Abstract
Background:
With a prevalence of 3.1%, approximately, 450 000 children in Bangladesh are having severe acute malnutrition (SAM). There is currently no national community-based program run by government to take care of these children, one of the reasons being lack of access to ready-to-use therapeutic food (RUTF).
Objective:
To develop RUTF using locally available food ingredients and test its acceptability.
Methods:
A checklist was prepared for all food ingredients available and commonly consumed in Bangladesh that have the potential of being used for developing a RUTF. Linear programming was used to identify the combinations of nutrients that would result in an ideal RUTF. To test the acceptability of 2 local RUTFs compared to the prototype RUTF, Plumpy’Nut, a clinical trial with a crossover design was conducted among 30 children in the Dhaka Hospital of International Centre for Diarrhoeal Disease Research, Bangladesh. The acceptability was determined by using the mean proportion of offered food consumed by the children themselves.
Results:
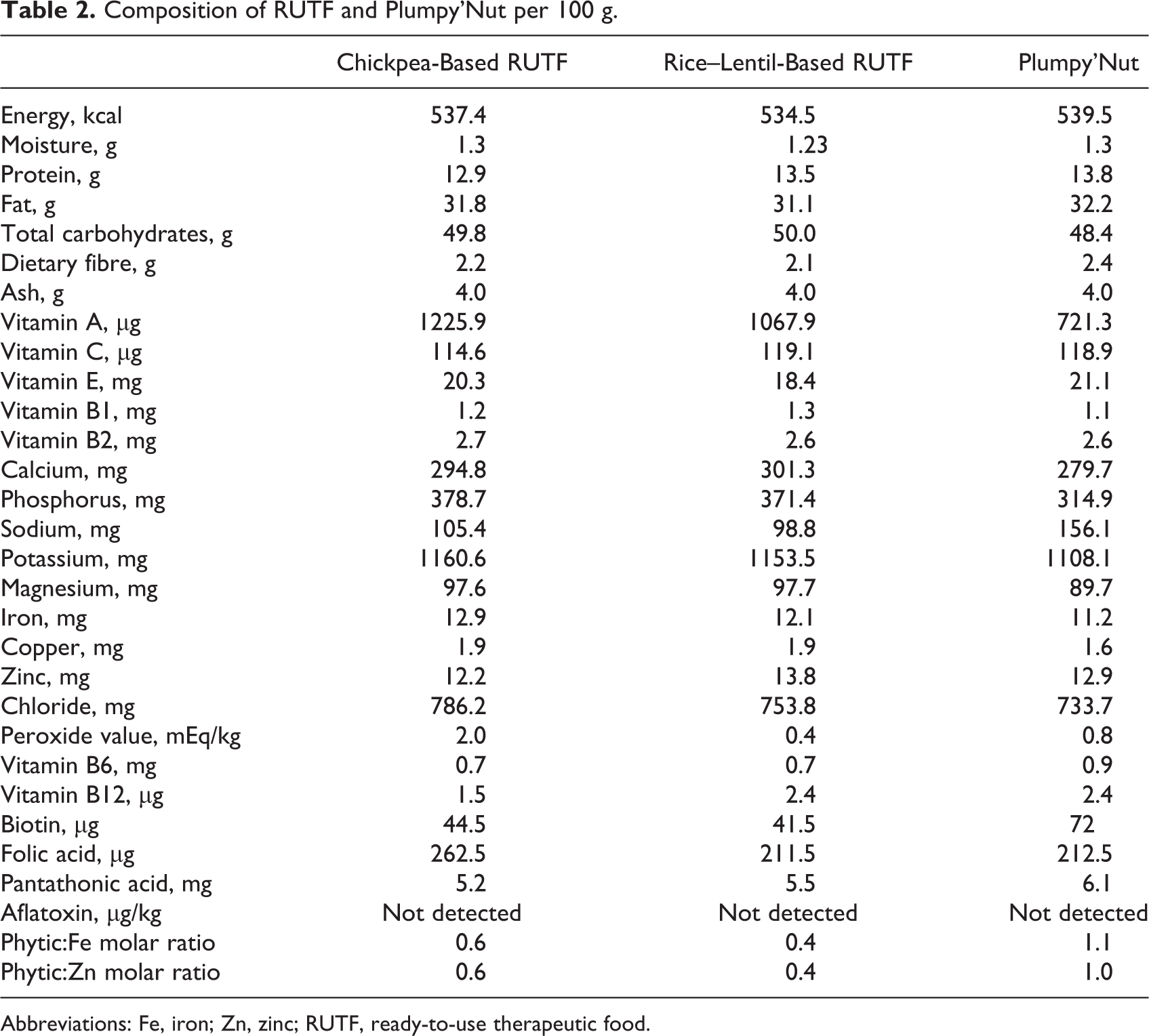
Two RUTFs were developed, one based on chickpea and the other on rice–lentils. The total energy content of 100 g of chickpea and rice–lentil-based RUTF were 537.4 and 534.5 kcal, protein 12.9 and 13.5 g, and fat 31.8 and 31.1 g, respectively, without any significant difference among the group. On an average, 85.7% of the offered RUTF amount was consumed by the children in 3 different RUTF groups which implies that all types of RUTF were well accepted by the children.
