Abstract
Background:
Globally, ready-to-use therapeutic food (RUTF) with peanut and milk as the primary source of protein is used to treat children having severe acute malnutrition (SAM). Valid Nutrition in collaboration with Ajinomoto Co., Inc has developed a nonmilk RUTF from soybean, maize, and sorghum (SMS-RUTF) and demonstrated its efficacy.
Objective:
To pilot SMS-RUTF in treatment of SAM within Community-Based Management of Acute Malnutrition (CMAM) program in Malawi, Africa.
Methods and Findings:
This was implemented from January to July 2018 and its performance was based on the SPHERE criteria and Ministry of Health CMAM guidelines. A total of 742 children were treated with SMS-RUTF. Of these, 94.5% (95% CI: 92.6-96.0) were successfully discharged to supplementary feeding program (SFP) with middle upper arm circumference (MUAC) ≥115 mm or directly to their homes with MUAC ≥125 mm; 3.6% (95% CI: 2.4-5.3) defaulted, 1.9 % (95% CI: 1.0-2.1) died, and 0.0% nonresponders. Analysis of 222 children who were discharged home with MUAC ≥125 mm gave a recovery rate of 88.3% (95% CI: 88.3-92.2), a defaulter rate of 6.8 % (95% CI: 3.8-10.9), a mortality rate of 1.3% (95% CI: 0.3-3.9), and a nonresponders rate of 1.8% (95% CI: 0.5-4.5). These outcomes exceed SPHERE minimum performance standards. The mean (standard deviation) length of stay of children discharged to SFP and discharged directly home were 42.0 (20.9) and 46.1 (21.1) days, respectively. These outcomes are within the recommended average duration of <60 days.
Conclusion:
The pilot CMAM program using SMS-RUTF recipe that contains no milk or peanuts achieved SPHERE minimum standards. Based on this evidence, SMS-RUTF should be encouraged for treatment of SAM in children between 6 and 59 months in routine CMAM programs in Malawi and globally.
Keywords
Introduction
Severe acute malnutrition (SAM) remains a global public health concern, as a leading cause of infant and child mortality in developing countries, resulting in an estimated 500 000 childhood deaths each year. 1 In 2018, the World Health Organization (WHO), the United Nations Children’s Fund (UNICEF), and the World Bank Group published findings which revealed that globally, over 49 million children younger than 5 years were malnourished (wasted) and nearly 17 million were severely wasted. 2 According to the report, about 68% and 28% of the world’s malnourished children were from Asia and sub-Saharan Africa, respectively.
In Malawi, Africa, in 2015, the estimated SAM caseload for children younger than 5 years was 79 301. 3 This prompted the Government of Malawi (GOM) to include management of malnutrition on the list of priorities for social development of the 2011 to 2016 Malawi National Growth and Development Strategy (MGDS) II 4 and was maintained as a priority in the MGDS III (2018-2022) 5 The GOM adopted the WHO/UNICEF/United Nations System Standing Committee on Nutrition model for treating acute malnutrition, Community-Based Management of Acute Malnutrition (CMAM). 6 This model was established as a core component of the Malawi Nutrition policy and Malawi CMAM operational plan. 7 The CMAM approach was developed to provide treatment and care to children through outpatient care outside of the nutrition rehabilitation units (NRUs). There are 4 components to the CMAM: community outreach, supplementary feeding program (SFP), outpatient management of SAM, and inpatient management of SAM. The program is developed to treat children with SAM without medical complications on an outpatient basis, using a ready-to-use therapeutic food (RUTF), whereas those with medical complications continue to be treated in 24-hour inpatient care facilities. 7 Although the CMAM approach has increased treatment coverage globally, still only 23% of children with SAM are able to access treatment. 8 One of the main reasons for this low treatment coverage is high cost of RUTF, a lipid-based, nutrient-dense paste that is currently the primary nutritional product for treatment.
The most commonly used RUTF is peanut and milk-based RUTF (PM-RUTF); a mixture of milk powder, sugar, vegetable oil, peanut butter, vitamins, and minerals. 9,10 The cost of PM-RUTF in treating SAM is the largest component and in Malawi accounts to 32% of the overall CMAM cost. 11 About 50% of PM-RUTF ingredients including milk are produced in developed countries. 12 The importation of these ingredients considerably impedes the ability to increase local production of the RUTF formulation in developing countries. Since the introduction of the CMAM program, considerable efforts have been made to develop alternative milk-free and peanut-free RUTFs, using locally grown and produced crops. However, to date, a large majority of the new recipes have been less effective than the PM-RUTF in the treatment of SAM among children younger than 5 years. 13 -15
A plant-based RUTF formulation, which is lower cost and produced predominantly from ingredients that can be grown in most sub-Saharan African countries, has recently been developed by Valid Nutrition and Ajinomoto Co, Inc. Although the efficacy study results from the use of SMS-RUTF for the treatment of SAM without medical complications was confirmed in a day-care setting in a randomized controlled efficacy clinical trial, 16 the translation of the results from children treated in a day-care setting to a routine outpatient treatment program (OTP) within a CMAM program is unknown. This determination is essential, as per the guidance of several regulatory bodies, to show whether efficacy of a new treatment observed in a controlled research environment remains effective in a pragmatic context. 17,18 Therefore, subsequent to the efficacy study conducted by Bahwere et al 16 above, this pilot was undertaken to evaluate whether the effectiveness of treatment of SAM among children using SMS-RUTF previously observed in a day-care setting is translated similarly into a routine CMAM outpatient program run as per the Malawi Guidelines for CMAM. 19
Methods
Program Objectives
The primary objective was to pilot SMS-RUTF in treatment of SAM in a CMAM program in 3 districts in Malawi. The assessment criteria were based on the SPHERE minimum standards performance criteria for recovery rate (>75%), default rate (<15%), and mortality rate (<10%). 20 The secondary objectives were nonresponse rate (<2%), weight gain velocity of 5.0g/kg/d, and length of stay (LOS) of 40 to 62 days in the treatment program. 19
Program Design
This was a pilot program conducted according to the established Malawi Guidelines for CMAM. 19 In contrast with the day-care approach employed during the previous simple randomized controlled efficacy clinical trial, this pilot was conducted in a routine CMAM program, implemented by the Malawi Ministry of Health (MOH).
