Abstract
This article describes current research on the development of alternative ready-to-use therapeutic foods (RUTFs) in the treatment of severe acute malnutrition. An innovative and versatile linear programming tool has been developed to facilitate the creation of therapeutic formulas that are determined acceptable on multiple levels: costs, ingredient acceptability, availability and stability, nutrient requirements, and personal preferences. The formulas are analyzed for ease of production by Washington University team members and for organoleptic properties acceptability to target populations. In the future, RUTF products that are cost-effective, acceptable, sustainable, and widely available will become a reality.
Keywords
Introduction
Severe acute malnutrition (SAM) remains one of the major causes of mortality worldwide for children younger than 5 years. 1 The current recommended treatment is a home-based treatment using a lipid-based paste, called ready-to-use therapeutic foods (RUTFs). 2 This highly caloric, high-energy, fortified peanut paste was developed as a superior alternative to the previous standard, a hospital-based treatment providing children with F100 or F75, a fortified milk supplement. 3 Standard RUTF is very effective, providing recovery rates of 75% to 95%; unfortunately, the comparatively high cost is a major reason why certain countries and producers have decided not to pursue future scale-up. The effective outcomes seen with RUTF have resulted in an increased demand. 4 To meet that demand, more cost-efficient formulations are needed. There is also a need to increase the acceptability among countries that have not previously recognized this method of treatment. These demands allow for opportunities to introduce locally available and acceptable ingredients and to increase production in developing countries. 5,6 An impetus to develop alternative formulations for select countries has propelled this concept forward.
Development of a Linear Programming Tool to Aid the Development of RUTF
The major question faced by many researchers in the RUTF world is, “how to treat children with SAM using RUTF, while minimizing the costs and without compromising the quality?” Working in collaboration with agricultural economists at the University of California, Davis, and with support from the Children’s Investment Fund Foundation, a multiphase alternative RUTF formulation project began in 2013. The work began with a systematic and comprehensive literature and nutrient database review and culminated in the subsequent development of a food formulation linear programming tool. 7 Four country-specific linear programming tools for Ethiopia, Ghana, Pakistan, and India were developed and used to develop individual products, which were then sent for in-country acceptability trials.
Linear programming tools have a long history of use, especially in low- and middle-income countries. 8 –10 Thus, developing a linear programming tool that could streamline the optimization process for therapeutic foods would be a cost-effective method without compromising quality. The objective was to formulate a least cost-optimal product while maintaining the nutritional integrity, known to be effective. Some of the decision variables that went into the linear programming tool were ingredient and nutrient constraints, including the way in which those ingredients are processed. The constraints set were to make sure the formulations align with RUTF nutrient specifications. The benefit of this process is that it eliminates some of the trial and error inherent in optimizing formulations.
Figures 1 and 2 provide an overview of the process. The first step was to identify all potential ingredients for the tool. This involved on-the-ground, in-country research, looking at different foods that were available, their nutrient information, the prices of those foods, their required processing, and the costs associated with processing. These data were further refined to create country-specific tools that can run to generate an unlimited number of formulas, all of which draw from the least cost-effective candidate ingredients. Some of these formulations may not be palatable products, so creativity and knowledge of food science and nutrition come into play. For example, a desirable formulation needs high-quality protein, such as dairy proteins. Although nonfat dried milk has a desirable taste, it is an expensive form of dairy protein. However, ingredient weight constraints can be set at a maximum amount to appear in the formulations and supplemented with other dairy ingredients, such as whey protein concentrates or milk acid whey to meet the RUTF nutrient specification of 50% dairy protein. Food scientists then reviewed these formulas to assess feasibility. The batches formulated at Washington University were then analyzed by an independent laboratory for nutrient composition. Further formula optimization occurred as needed, until 3 or 4 final formulas were selected for scale-up at a university processing facility. The final products were then delivered to the target countries for a 2-week in-country acceptability trial.
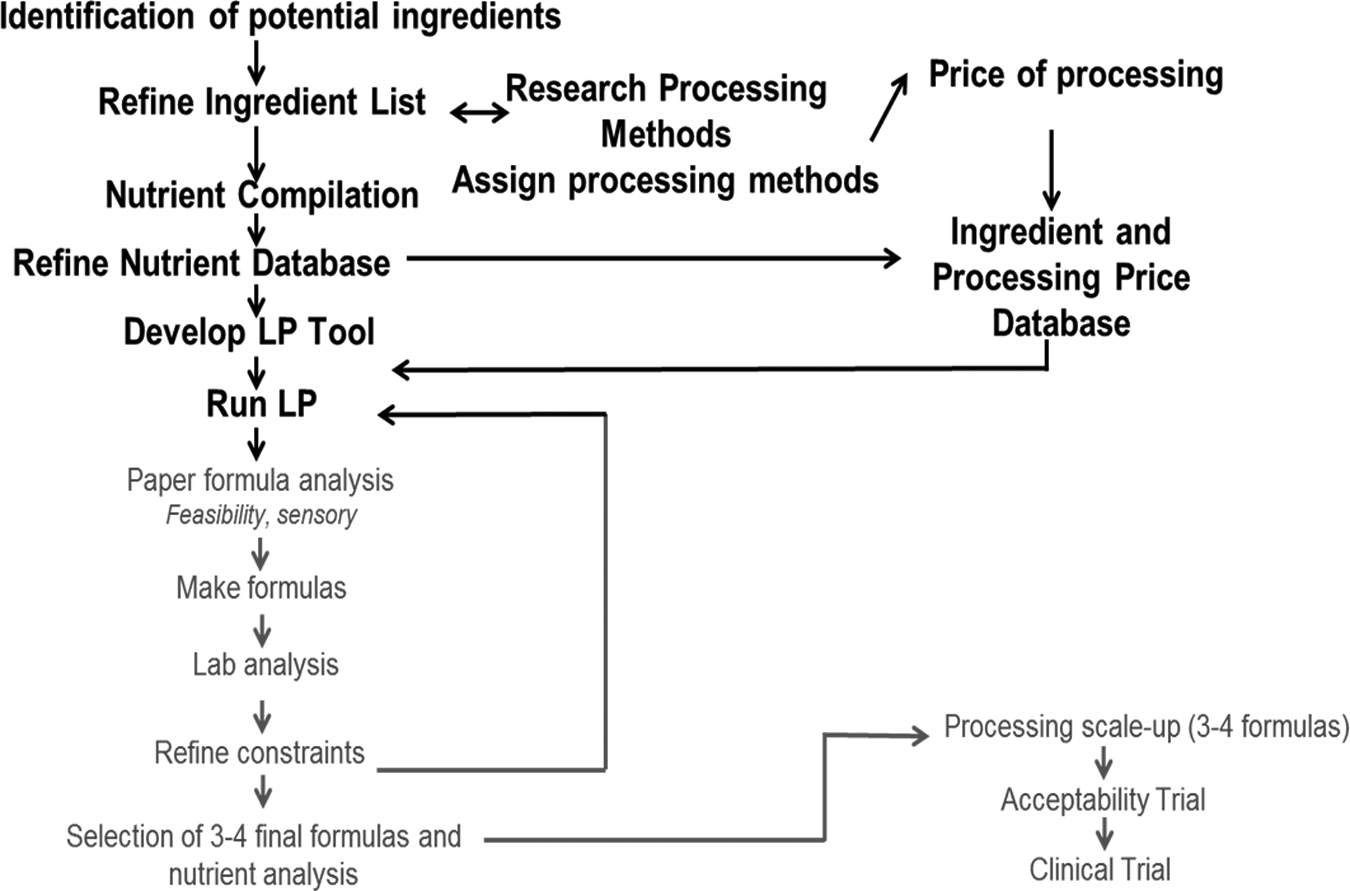
An overview of the linear programming tool development process.