Conclusion:
Ready-to-use therapeutic foods were developed using locally available food ingredients—rice, lentil, and chickpeas. Chickpea-based and rice–lentil-based RUTF were well accepted by children with SAM.
Keywords
Introduction
The World Health Organization (WHO) and United Nations Children’s Fund (UNICEF) diagnostic criteria for severe acute malnutrition (SAM) in children aged 6 to 59 months include any of the following: (1) weight-for-height Z scores below −3 standard deviation (SD) of WHO child growth standards, (2) the presence of bilateral pedal edema, and (3) mid-upper arm circumference (MUAC) below 115 mm. 1 With any of these criteria, globally close to 2.4% (16 million) of all children younger than 5 years are affected by this serious nutritional disorder and who have a 9.4-fold increased risk of death compared to healthy peers. 2 Bangladesh has an estimated 450 000 children (3.1%) with SAM. 3
In view of the burden of acute malnutrition, a national guideline for a community-based approach to manage acute malnutrition has been developed to complement the inpatient guidelines for the management of children having SAM. 4,5 Only a few tertiary care hospitals in Bangladesh are currently competent to provide appropriate treatment and care for SAM children. 6 The major constraints for the management of SAM in a hospital setting in Bangladesh are identified as shortage of hospital ward space and trained doctors and nurses, inadequate supply of F-75, F-100, Re-hydration Solution for Malnourished Children (ReSoMal), and combined mineral vitamin mix. 6,7 Most important is the absence of separate arrangement for management of SAM; children with SAM and complications such as diarrhea or pneumonia are usually treated in the same way as children with diarrhea or pneumonia who are not having SAM. It is also well established that children with SAM without any medical complication can be effectively treated in the community. 5 In the context of community-based management of acute malnutrition (CMAM), the corner stone of SAM treatment is the use of ready-to-use-therapeutic food (RUTF). Ready-to-use therapeutic food (PlumpyNut) is an energy-dense, lipid-based paste enriched with vitamins and minerals and is administered to children with SAM in a dose of 175 to 200 kcal/kg body weight per day, once they demonstrate a reasonable appetite. 8 There has been speculation whether RUTF should be produced primarily offshore, locally, or both. 9 It is now evident, however, that local production of gold standard RUTF remains costlier than offshore production. 9 Indigenous or locally produced RUTF using local ingredients is necessary to meet this gap. This local production can prevent and treat malnutrition within a country’s borders and move toward self-sufficiency. Like many other African countries, India has developed an indigenous RUTF and tested acceptability in their local context. 10 Policy makers and other stakeholders in Bangladesh are hesitant to take up a program for CMAM using RUTF on the premises that importation of RUTF will never be a sustainable option. There is no evidence of locally produced RUTF using local ingredients and its acceptability among SAM children in Bangladesh. 11 A research was initiated by International Centre for Diarrhoeal Disease Research, Bangladesh (icddr, b), with support from UNICEF, Bangladesh, and Nutriset, to develop RUTFs made of locally available food ingredients and are culturally acceptable. Such RUTFs will contribute to effective scale-up of a program for treating children with SAM with good appetite and free from any acute illnesses in their communities while also convincing the policy makers to promote community-based management of SAM in this country. This article aims to describe the formulation of newly developed RUTF recipes and to assess whether these RUTFs are acceptable compared to the standard Plumpy’Nut among SAM children aged 6 to 59 months through a clinical trial at icddr, b.
Methods
Development of RUTF
Selection of ingredients
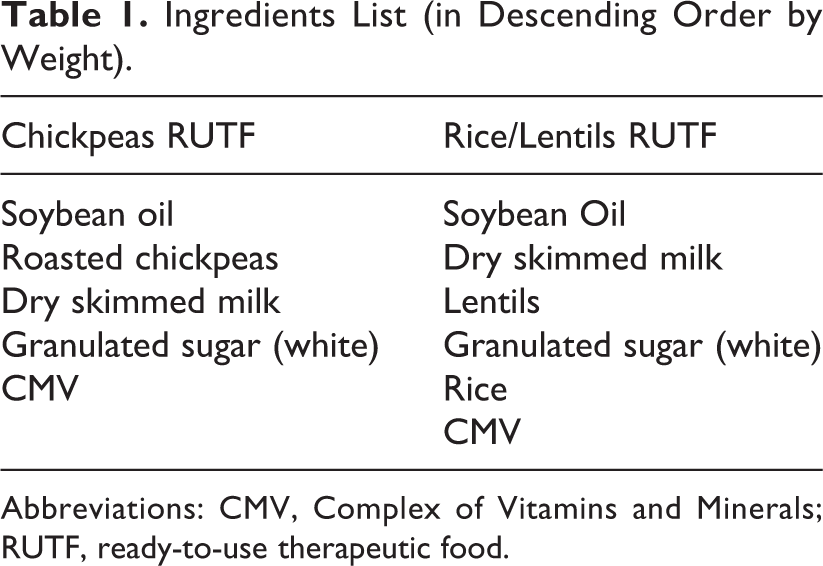
As part of the development of RUTF, a checklist was first prepared for all food ingredients available and commonly consumed in Bangladesh that have the potential of being used as components of RUTF. A final selection was made based on the nutrient content, local availability, acceptability, and cost of the local ingredients. Rice, lentils, and chickpea were chosen as they are widely grown and consumed in Bangladesh and other South Asian countries. During development of the RUTF, all ingredients, such as, rice, lentils, chickpea, skimmed milk powder, soybean oil, and sugar, were purchased from the local market.