In Malawi, CMAM is fully integrated into the national health delivery system and services provided by all the public health facilities using the Malawi Guidelines for CMAM updated in 2016. 19
Ethical considerations
Ethical approval for the CMAM pilot was obtained from the National Health Sciences Research Committee (NHSRC) of the Malawi MOH (Approval Number 1937, January 25, 2018). Participation in the study was voluntary. At the time of admission, each child’s parent or caregiver was informed in their local language about the nature and purpose of the study. Parents or caregivers were invited to have their children participate in the study after verbal and written consent were obtained. If a parent or caregiver withheld consent for participation, children were referred to the PM-RUTF outpatient program as per the Malawi Guidelines for CMAM. 19
Location
The pilot was conducted in Dedza, Mchinji, and Salima districts located in the central region of Malawi. These districts had the highest levels of undernutrition as per the 2015 to 2016 Malawi Demographic Health Survey. 21 The 3 districts were selected based on: (1) high numbers of SAM cases (average of 600 new SAM admissions from September 2015 to February 2016);(2) prior extensive experience of the MOH in running CMAM programs; (3) good accessibility allowing for close supervision; and (4) the pre-existence of collaboration with World Vision Malawi. In the districts, all health facilities totaling 69 implementing CMAM participated in the pilot study (ie, 34 in Dedza, 17 in Mchinji, and 18 in Salima).
Production and distribution of SMS-RUTF
Valid Nutrition (VN) produced the SMS-RUTF at its UNICEF-certified factory in Lilongwe, Malawi. The product was verified to meet the required specifications. The detailed description of the SMS-RUTF product has been presented by Bahwere et al. 16 The SMS-RUTF product was distributed by VN in collaboration with World Vision and MOH to each of the locations for the study.
Program criteria for admission and discharge
Participants in the pilot program were children diagnosed with SAM, who were admitted into CMAM programs implemented by the MOH and Christian Association of Malawi (CHAM) in the selected districts.
Admission criteria into OTP were per the Malawi Guidelines for CMAM (Table 1). Children were admitted to the OTP if they had (1) an MUAC of <115 mm, (2) weight-for height/length (WFH/L) z score <−3, or (3) a grade 1 or 2 of bilateral pitting edema, and they had an appetite for RUTF and they did not have medical complications. Children with medical complications were referred to the NRU to receive inpatient care until their condition stabilized and they were then referred to the OTP. Additionally, during follow-up, children identified as having medical or nutritional complications were also referred to the NRU for appropriate treatment, after which they were readmitted to the OTP. Nutrition rehabilitation was accomplished as per the Malawi Guidelines for CMAM; therapeutic milks F-75 and F-100 were provided during inpatient stay, and SMS-RUTF was used during OTP treatment.
Outpatient Therapeutic Program Admission and Discharge Criteria.a
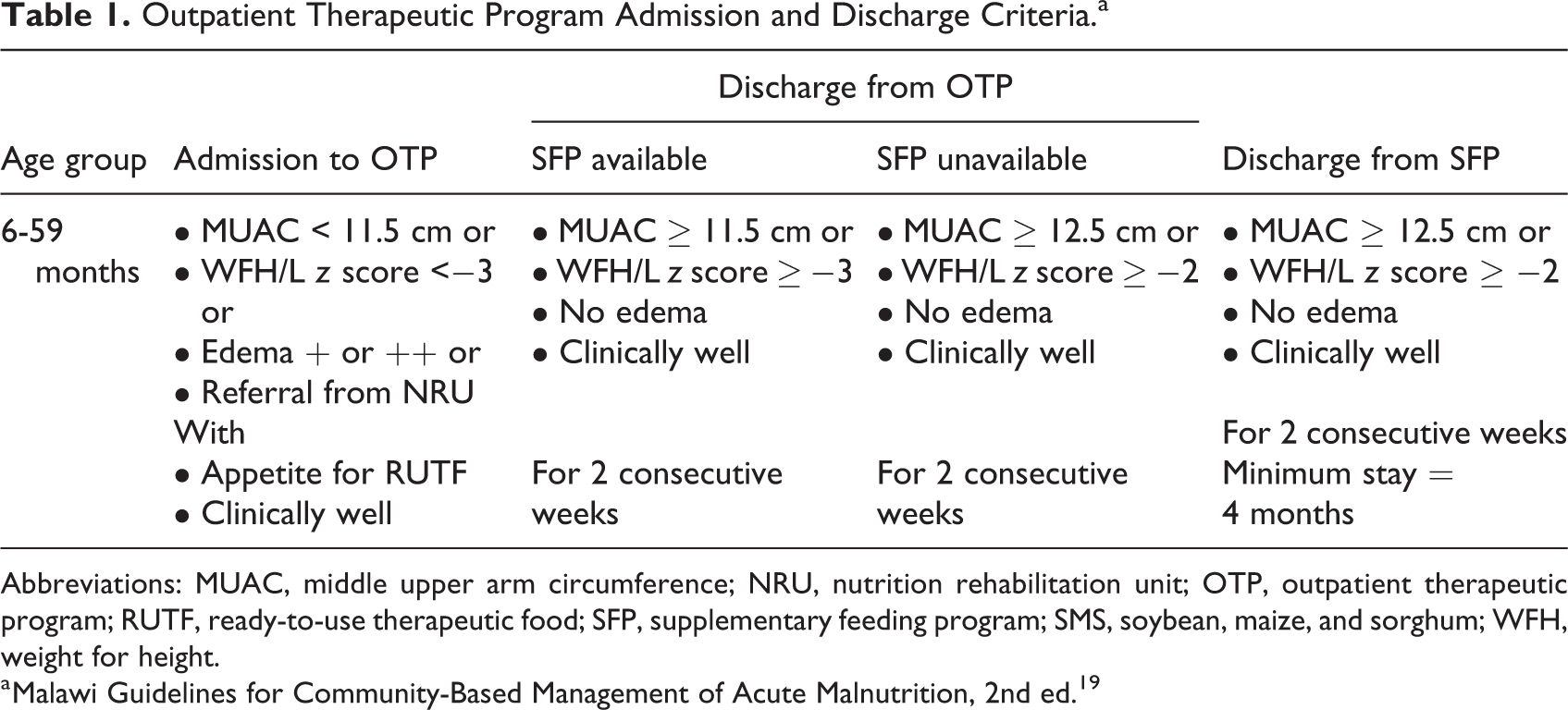
Abbreviations: MUAC, middle upper arm circumference; NRU, nutrition rehabilitation unit; OTP, outpatient therapeutic program; RUTF, ready-to-use therapeutic food; SFP, supplementary feeding program; SMS, soybean, maize, and sorghum; WFH, weight for height.