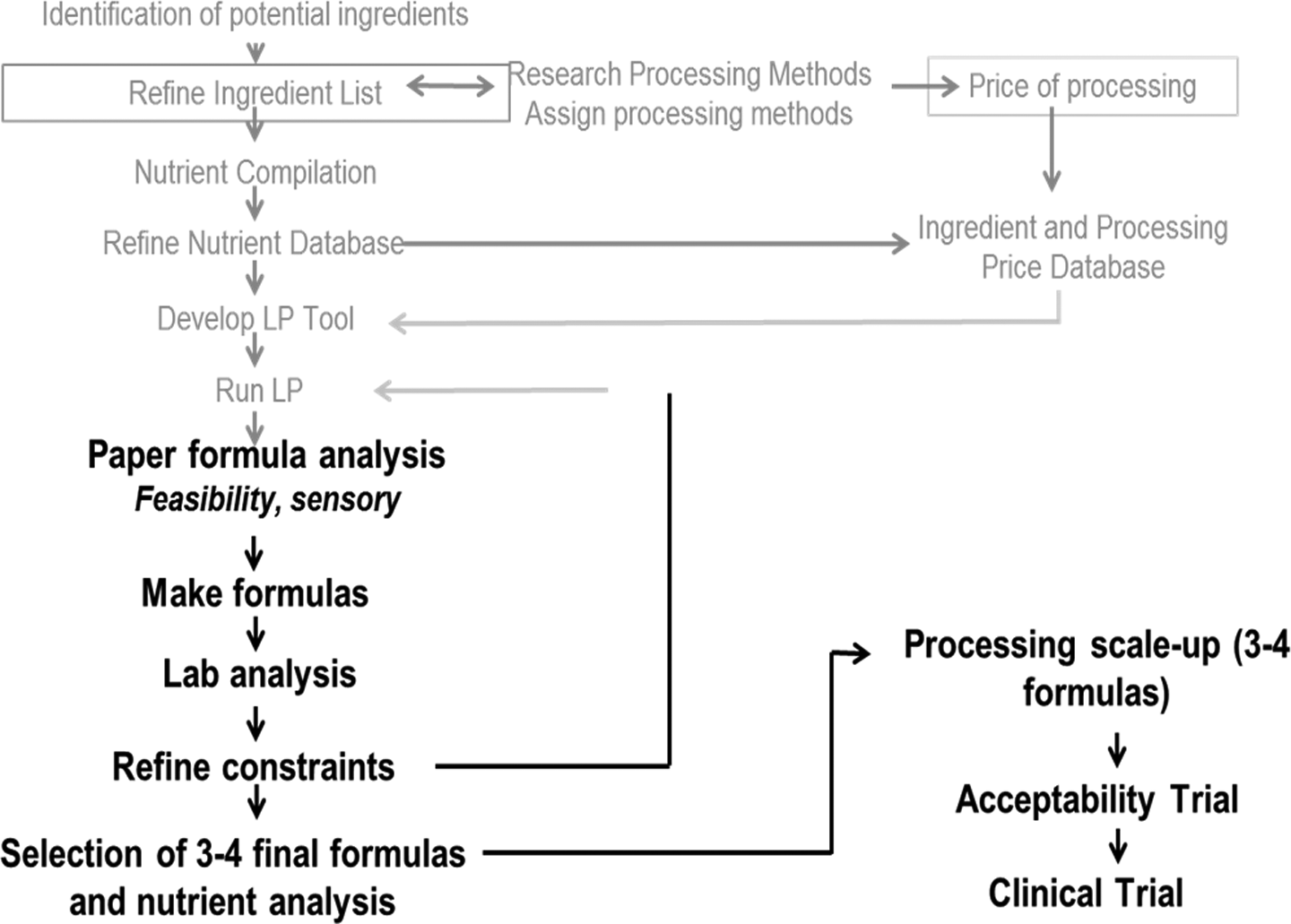
An overview of the linear programming tool process for developing alternative ready-to-use therapeutic food (RUTF).
Figure 3 provides an in-depth explanation of the process with examples. First, note that the linear processing tool is a Microsoft Excel-based database. Figure 3 is the cover sheet, which is divided into 5 subsections. The first subsection contains all the inputs for the ingredients and their associated nutrient information. The second describes the nutrient constraints that were set by regulatory agencies and can be manipulated to create the desired final formulation. The third is optimization and results, the heart of the linear programming tool. This is where the solver database takes into account all the inputs and all the constraints that were previously set and then generates the most cost-effective formula using the least expensive ingredients. The fourth subsection contains the underlying data. Although this subsection is rarely used after the linear programming tool is created, it is still important because it contains all the information about ingredient pricing, local ingredient availability, transportation costs of the ingredients, and the overall production of the product. The fifth subsection, ingredients considered, contains all of the potential ingredients that could be used in an RUTF formula. Some realistic ingredients include peanuts, milk powders, and vegetables, whereas unrealistic ones include meat products, cockroaches, and snails.