Recipe formulation
Linear programming was used to identify the combinations of nutrients that would result in an ideal RUTF. Based on an iterative approach involving multiple calculations of products and sums using nutritive values of food ingredients that can be performed on a personal computer, linear programming is a logical approach for identifying low-cost nutritionally adequate diets. 12,13 As per international specifications, the energy density was targeted at 500 kcal/100 g, and caloric distribution to be 45% to 50% from fat and 10% to 12% from protein. 14 Potential recipes were produced in small batches by mixing all ingredients in an electric kitchen blender. When necessary, consistency of the recipe was adjusted by varying the amount of dry ingredients and soybean oil. Furthermore, a small amount (1%) of soy lecithin was added to the recipe in order to improve the consistency and prevent oil separation.
Ready-to-use therapeutic food production
Experiments for developing recipes and preparation of samples were done at the icddr, b Food Processing Laboratory. A standardized production procedure was followed to control the quality of RUTF from each production batch and to ensure that no unexpected contamination and nutrient losses occur during preparation. Preparation of RUTF underwent different steps, that is, roasting, particle size reduction, homogeneous blending, and packaging. After the initial formulation (grinding and mixing of ingredients to be used for making the RUTFs) by icddr, b, experts from Nutriset and icddr, b further optimized the recipes so that they conform to the specifications as per the 2007 UN statement for the CMAM. 14 The vitamins and mineral premixes were designed to match the requirements of RUTF as per international guidelines. Nutriset (S.A.S, Bois Ricard, Malaunay, France) prepared coded sachets of local RUTFs for the double-blind acceptability trial. The RUTFs used in this study were similar in appearance and were distinguishable only by codes imprinted on the sachets.
Determination of RUTF quality and stability
Microbiological tests (total viable count, yeasts, moulds, coliforms, Escherichia coli, Bacillus cereus, Staphylococci, Listeria monocytogenes, and Cronobacter sakazakii) were done at icddr, b Food Safety Laboratory. Chemical properties (pH, water activity, moisture, peroxide value, and total aflatoxin), nutritional composition (protein, fat, energy, and carbohydrates), and micronutrient composition (vitamins and minerals) were determined at the Institute of Nutrition, Mahidol University, Bangkok, Thailand, as well as at the Nutriset laboratory following standard procedures.
To ensure that the level of micronutrients added were as per the declared premix formulation, iron (Fe) and zinc (Zn) levels were analyzed. Additionally, total phytic acid (phy) amount (mg/100 g) was analyzed and the results were used to calculate phy to Fe and phy to Zn molar ratios.
Acceptability Trial
The aim of this trial was to measure the acceptability of newly developed local RUTFs compared to Plumpy’Nut. We defined the RUTF would be acceptable if a child ingests ≥40% of the offered amount. Acceptability was assessed among SAM children aged 6 to 59 months in Dhaka Hospital of icddr, b by means of a double-blind randomized clinical trial. The study participants were clinically stable and had a good appetite after successful completion of acute stabilization phase using the routine treatment protocol practiced in the hospital. 15 Weight, height, presence or absence of bilateral pedal edema, MUAC, and socioeconomic information were collected on the day of randomization (day 1), whereas 24-hour morbidity and data related to acceptability were collected daily. Our assumption was that there is not a lot of day-to-day variation in feeding behavior in a child and based on this assumption we selected the second day data for the analysis. During the study period children were resided in nutrition rehabilitation unit (NRU), and duty doctors clinically evaluated the participants on a day-to-day basis and recorded morbidity data. Data on the second day for each diet was included in the analysis. A 7-point Hedonic scale was also used to assess the characteristics of 2 local RUTFs and Plumpy’Nut (1 = dislike extremely and 7 = like extremely).