a Malawi Guidelines for Community-Based Management of Acute Malnutrition, 2nd ed. 19
Some children who were already admitted into CMAM program on PM-RUTF were shifted to SMS-RUTF due to stock out of PM-RUTF. Once the PM-RUTF was restocked, children were allowed to continue consumption of SMS-RUTF if they wanted to. At the end of the pilot or at any point SMS-RUTF supplies were exhausted, children who had not reached the discharge criteria continued treatment with PM-RUTF.
Discharge criteria were as per the Malawi Guidelines for CMAM (Table 1). The OTP discharge criteria is MUAC ≥115 mm or no edema or WFH/L z score ≥−3 into SFP where available. They were discharged at MUAC ≥125 mm or no edema or WFH/L z score ≥−2 where there was no SFP, and children were only discharged when the discharge criteria were met for 2 consecutive weeks.
Program size
The program was designed to cover all the 3 selected districts during the months of January to July 2018. This period is usually associated with increased SAM cases. A convenient sample of 1000 SAM admissions of 6 to 59 months old children was expected based upon 2017 MOH SAM admission data (2017 National CMAM Report, MOH; unpublished). All children classified as being severely malnourished without complications were eligible to be included in the pilot program.
Monitoring and follow-up
All children in the OTP program were monitored on weekly basis, for medical checkup, nutrition assessment (ie, measures of MUAC, weight, height or length, and edema), response to treatment, and restocking of the RUTF ration. Information pertaining to the medical checkup and nutrition assessment was recorded on each child’s OTP card, which were maintained at the health facility. At the time of the pilot implementation, Malawi was rolling out use of the Malawi Guidelines for CMAM, as such, no OTP cards were available for new patients with SAM in some of the health facilities where training in the revised CMAM guidelines had not yet been done. This affected weekly recording of weight, MUAC, height/length, health status, and sociodemographic data. The affected patients are labelled as OTP card N/A in Figure 1. Following each OTP weekly session, personnel at the health facility recorded the program’s outcome summary data (recovery rate, defaulter rate, mortality rate, and nonresponse rate) on a tally sheet.
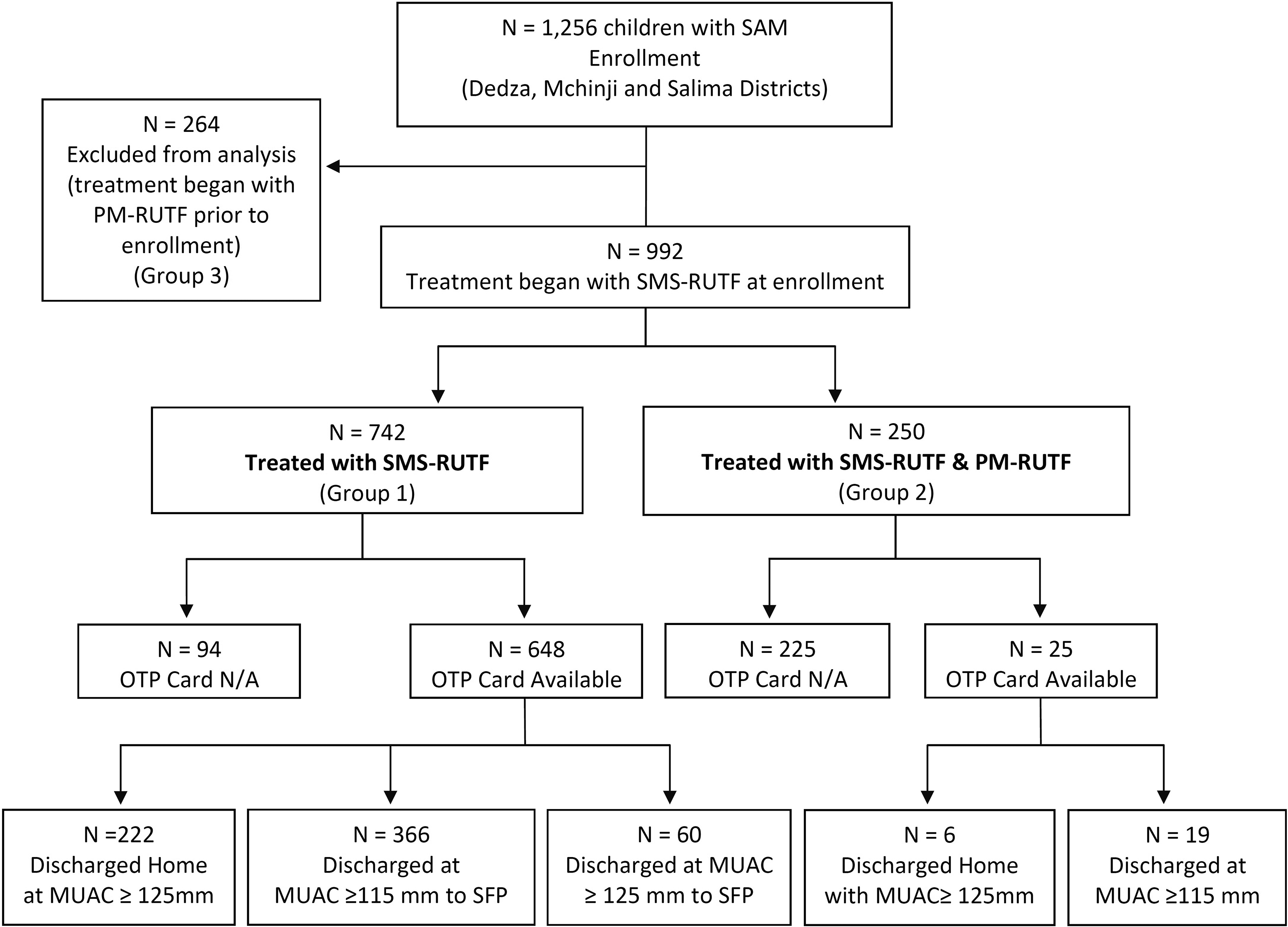
Program participant flow diagram.
