Abstract
INTRODUCTION:
Crohn’s disease (CD) is characterized by a chronic inflammation of the gastrointestinal tract causing abdominal pain, diarrhea, weight loss and systemic symptoms. Although the etiology of this disease is unknown, current knowledge suggests a multifactorial genesis involving genetic, environmental and immunological factors.
EVIDENCE ACQUISITION:
We focused our attention on critical analysis of the recent literature on the role of gut microbiota in inflammatory bowel disease (IBD), by evaluating the differences of composition, functions and role of intestinal flora. In particular, we focused on evidences about the interaction between gut microbiota and pathogenesis of IBD. In this setting, we conducted a PUBMED search for guidelines, systematic reviews (SR) and primary studies.
EVIDENCE SYNTHESIS:
Some data suggest that, in a significant percentage of patients, the microbiota plays an important role in the genesis and maintenance of CD. Probiotic supplementation and antibiotic treatment appear to be a valid therapeutic approach, although the clinical data remain controversial.
CONCLUSIONS:
Despite the exciting and growing research on the role of gut microbiota in IBD, our knowledge remains fairly limited. Further studies are needed to measure the diversity, function and resistance to antibiotics of the intestinal microbiota in CD.
Keywords
Introduction
Crohn’s disease [CD] is a life-long and disabling disease. It could affect any portion of the gastrointestinal tract from mouth to anus, with a propensity for the distal small intestine and proximal large bowel [1]. Inflammation is discontinuous along the bowel longitudinal axis and can involve all layers from mucosa to serosa (transmuralphlogosis) [1]. The precise etiology is unknown and therefore a causal therapy is not yet available. Probably CD arise from an interaction between genetic and environmental factors:inparticular, the role of gut microbiota has been highlighted by recent evidences [2].
Gut microbiota
It is estimated that the human microbiota contains about 1014 cells [3] and most of these cells are found in the gastrointestinal (GI) tract, with a range from 101–103 bacteria per gram of content in the stomach and duodenum to 1011–1012 cells per gram in the large intestine [4]. Currently more than 50 bacterial phyla have been described, although some phylotypes predominate: the
Moreover, it has been established not only that the microbial composition varies between upper and lower GI tract [6],but also that gut microbiota significantly differs between its composition existing in the lumen and that fixed to imbedded in the mucus layer of the GI tract [7].
The microbial composition of the small intestine differs from that of the colon. For this reason, faecal samples are considered to be more representative of the luminal colonic microbiota than of the microbial residents of the small intestine, while, intraluminal catheter samples and biopsies from locations in the small intestine are regarded as more representative of the small bowel microbiome. Furthermore, stool samples are poor determinants of the microbes associated with the bowel mucosa [8, 9]. Bacteria belonging to the
Physiological role of the gut microbiota
The intestinal microbiome is involved in a wide number of physiological processes, including digestion, metabolism, immune system development and immune response. Moreover, animal studies have suggested that intestinal microbes are implicated in brain development and central nervous system signalling [12]. The digestive enzymes found in the human GI tract are unable to degrade certain carbohydrates such as resistant starch and cellulose; however, intestinal microbes convert these compounds through fermentation, generating metabolites that are favourable to the host. One of the end-products of the fermentation process is short chain fatty acids (SCFAs) that possess anti-inflammatory properties and act as a source of energy. Moreover, the gut microbiota can degrade proteins, which results in various toxic metabolites but also SCFAs. Additionally, intestinal microbes synthesize indispensable amino-acids [13]. Also, certain vitamins are produced by the microbiota, including vitamin K and components of vitamin B [14].