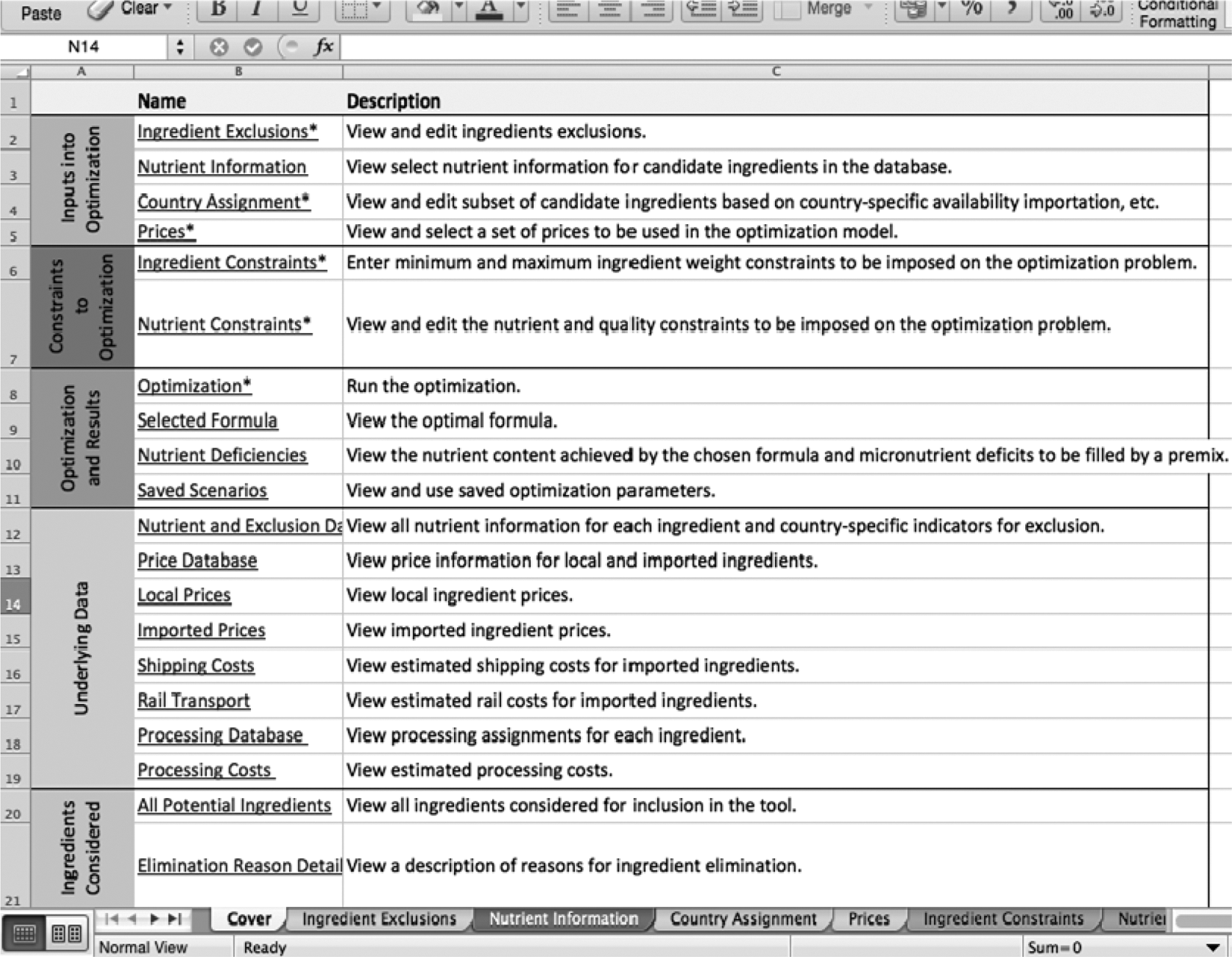
Screenshot of the cover sheet in the linear programming tool.
Figure 4 shows a country assignment sheet, using Pakistan as an example. Note, the specific tabs used to optimize an RUTF formula for Pakistan. All the potential ingredients for Pakistan can be identified by their availability in Pakistan or their possibility of importation. This knowledge allows the user to determine whether a particular ingredient is worth carrying through the formulation. Many ingredients are identified as unsuitable for inclusion in RUTF formulations for Pakistan for lack of availability reasons. As a result, it may not be beneficial to include these ingredients in the final formulation.
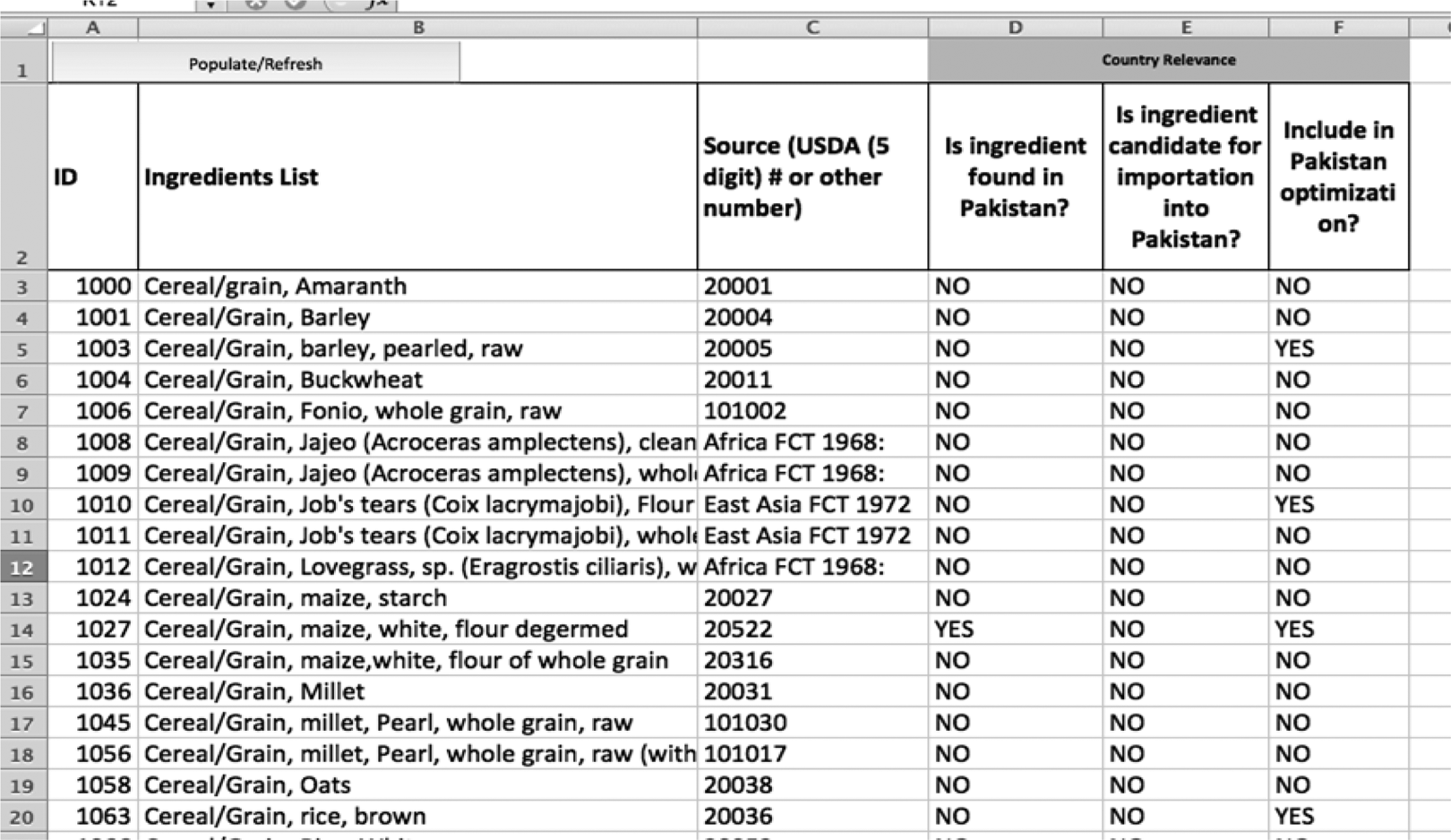
A screenshot of the country assignment sheet in the linear programming tool for Pakistan.