Data collection
Data collection was conducted and compiled by Health Surveillance Assistants (HSAs) at each health facility and conveyed to the district health office for district level consolidation. Project monitoring and evaluation team members collected copies of compiled health facility data for monthly monitoring of performance of all children with SAM.
Outcomes of interest and definitions
Program performance: The principal performance criteria used for describing effectiveness of the SMS-RUTF pilot program were based on SPHERE minimum standards: The proportion of discharges from OTP who recovered—those who reached MUAC ≥125 mm or absence of edema or WFH/L z score ≥ −2 for 2 consecutive weeks (ie, effectiveness was confirmed acceptable if the recovery rate was >75% for children exclusively treated with SMS-RUTF and the lower limit of the corresponding 95% CI was >75%); The proportion of discharges from OTP who defaulted—those who were absent for 3 consecutive weeks (ie, default rate was confirmed as acceptable if it was <15% for those treated exclusively with SMS-RUTF and the upper limit of the corresponding 95% CI was <15%); The proportion of discharges from OTP who died in program (ie, mortality rate was confirmed acceptable if it was <10% for those treated exclusively with SMS-RUTF and the upper limit of the corresponding 95% CI was <10%).
The secondary performance criteria included: The proportion of discharges from OTP who did not respond to treatment for more than 12 weeks (ie, nonresponse rate was <2% for children exclusively treated with SMS-RUTF and the upper limit of the corresponding 95% CI was <2%); The average weight gain of children with nonedematous SAM at admission, but discharged as recovered (ie, average weight gain was considered as acceptable if 4-5 g/kg/d in children treated exclusively with SMS-RUTF)
19
; and The average LOS of children discharged as recovered (ie, average LOS was considered acceptable if it was 40-62 days in children treated exclusively with SMS-RUTF).
19
Data management
All staff involved in data management were trained taking anthropometric measurements and were standardized. On admission and at each subsequent visit, the child’s weight was measured (naked or wearing light undergarments and without shoes) to the nearest 100 g using Salter scales (Salter-Brecknell 235-6 S series). Middle upper arm circumference was measured to the nearest millimeter using a standard nonelastic MUAC tape (UNICEF supply code S0145620 “MUAC, Child 11.5, Red/PAC-50”). On admission and discharge, the child’s height was measured to the nearest millimeter using a standard pediatric height board (supine length was measured for children <24 months of age).
Data management was performed by the data quality manager and the principal investigator in collaboration with project field officers, district nutrition coordinators, and health facility CMAM focal persons. This team spot checked the quality of measurements for the parameters being assessed during field supervision visits. These included checks on edema diagnoses, outcomes analysis, individual data collection forms, and completion of program questionnaires, and mentoring of health facility staff (HSAs and project field assistants). Individual data collection forms were double-checked for accuracy and completeness at the time of the child’s discharge from the pilot program. The verified forms were then collected for data entry using Epi Data version 3.2. Quality of data was monitored through double entry and verification.
Data analysis
Data were cleaned and exported to STATA 13 software (Stata Corp LLC) for analysis. Sociodemographic and nutritional characteristics of children who were treated with SMS-RUTF at one point or the other were determined from OTP cards on admission (Table 2). This group was further divided into those who were treated exclusively on SMS-RUTF (group 1) and those who combined SMS-RUTF and PM-RUTF (group 2). Program performance (recovery, defaulter, mortality, and nonresponse rates) was determined by analysis of anthropometric measurements (weight, height, and MUAC), OTP attendance, and deaths from weekly tally sheets and OTP cards (Table 3). In addition, children were stratified by age because in our previous studies in Zambia and Democratic Republic of Congo, children younger than 2 years showed lower performance than other age-groups using similar RUTF formulation without added amino acids. 14 Further analysis was done on children who fully recovered and were discharged home to confirm performance of SMS-RUTF where there is no SFP, which is common in most of the districts and other countries.