Randomization
A total of 78 children with SAM were screened for eligibility in icddr, b, Dhaka hospital. They were then transferred to the NRU after completion of the stabilization phase of management and fulfillment of the enrollment criteria: aged between 6 and 59 months, either sex, no acute illness, and not having lethargy, poor feeding, loose stools, fever, tuberculosis, or any congenital disorder. An appetite test was performed among these children. For appetite test, Plumpy’Nut was offered to each child. A schematic diagram of the randomization sequence of 1 study participant is shown in Figure 1. If a child had a body weight of <4 kg, then she/he was offered an amount of 11.5 g, likewise 23 g was offered if she/he had body weight of 4 to 9.9 kg, 46 g for 10 to 14.9 kg body weight, or 69 g for ≥15 kg. The child was considered to have passed the appetite test if she/he consumed the required amount according to his/her body weight within 45 minutes. If a child passed the appetite test, then he or she could have enrolled into the study. Our rationale behind the use of 45 minutes as the duration for the appetite test was based on previously published articles. 16,17
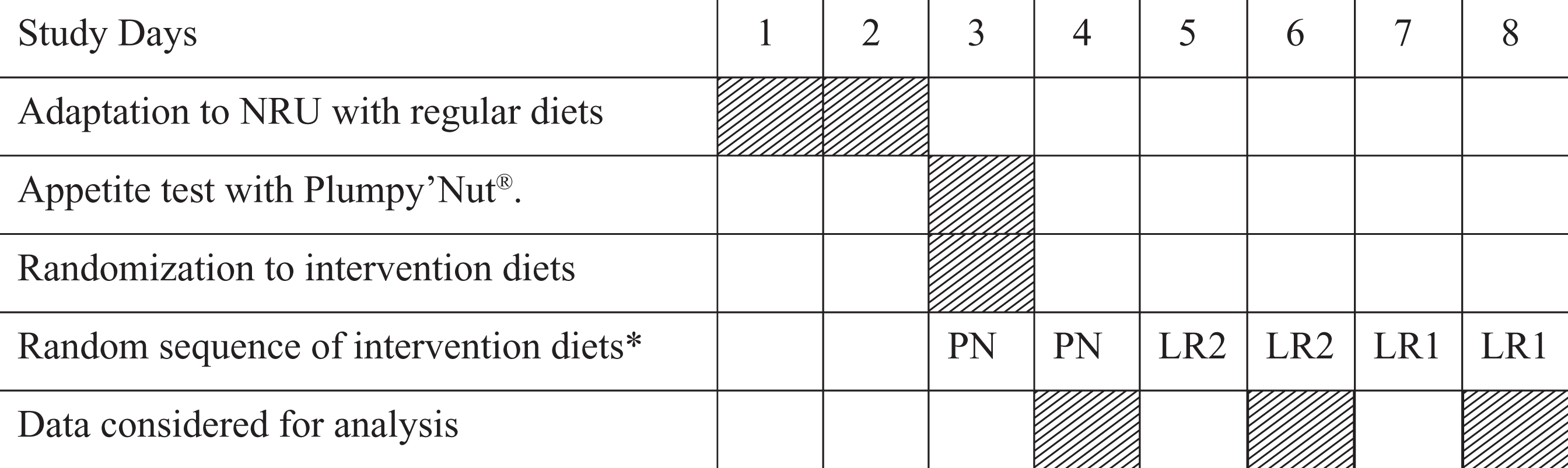
Schematic diagram of randomization sequence of 1 study participant. LR1 indicates local RUTF1; LR2, local RUTF2; NRU, Nutrition Rehabilitation Unit; PN, Plumpy’Nut; RUTF, ready-to-use therapeutic food. *Allocation of sequences of the first, second, and third types of RUTF was randomly predetermined and evenly distributed.
Intervention
An adjustment period of 2 days after arrival in the NRU was allowed for the children and their mothers to adapt to the NRU environment. At this stage, children consumed the regular standardized diet for nutritional rehabilitation as followed in the NRU, that is, milk suzi, khichuri (a gruel based on rice, lentils, vegetables, and soybean oil; 1.4 kcal/g), and halwa (a sweet diet cooked with wheat flour, lentils, molasses, and soybean oil; 2.4 kcal/g). 14 On day 3, the children were randomly allocated to 1 of the 3 types of RUTF (2 local RUTFs and Plumpy’Nut) in a crossover design. Allocations of sequences of the first, second, and third types of RUTF were randomly predetermined. Each of the diets was offered for 2 days over a period of 6 days. We considered the first day of the tasting as a practice day to allow the child and mother to familiarize them with the entire procedure. Therefore, the acceptability data recorded on the first day were not included in the analysis. Each child received all varieties of RUTF but with a different sequence. Each diet was offered at ∼200 kcal/kg divided in 7 meals per 24 hours: a total of 7 times a day at 6:00, 9:00, 12:00, 15:00, 18:00, 21:00, and 24:00 hours.
Sample size
The sample size was used to test the hypothesis that the mean consumption of RUTF during the acceptability test would be at least 40% of the amount offered. 17 We assumed that the SD of consumption would be 15% of the amount offered. The sample size of 30 for each diet would therefore allow us to reject the null hypothesis with 80% power if the true means were at least 50%. The sample size was also adjusted for multiple comparisons using Bonferroni correction.
Sampling
During March to June 2013, children aged 6 to 59 months with SAM who were admitted to the NRU and passed the appetite test were randomly assigned to one of the local RUTFs or Plumpy’Nut according to computer-generated random numbers.
Observation of feeds and interviews with caregivers
During the feeding time, the mothers were asked to spoon-feed their children the assigned diet until the child refused to eat. After a 2-minute pause, the same food was offered a second time until she/he refused again. After a second 2-minute pause, the food was offered a third time until refused again. After this third refusal, the feeding episode was considered terminated. The duration of the feeding (excluding the intervening “pause periods”) was recorded by stopwatch, and the total duration of the feeding was noted. The feeding episode took place under the direct supervision of a trained research assistant. Children were considered as refusing intake if they moved their head away from the food, cried, clamped the mouth shut or clenched the teeth, or become agitated, spit out the food, or refused to swallow. The amount of food ingested was calculated by subtracting the left over from the offered amount. Preweighed napkins were provided; any food that was regurgitated, vomited, or spilled was swabbed, and the napkin weighed and subtracted from the weight of the amount offered. For the weighing instrument that was used to measure the amounts of food was Sartorius model 4101, sensitivity 100 mg. Using a 7-point Hedonic scale in which each point (1 = dislike extremely, 2 = dislike moderately, 3 = dislike, 4 = neither dislike nor like, 5 = like slightly, 6 = like moderately, 7 = like extremely) was depicted by a facial drawing, we asked mothers to rate the food’s color, flavor, mouth feel, and overall acceptability.