Figure 5 shows the next step in the evaluation. Having excluded ingredients for the lack of availability in Pakistan, ingredients can now be excluded based on other criteria. For example, identical foods that are called different names in different countries (eg, chickpeas and garbanzo beans) can be eliminated such that one ingredient is represented. Ingredients can also be excluded because they have a short shelf life or because they have a poor nutrient content. If the nutrient content for an ingredient specific to a country is unknown, it may be excluded from the final formulation. Finally, an ingredient may be excluded for religious reasons or personal beliefs.
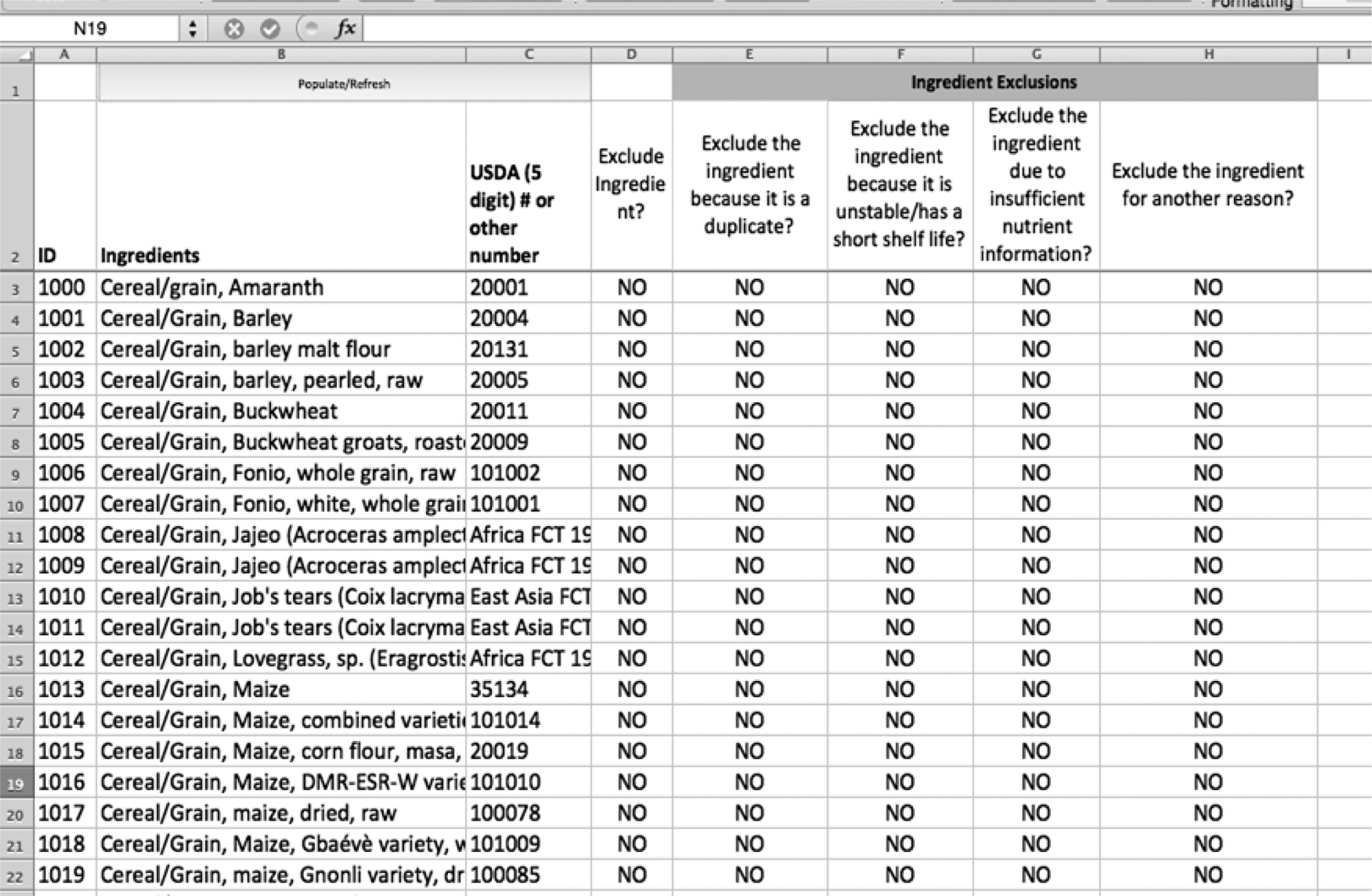
A screenshot of the ingredient exclusion sheet in the linear programming tool.
The initial exclusions steps identified not only the ingredients that could not be used but also the ingredients that can be used. Thus, the next phase involves working creatively with these ingredients to develop the final formulation. Figure 6 shows an example of ingredient constraints, which are minimums or maximums depending on what is needed in the final formulation. Minimums are typically set for ingredients that have a higher associated cost because the tool will always select the cheapest ingredients. For example, if peanuts are desired in a formulation, a minimum weight is set so the tool will select it. Maximums also can be set to limit the amount the tool will select for this ingredient.
