Abstract
Background:
Dairy is recommended in specially formulated supplementary foods to treat children with moderate acute malnutrition (MAM) but with limited evidence and added cost.
Objective:
Review studies of ready-to-use foods (RUFs) versus fortified blended foods (FBFs) to determine whether inclusion of dairy modifies the comparative effectiveness and cost.
Methods:
We reviewed literature comparing FBF and RUF in treatment of MAM among children younger than 5 years in developing countries. Outcomes of recovery from MAM, weight, and length gain were compared among treatment categories: FBF with dairy (FBF+), FBF without dairy (FBF−), RUF with dairy (RUF+), and RUF without dairy (RUF−). Supplement cost was compared per 500 kcal.
Results:
Eight studies were included. Rations were heterogeneous in energy and type of dairy. Overall, RUF+, RUF−, and FBF+ performed similarly, with higher recovery and weight gain compared with FBF−. RUF+ had higher recovery (in 5 of 6 comparisons), weight gain (4 of 4), and length gain (1 of 4) versus FBF−. The RUF+ had higher recovery (1 of 2) versus FBF+, with no other differences. The RUF− versus FBF+ had no differences (0 of 2). The RUF− had higher recovery (1 of 2), weight gain (2 of 2) versus FBF−. Four studies reported supplement costs, which averaged US$0.15 (FBF−), US$0.18 (FBF+), US$0.18 (RUF−), and US$0.37 (RUF+) per 500 kcal.
Conclusions:
There is a consistent benefit of FBF that include dairy in treatment of children with MAM. Benefits of dairy in RUF require further investigation. Evidence from rigorous quantitative analysis of existing data, cost-effectiveness, and prospective trials will be essential in determining policy on treatment for children with MAM.
Keywords
Introduction
In 2013, an estimated 51 million children younger than 5 years worldwide had moderate acute malnutrition (MAM), 1 defined as weight-for-height between −3 and −2 z-scores of the median of the World Health Organization (WHO) child growth standards, without edema. 2 Children with MAM are at an increased risk of morbidity, mortality, and poor cognitive development 3 and have an increased need for energy and essential nutrients beyond the nutritional requirements of nonmalnourished children.
World Health Organization recommends the use of optimal locally available nutrient-dense foods to prevent children from becoming severely malnourished; however, in situations where this is not be possible, specially formulated foods can complement the diet and provide the additional nutrients required to treat children with MAM. 4 Introduced in the 1960s, fortified blended foods (FBFs), such as corn–soy blend (CSB), are cereal based, with soy or other legumes added to improve the nutrient content, including protein quality. 5 Fortified-blended foods may also include additional ingredients such as vegetable oil, whey or skim milk powder, and micronutrients 5 and are typically prepared into porridge by boiling with water. A relatively newer category of specially formulated foods known as ready-to-use therapeutic or supplementary foods (RUTFs or RUSFs, henceforth referred to together as RUFs) are high-fat, energy-dense, ready-to-eat pastes often made from a combination of peanuts, milk powder and/or soy, sugar, oil, and micronutrients. 6,7
Scientific evidence shows a link between dairy and growth in children, 8 and many scientists and policy groups are now advocating inclusion of dairy in these specially formulated foods to treat child malnutrition. 5,9,10 However, there is sparse evidence on the actual effectiveness of including dairy ingredients in treatment of MAM, especially when comparing between and within various types of supplementary foods. A recent review found only limited evidence for the effectiveness of animal source foods and dairy in improving growth outcomes, including height and weight gain. 11 Our own systematic review 12 agreed with 2 other recent meta-analyses 13,14 that found RUF to be marginally more effective in treating MAM, but none examined whether the addition of dairy components could have had an impact on the effect size.
Data on the relative effectiveness of dairy in supplementary foods to treat MAM are especially important as dairy may add considerably to the cost of supplementary foods. 5,6,9 Cost-effectiveness data—that is the cost per recovered child, rather than cost per treated child—are also lacking. 5,15 Although more recent studies have started to include costs, this is usually reported as cost per food ration, not true cost-effectiveness. In addition, dairy production and consumption are increasing worldwide but are still not common in parts of Africa and Asia. 16 In the context of supplementary foods to treat malnutrition, this may create reliance on imported dairy products (for local supplementary food production) or importing fully manufactured supplementary foods.