Analysis
We performed data analysis using SPSS version 16.0 for Windows (SPSS Inc, Chicago, Illinois). Background characteristics of the participants were evaluated by using descriptive statistics. For the acceptability test, we first tested whether the data are normally distributed by Shapiro-Wilk test. We found statistically all variables are not normally distributed. Therefore, we calculated the median and interquartile range of the amount of the RUTF consumed. We used Kruskal-Wallis test to detect differences in continuous variables. Data from the Hedonic scale questions are presented as median and interquartile range.
Ethical approval
Ethical approval was obtained from icddr, b institutional review board. Informed and signed consent were obtained individually from the caregivers of the participants in the study, and all data were coded to remove identifying information and secure confidentiality. The trial was registered at Clinicaltrials.gov and the registration number of this trial is NCT01889329.
Results
Development of RUTF
Two varieties of RUTFs were developed using locally available food ingredients—one was based on chickpea and the other on rice and lentil. Dried skimmed milk powder, sugar, soybean oil, and vitamin mineral premix were the common ingredients for both the RUTF (Table 1). The total energy obtained from 100 g of chickpea
Ingredients List (in Descending Order by Weight).
Abbreviations: CMV, Complex of Vitamins and Minerals; RUTF, ready-to-use therapeutic food.
Composition of RUTF and Plumpy’Nut per 100 g.
Abbreviations: Fe, iron; Zn, zinc; RUTF, ready-to-use therapeutic food.
Acceptability Testing
Thirty SAM children completed the study (Figure 2). The mean age of the study participants were 13.1 (5.9) months, whereas 57% of them were female. Anthropometric indices and general characteristics of the study participants are detailed in Table 3.
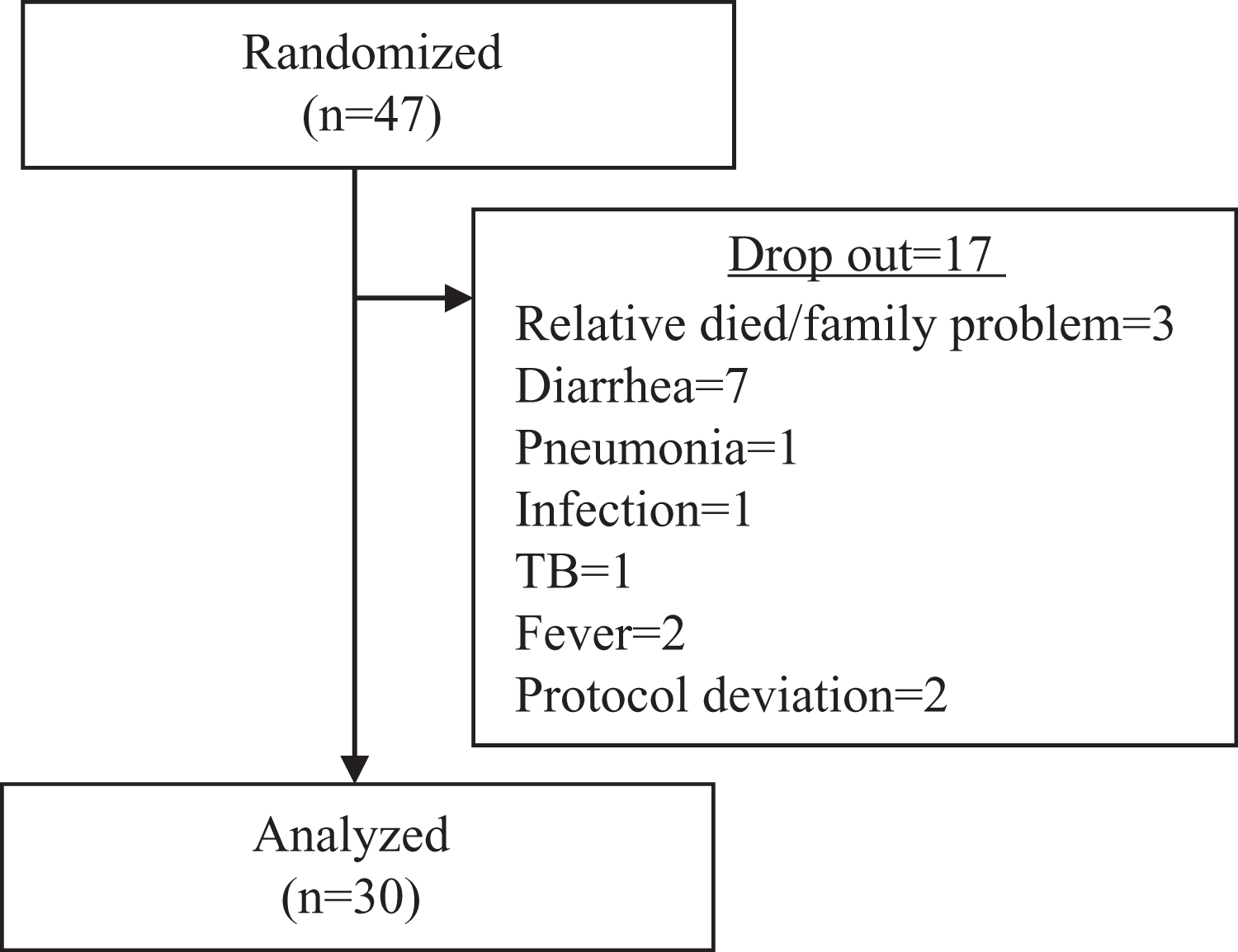
Profile of trial of ready-to-use therapeutic foods among children with severe acute malnutrition.