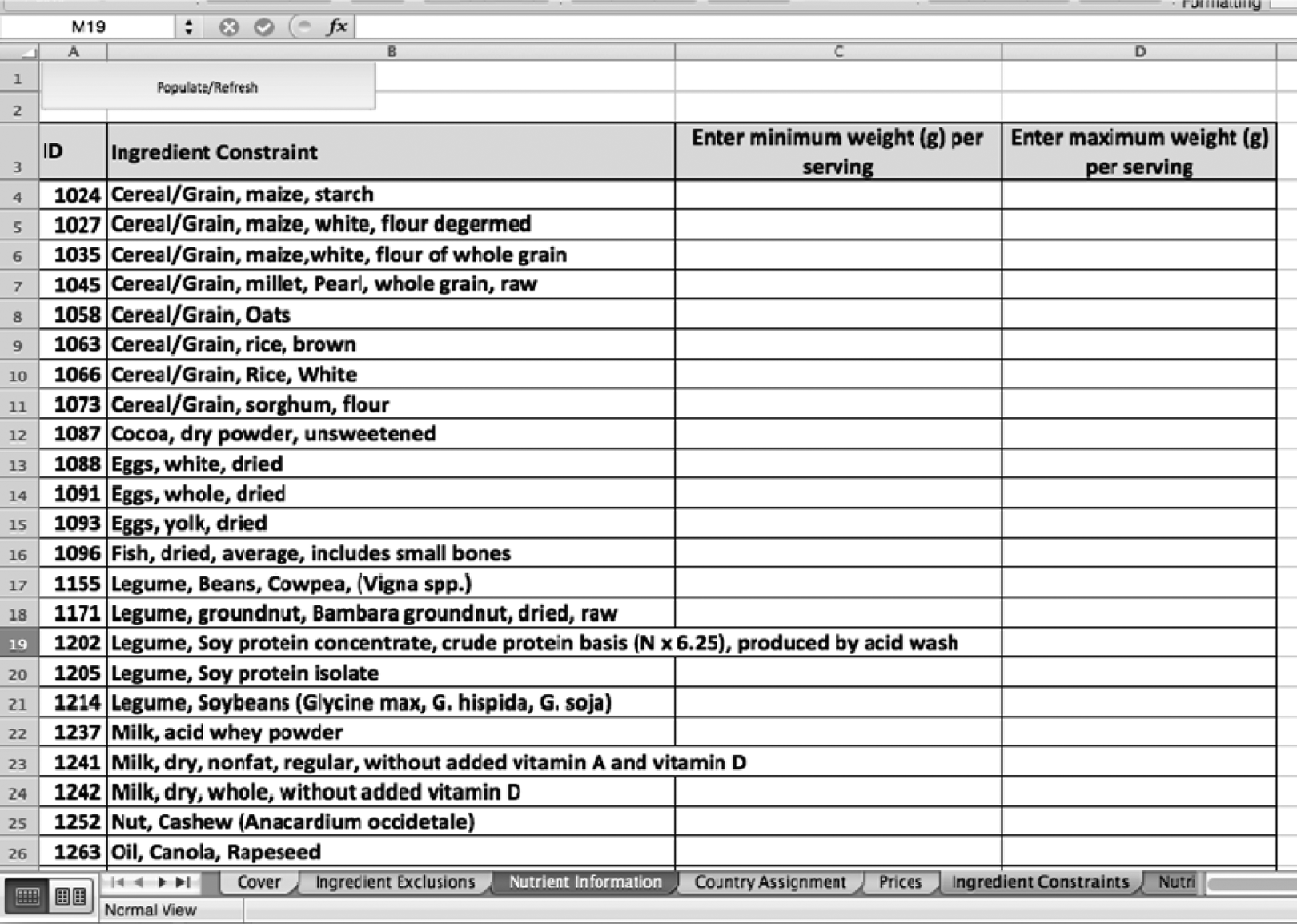
A screenshot of the ingredient constraint sheet in the linear programming tool.
The versatility of the tool is demonstrated by the requests made by Washington University’s collaboration with Aga Khan University in Pakistan. The first steps of the process resulted in multiple exclusions, but even after the initial limits, there were still 100+ candidate ingredients. The collaborators at Aga Khan requested a formulation that excluded peanuts but included more local, indigenous foods. Thus, lentils were substituted for peanuts. However, to enhance the “bland” flavor of lentils and coarse texture, almonds were included. Almonds impart a similar fatty mouthfeel like peanuts while complementing the mild lentil flavor. Figure 7 shows that minimum weight constraints were made for lentils and almonds. Once solver calculates, the additional constraints will reflect the minimum weight (%) constraint in the final formulation.
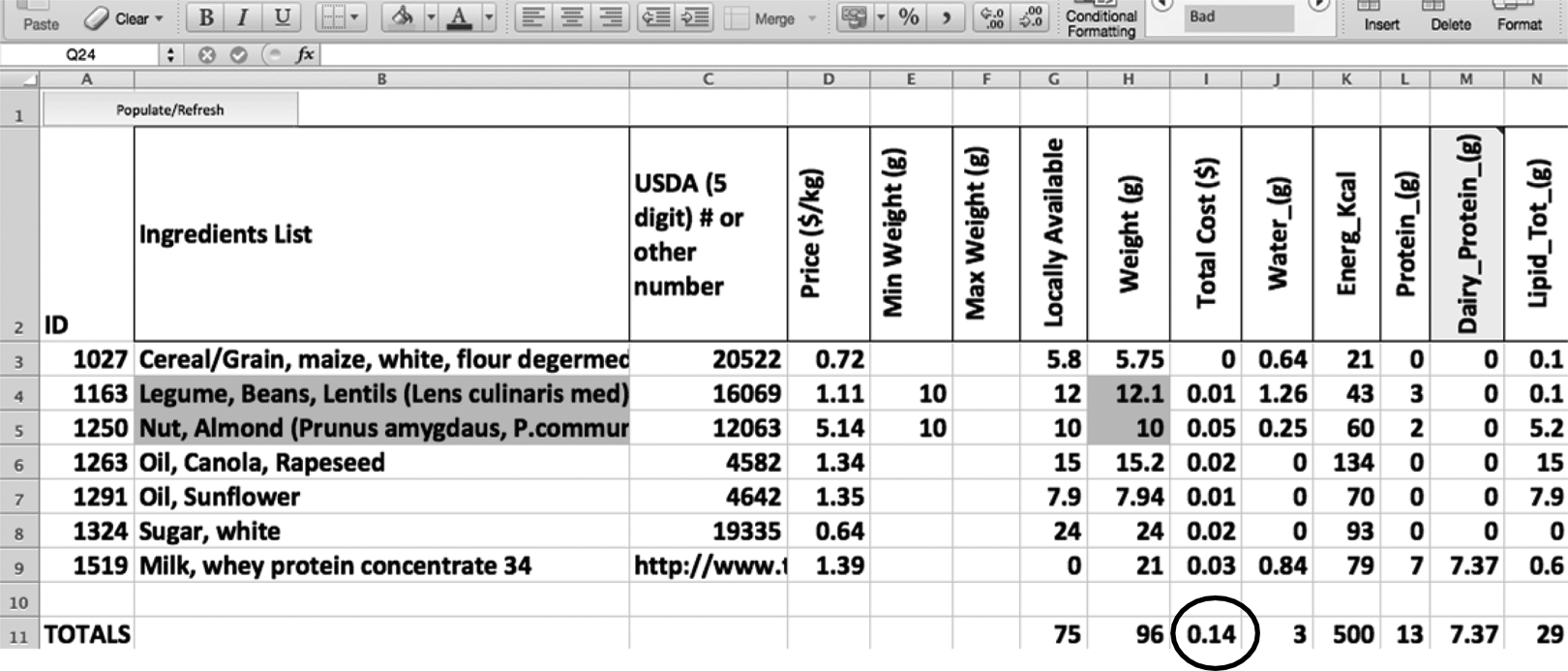
Screenshot of the low-cost formulation output when lentil and almond ingredient constraints were set.