Some gut microbes are able to indirectly reduce serum cholesterol through conversion of bile salts, thereby reducing the enterohepatic bile reabsorption and increasing the secretion of bile salts in the faeces [14]. The development and maturation of the human immune system are influenced by both host genetics and environmental elements, including the interaction of microbes with the host. The maturation of gut-associated lymphoid tissue (GALT) (e.g. mesenteric lymph nodes and Peyer’s patches) relies upon post-natal interaction with microbes, which leads to the expansion of lymphoid tissue and formation of germinal centres containing immunoglobulin A (IgA) secreting B cells [15]. Moreover, the intestinal microbiotais also involved in the development of T cells [16].
Microbiota in Crohn’s disease
Several studies have reported differences between the microbiota of CD patients and healthy subjects. In particular, some authors showed that CD patients had lower amounts of
The role of specific microbes in the pathogenesis of CD has been studied extensively, and two bacteria have been suggested to play a role in CD development: AIEC and
The involvement of MAP in the pathogenesis of CD is controversial. Some studies have shown the presence of this bacterium in tissues from CD patients, others have not [28, 32]. The hypothesis of MAP is the causative agent of CD suggests that antimycobacterial treatment would lead to eradication of the bacterium from the gut, and thereby to the induction of remission. However, antimycobacterial therapy did not induce a long-term improvement of disease in CD patients, thus showing that MAP is not the causative agent of CD [33].
In addition, as above reported, the gut microbiotais associated with several aspects of the human immune system. For instance, the normal resident flora is involved in the development and maturation of GALT, and it is associated with mucosal secretion of immunoglobulins [15, 16]. Furthermore, animal studies have suggested that the microbiota is implicated in the development, regulation and induction of immune cells; including T cells and APCs [34–36]. Additionally, members of the microbiota may influence the integrity of the intestinal epithelial barrier, and produce anti-inflammatory metabolic compounds that are utilized by intestinal epithelia (e.g. SCFAs) [13] and [22]. Also, certain microbes may influence the inflammatory process itself, causing either an increase [37–39] or a decrease [40, 41] in bowel inflammation, and inflammation-associated cancer development [42].