Characteristics of Children Who Completed the Study.
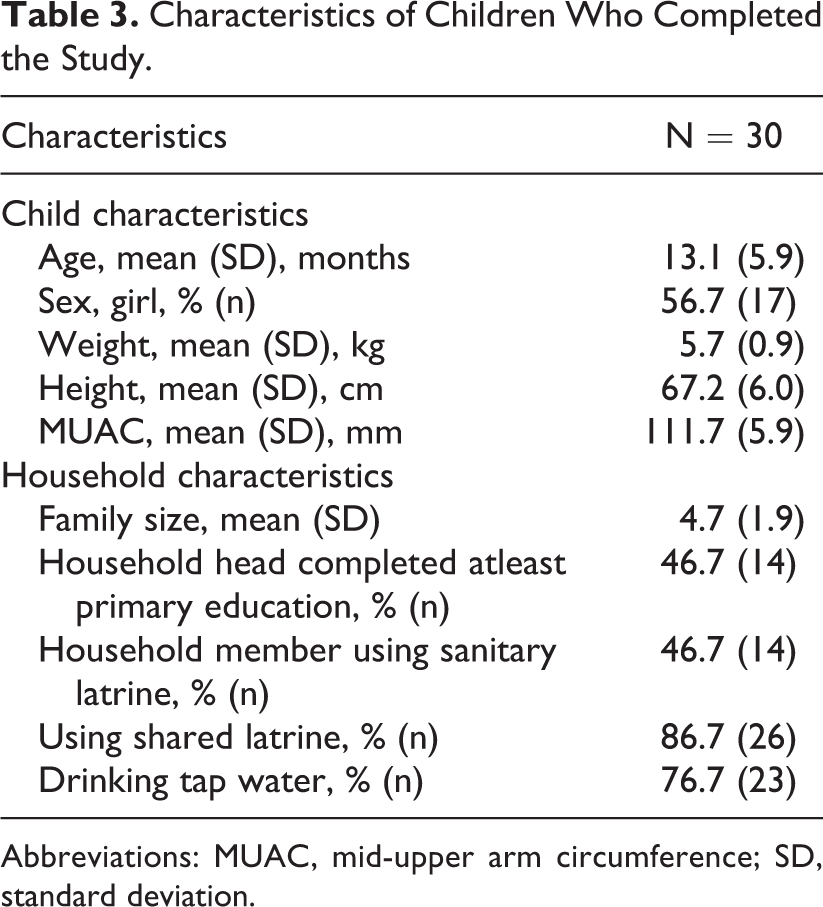
Abbreviations: MUAC, mid-upper arm circumference; SD, standard deviation.
Duration of stabilization period was 7.93 (5.51) days. A total of 47 children and their caregivers were found eligible for the trial and were randomized. A total of 17 mother/child pairs left the study after the adaptation period either due to their illness or family problem. From these 17 mother/child pairs, 1 child was diagnosed to have tuberculosis during this period. Due to protocol deviation, we could not recruit 2 child/mother pair; thus, 30 children completed the acceptability trial.
Children were offered ∼30 g RUTFs to assess the acceptability of RUTF. They consumed 25.6 (7.9) g chickpea
Intake of Local RUTF Compared to Plumpy’Nut.a
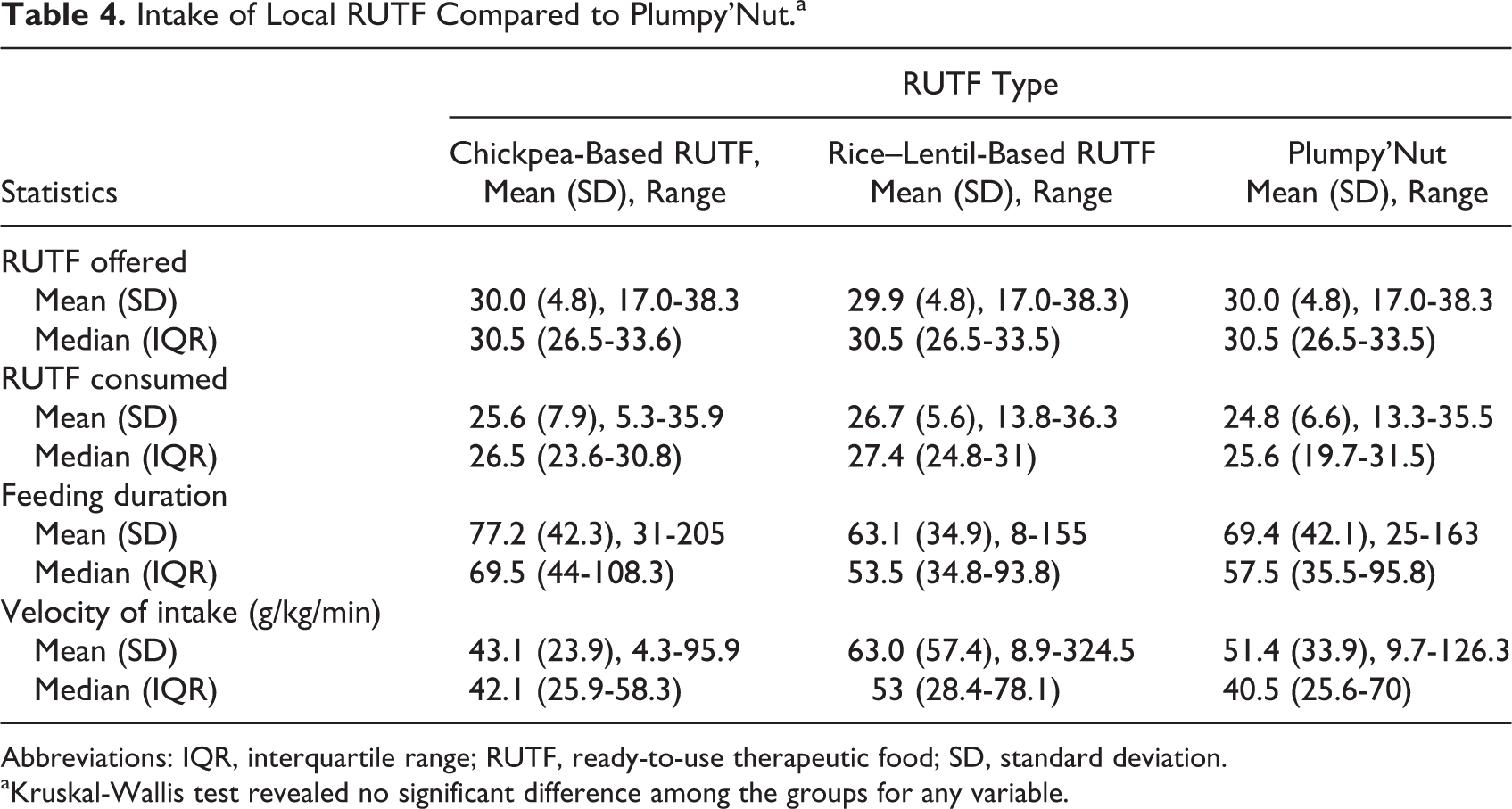
Abbreviations: IQR, interquartile range; RUTF, ready-to-use therapeutic food; SD, standard deviation.
aKruskal-Wallis test revealed no significant difference among the groups for any variable.
Degree of Liking Rating on the 7-Point Hedonic Scale for Appearance, Flavor, Texture, and Overall Degree of Liking by Type of RUTF.a
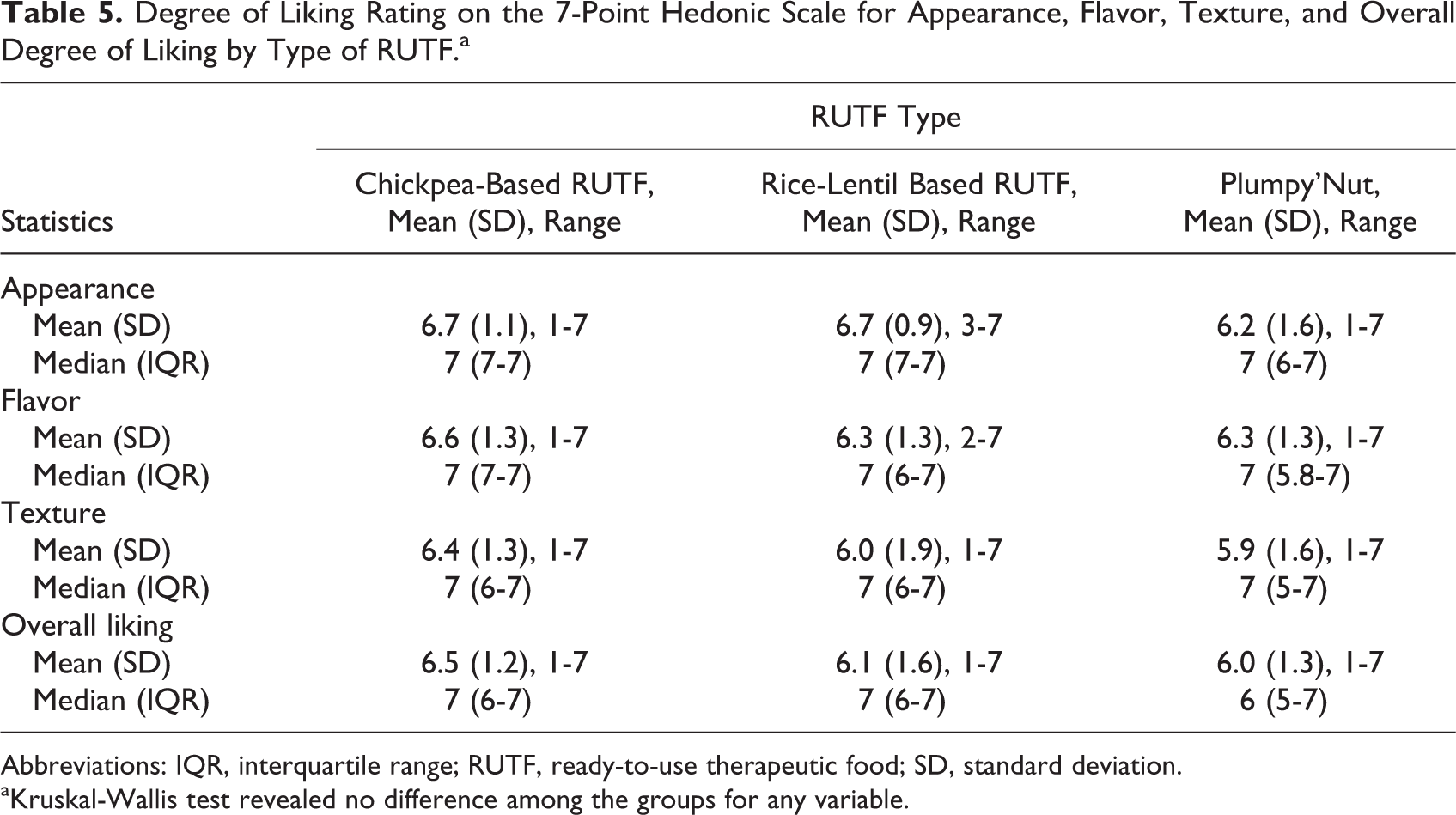
Abbreviations: IQR, interquartile range; RUTF, ready-to-use therapeutic food; SD, standard deviation.
aKruskal-Wallis test revealed no difference among the groups for any variable.
Discussion
We formulated 2 RUTF recipes based on locally available, culturally appropriate food ingredients, that is, chickpea and rice–lentil RUTFs that meet international standards. Chickpea and rice–lentil RUTFs were equally acceptable compared to Plumpy’Nut. The assessment of the acceptability of the 3 RUTFs was determined by using the mean proportion of offered food consumed by the children themselves and velocity of intake as proxy indicators. Hedonic scale was also used as other outcome variables. Our rationale was that with no forced feeding, the