The next step is to set nutrient constraints. As shown in Figure 8, it is possible to manipulate nutrient constraints for different target populations by changing the overall weight of the product. For example, a formula for malnourished pregnant women or a supplementary food product will have a different dosing weight. Regulatory agencies dictate what nutrient properties are present in each formula. One of those specifications is a minimum of 50% of the protein must come from dairy protein. By manipulating the minimums and maximums of calcium, phosphorus, and candidate dairy ingredients, various dairy ingredients will be chosen, providing more variety and therefore allowing a reduction in cost.
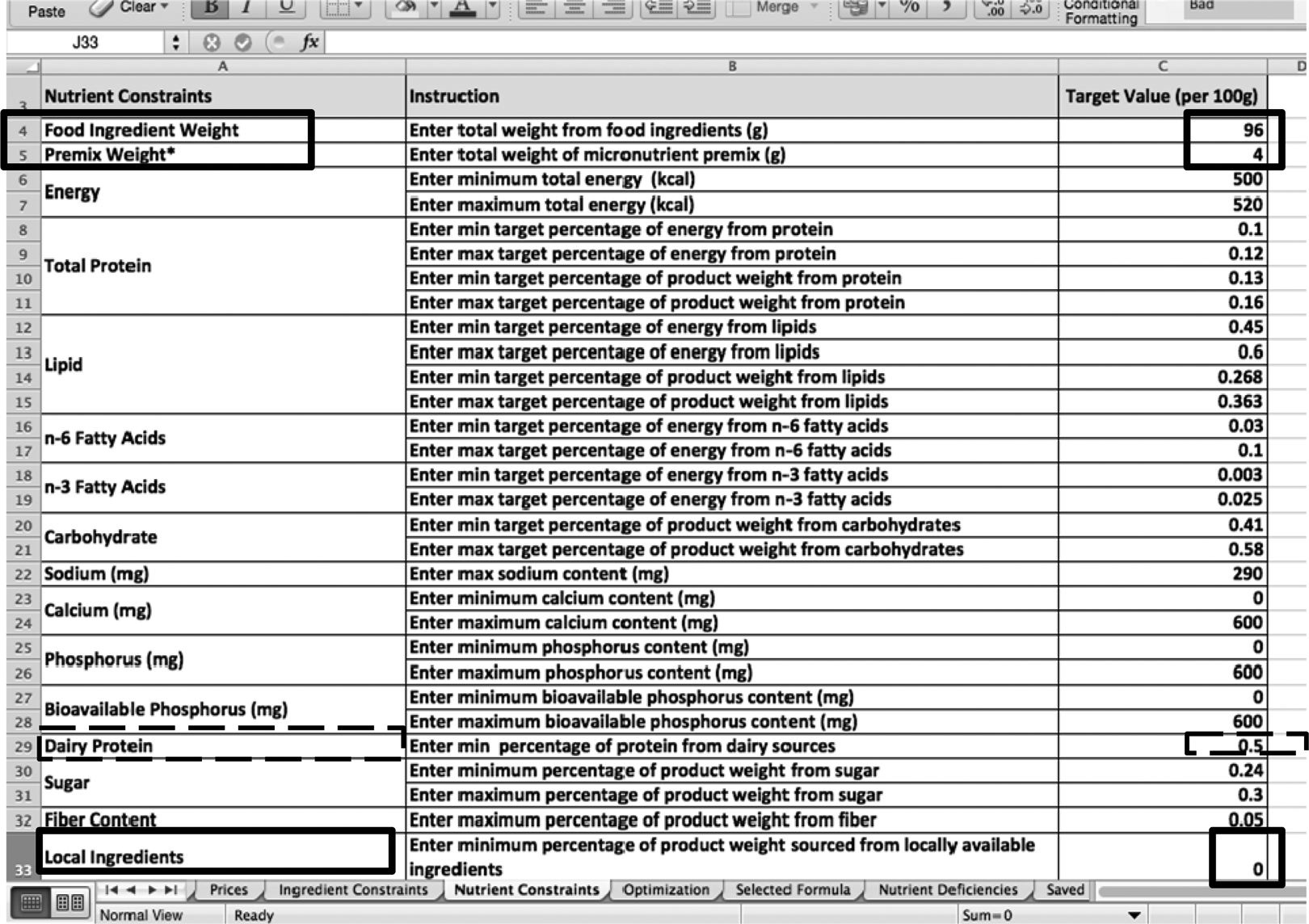
A screenshot of the nutrient constraint sheet highlighting key manipulation features and the built-in ready-to-use therapeutic food (RUTF) dairy specification in the linear programming tool.
To produce the most cost-effective formula, containing no ingredient constraints or the ingredient elimination process, the formula shown in Figure 9 will be generated. It is made primarily of vegetable oil, whey, and sugar, with small amounts of soybeans and egg yolk. It costs only 8 cents to provide 520 kcals, but it is very hard to produce and it does not have an appealing flavor. For comparison, a standard RUTF formula costs anywhere from 21 to 27 cents.
