Abstract
Background:
In Pakistan, nearly half of children younger than 5 years are stunted, and 1 in 3 is underweight. Micronutrient deficiencies, a less visible form of undernutrition, are also endemic. They may lead to increased morbidity and mortality as well as to impaired cognitive and physical development.
Objective:
To estimate the lifetime costs of micronutrient deficiencies in Pakistani children aged between 6 and 59 months.
Methods:
We develop a health economic model of the lifetime health and cost consequences of iodine, iron, vitamin A, and zinc deficiencies. We assess medical costs, production losses in terms of future incomes lost, and disability-adjusted life-years (DALYs). The estimation is based on large population surveys, information on the health consequences of micronutrient deficiencies extracted from randomized trials, and a variety of other sources.
Results:
Total societal costs amount to US$46 million in medical costs, US$3,222 million in production losses, and 3.4 million DALYs. Costs are dominated by the impaired cognitive development induced by iron-deficiency anemia in 6- to 23-month-old children and the mortality caused by vitamin A deficiency. Costs are substantially higher in poorer households.
Conclusions:
Societal costs amounted to 1.44% of gross domestic product and 4.45% of DALYs in Pakistan in 2013. These costs hinder the country's development. They could be eliminated by improved nutrition of 6- to 59-month-old children and public health measures. Our results may contribute to the design of cost-effective interventions aiming to reduce micronutrient deficiencies in early childhood and their lifetime consequences.
Keywords
Introduction
Micronutrient deficiencies (MNDs) are a serious but often invisible form of undernutrition, also called hidden hunger. MNDs increase the morbidity and mortality in children younger than 5 years. They also impair a child’s cognitive and physical development and long-term outcomes, thus leading to adverse health outcomes, lower productivity, and lower incomes in adult life. 1,2 Widespread MNDs will therefore hinder a country’s economic development.
MNDs are endemic in children younger than 5 years in Pakistan with 43.8% with iron deficiency anemia (IDA), 54.0% with vitamin A deficiency (VAD), and 39.2% with zinc deficiency (ZnD) in 2011. Among school-age children, 36.7% may be at risk of iodine deficiency (IoD). 3 These high levels of deficiencies have been recognized for decades, but except for a substantial improvement in iodine status, very little progress has been made in eradicating them. 3
The high costs of nutritional deficiencies and the substantial economic benefits of nutritional interventions have been previously assessed. 4 –6 However, there is still a need for a deeper understanding of the societal costs of MNDs in the countries affected, as it may increase the awareness of decision makers for their severe and multiple consequences. It may also contribute to the design of effective and cost-effective nutritional interventions by helping to identify the socioeconomic strata (SES), age-groups, and micronutrients with the highest potential for cost reduction in a specific population.
The aim of this study was to assess the societal costs of MNDs in a birth cohort of Pakistani children affected by MNDs between the age of 6 and 59 months. The micronutrients considered include iodine, iron, vitamin A, and zinc. We assessed medical costs, production losses in terms of future incomes lost, and healthy life-years lost. The study follows an incidence-based lifetime cost approach and thus calculates cost consequences of MNDs in childhood over the whole lifetime of the children affected.
Methods
We developed a health economic simulation model that estimates the lifetime health and cost consequences of IoD, IDA, VAD, and ZnD in 6- to 59-month-old Pakistani children. Infants younger than 6 months were excluded due to lack of data. The model is an extension of a previously published model 7,8 to Pakistan and additionally integrates the consequences of IoD and xerophthalmia as a consequence of VAD. Figure 1 and the following sections give an overview of the model and the data used. Appendix 1 illustrates further details of the model.

Overview of health economic model of societal costs of selected micronutrient deficiencies (MNDs). The social costs of MNDs are calculated in a health economic model proceeding in 4 steps: Step 1 stratifies 6- to 59-month-old Pakistani children in 20 groups (by age-group and socioeconomic status). Step 2 assesses the levels of MNDs in each of these groups. Step 3 assesses the health consequences of the current levels of MNDs. Step 4 assesses the societal costs of these health consequences in 3 cost dimensions.
The model is based on a variety of data sources. They include 2 large population surveys (the 2012-2013 Pakistan Demographic and Health Survey [PDHS] 9 and the 2011 Pakistan National Nutrition Survey [NNS] 3 ), a survey on the treatment costs in Pakistan of diseases induced by MNDs (see Appendix 2), data from national and international agencies, and data extracted from systematic reviews and other publications.
Model Population
In a
Prevalence of MNDs
In a
Biomarkers for Micronutrient Status and Threshold Values for Deficiencies.a
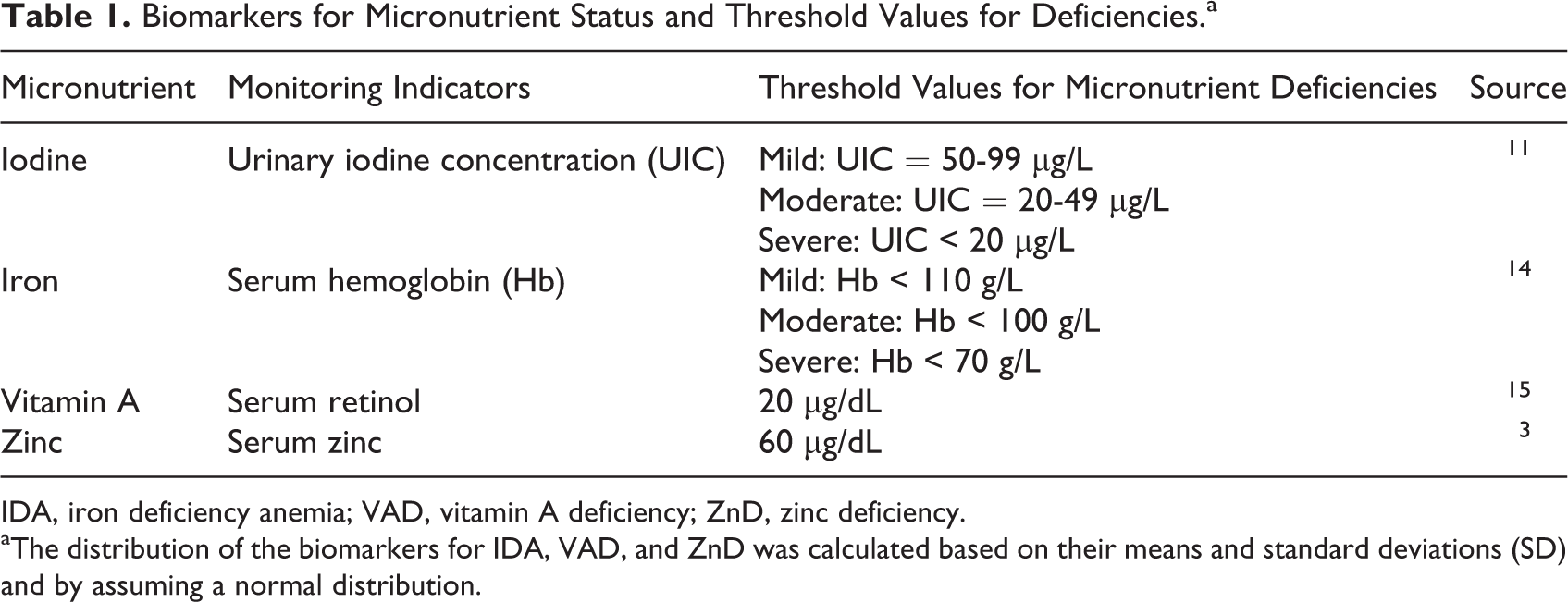
IDA, iron deficiency anemia; VAD, vitamin A deficiency; ZnD, zinc deficiency.
aThe distribution of the biomarkers for IDA, VAD, and ZnD was calculated based on their means and standard deviations (SD) and by assuming a normal distribution.