Given the potentially limiting factors of cost and/or availability of dairy ingredients, the question of whether and to what extent dairy ingredients increase effectiveness of specially formulated foods to treat children with MAM becomes critically important. The objective of this article is to review the data on the effect of dairy in supplementary foods to treat MAM, comparing outcomes between FBF and RUF with and without dairy ingredients to identify discernable trends in recovery outcomes and potential impacts on cost-effectiveness.
Methods
We performed a literature review, searching published and gray literature for the key words “moderate acute malnutrition,” “fortified-blended foods,” “corn-soy blend,” “ready-to-use therapeutic foods,” and “ready-to-use supplementary foods.” Studies were selected if they were effectiveness trials with study arms that compared at least 1 FBF with at least 1 RUF in treatment of MAM among children 6 to 60 months of age in a developing country setting.
We undertook a qualitative synthesis of the studies. Within each study, intervention groups were categorized as FBF with dairy (FBF+), FBF without dairy (FBF−), RUF with dairy (RUF+), or RUF without dairy (RUF−). The outcomes compared were percentage of children recovered from MAM, weight and length gain during the treatment period, and time to recovery if reported. For each study, details on the type of food supplement, dairy ingredients, daily ration (kcal), cost (if available), and growth outcomes were extracted and tabulated in Microsoft Excel (2011). Costs for each food supplement were converted to 2015 US dollars if necessary, and for comparison normalized to cost per 500 kcal of supplement and averaged over each food supplement/dairy category.
Results