Sociodemographic and Nutritional Characteristics of Children With Severe Acute Malnutrition Enrolled in the Pilot Program (Groups 1 and 2) With Completed OTPa Cards on Admission.b
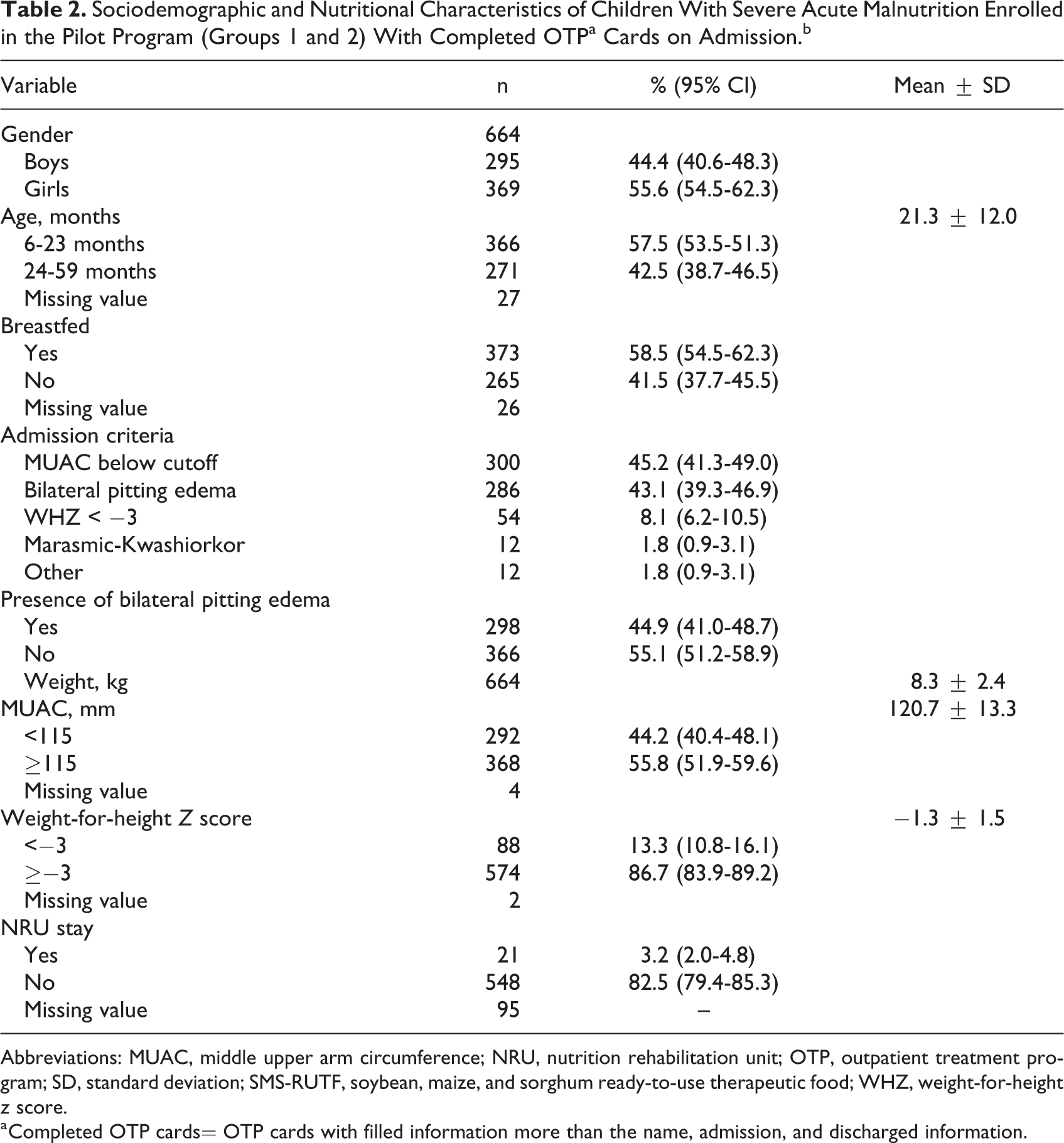
Abbreviations: MUAC, middle upper arm circumference; NRU, nutrition rehabilitation unit; OTP, outpatient treatment program; SD, standard deviation; SMS-RUTF, soybean, maize, and sorghum ready-to-use therapeutic food; WHZ, weight-for-height z score.
a Completed OTP cards= OTP cards with filled information more than the name, admission, and discharged information.
Key Performance Indicators of the Pilot Program Based on Tally Sheet Records.