amount of the offered food consumed by children would depend largely on the extent to which they liked the food and their appetite given that none of the children were fed for at least 2 hours prior to the feeding session. On an average, 85.7% of the offered RUTF amount was consumed by the children in 3 different RUTF groups which implies that all types of RUTF were well accepted by the children.
India has made their own local RUTF to treat SAM. 10 Thapa et al compared the local RUTF Nutreal with a defined food. Their objective was not only to see the acceptability but also efficacy in terms of weight gain. For our present study, however, we cannot directly compare with Thapa’s study because we did not measure efficacy in terms of weight gain rather we only measured the acceptability of local RUTF among SAM children by using “amount consumed.” On the other hand, Dube et al compared RUTF with khichri. 18,19 They found khichri was a culturally acceptable and palatable meal and widely consumed by families and children in their study setting when the outcome variable was “amount consumed.” The intake of RUTF was lower but the energy intake from that was significantly higher. The energy density for RUTF was 5-fold higher and this would explain the lower consumption as well as the substantially higher energy intake. The rationale behind to introduce RUTF in the community was proven again by Dube’s study. Although Dube’s study showed the average intake of RUTF was higher (56 g) compared to our present study (∼26 g); however, among 3 RUTF groups within our present study was not significantly different. In this study, it should be noted that we only offered 30 g to see the acceptability.
Children consumed similar amounts of chickpea