Health Consequences
In a
Costs of Health Consequences
In a
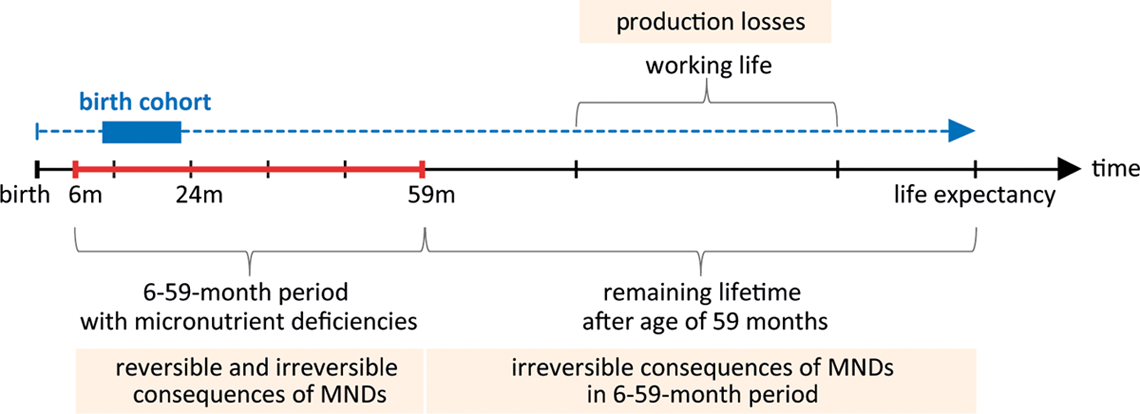
Time dimensions of the health economic model. The health economic model follows an incidence-based approach and assesses the lifetime costs of micronutrient deficiencies (MNDs) in a birth cohort exposed to MNDs from the age of 6 to 59 months. These costs include reversible and permanent effects and are modeled over the whole lifespan of the birth cohort. Future production losses are discounted to present value.
Production losses arise due to lower future wages as children affected by impaired cognitive or physical development will be less productive as adults. These losses were measured as future gross income losses based on SES-specific wages. Current wages according to the International Labor Organization 18 were extrapolated to the future by assuming an income growth equal to the yearly average in the previous decade. Future wage differences between SES were calculated by adjusting the current income distribution 9 for the social mobility between SES according to a recent study based on the Pakistan Panel Household Survey. 19 Production losses were discounted to present values with a discount rate of 3% 20 and converted to US Dollars with the mean exchange rate of 2013.
Life-years lived with disability and lost due to premature mortality were measured in disability-adjusted life-years (DALYs). 21 Following the recent practice by the Institute of Health Metrics and Evaluation, we did not discount future DALYs. 21 Note that reporting both production losses and DALYs does not lead to double counting, as a number of health economic studies have shown that measures of production losses and of health-related quality of life do not overlap. 22
Sensitivity Analysis
In order to test the robustness of the results, we ran a probabilistic and univariate sensitivity analysis (SA). Although the univariate SA shows the impact of the most important model parameters, the probabilistic SA creates a range of possible model outcomes. It also determines the confidence intervals of the main model outcomes. The model was implemented in R version 3.1.1.
Ethics Statement
The data sets used in this study were obtained from the PDHS 2012-2013 and the NNS 2011. 3,9 Ethical review of the analysis was not considered necessary as per guidelines from the National Bioethics Committee, Pakistan, as the data set was anonymous and available for public use with no identifiable information on the survey participants. The survey used to assess medical costs was approved by the National Bioethics Committee, Pakistan (No.4-87/14/NBC-152/RDC/434–2014).
Results
Model Population and Child Mortality
The model population consisted of 21.5 million children stratified in 20 age and SES subgroups (Table 2). The average size of a birth cohort was 4.7 million children with a higher number of children per household in poorer SES. All-cause mortality rate was almost 4 times higher in the younger than in the older age-group and differed substantially between the poorest and the wealthiest SES, with an 8 times higher mortality among the poorest 6- to 23-month-old and a 6 times higher mortality among the poorest 24- to 59-month-old (Figure 3).
Distribution of Number of Children (in 1,000) of Birth Cohort by Age-Group and SES.a
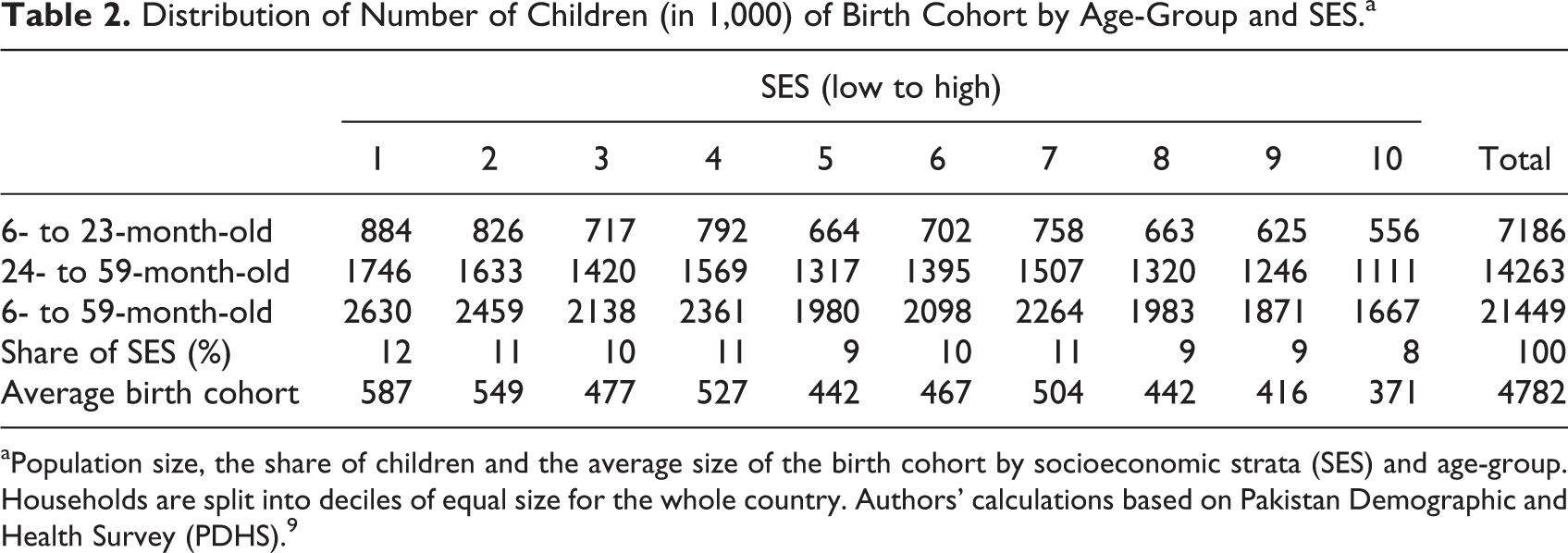
aPopulation size, the share of children and the average size of the birth cohort by socioeconomic strata (SES) and age-group. Households are split into deciles of equal size for the whole country. Authors’ calculations based on Pakistan Demographic and Health Survey (PDHS). 9
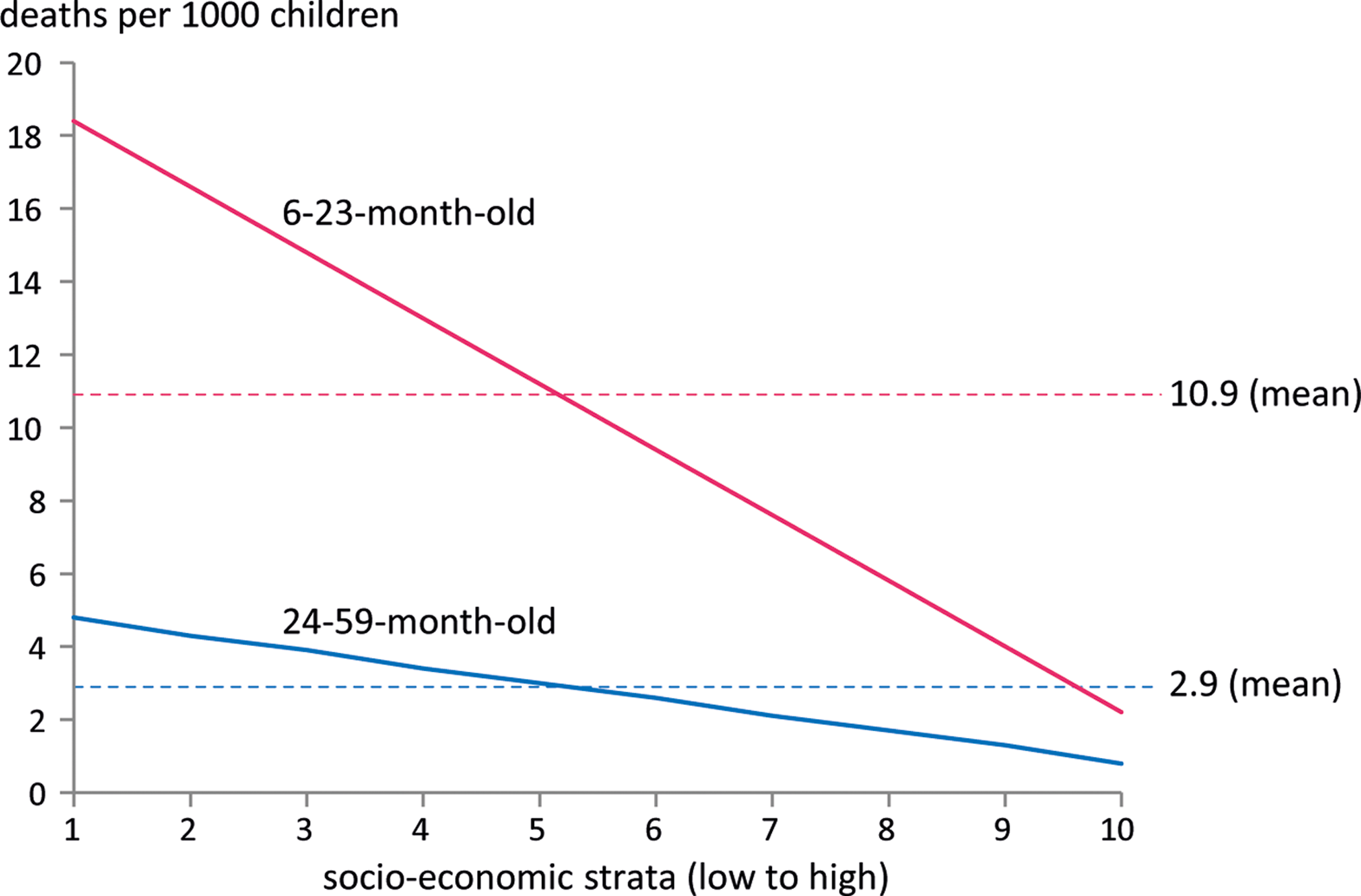
Yearly, all-cause mortality rates per 1000 children. The figure shows the smoothed all-cause mortality rates by SES and age-group. Households are split into deciles of equal size for the whole country. Authors’ calculations based on PDHS. 9 SES indicates socioeconomic strata; PDHS, Pakistan Demographic and Health Survey.