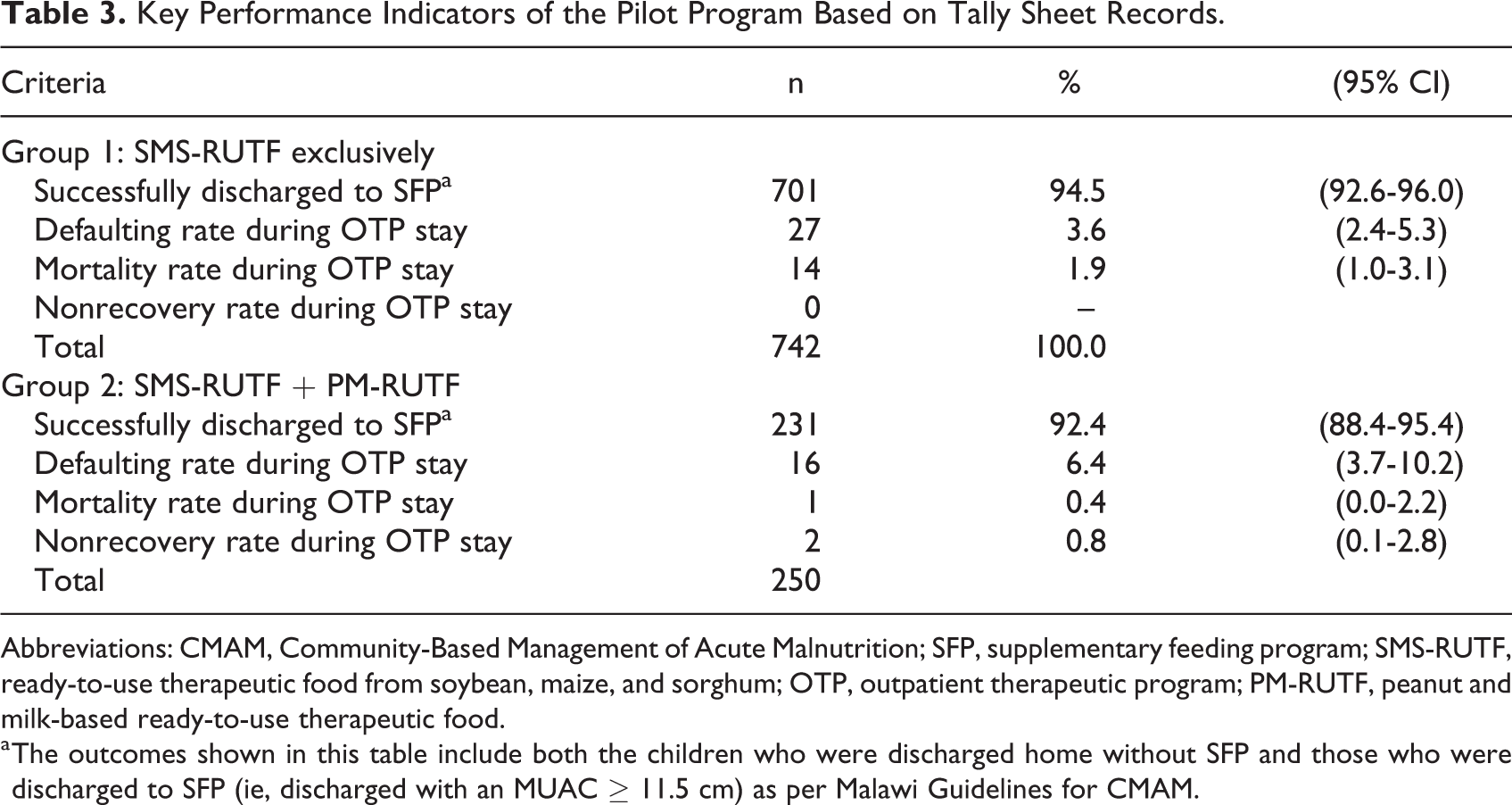
Abbreviations: CMAM, Community-Based Management of Acute Malnutrition; SFP, supplementary feeding program; SMS-RUTF, ready-to-use therapeutic food from soybean, maize, and sorghum; OTP, outpatient therapeutic program; PM-RUTF, peanut and milk-based ready-to-use therapeutic food.
a The outcomes shown in this table include both the children who were discharged home without SFP and those who were discharged to SFP (ie, discharged with an MUAC ≥ 11.5 cm) as per Malawi Guidelines for CMAM.
Means and standard deviations, medians and proportions, and 95% CIs were used to describe the admission and exit parameters, as appropriate. We used the t test to compare average weight gain with recommended standard and Pearson chi-square test to compare proportions of LOS to the Malawi Guidelines for CMAM standard of 40 to 62 days.
Results
Admissions of children with SAM into SMS-RUTF pilot program started in January 2018 after ethical approval by NHSRC and ended in July 2018.
Participants
Participants in the pilot program were children aged 6 to 59 months diagnosed with SAM, who were admitted into CMAM programs in 69 health facilities implemented by the MOH and Christian Association of Malawi (CHAM) in Dedza, Salima, and Mchinji districts. Active case finding of children with SAM was done routinely in all health facility catchment areas through household visits and monthly mass screening by care group volunteers and HSAs as part of community mobilization for CMAM. During the course of the pilot, the MOH increased the number of health facilities implementing SFP which led to more children aged 6 to 59 months discharged into SFP with an MUAC of ≥115 mm.
A total of 1256 children with SAM were enrolled in the SMS-RUTF pilot program in all the 3 districts (Figure 1). Of these, 264 children (group 3) started treatment with PM-RUTF prior to the initiation of the pilot program and were shifted to SMS-RUTF due to PM-RUTF stock out. This group contributed to the increase of the sample size from an estimated 1000 to 1256 and stock out of the SMS-RUTF toward the end of the program. These children were not included in the analysis. Of the remaining 992 children, 742 were treated with SMS-RUTF exclusively (group 1) and 250 children were later shifted to PM-RUTF due to stock out of SMS-RUTF toward the end of the pilot program (group 2). The SMS-RUTF production was only for 1000 children with 10% contingency.
Of 742 children with SAM exclusively treated with SMS-RUTF, 648 OTP cards were available with detailed discharge criteria, and 222 children with SAM were discharged from the OTP directly to their homes where there was nonavailability of SFP (Figure 1). Table 2 describes the characteristics of children enrolled in SMS-RUTF groups (groups 1 and 2) based on the analysis of data from the OTP cards.
Program Performance
Program performance for groups 1 and 2 was determined by analysis of the weekly tally sheets (Table 3). Group 1 (742 exclusively on SMS-RUTF) showed (95% CI: 2.4-5.3) a recovery rate of 94.5% (95% CI: 92.6-96.0), defaulter rate of 3.6% , and mortality rate of 1.9% (95% CI: 1.0-3.1). Group 2 showed a recovery rate of 92.4% (95% CI: 88.4-95.4), defaulter rate of 6.4% (95% CI: 3.7-10.2), and mortality rate of 0.4% (95% CI: 0.0-2.2). These performance indicators exceeded the SPHERE minimum standards. 20
Subgroup analysis
Table 4 presents outcomes for the 222 children who were exclusively treated with SMS-RUTF and were discharged from OTP to home. The results indicate that the observed recovery, default, and mortality rates exceeded the SPHERE minimum standards for both the overall analysis and for the analyses stratified by age and bilateral pitting edema.