A total of 8 comparative studies of treatment of MAM with FBF versus RUF were included in the review (Table 1). The studies were conducted among 7 countries in sub-Saharan Africa and ranged 8 to 16 weeks of treatment, with 1 study that had no time limit. Descriptions of the supplementary foods used in the arms of the studies are shown in Table 2. The rations were heterogeneous, ranging from about 250 to 1400 kcal/d (fixed or by child’s weight), with 4 studies comparing isocaloric amounts and 4 comparing differing caloric levels between study arms. Among the 8 studies, 10 RUFs were tested, of which 6 contained dairy, and 10 FBFs were tested, of which 3 contained dairy. When dairy was included in FBF, it was always from dried skimmed milk (DSM), while dairy added to RUF included DSM, whey, whey protein isolate, or “powder milk.” Few studies reported the dairy content of the foods, or only specified protein from dairy per ration, so it was not possible to compare amounts of dairy in supplements among the studies.
Description of Studies Included in Review.
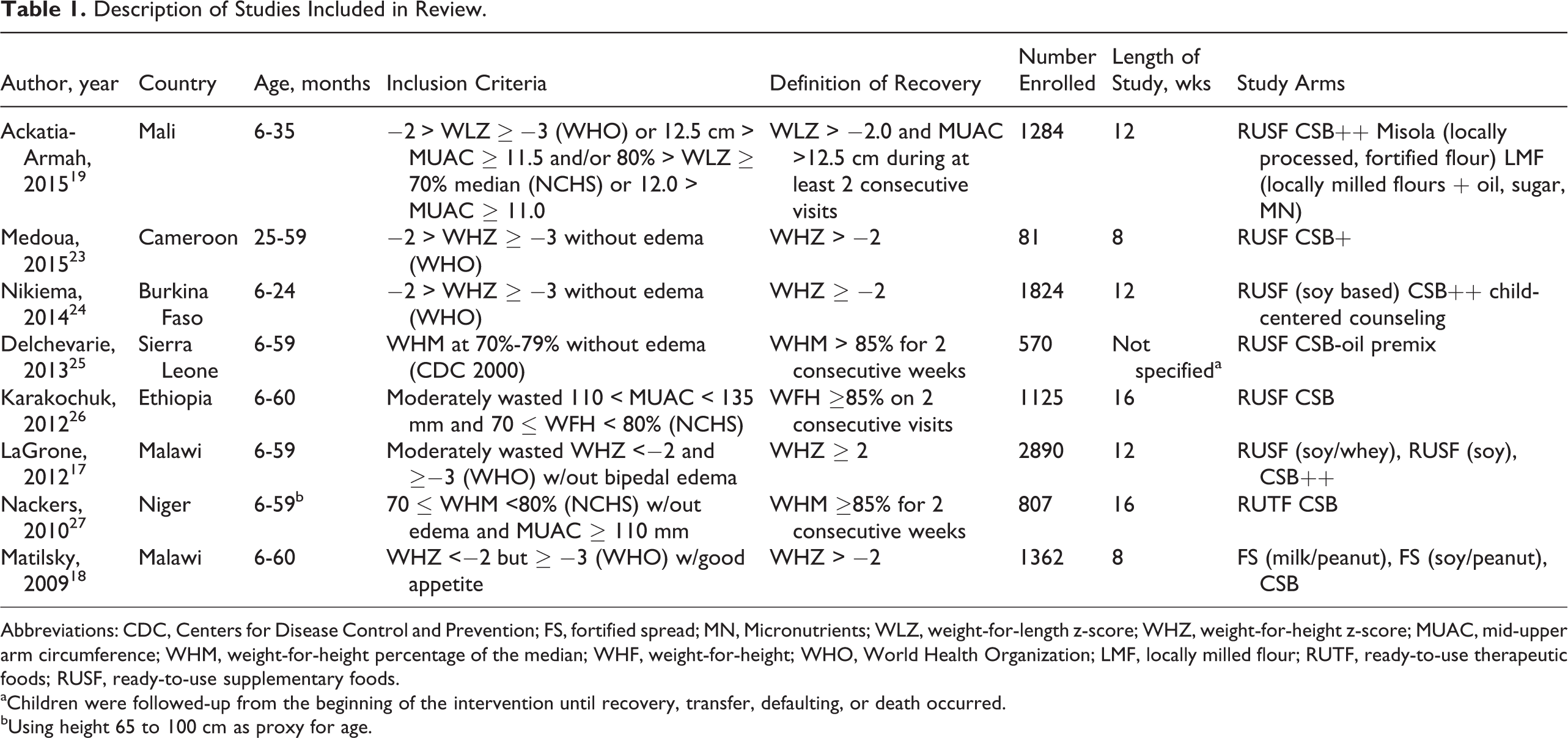
Abbreviations: CDC, Centers for Disease Control and Prevention; FS, fortified spread; MN, Micronutrients; WLZ, weight-for-length z-score; WHZ, weight-for-height z-score; MUAC, mid-upper arm circumference; WHM, weight-for-height percentage of the median; WHF, weight-for-height; WHO, World Health Organization; LMF, locally milled flour; RUTF, ready-to-use therapeutic foods; RUSF, ready-to-use supplementary foods.
aChildren were followed-up from the beginning of the intervention until recovery, transfer, defaulting, or death occurred.
bUsing height 65 to 100 cm as proxy for age.
Description of Food Supplements Used in Reviewed Studies.
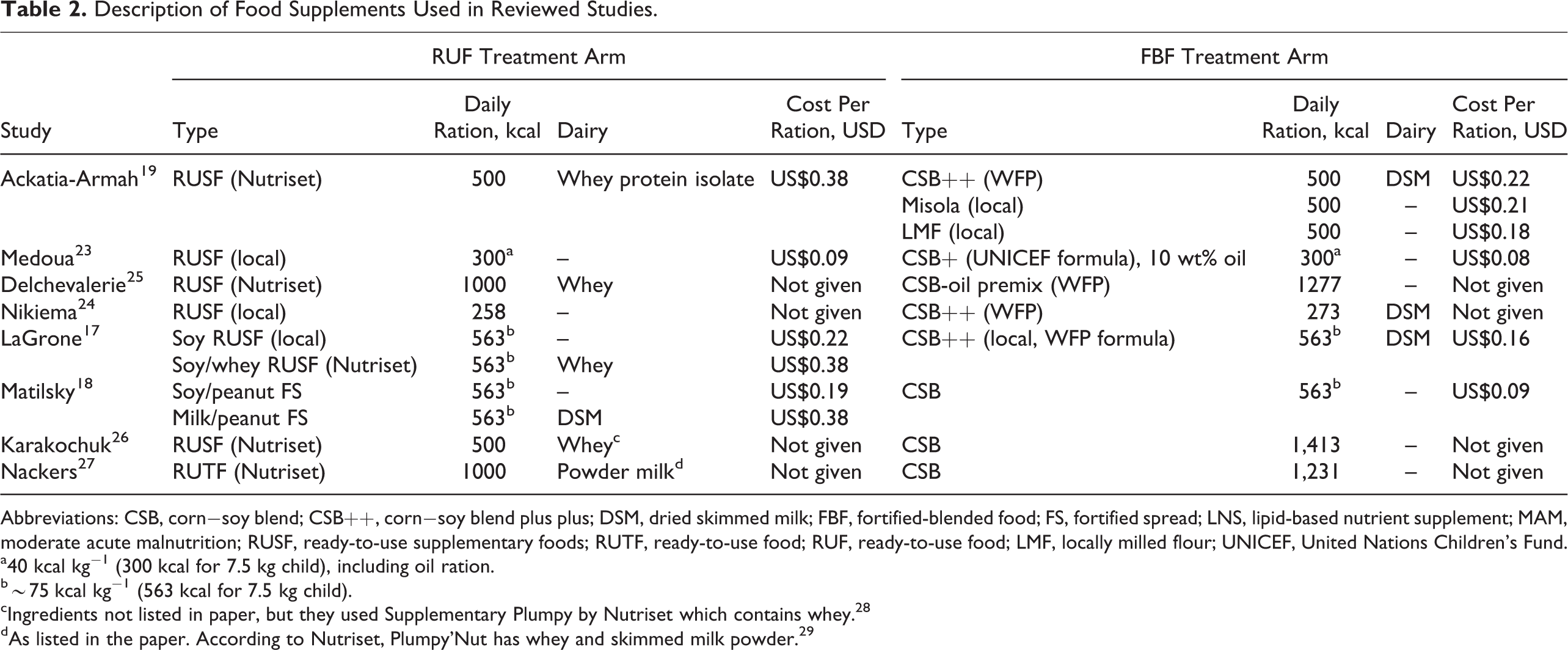