Prevalence of MNDs
The prevalence of MNDs was stratified by age-group, SES, and degree of severity (Table 3, Figure 4). The overall prevalence of IoD was 37% and ranged from 2.30% for severe IoD to 24.45% for mild IoD. IDA affected 39% of children and was higher in the 6- to 23-month-old group. Nearly half of the children were affected by VAD and 1 in 3 children by ZnD. Although prevalence of IDA and VAD decreased with increasing wealth, we could not stratify ZnD and IoD by SES due to data limitations.
Prevalence of MNDs by Age-Group and Severity in % (SE).a
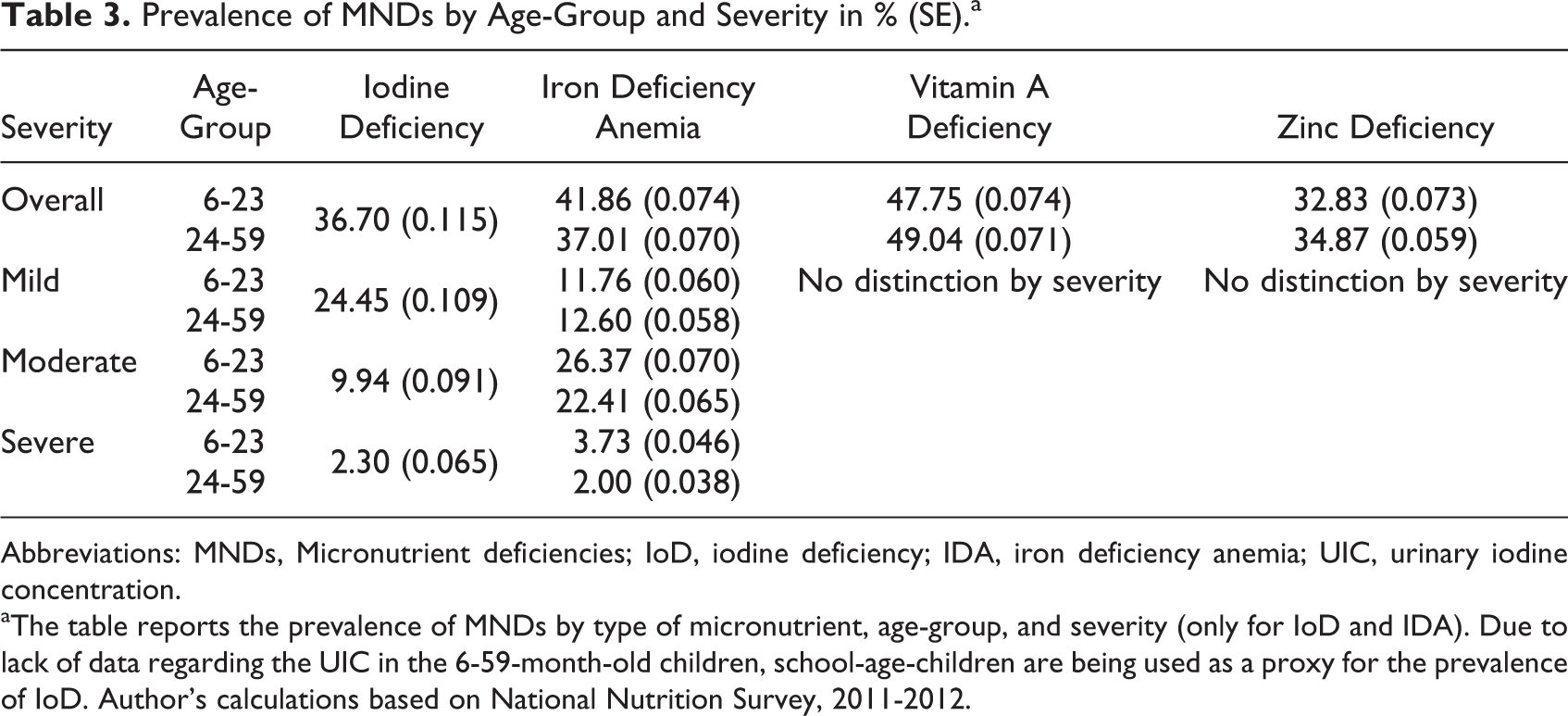
Abbreviations: MNDs, Micronutrient deficiencies; IoD, iodine deficiency; IDA, iron deficiency anemia; UIC, urinary iodine concentration.
aThe table reports the prevalence of MNDs by type of micronutrient, age-group, and severity (only for IoD and IDA). Due to lack of data regarding the UIC in the 6-59-month-old children, school-age-children are being used as a proxy for the prevalence of IoD. Author’s calculations based on National Nutrition Survey, 2011-2012.
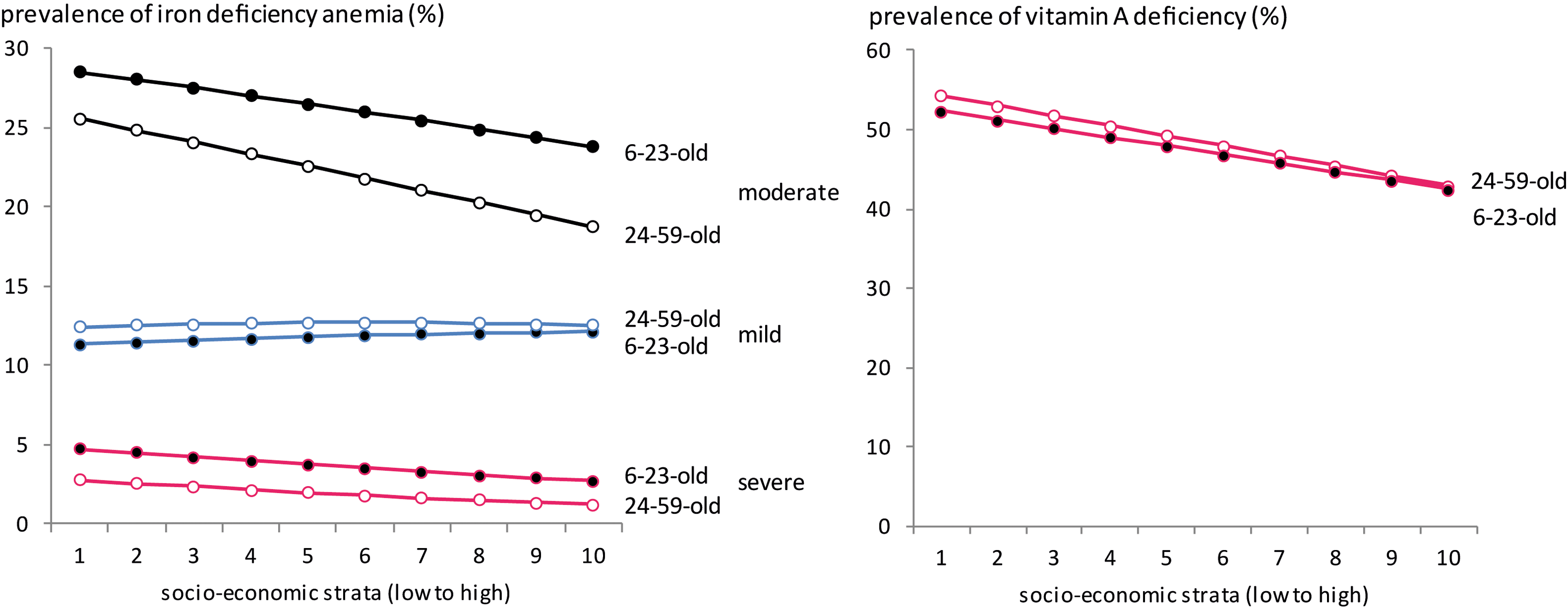
Prevalence of iron-deficiency anemia and vitamin A deficiency by age-group, severity, and SES (%). Households are split into SES deciles of equal size for the whole country. Authors’ calculation based on NNS 2011-2012. SES indicates socioeconomic strata; PDHS, Pakistan Demographic and Health Survey; NNS, Pakistan National Nutrition Survey.