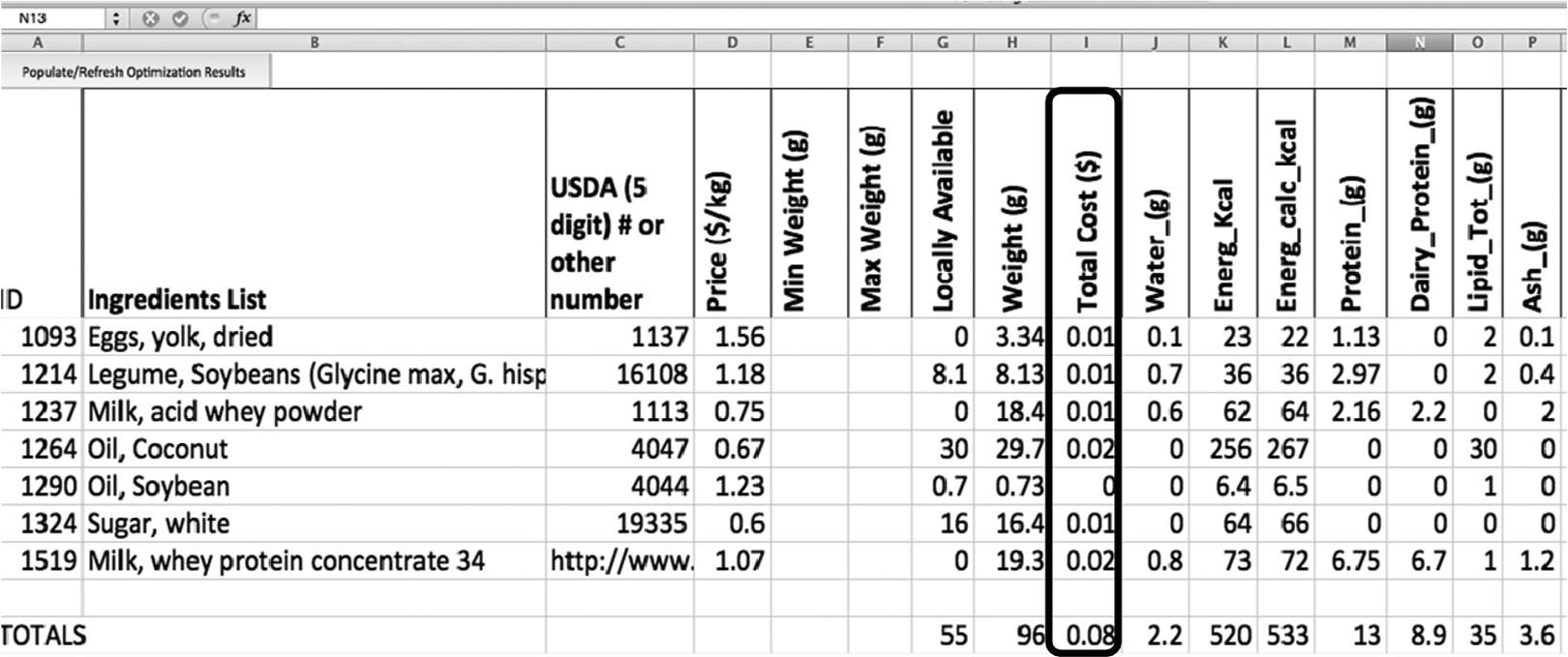
A screenshot of the most cost-effective formulation generated by the linear programming tool.
Figure 10 shows the formula developed for Pakistan. Minimums for lentil and almond, both providing 10%, were set, and the solver function assured these minimums were present in the final formula. Although the price, 14 cents for 500 kcals, is more expensive than the 8 cents for the most cost-effective formula, it is still significantly cheaper than the current RUTF standard. The final optimized formulation also has significantly more appealing organoleptic properties than the most cost-effective formulation.
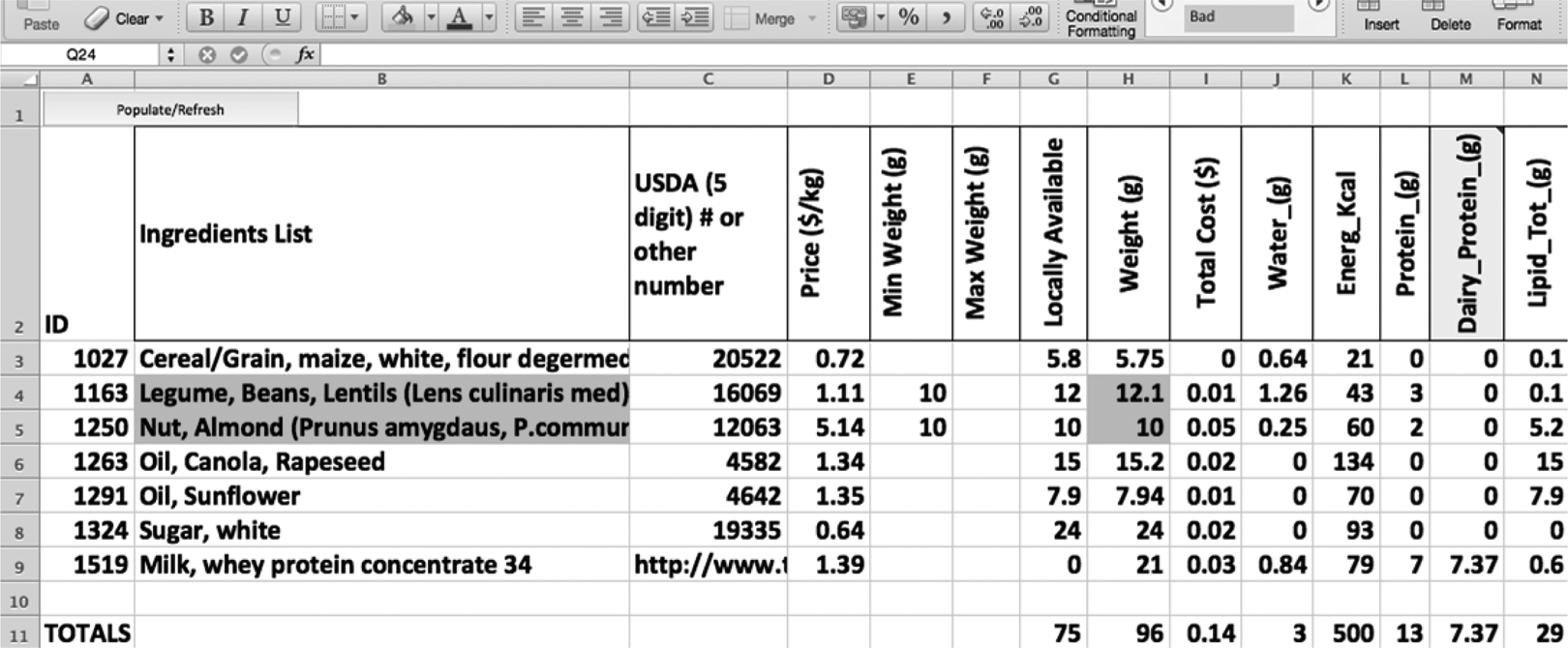
A screenshot of the cost-optimal Pakistan formulation reflecting the ingredient constraints for lentils and almonds with minimum weights of 10 g.