Abbreviations: CSB, corn−soy blend; CSB++, corn−soy blend plus plus; DSM, dried skimmed milk; FBF, fortified-blended food; FS, fortified spread; LNS, lipid-based nutrient supplement; MAM, moderate acute malnutrition; RUSF, ready-to-use supplementary foods; RUTF, ready-to-use food; RUF, ready-to-use food; LMF, locally milled flour; UNICEF, United Nations Children’s Fund.
a40 kcal kg−1 (300 kcal for 7.5 kg child), including oil ration.
b∼75 kcal kg−1 (563 kcal for 7.5 kg child).
cIngredients not listed in paper, but they used Supplementary Plumpy by Nutriset which contains whey. 28
dAs listed in the paper. According to Nutriset, Plumpy’Nut has whey and skimmed milk powder. 29
Treatment Effects of RUF and FBF With and Without Dairy Ingredients
There were a total of 6 comparisons from 5 studies between study arms of RUF+ versus FBF−; among the other 3 supplement/dairy categories, there were a total of 2 comparisons in each (Table 3). Among the 6 comparisons of RUF+ versus FBF−, children treated with RUF+ were found to have significantly higher recovery rates in all but one. There were 4 comparisons of weight and height gain; all 4 were significantly higher for weight gain in the RUF+ group, while only 1 of 4 was significantly higher for length gain in the RUF+ group, with no differences among the other 3 comparisons. In the 2 comparisons between RUF+ and FBF+, children treated with RUF+ were found to have significantly higher recovery rates in one, with no significant differences in weight or length gain in either. There were 2 comparisons of treatment using RUF− and FBF+; no significant differences were found in recovery rates, weight, or length gain among these arms. Finally, between the 2 comparisons of treatment using RUF− versus FBF−, RUF− was found to be associated with significantly higher recovery rates compared with FBF− in one, significantly higher weight gain in both, but no differences in length gain in either.