Health and Cost Consequences
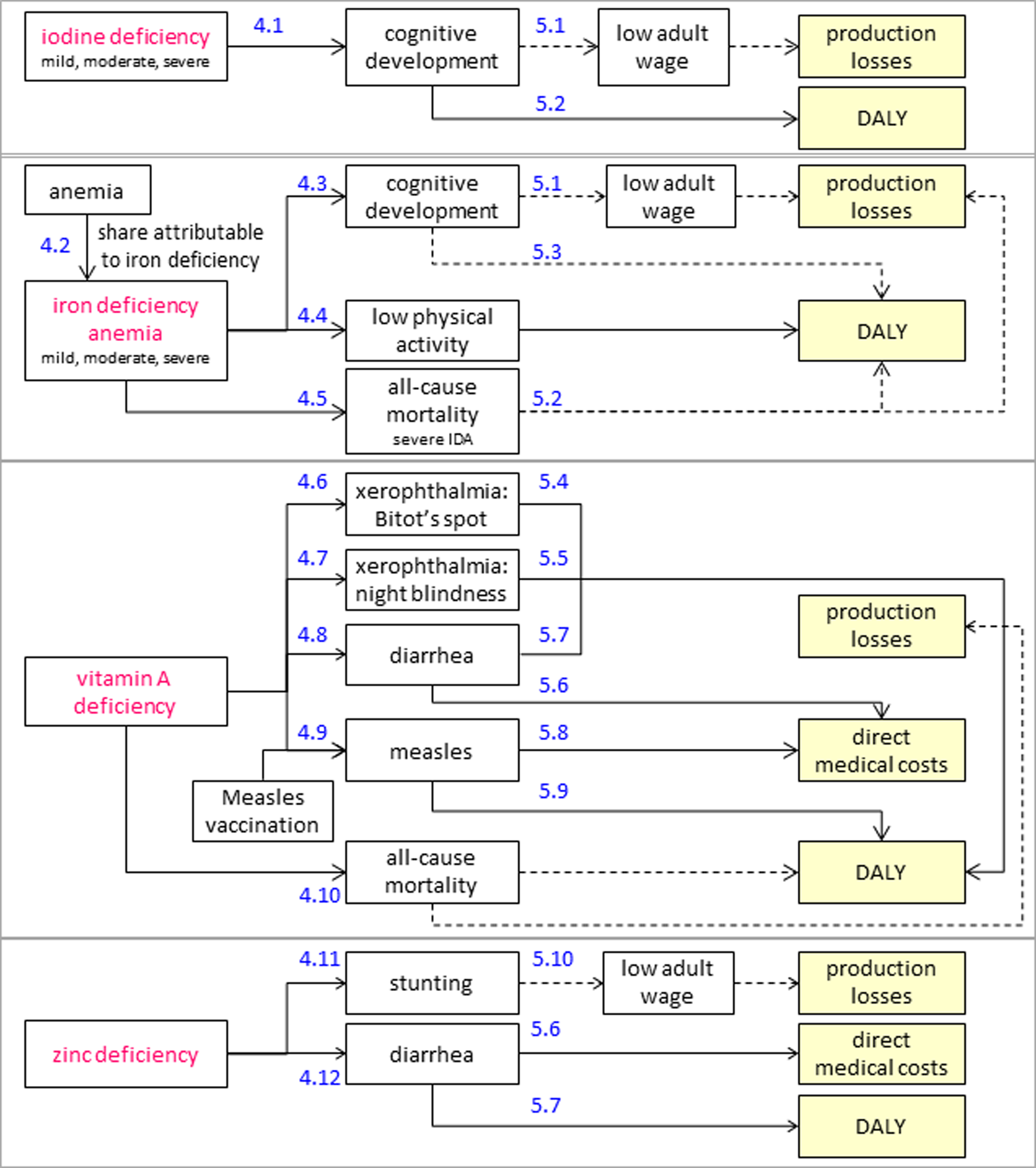
The health and cost consequences of MNDs in 6- to 59-month-old children are summarized in Figure 5 and Tables 4 and 5. IoD in this age-group of children may lead to mental impairement. 11 Mild to severe IoD may cause functional and/or structural changes in the brain leading to reversible and to permanent (nonreversible) loss of IQ points. 23 As there are no data quantifying the ratio of reversible to permanent damage of IoD on brain development, for the purposes of this analysis, we assume that the permanent effect is equal to one-third of the total effect on mental impairment and occurs only in 6- to 23-month-old children. We also assume that permanent loss of IQ points leads to future production losses and DALYs, whereas reversible loss of IQ points leads only to current DALYs (see Appendix 1).
Health Consequences of Micronutrient Deficiencies.a
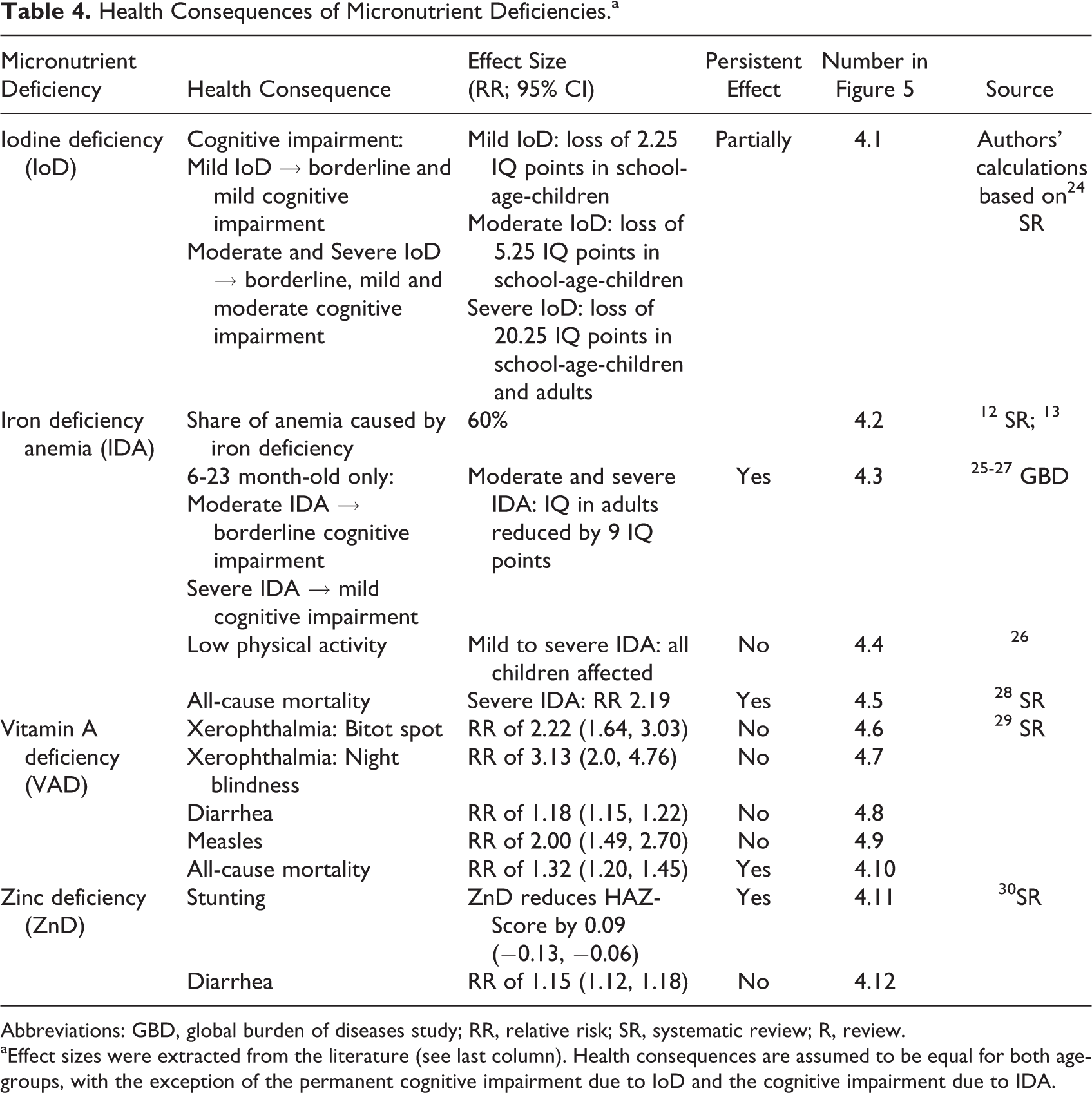
Abbreviations: GBD, global burden of diseases study; RR, relative risk; SR, systematic review; R, review.