Key Performance Indicators of the Pilot Program Based on Analysis of OTP Cards for Children Discharged Home From OTP as per National CMAM Guidelines.
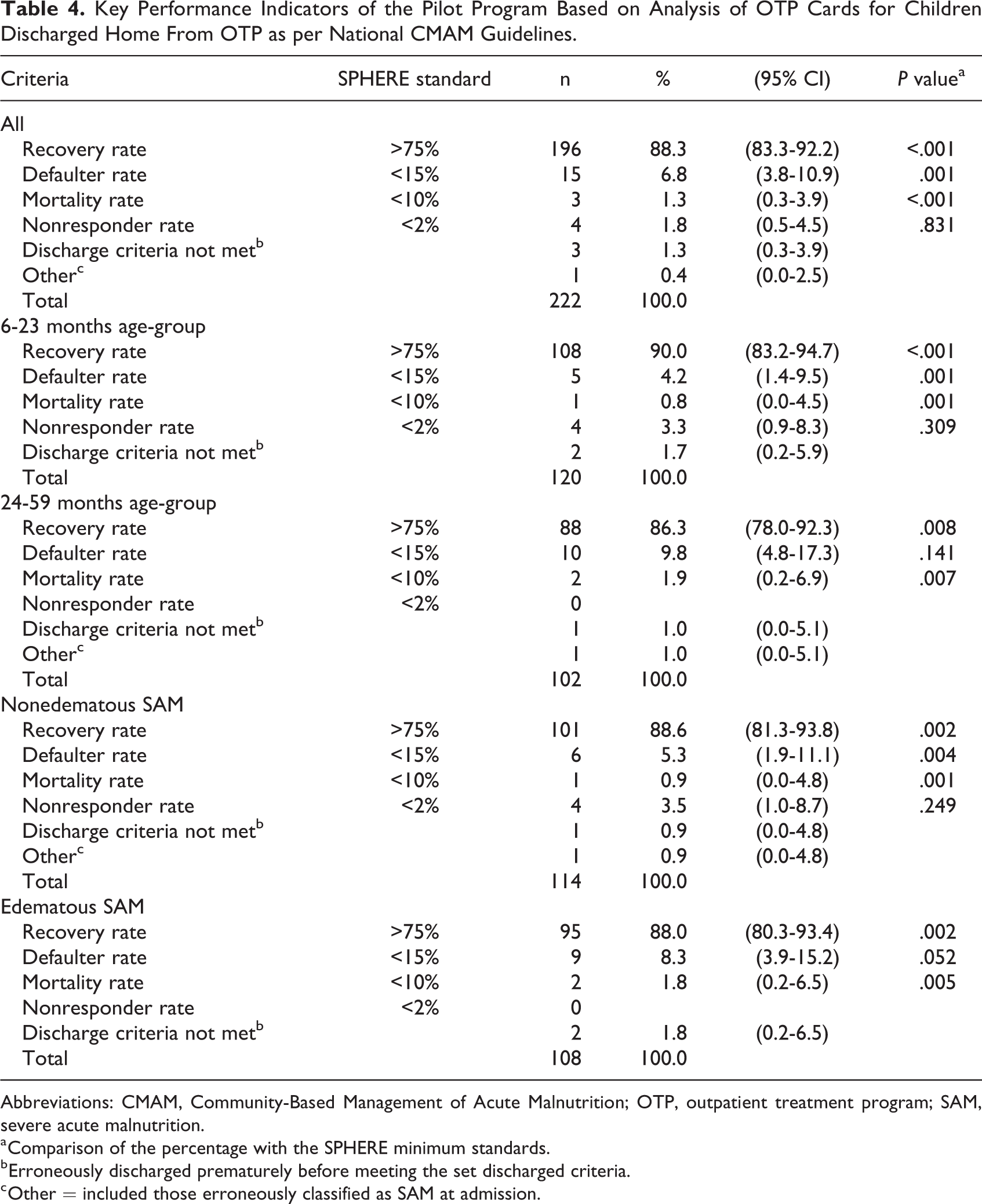
Abbreviations: CMAM, Community-Based Management of Acute Malnutrition; OTP, outpatient treatment program; SAM, severe acute malnutrition.
a Comparison of the percentage with the SPHERE minimum standards.
b Erroneously discharged prematurely before meeting the set discharged criteria.
c Other = included those erroneously classified as SAM at admission.
Patient Outcomes
All children whose OTP card information was available and who were successfully discharged to SFP showed a mean daily weight gain velocity of 3.9 ± 4.4 g/kg/d. Children without edema at admission had a weight gain velocity of 4.4 g/kg/d, which was 1.1 (0.3-1.8) g/kg/d (P = .004) higher than that of those admitted with edema. Overall LOS for groups 1 and 2 who were successfully discharged to SFP was 43.1 ± 21.2 days. Children admitted with edema had a shorter LOS than those admitted without edema, and the difference between the 2 groups was 3.6 (0.1-7.0) days (P = .041).
Subgroup analysis
A total of 222 children discharged from OTP to home had an average mean weight gain of 3.9±5.0 g/kg/d. This estimate is statistically equivalent to the recommended range for CMAM programs of 4 to 5 g/kg/d (P = .820). 22 Children without edema at admission had similar weight gain velocity to that of those admitted with edema with a difference of 0.9 (-0.5-2.3) g/kg/d (P = .205). The overall LOS for the entire samples was 43.1 ± 21.2 days. This is within the 40 to 62 days recommended in the Malawi Guidelines for CMAM. Additionally, LOS was shorter for children admitted with bilateral pitting edema than those admitted without edema with a difference of 7.4 (1.4-13.4) days (P = .016).