Having completed the final formulations (Table 1) and passed informal acceptability testing in the Manary Food and Nutrition Lab, the products were scaled up for the formal acceptability testing in Ethiopia, Ghana, and Pakistan. These formal acceptability trials took place over a 2-week period with moderately malnourished children between the ages of 8 and 24 months. The children were given one of the test foods, the new alternative optimized formula, or the standard RUTF. The outcome measurements were (1) amount eaten, (2) likeability perceived by the caregiver, and (3) any adverse side effects. As shown in Table 1, the formulations contained varying and unique ingredients, for instance, oats in Ethiopia, cocoa and soybeans in Ghana, and almonds and lentils in Pakistan. These unique ingredients have never been used in an RUTF formula. Additionally, the formulas used different milk powders: nonfat dried milk, whey protein concentrate 34, whey protein concentrate 80, and milk acid whey. These ingredients helped decrease the overall price of the formulas compared to the standard RUTF by about 40%. Price reduction and sustainability are key goals where there is an increased need.
The Ingredients and Costs for 3 Final Alternative RUTF Formulations and the Standard RUTF Formulation Used in Acceptability Trials.
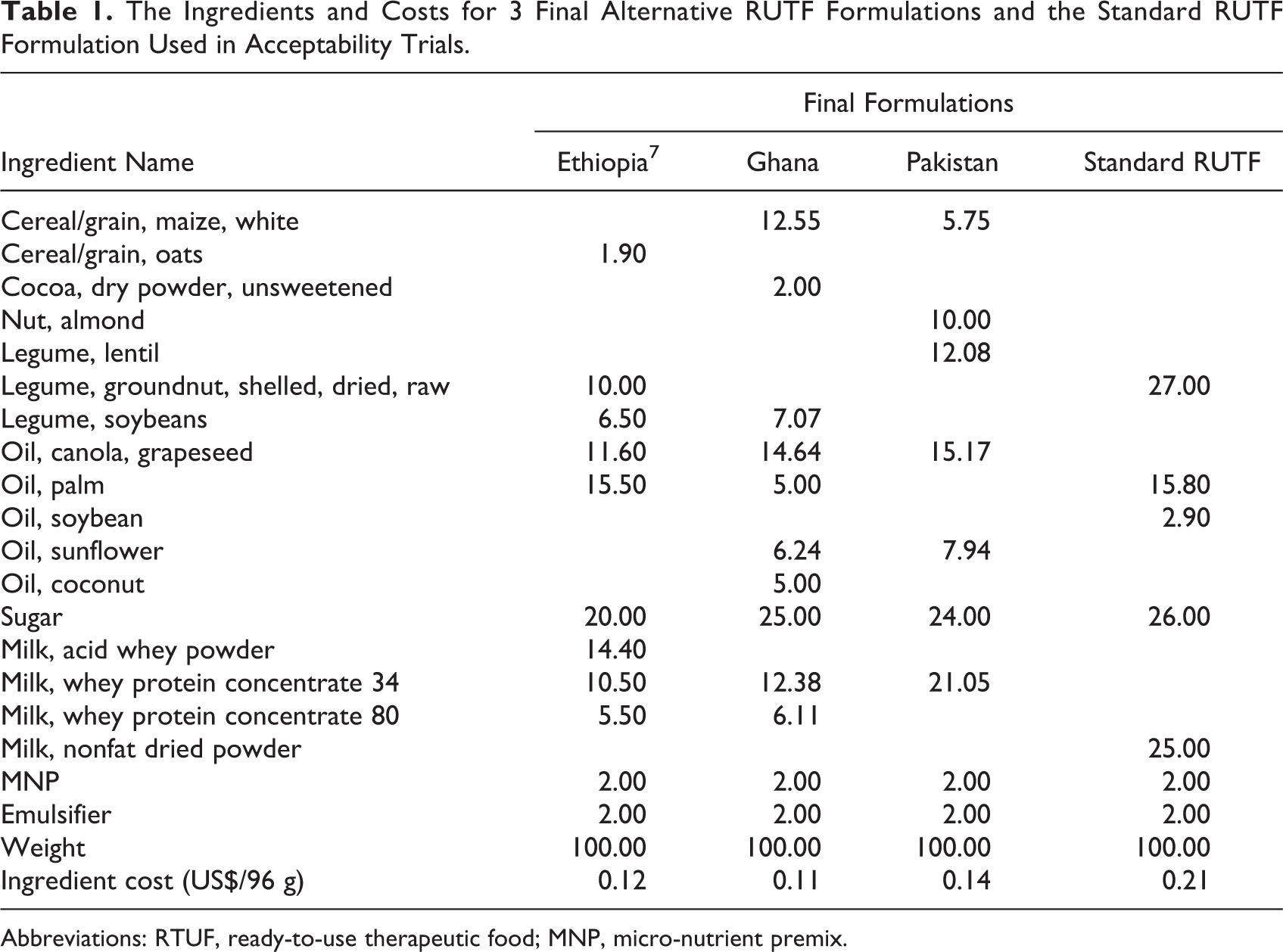
Abbreviations: RTUF, ready-to-use therapeutic food; MNP, micro-nutrient premix.
Figure 11 shows the results of the acceptability trials. Likability scores were around 4 on a 5-point scale, meaning the children generally liked the formulas “a little.” Amounts consumed were in the high 80s to low 90s percentage. These results are positive and reassuring.

Results of the 2-week acceptability trials based on the shown 1 through 5 likeability scoring system.
In conclusion, an innovative and flexible linear programming tool was developed to facilitate the creation of feeding formulas that are acceptable on multiple levels: costs, ingredient acceptability, availability and stability, nutrient requirements, and personal preferences. It presents an opportunity to apply it to other areas, such as the creation of formulas for special populations, such as pregnant, malnourished women who may have greater nutrient needs. In the future, RUTF that are cost-effective, acceptable, sustainable, and widely available will become a reality.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