aEffect sizes were extracted from the literature (see last column). Health consequences are assumed to be equal for both age-groups, with the exception of the permanent cognitive impairment due to IoD and the cognitive impairment due to IDA.
Cost Consequences of Adverse Health Effects.
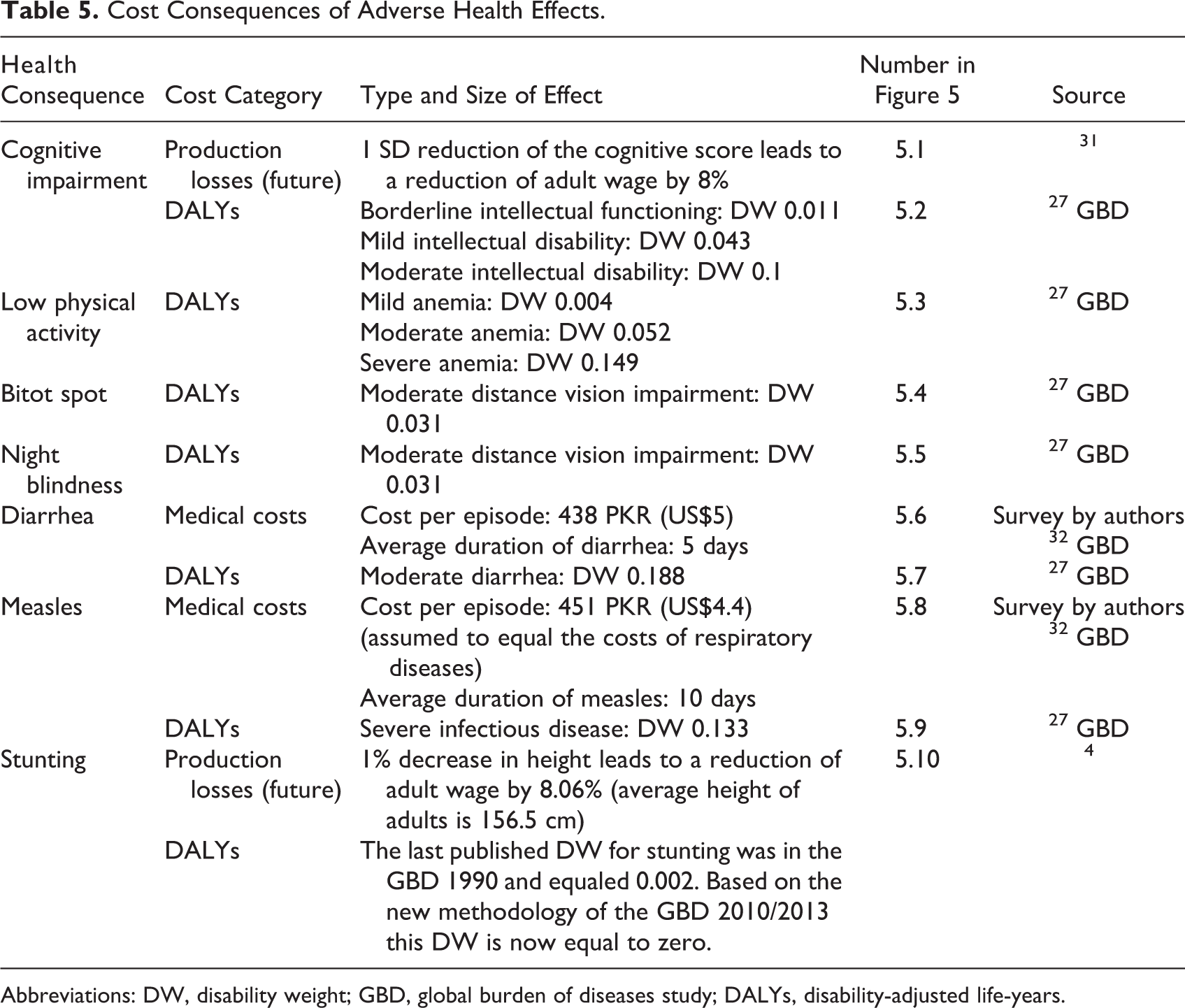
Abbreviations: DW, disability weight; GBD, global burden of diseases study; DALYs, disability-adjusted life-years.
The health consequences of IDA vary by severity and age-group. IDA may lead to impaired cognitive development, low physical activity, and increased all-cause mortality (severe IDA only). Moderate and severe IDA may cause permanent cognitive impairment in 6- to 23-month-old children affecting their productivity in adulthood. Although several studies have found a substantial effect of iron deficiency on cognitive ability, 33 –35 only Lozoff et al 33 have assessed the long-term effect on IQ and estimated it separately for children in mid-income and low-income households. Our model is based on the lower effect estimated in the mid-income group, as the higher effect in the low-income group might be biased due to adverse circumstances not purely attributable to IDA. IDA also reduces physical activity leading to reversible DALYs. Premature mortality due to severe IDA leads to future production losses and DALYs.
The health consequences of VAD include xerophthalmia and increased risk of diarrhea, measles, and all-cause mortality. 29 Bitot spot and night blindness are the first and most pervasive symptoms of xerophthalmia in young children. 36 They lead to current DALYs but are fully reversible with vitamin A supplements. Diarrhea and measles lead to direct medical costs and DALYs. Premature all-cause mortality leads to future production losses and DALYs.
The health consequences of ZnD include stunting and diarrhea. 30 Stunting has a long-term negative effect on adult wages. The cost consequences of diarrhea were calculated as for VAD. Table 6 presents additional economic parameters used in the health economic model.
Economic Model Parameters.

Abbreviation: DALYs, disability adjusted life years.
Societal Costs of MNDs
Combining the information on the prevalence of MNDs with the information on the health and cost consequences of these deficiencies, we estimated the total lifetime costs of MNDs in 6- to 59-month-old children in Pakistan in 2013 (Table 7). The costs accounted for US$46 (95% confidence interval [CI] 26.1-71.5) million in medical costs, US$3,222 (95% CI 1135-7725) million in production losses, and 3.4 (95% CI 2.5-4.4) million DALYs. They represent the lifetime costs of a birth cohort affected by IoD, IDA, VAD, and ZnD between the age of 6 and 59 months and may also be interpreted as the costs that could be prevented every year by completely eliminating these MNDs in this age-group.