Discussion
Children with SAM need safe, palatable foods with energy, protein, fats, minerals, and vitamins tailored to their requirements. Many studies in Malawi and elsewhere have shown that providing PM-RUTF tailored to body weight successfully supports recovery in these children. 23 -26 The results presented in this report describe a “real-world” program performance in which SMS-RUTF is used instead of PM-RUTF and complement those of a recently published efficacy study. The efficacy study showed in a controlled research context that the milk-free formulation based on soya, maize, and sorghum and enriched with amino acids is not inferior to PM-RUTF for SAM treatment in children aged 6 to 59 months with respect to recovery rate and LOS. 16
The results from this pilot program which assessed the effectiveness in routine CMAM program confirm that the use of SMS-RUTF in management of SAM implemented ambulatory with routine MOH staff and community volunteers not only achieves but exceeds the SPHERE minimum standards of recovery, default, and mortality rates regardless of age and edema status. For recovery indicator, both SPHERE minimum standards (of >75%) and the national target for Malawi (of >85%) were exceeded. These results indicate that a CMAM program using SMS-RUTF and run by the MOH within normal delivery system with staffing and other operational challenges achieves similar performance and even better in some other instances than those of the controlled clinical efficacy trial. 16 Importantly, this pilot program experienced a low mortality rate below the minimum SPHERE standards of 2% and matching also mortality rates reported for well-run CMAM programs that used PM-RUTF for treating SAM in children younger than 5 years.
Contributing factors to such low mortality in addition to the effectiveness of the therapeutic food is certainly the active case finding through mass screening and care group volunteer household visits. These were deployed by MOH to maximize early detection and initiation of SAM treatment across the country. The default rate is also lower than what is usually observed in routine programs. 27,28 This could be due to consistent supply of RUTF regardless of the type in this pilot and community mobilization activities within health facility catchment areas.
The rate of weight gain observed in this pilot program is lower than what was observed in the efficacy trial cited above but is comparable to the weight gain reported in the literature for CMAM programs using PM-RUTF. 27 -29
These findings have important practical implications. The SMS-RUTF is made from ingredients that can be sourced locally in Malawi, with a lower proportion of ingredients that need to be imported for local production. As CMAM services are integrated in the MOH health delivery system, timely local production and availability of RUTF would have a significant positive impact on decentralized service provision and procurement of RUTF. In addition, use of SMS-RUTF at scale is likely to contribute towards strengthening local food manufacturing and improving local agricultural markets, both of which are important if malnutrition is to be prevented.
The findings of the pilot program presented in this report should be interpreted taking into account the fact that Malawi implements all the 4 components of CMAM (Community mobilization/active case finding, OTP, NRU/in patient, and SFP) as such SAM cases from OTP are discharged to SFP for nutritional support until they reach traditional cure discharge criteria. This was the case for the large majority of cases as only 222 of the 648 children with SAM who were exclusively treated with SMS-RUTF and whose OTP cards were accessed were kept in OTP until reaching the traditional exit criteria of cure, default, or nonresponse because of nonavailability of SFP within their health facilities. We were unable to get information on final outcomes at the time of exiting SFP for these children. Thus, we cannot ascertain that the trend and outcome were the same at the time of exiting OTP although SFP national reports from January to August 2018 showed that 80% to over 90% of the children in SFP reached discharge criteria (The Malawi MOH CMAM Report 2018; unpublished). Also the time they spent in SFP before exiting is not accounted for in our calculation of LOS. However, the results of the analysis of the 222 children discharged according to the traditional criteria confirm the effectiveness of SMS-RUTF as all the key performance indicators showed superiority when compared to SPHERE minimum standards. Also, as there were no reports of any relapse and referral back to OTP or NRU for these children, we assume that most of them were successfully discharged as cured and that the program performances presented in this report are close to the actual final performances of OTP and SFP combined data.
There were some few inaccuracies in captured data, for example, difference in performance as estimated by the data from tally sheets and those from OTP cards; some children were discharged as cured while the analyzed data from OTP cards indicated that their discharge was premature and only 664 of 673 cards were completed. This is one of the main reasons why there are differences in figures reported discharged and their performance indicators between tally sheets and OTP cards. These are some operational challenges that the MOH encounters and are dealt with through trainings and mentoring of staff. The other limitation was the scaling up of SFP within the pilot health facilities leading to 366 children discharged into SFP at an MUAC of ≥11.5 cm instead of the planned MUAC of ≥12.5 cm similar to the WHO criteria of cure during the OTP stay. 30 The scaling up of SFP in the 3 districts was not anticipated at the beginning of the pilot.
In conclusion, this pilot program strengthens the evidence on the effectiveness of the use of SMS-RUTF in treating SAM. It demonstrates that a program run using SMS-RUTF instead of PM-RUTF does meet and surpass the SPHERE minimum standards and that this formulation which has neither milk nor peanuts can be used to treat SAM in children aged 6 to 59 months in routine CMAM program facing usual implementation challenges. The production and use of SMS-RUTF in CMAM programs should therefore be encouraged in Malawi and globally. It is also encouraged to conduct a scaled up comparative effectiveness study with cost component to evaluate the comparative cost-effectiveness of SMS-RUTF to traditional PM-RUTF in a programmatic setting.
Footnotes
Acknowledgments
We thank Peter Akomo, Dr Paluku Bahwere, Chrissy Banda, and Dr Steve Collins from Valid Nutrition who provided technical expertise that greatly assisted the program. We also thank our colleagues from World Vision, Chisomo Kamchacha, Mwandida Mvuma, Thokozani Chibwana, Mavuto Kholowa, Gladson Chataika, and from Malawi government (Caisious Mkandawire, Martin Mkandawire, Kingsley Chizeze) for support to implement the program. We would like to extend our gratitude to all Health Surveillance Assistants, mothers/caregivers, and children involved in the pilot.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The program was funded by Ajinomoto Co, Inc. The organization had no role in the study design, data collection, or analysis and interpretation.