Therapeutic potential of microbiota
CD patients exhibit alterations in their microbiota function and structure and these microbial alterations are thought to participate in the pathogenesis of the disease (Fig. 1). In this setting, therapies aimed at restoring the CD microbiota into a non-dysbiotic one would prove beneficial effects. Besides antibiotics recent treatment options include the pro- and prebiotics supplementation, and faecal microbiota transplantation.
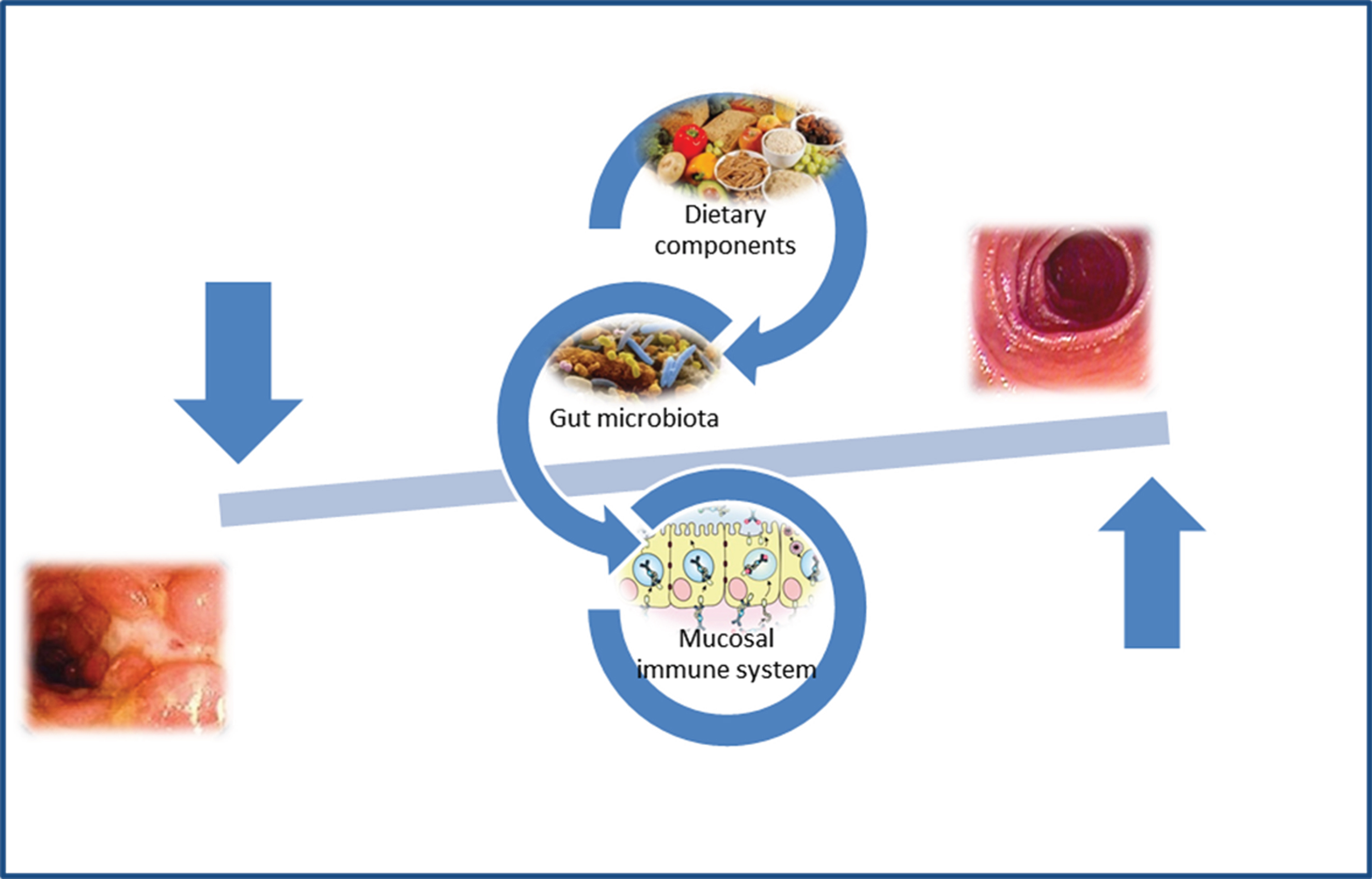
Gut microbiota and pathogenesis of Crohn’s disease.
Prebiotics are non-digestible dietary components that promote the growth of beneficial intestinal microbes (i.e. probiotics like
A few trials have conducted to evaluate the use of synmbiotics (synergistic combination of a probiotic and prebiotic) in CD [47, 48]. Steel et al. conducted a randomized, double-blind placebo-controlled trial involving 35 patients with active CD, using a synbiotic comprising Synergy 1 and Bifidobacterium longum [47]. Synbiotic determined significant improvement in clinical outcomes, with reductions in both Crohn’s disease activity indices (
As discussed above, the use of pre- and probiotics and synbioticsappears to be of little benefit in the treatment of CD; however, scientific data on their application and effect in this disease are limited, so further investigations are needed in order to determine the real role of these products in the treatment of CD.
Fecal microbiota transplantation
Faecalmicrobiota transplantation (FMT) wasfirst used 1958 in 4 cases of pseudomembranous colitis, thought to be caused by
Conclusions
A growing number of patients are diagnosed with IBD worldwide. The incidence is increasing, especially in newly industrialized countries, probably due to the relatively recent westernization of these societies. The recognition of several susceptibility genes and environmental risk factors has focused on the intestinal microbiota, that appear to play an essential role in disease pathogenesis, probably because of inappropriate interactions with the gut innate immune system. Initial findings regarding microbiota modulation are promising and several studies are elaborating on whether or not it can be useful in the treatment of CD. As numerous disease conditions appear to be related to the intricate microbial communities residing in human body, the future of this “new field” of medicine is certainly interesting and stimulating.