Total Costs by Cost Category, Time Dimension, MND, and Age-Group.a
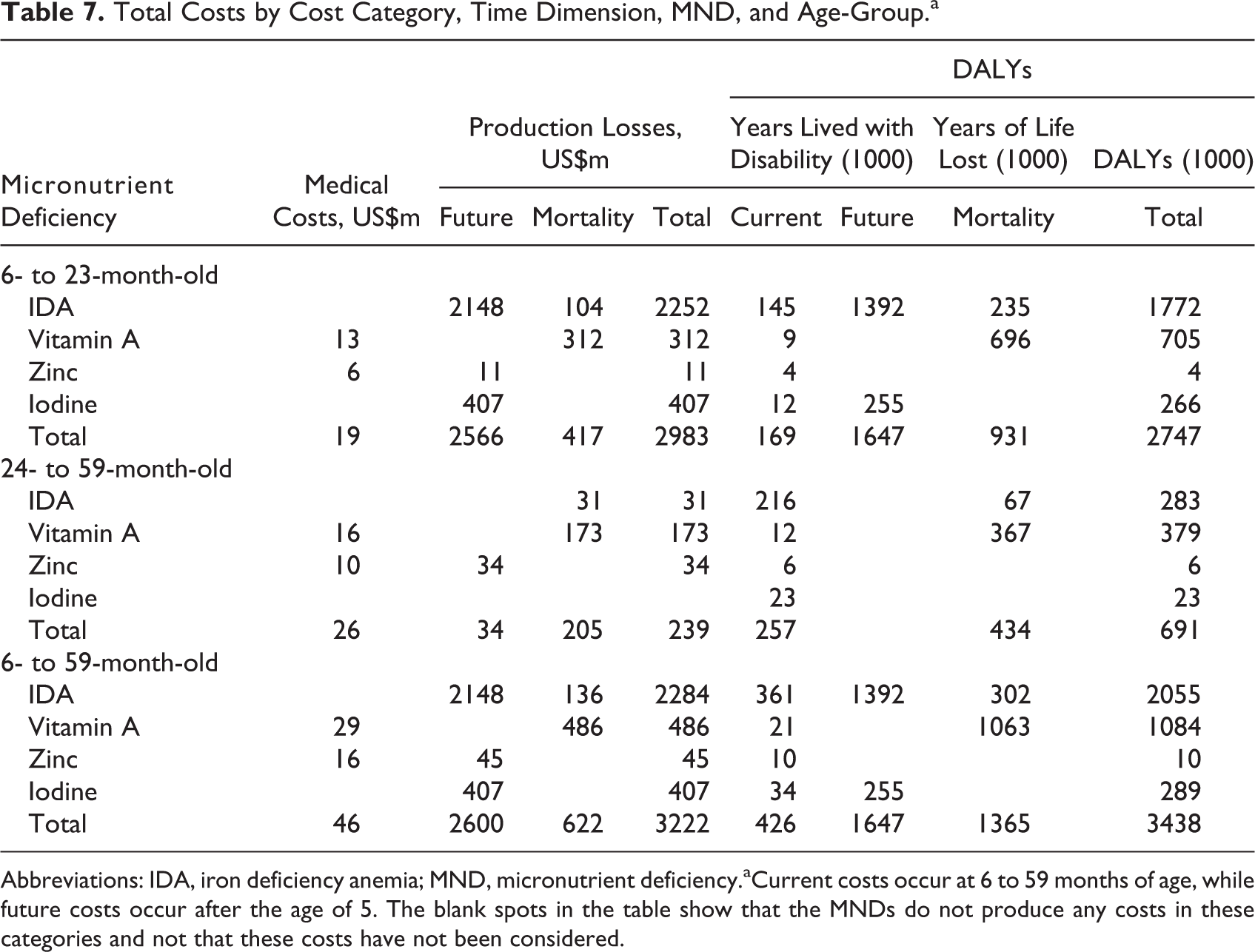
Abbreviations: IDA, iron deficiency anemia; MND, micronutrient deficiency.aCurrent costs occur at 6 to 59 months of age, while future costs occur after the age of 5. The blank spots in the table show that the MNDs do not produce any costs in these categories and not that these costs have not been considered.
Costs were dominated by MNDs in 6- to 23-month-old children, with 92.6% of production losses and 79.9% of DALYs (Figure 6). The high production losses in the 6- to 23-month-old children were due to the impaired cognitive development induced by IDA and IoD. The IDA was responsible for over two-thirds of all production losses and over half of all DALYs. The VAD and IoD were respectively the second and third most important causes for both production losses and for DALYs. ZnD played only a minor role for production losses and was negligible for DALYs.
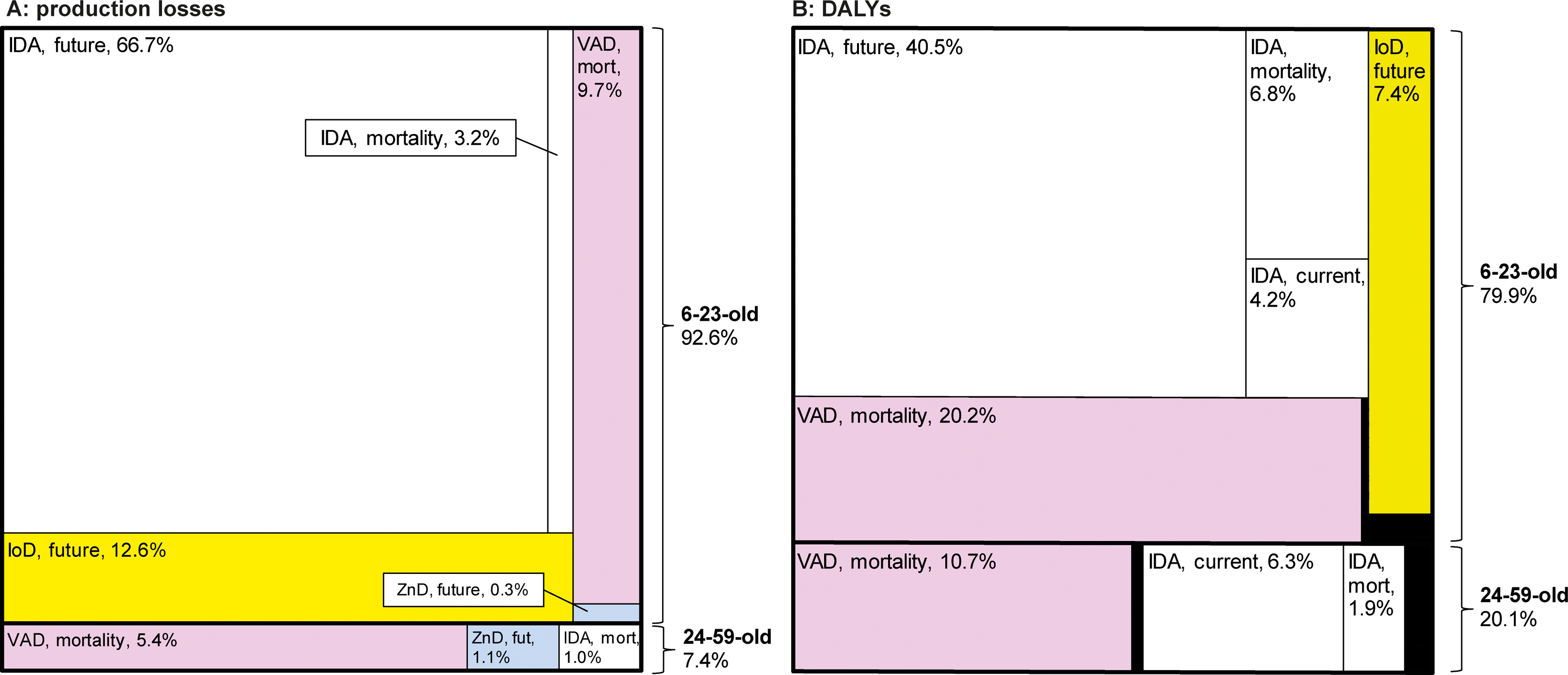
Production losses and DALYs by micronutrient deficiency and cost category (share of respective total). Shares of the production losses (A) and DALYs (B) by age-group (6- to 23-month-old and 24-59-month-old), type of micronutrient deficiency and type of cost (current, future life, and mortality). Areas in black represent minor cost categories. IDA, iron deficiency anemia; IoD, iodine deficiency; VAD, vitamin A deficiency; ZnD, zinc deficiency; mort, mortality; fut, future; DALYs, disability adjusted life years.
The comparison between the societal costs of MNDs in children living in the poorer and wealthier households is presented in Figure 7. Costs were higher in poorer households due to the higher prevalence and severity of deficiencies and the higher number of children. Differences were particularly strong for VAD with 4 times higher production losses and 6 times higher DALYs in the poorest than in the wealthiest third of households.
