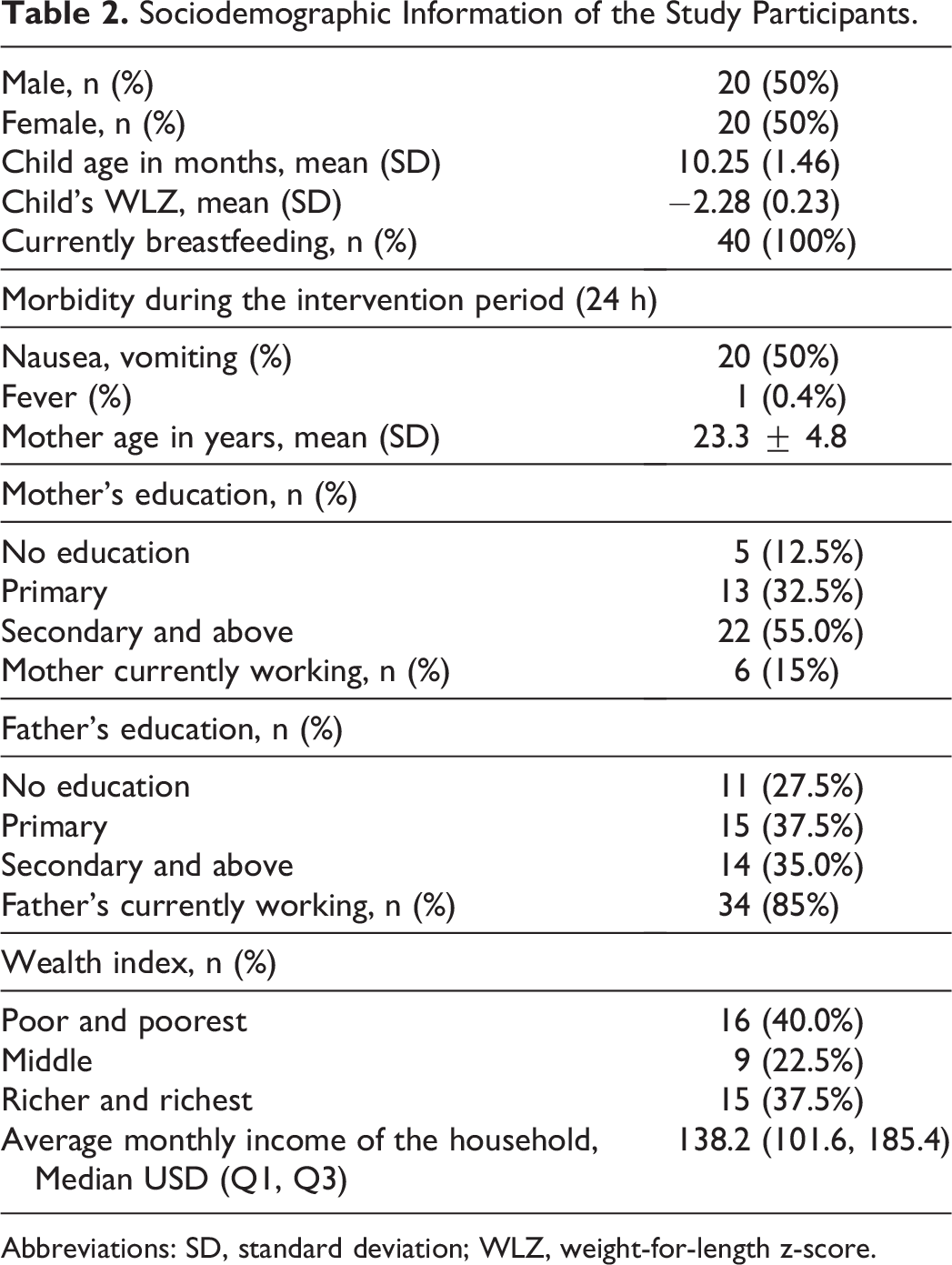
Abbreviations: SD, standard deviation; WLZ, weight-for-length z-score.