Comparison of Percentage of Children Recovered From MAM, Weight Gain, and Length Gain Outcomes Among Arms of 8 Studies Categorized by Food Supplement Category and Dairy Content.a
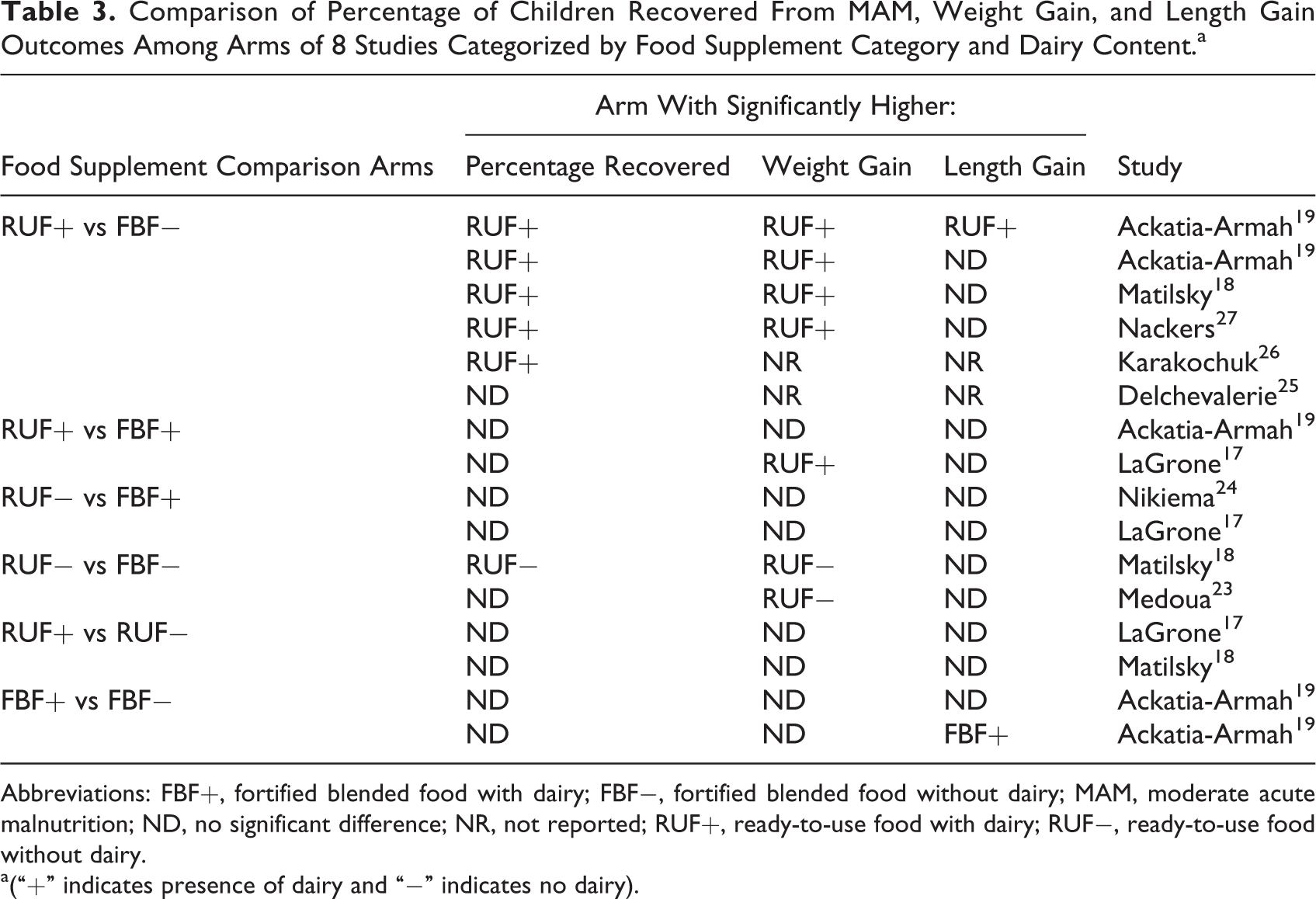
Abbreviations: FBF+, fortified blended food with dairy; FBF−, fortified blended food without dairy; MAM, moderate acute malnutrition; ND, no significant difference; NR, not reported; RUF+, ready-to-use food with dairy; RUF−, ready-to-use food without dairy.
a(“+” indicates presence of dairy and “−” indicates no dairy).
It was also possible to examine the potential effect of dairy inclusion within each food supplement category in 3 of the studies (Table 3). Two studies 17,18 included 2 RUF arms, 1 RUF+ and 1 RUF−; neither study found any significant difference in recovery, weight, or length gain among children treated with either RUF. Ackatia-Armah et al 19 included 3 FBF arms, 1 FBF+ and 2 FBF−; comparing the FBF+ with each FBF− showed no differences in recovery or weight gain, with only length gain significantly higher in the FBF+ arm in one of the 2 comparisons.
Recovery Rates and Time to Recovery
The percentage of children recovered from MAM among all the study arms ranged 58% to 94% (Table 4); both the highest and lowest recovery rates were found among children treated with FBF−. Percentage recovery ranged 74% to 88% among children treated with RUF−, 75% to 86% for FBF+ , and 71% to 93% for RUF+. Figure 1 shows that the recovery rates found among the supplement categories of the reviewed studies are very similar for children treated with RUF−, FBF+, or RUF+, while recovery tends to be slightly lower among those treated with FBF−. Four studies reported mean time to recovery (means ranged 23-68 days among supplement categories), 1 study reported median duration of supplementary feeding (medians ranged 14-28 days among the arms) and two studies reported mean “length of stay” in the program (means ranged 28-56 days among the supplement categories). Generally, time to recovery or time in the program was higher in the FBF− arms, while it had similar ranges among RUF−, FBF+ and RUF+ arms (Table 4).