We developed 2 types of RUTF based on locally available food ingredients. We conclude that the newly developed chickpea-based and rice–lentil-based RUTFs are as acceptable to children and their caregivers as is Plumpy’Nut, the standard RUTF. This is the first study presenting acceptability data on RUTFs made from locally available food ingredients for children in South Asia, which is a novel way to ensure nutritional treatment of children with SAM in this region. This acceptability trial will not give the suffice evidence for CMAM implementation unless it is done the efficacy trial and effectiveness trial of these local products.
Limitation
Crossover designs usually demand a washout period. However, the washout period in our case might not have been sufficient and may have introduced some bias in this study. Moreover, data was obtained from the NRU which was the study personnels’ working area and that might have introduced bias.
Footnotes
Authors’ Note
Nuzhat Choudhury and Tahmeed Ahmed were joint first authors. T. A., M. I. H., and M. M. I. conceptualized the work and the manuscript. N. C. analyzed the data and drafted the manuscript. M. Z., J. D. C., and S. A. S. helped in the interpretation of findings, and contributed to the critical revision of the manuscript for making the final draft for submission. All authors went through the final draft and approved it for submission. This study was approved by the Research Review Committee and Ethical Review Committee, the 2 obligatory components of the institutional review board of icddr, b. Written informed consent was taken from the study participants.
Acknowledgments
The authors acknowledge the following donors for providing unrestricted support to icddr, b’s research efforts and advancement to its strategic plan: The Government of Bangladesh, Global Affairs, Canada, Sweden (Sida), and Department for International Development (UKAid). Nutriset prepared and supplied the coded sachets of the 3 RUTFs for the trial.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research activity was funded by UNICEF. icddr, b acknowledges with gratitude the commitment of UNICEF to its research efforts.