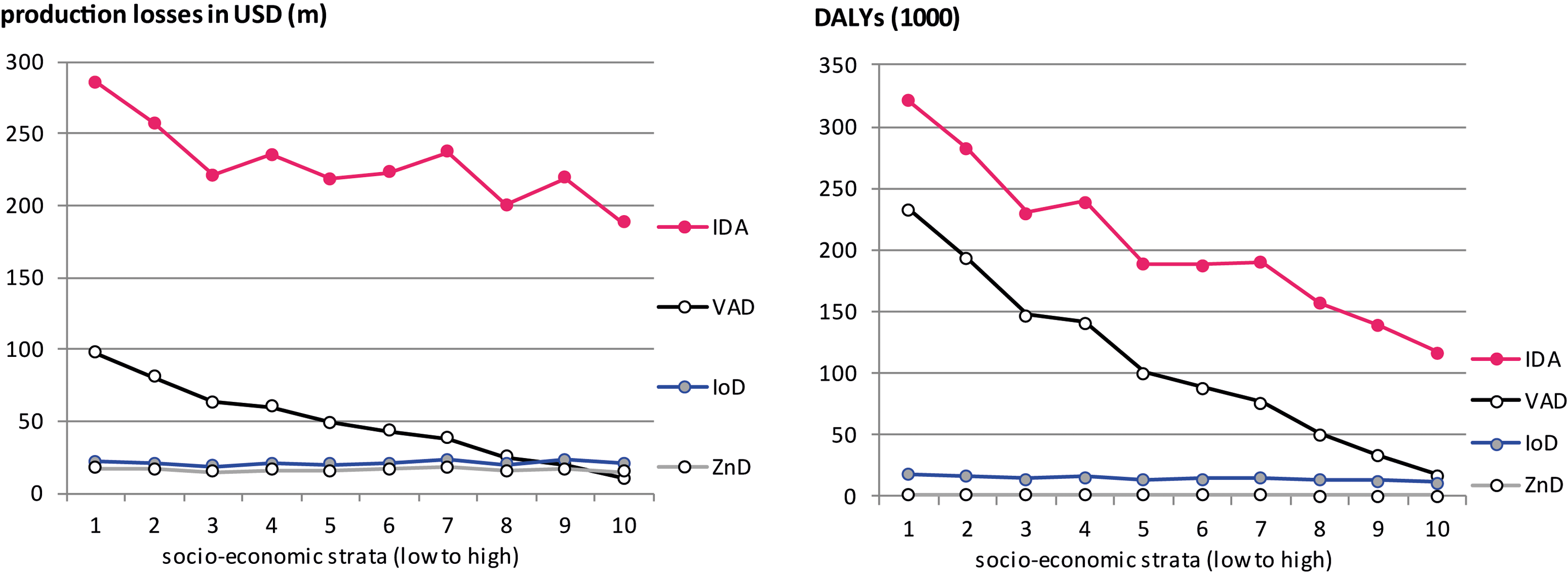
Production losses and DALYs due to iron deficiency anemia and vitamin A deficiency by socioeconomic strata. Households are split into deciles of equal size for the whole country. The societal costs of zinc deficiency and iodine deficiency are not represented as their prevalence could not be calculated by socioeconomic strata. IDA, iron deficiency anemia; VAD, vitamin A deficiency; DALYs, disability adjusted life years.
Results of SA
The tornado diagrams show the effect of a 10% increase or decrease in the model parameters (Figure 8). Production losses were most influenced by the discount rate, the work-related parameters (work force participation rate, wage, income growth), parameters relating to the effect of IDA on cognition, and by the relative risk of mortality with VAD. DALYs were most influenced by the relative risk of mortality with VAD and the parameters related to the impact of IDA (share of anemia attributable to IDA, disability weights of intellectual disability, and relative risk of mortality with IDA).
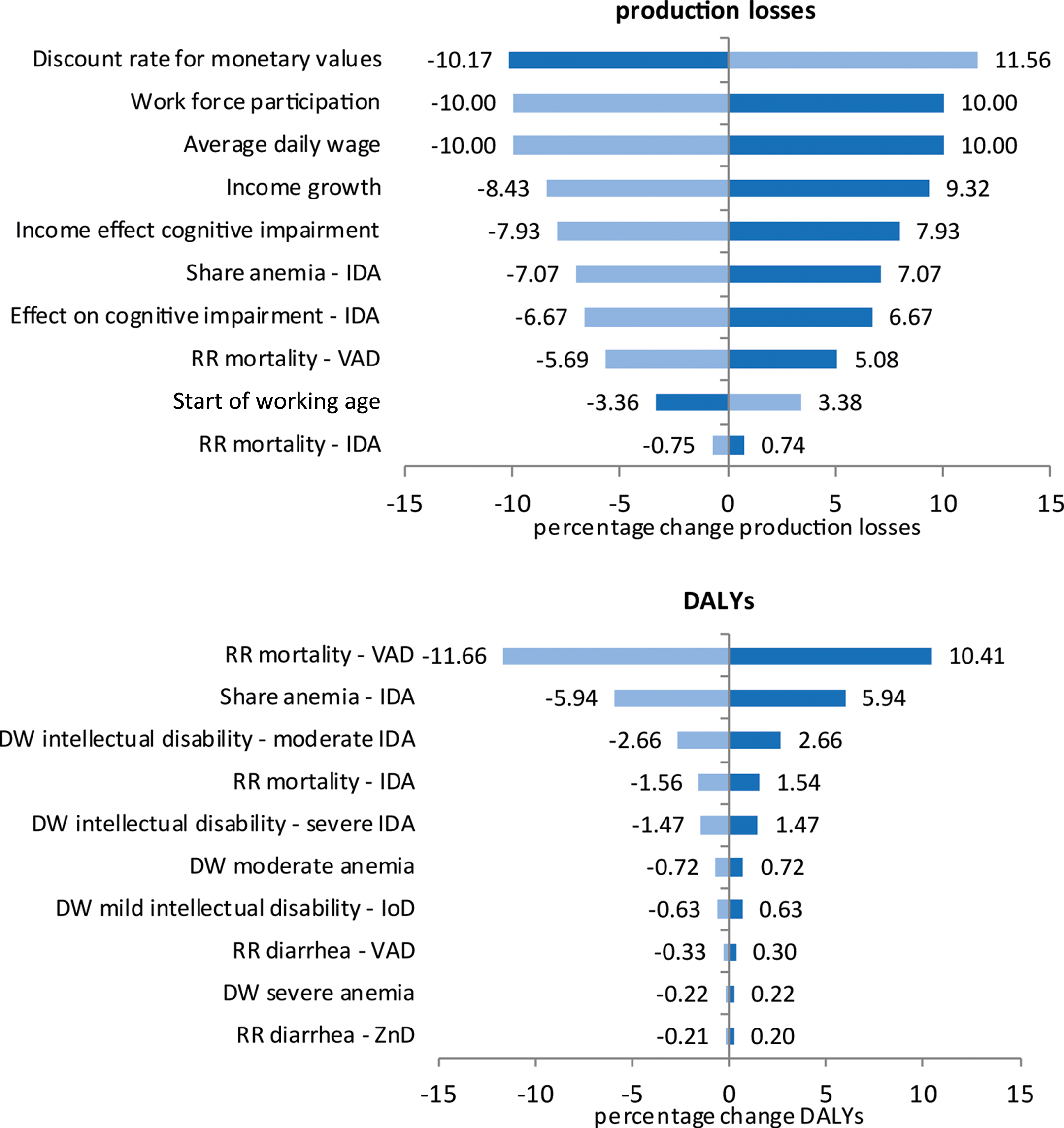
Univariate sensitivity analysis of production losses and DALYs. Dark blue bars show the effect of a 10% increase in a parameter, while light blue bars show the effect of a 10% decrease in a parameter. RR, relative risk; DW, disability weight; IoD, iodine deficiency; IDA, iron deficiency anemia; VAD, vitamin A deficiency; ZnD, zinc deficiency; DALYs, disability adjusted life years.
For the probabilistic SA, we randomly varied all model parameters within predefined distributions and then ran the model 10 000 times. Where information on the variability of the parameters was not available from the literature, we chose an SD equaling 20% of the mean. Figure 9 shows the distribution of total production losses and total DALYs together with 95% CIs according to the multivariate SA. The CIs around our estimates are relatively narrow with 95% of all model runs demonstrating production losses between US$1.1 and US$7.6 billion and 95% of DALYs between 2.5 and 4.4 million DALYs.