Percentage of children recovered from moderate acute malnutrition (MAM) among reviewed studies by supplementary food arm and dairy categories. FBF− includes 7 study arms (2 at 64% recovery); RUF− includes 4 study arms; FBF+ includes 3 study arms; RUF+ includes 6 study arms (2 at 79% recovery). FBF+ indicates fortified-blended food with dairy; FBF−, fortified blended food without dairy; MAM, moderate acute malnutrition; RUF+, ready-to-use food with dairy; RUF−, ready-to-use food without dairy.
Number of Children Treated, Number Recovered, Percent Recovered, and Time to Recovery or Time in Program by Study and Supplement Category, With Cost Per 500 kcal if Available.
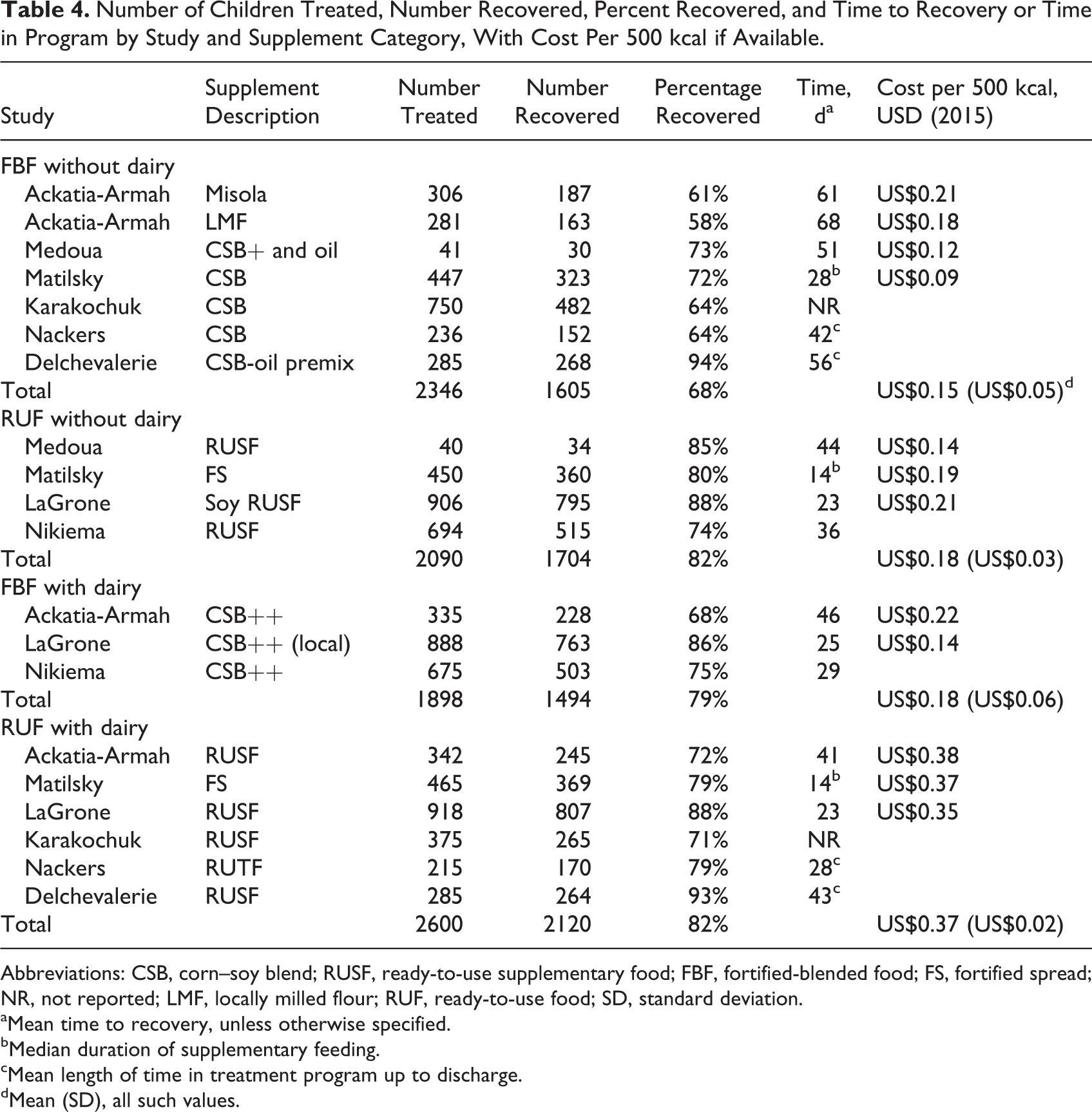
Abbreviations: CSB, corn–soy blend; RUSF, ready-to-use supplementary food; FBF, fortified-blended food; FS, fortified spread; NR, not reported; LMF, locally milled flour; RUF, ready-to-use food; SD, standard deviation.