There were no significant differences in the amount of each formulation consumed (P value = 0.676; 1-way ANOVA test); from the 100 g provided over 2 feeding sessions, the mean consumption of kitchen-prepared MDCF-2 was 82.5 ± 7.84 g, for “Jumpstart” MDCF-2 it was 85.4 ± 7.15 g, for MDCF-2 with green banana it was 85.4 ± 8.70 g, and for MDCF-2 with sweet potato, it was 86.2 ± 4.26 g. All prototypes, therefore, met the basic requirement of more than 40% consumption. 13
Using the 7-point hedonic scale, mothers rated the overall acceptability of the formulations similarly. The kitchen-prepared MDCF-2 achieved an overall acceptability score of 5.67 ± 1.05, “Jumpstart” MDCF-2 achieved 6.02 ± 0.81 score, MDCF-2 with green banana powder 5.57 ± 0.79, and 6.05 ± 0.91 score were for MDCF-2 with sweet potato. These differences were not statistically significant (P = 0.543, one-way ANOVA; see Table 3 for the mean hedonic scores of the 4 different MDCF-2 formulations across each organoleptic parameter).
Mean Hedonic Scores of MDCF-2 Prototypes.
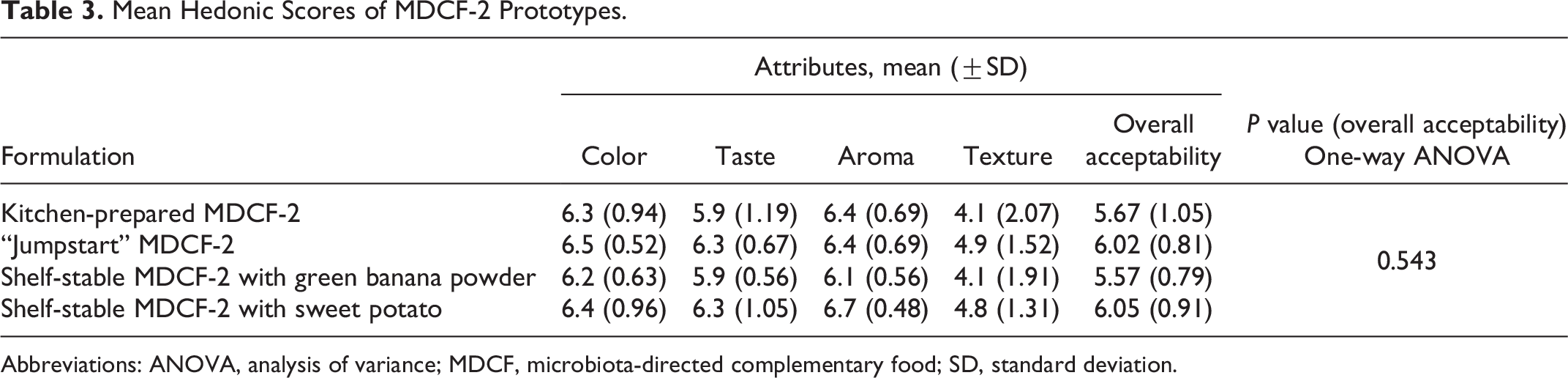
Abbreviations: ANOVA, analysis of variance; MDCF, microbiota-directed complementary food; SD, standard deviation.
Discussion
Large scale controlled clinical studies designed to compare the efficacy of supplementary/therapeutic foods to improve nutritional status require test products that can be produced at scale to consistent specifications, including nutritional and microbiological stability. Moreover, the products should retain their overall palatability to maximize compliance. In this study we examined the shelf life of 2 alternative industrially manufactured prototypes of a MDCF-2. MDCF-2 was formulated with a specific combination of plant-based ingredients that are affordable, commonly consumed, and culturally acceptable in Bangladesh. 10 Furthermore, our previous study involved a randomized controlled trial conducted on children aged 12-18 months in Bangladesh who were experiencing MAM. We found that providing these children with a daily freshly prepared formulation of MDCF-2 as a dietary supplement resulted in a significantly greater increase in weight gain over a period of 3 months compared to the use of a standard RUSF, despite the RUSF having a higher caloric density. 11
In the food processing industry, thermal and nonthermal sterilization methods are commonly used to increase the shelf life of processed, packaged foods. Thermal sterilization uses high temperatures to eliminate microorganisms, but this in turn may cause the migration of small molecules from the packaging into the food. 17 In our shelf-stable MDCF-2 prototypes, we used trilaminar, retort-stable aluminum foil pouches, which can resist high pressure and temperature while possessing low gas and moisture permeability and resistance to fat, oil, and other food component penetration. 18 This combination of packaging and heat/pressure treatment produced prototypes that had microbiological and chemical stability for a minimum of 10 months.