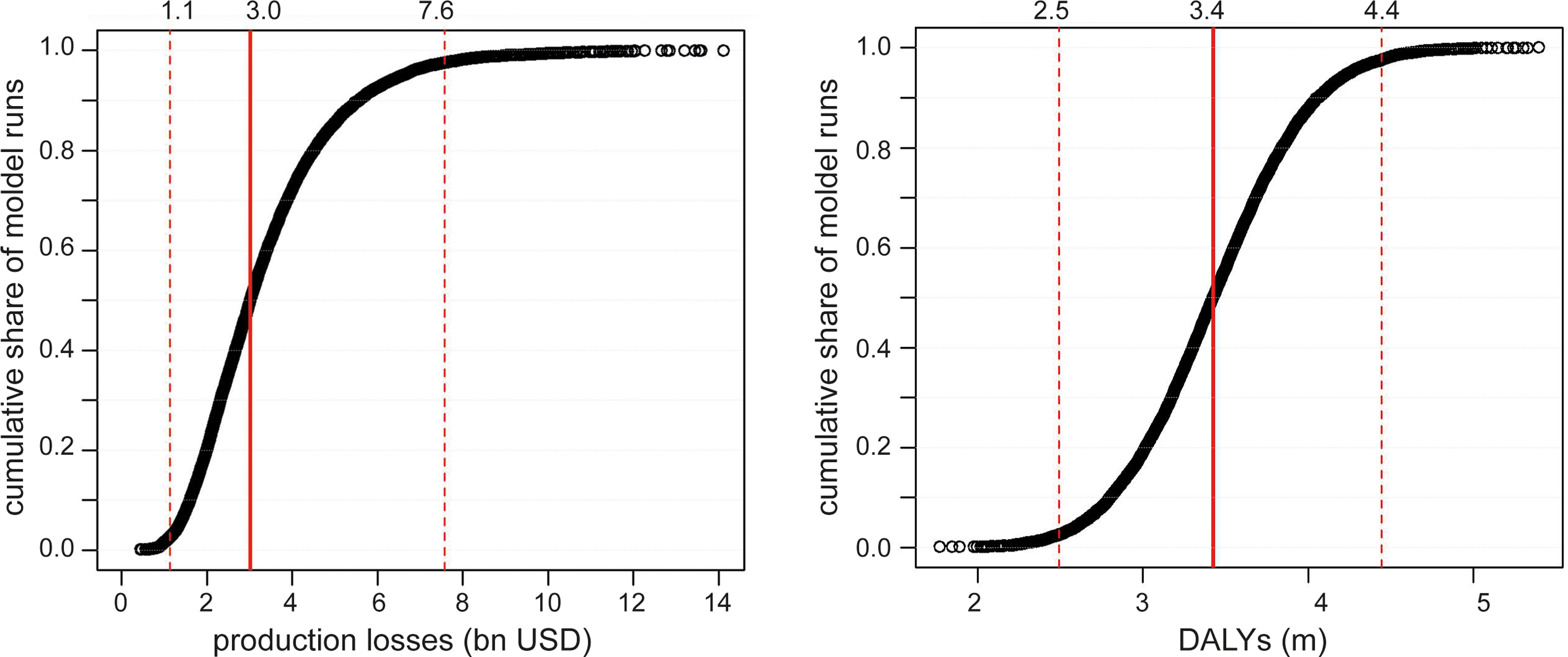
Multivariate sensitivity analysis by type of societal cost. Distribution of total production losses and total DALYs in 10 000 model runs. The dotted lines represent 95% confidence intervals. DALYs indicate disability adjusted life years.
Discussion
We assessed the societal costs of MNDs in 6- to 59-month-old Pakistani children in 2013. The costs were calculated as the lifetime cost consequences of the current levels of iodine, iron, vitamin A, and zinc deficiencies in a yearly birth cohort. Total societal costs amounted to medical costs of US$46 million for the treatment of illness attributable to MNDs, to production losses of US$3,222 million due to future incomes lost, and to 3.4 million DALYs due to premature deaths and years lived with disability. These costs represent a substantial burden for Pakistan and correspond to 1.44% of GDP in 2013 lost due to lower future productivity and to 4.45% of the overall burden of 77.3 million DALYs in 2013. 21 These costs can be interpreted as the costs that could be prevented every single year if MNDs could be completely eliminated in 6- to 59-month-old children.
Costs occurring at the age of 6 to 23 months were substantially higher than those at the age of 24 to 59 months because of the higher prevalence of MNDs and the more severe health consequences at younger age. The same reasons led to higher costs in poorer households. However, costs were substantial even in the wealthiest household decile. IDA had the most severe cost consequences accounting for two-thirds of total costs. The costs of VAD and IoD were also substantial, while the costs of ZnD were limited.
Production losses were dominated by future income losses due to impaired cognitive and physical development. They affect the country’s economic growth and contribute to an undernutrition-based poverty trap in which children born in poor households are more likely to become poor parents themselves.
Our study is based on a health economic model simulating the lifetime consequences of MNDs in young children by combining information from clinical and epidemiological studies with economic data. 44 The model incorporates the currently available evidence on the prevalence of MNDs in Pakistan, on their health consequences in young children, and on their long-term effects. The model might be criticized for the long causal chains linking MNDs in early childhood to health and cost outcomes over the whole lifespan. However, there is no alternative to this approach, as randomized controlled studies tracking outcomes over such a long time period are not available.
A previous study has estimated the lifetime consequences of IDA in Pakistani children on production losses at 0.75% of GDP in 1996. 45 This result is of the same magnitude as ours, although it based on older literature on the impact of IDA on cognition and not limited to 6- to 59-month-old children.
Our model differs from other epidemiological models describing the consequences of MNDs such as the Life Saved Tool 46 or the PROFILES model, 47 as it considers morbidity as well as mortality and assesses the cost consequences in the 3 dimensions (medical costs, production losses, and DALYs). Furthermore, it differentiates by the socioeconomic status of households and between 6- to 23-month-old and 24- to 59-month-old children. We believe that this model is particularly well suited to capture the multiple often irreversible health and cost consequences of MNDs in these children.
Several limitations of our analysis should be recognized.
The high levels of MNDs in Pakistan have been recognized for decades, but with the exception of IoD, very little progress has been made in eradicating them. 3 Although economic growth may contribute to improve the nutritional content of diets, it is not sufficient to solve the problem as illustrated by the considerable levels of MNDs in children living in relatively wealthy Pakistani households. The micronutrient status of the population must thus be improved by increasing micronutrient intake through food fortification and supplementation.
The Pakistan Standards and Quality Control Act has issued specifications for the mass fortification of staple foods such as wheat flour, edible oil and ghee, and iodized salt. 48 So far, only iodized salt has been successful by reaching a household coverage of 69% in 2011. 3 Efforts for the mass fortification of other staple foods, such as the fortification of edible oil with vitamin A or of wheat flour with iron and folate, have been largely unsuccessful due to weak legislative support and a poor regulatory environment. 48 The provision of micronutrient supplements to populations with particularly high nutritional needs is another strategy for reducing MNDs. 49 Vitamin A supplementation has reached a high coverage in 6- to 59-month-old children in Pakistan with 72%. 9 However, the persistently high levels of VAD among these children show that supplementation, although effective in controlled trials, may not be sufficient.
Our results confirm the urgent need for policies aiming at an improvement in the micronutrient status of 6- to 59-month-old children. Interventions should in particular target 6- to 23-month-old children, as social costs of MNDs are dominated by the costs occurring in this age-group. Such policies may include the promotion of dietary diversity and complementary feeding, food fortification as well as multiple micronutrient supplementation. 49 Furthermore, interventions aiming to eradicate MNDs in young children should not only be aimed at children but also at their mothers, as the low body stores of micronutrients in young children may also be the consequence of maternal undernutrition in pregnancy and while breastfeeding.
In conclusion, our study reveals the severe cost consequences of iodine, iron, vitamin A, and zinc deficiencies in young Pakistani children. Most of these costs are invisible to the parents, as they will occur over the future lifetime of the child. As our results identify the areas of highest socieatal costs of MNDs, they may be useful for the design and the implementation of interventions aiming to maximize the effect of the available financial resources. Future research should focus on further improving the knowledge on the living conditions and on the inadequate nutrition responsible for MNDs. This research could be strengthened with refined population surveys collecting richer data on health, nutrition, exposition to policy measures, and socioeconomic conditions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded through an unrestricted grant from Nestlé Research Center. Additional direct and in-kind support was received from the Aga Khan University, the ETH Zurich, and the Zurich University of Applied Sciences.