aMean time to recovery, unless otherwise specified.
bMedian duration of supplementary feeding.
cMean length of time in treatment program up to discharge.
dMean (SD), all such values.
Costs
Four studies included costs of the food supplements, either per weight or per kJ of energy (Table 2). Among these studies, there were 4 FBF− study arms, 3 of RUF−, 2 of FBF+, and 3 of RUF+. RUF+ was consistently more than double the cost of the other 3 supplement categories. Among the 4 studies, the mean costs (± standard deviation) by supplement category, per 500 kcal, were FBF− US$0.15 (US$0.05), RUF− US$0.18 (US$0.03), FBF+ US$0.18 (US$0.06), and RUF+ US$0.37 (US$0.02; Table 4 and Figure 2).
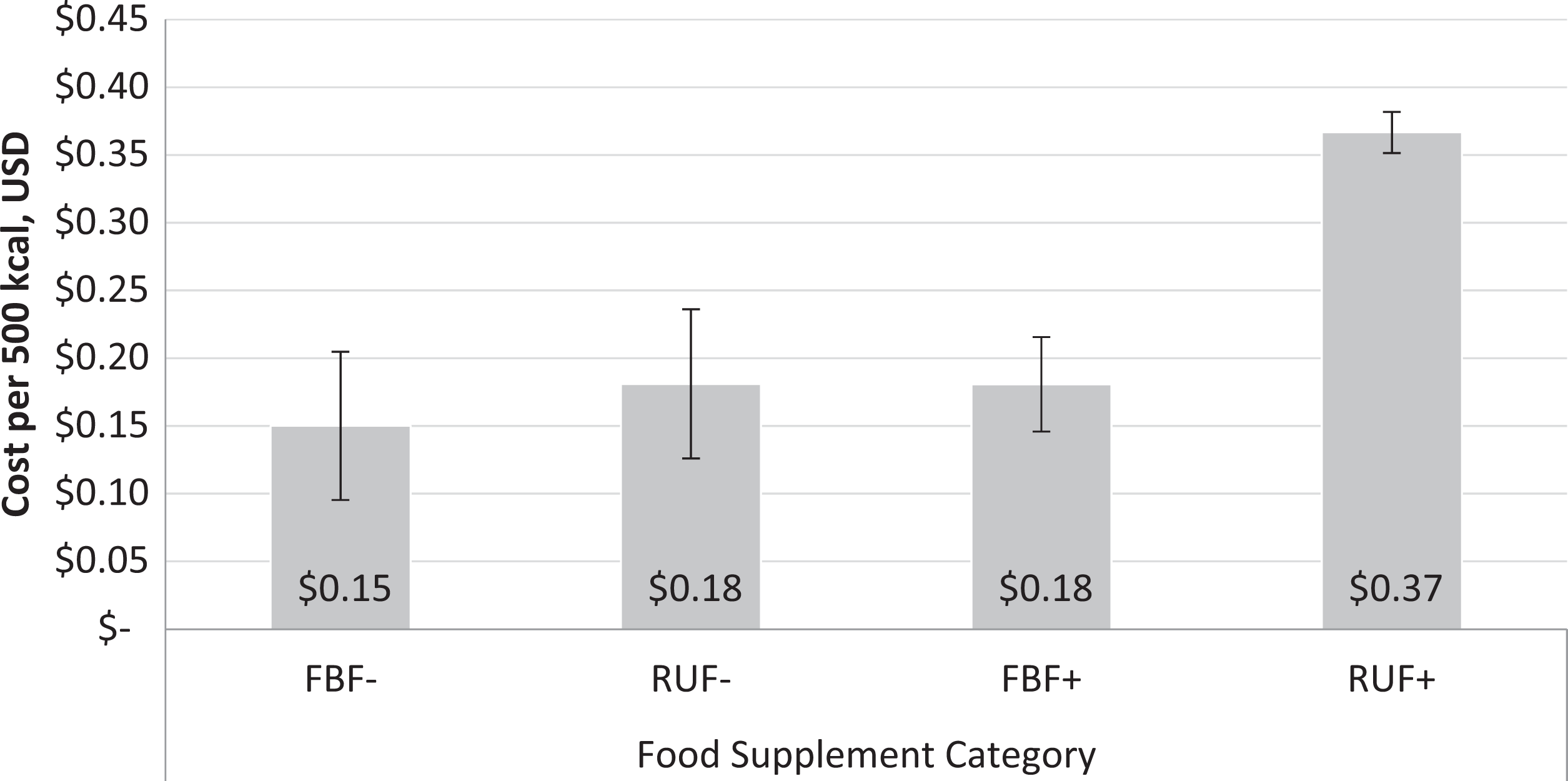
Average cost per 500 kcal of food supplement, by supplementary food arm and dairy category among studies that reported cost data, in 2015 US dollars. Error bars are standard deviations. FBF−, n = 4; RUF−, n = 3; FBF+, n = 2; RUF+, n = 3. FBF+ indicates fortified blended food with dairy; FBF−, fortified-blended food without dairy; RUF+, ready-to-use food with dairy; RUF−, ready-to-use food without dairy.