We conducted a randomized acceptability trial in children with MAM to compare the shelf-stable MDCF-2 prototypes with the kitchen-prepared MDCF-2. This test used 2 industrially made MDCF-2 prototypes and the “Jumpstart” MDCF-2 to compare their organoleptic acceptability (how much was eaten) to that of MDCF-2 made in the kitchen. Caregivers/mothers provided their own feedback using a 7-point hedonic scale, given that they primarily decide what to feed their children, and may select foods for their children based on their preferences. 19 The combination of the amount of a given formulation consumed by children, together with the mothers’ hedonic scores, has been shown to provide a more accurate view of the acceptability/liking of the alternate formulations. 20 In our study of the shelf stable formulations, these parameters were rated as not significantly different from those of the kitchen-prepared MDCF-2, thus providing support for their further development/scale up for future clinical trials.
Strengths and Limitations
Our studies provide an approach for evaluating the shelf-stability and organoleptic properties of industrially prepared MDCF-2 prototypes. These data enabled us to proceed into a 4-week pilot clinical (efficacy) component of the study with sufficient confidence in the safety and acceptability of prototypes. Our sample size for the acceptability study was relatively small, including 40 participants and limited to children living in an urban slum in Dhaka, Bangladesh. As the sample size was small, the results may not fully translate to larger cohorts of Bangladeshi children; however prior studies with similar sample sizes have proven to generate results that are not unrepresentative of the broader study population. 21,22
Acceptability in different geographies/cultural settings will likely differ from that observed in Bangladesh. Local acceptability tests will therefore be needed prior to large scale studies in different study populations. Since the design was not cross-over, each child tested only one product. In addition, longer term acceptability is not necessarily predicted by a single feeding experience, so careful monitoring of consumption will be essential in follow-up clinical studies.
Conclusions
We have developed industrially prepared, shelf-stable formulations of MDCF-2 whose organoleptic properties are comparable to those of the existing, kitchen-prepared formulation for which clinical efficacy has been demonstrated. 10 These results set the stage for a pilot clinical study to compare the efficacy of the newly developed formulations with MDCF-2. 12 We anticipate that this will be a prelude to future larger scale efficacy and effectiveness trials of shelf-stable MDCF-2 in other undernourished populations in Bangladesh, as well in other countries where the burden of malnutrition is high.
Supplemental Material
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721241250104 - Development and Acceptability of Shelf-Stable Microbiota Directed Complementary Food Formulations
Supplemental Material, sj-pdf-1-fnb-10.1177_03795721241250104 for Development and Acceptability of Shelf-Stable Microbiota Directed Complementary Food Formulations by Ishita Mostafa, Umme Habiba Lamiya, Md. Golam Rasul, Nurun Nahar Naila, Shah Mohammad Fahim, S. M. Tafsir Hasan, Michael J. Barratt, Jeffrey I. Gordon and Tahmeed Ahmed in Food and Nutrition Bulletin
Supplemental Material
Supplemental Material, sj-pdf-2-fnb-10.1177_03795721241250104 - Development and Acceptability of Shelf-Stable Microbiota Directed Complementary Food Formulations
Supplemental Material, sj-pdf-2-fnb-10.1177_03795721241250104 for Development and Acceptability of Shelf-Stable Microbiota Directed Complementary Food Formulations by Ishita Mostafa, Umme Habiba Lamiya, Md. Golam Rasul, Nurun Nahar Naila, Shah Mohammad Fahim, S. M. Tafsir Hasan, Michael J. Barratt, Jeffrey I. Gordon and Tahmeed Ahmed in Food and Nutrition Bulletin
Supplemental Material
Supplemental Material, sj-pdf-3-fnb-10.1177_03795721241250104 - Development and Acceptability of Shelf-Stable Microbiota Directed Complementary Food Formulations
Supplemental Material, sj-pdf-3-fnb-10.1177_03795721241250104 for Development and Acceptability of Shelf-Stable Microbiota Directed Complementary Food Formulations by Ishita Mostafa, Umme Habiba Lamiya, Md. Golam Rasul, Nurun Nahar Naila, Shah Mohammad Fahim, S. M. Tafsir Hasan, Michael J. Barratt, Jeffrey I. Gordon and Tahmeed Ahmed in Food and Nutrition Bulletin
Footnotes
Authors’ Note
All supplementary and metadata relevant to this manuscript are available upon request to the corresponding author.
Acknowledgments
The authors would like to thank all the participants and their parents for sharing their time and providing consent and information necessary for the successful completion of the study. The authors also acknowledge the contribution of icddr, b’s core donors including the Government of the People’s Republic of Bangladesh, Global Affairs Canada, for their continuous support and commitment to the icddr, b’s research efforts.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This project is funded by Bill & Melinda Gates Foundation (BMGF).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