Discussion
This review found that generally young children with MAM treated with RUF, regardless of dairy content, had higher, faster recovery rates, and weight gain outcomes compared with nondairy FBF; however, when FBF contained dairy, those differences largely disappeared. Few studies found differences in length gain (perhaps not surprisingly due to the short duration of the treatments), but the 2 comparisons that did were between a supplement with dairy versus 1 without, an effect of dairy on length gain that has been documented previously. 8,10,20 While 3 recent meta-analyses reached similar conclusions for these outcomes between more broadly categorized RUF and FBF, 12 –14 our findings are novel in the distinction between food supplements with and without dairy.
Somewhat surprisingly, we did not find indications of differential effectiveness between RUFs with and without dairy. However, the RUFs in the included studies were heterogeneous, containing varying protein sources. Dairy ingredients may have differential biological effects depending on levels of whey, lactose, and other growth-promoting compounds. 10 The amount of dairy per ration was not reported among the reviewed studies but a dose–response or optimal quantity of dairy needed to have discernable effects on growth outcomes have been examined in the context of treatment of severe acute malnutrition and a preschool feeding program. 21,22 In addition, daily rations were heterogeneous in caloric value within and among studies but it was beyond the scope of this review to examine the quantitative relationship between caloric ration and recovery rate. Subsequent studies will need to examine ration size, the dose–response effect of dairy content, and type of dairy in RUFs and FBFs for treatment of MAM, through more detailed analysis of existing data (eg, meta-analysis) and future trials designed specifically to test these factors.
While these studies varied in details such as location, duration, and sample size, there was actually little variation in the food supplement costs per 500 kcal among FBF−, FBF+, or RUF−. However, RUF+ was consistently the most expensive in all studies, on average almost double the cost of the other 3 food supplements per kcal. While we could not estimate cost-effectiveness—which requires a total cost of treatment per child taking into account duration of treatment and ideally including costs not only of food products but of transportation, distribution, and other programmatic costs—if isocaloric rations and equal treatment duration were assumed, the cost per recovered child would be much higher in RUF+. However, differences in ration size, duration of treatment required to achieve recovery, and other factors make it difficult to estimate true cost-effectiveness and underscore the need to conduct cost-effectiveness analysis in conjunction with effectiveness when evaluating these food supplements.
Overall, this review finds that there is a consistent benefit of fortified-blended foods that include dairy in treatment of children with MAM. The benefits of including dairy in RUF may depend on the amount of dairy as well as other ingredients and protein sources. For both FBF and RUF, the optimal amount and type of dairy remain research priorities, including more detailed quantitative analysis of existing data. Additionally, evidence is urgently needed on food supplement and program costs combined with duration of treatment in order to guide policy on the most cost-effective treatments, which may vary by location and context of treatment programs. Continued research into optimal, cost-effective treatment of children with MAM should be carried out in parallel with ongoing sustainable preventative strategies to reduce the heavy burden of child malnutrition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication is made possible by the generous support of the American people through the United States Agency for International Development (USAID). The contents are the responsibility of Tufts University under the terms of Contract AFP-C-00-09-00016-00 and do not necessarily reflect the views of USAID or the United States Government.
