Abstract
Background:
Incorporation of omega-3 polyunsaturated fatty acid (PUFA) sources to lipid-based nutrition supplement (LNS) formulations for undernourished populations presents a challenge due to reduced shelf life and poor consumer acceptability.
Objective:
To systematically develop an omega-3-fortified LNS formulation, using flaxseed oil (FO), by optimizing its processing stability and evaluating its sensory acceptance.
Methods:
LNS formulations were formulated to yield approximately 452 kcal, 13.2 g of protein, 30 g of fat, and 53 g of carbohydrates per 100 g. Response surface methodology (RSM) with 4-factor-3-level: omega-3 fatty acid source (FO; 0%-10%), antioxidant (ascorbyl palmitate: 0.0%-0.03%), emulsifier (soy lecithin: 0.5%-1.5%), and storage time (0-6 months at 40°C) was used to optimize LNS’s functionality and storage stability. Factor effects were evaluated for peroxide value, oil separation, water activity, moisture content, hardness, and vitamin C content of the samples. Consumer acceptability was assessed using a 9-point hedonic scale.
Results:
After 6 months of accelerated storage, the addition of FO to LNS formula resulted in increased peroxide values (7.75 mEq/kg fat) and lipid separation and a reduction in vitamin C and water activity. Optimal levels to maximize storage time and FO and minimize oxidation were 0.02% antioxidant, 1.5% emulsifier, and 4.9% FO. Indian women and students accepted LNS formulations with or without FO similar to other commercial supplements.
Conclusion:
An LNS containing FO with improved omega-6/omega-3 PUFA ratio was successfully formulated with staple Indian ingredients and optimized for storage stability using RSM.
Keywords
Introduction
Moderate acute malnutrition (MAM) afflicts 33 million children worldwide of which 20 million children live in India alone. 1 Moderate acute malnutrition is characterized by weight for height score between −3 and −2 Z scores compared to a reference population or by mid-upper arm circumference <12.5 cm. 2 Supplementary feeding programs are widely used to tackle MAM and avoid mortality by reducing deterioration into severe acute malnutrition (SAM), despite insufficient evidence on their effectiveness. 3 In India, MAM is addressed through take-home rations such as mixed flours (sattu), sweets (sheera), and porridge (upma) or sometimes raw ingredients such as rice and pulses. Although such interventions have been useful in improving some health outcomes, there are still issues with nutritional adequacy and consumption compliance due to the low acceptability of products, and additional food preparation requirements. 4,5
Lipid-based nutrient supplements (LNS) are ready-to-use energy- and nutrient-dense food products that are suitable to complement most food rations, especially due to their longer shelf life and convenience. Existing LNS have been widely successful in treating SAM among at-risk children and has instigated the development of similar or improved alternative foods for addressing MAM to overcome the issues with existing supplementary feeding program. 6,7
Recently, there has been an increased interest in improving the omega-3 fatty acid profile of conventionally used peanut-based formulations, which are rich in omega-6 linoleic acid (LA) but contain negligible omega-3 alpha linolenic acid (ALA). 8 -10 Balanced omega-6/omega-3 polyunsaturated fatty acids (PUFAs) are beneficial for children having SAM or MAM, as omega-3 ALA serves as precursors for eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), which play a significant role in modulation of tissue inflammation, immune function, and overall neural development. 11,12 In addition, omega-3 PUFA modulates intestinal inflammation, common in malnourished children, that results in malabsorption of nutrients from the diet. 13,14 Currently, there are no specifications for omega-6–omega-3 PUFA ratio in LNS; however, the importance of balanced PUFAs in LNS is now well recognized. 8
Incorporating omega-3 PUFA sources to LNS formulation is challenging due to storability, oxidative stability, and off-flavor issues resulting in reduced shelf life and poor consumer acceptability. 15 The aim of this study was to systematically develop an omega-3 fortified LNS formulation using flaxseed oil (FO) by optimizing process variables based on stability parameters, characterize its physicochemical properties, and evaluate its consumer acceptance. This process will result in a stable and acceptable complementary spread that is highly palatable, cost-effective, and locally produced and that provides required micronutrients, which could be included in the daily meals of children at schools or supplementary feeding centers in India.
Materials and Methods
Ingredients
Ingredients included chickpea flour (Spicy World, Houston, TX), soy flour (Bob’s Red Mill, Milwaukee, OR), maltodextrin (Honeyville, Inc, Brigham City, UT), FO (Spectrum Naturals, Inc, Melville, New York), clarified butter (ghee; AMUL, Anand, India), skim milk powder (Barry Farm, Cridersville, OH), soybean oil (Crisco, Orville, OH), coconut oil (Spectrum Naturals, Inc, Melville, New York), palm oil shortening (Crisco, Orville, OH), vitamin mineral mix (Watson Inc, West Haven, CT), soy lecithin (Now, Bloomingdale, IL), antioxidant ascorbyl palmitate (Spectrum Chemical, Gardena, CA), and cocoa powder (Hershey’s, Hershey, PA). LNS A20 was donated by USAID, and Sattu Maavu was procured from a commercial supplier in India. Three-layer minipouches (PAKVF4) were purchased from IMPAK Corporation (Los Angeles, CA).
Chemicals
Hexane, acetic acid, potassium iodide, sodium thiosulphate, starch solution, and sand were procured from Fisher Scientific. Chloroform, metaphosphoric acid, EDTA, sodium phosphate monobasic, and
Recipe Formulation
Recipes for LNS were designed using ESHA food processor diet analysis and fitness software (ESHA research version 10.10.0) and the Indian nutrient database to yield approximately 13% protein (ie, dairy, chickpea, and soy), 40% carbohydrate (ie, sucrose and maltodextrin), 38% fat (different blends of ghee, soybean oil, coconut oil, and FO), and 3% complete vitamin and mineral mix (Table 1). 16 Flaxseed oil was incorporated at 5% and 10% level by replacing soybean oil to prevent any textural changes, as they have a similar viscosity (800 mPa·s) and specific gravity (0.91 at 25°C). 17 Formulations were designed to meet the World Food Program LNS technical specifications 18 and specifications described by Chaparro and Dewey. 19 Ready-to-use therapeutic food (RUTF) specifications were obtained from Medecins Sans Frontieres/United Nations Children’s Fund (UNICEF) 20 and Commercial Item Description 21 for standards not listed but specified by Nguyen and Bouzambou. 18
Recipe Formulation for LNS.
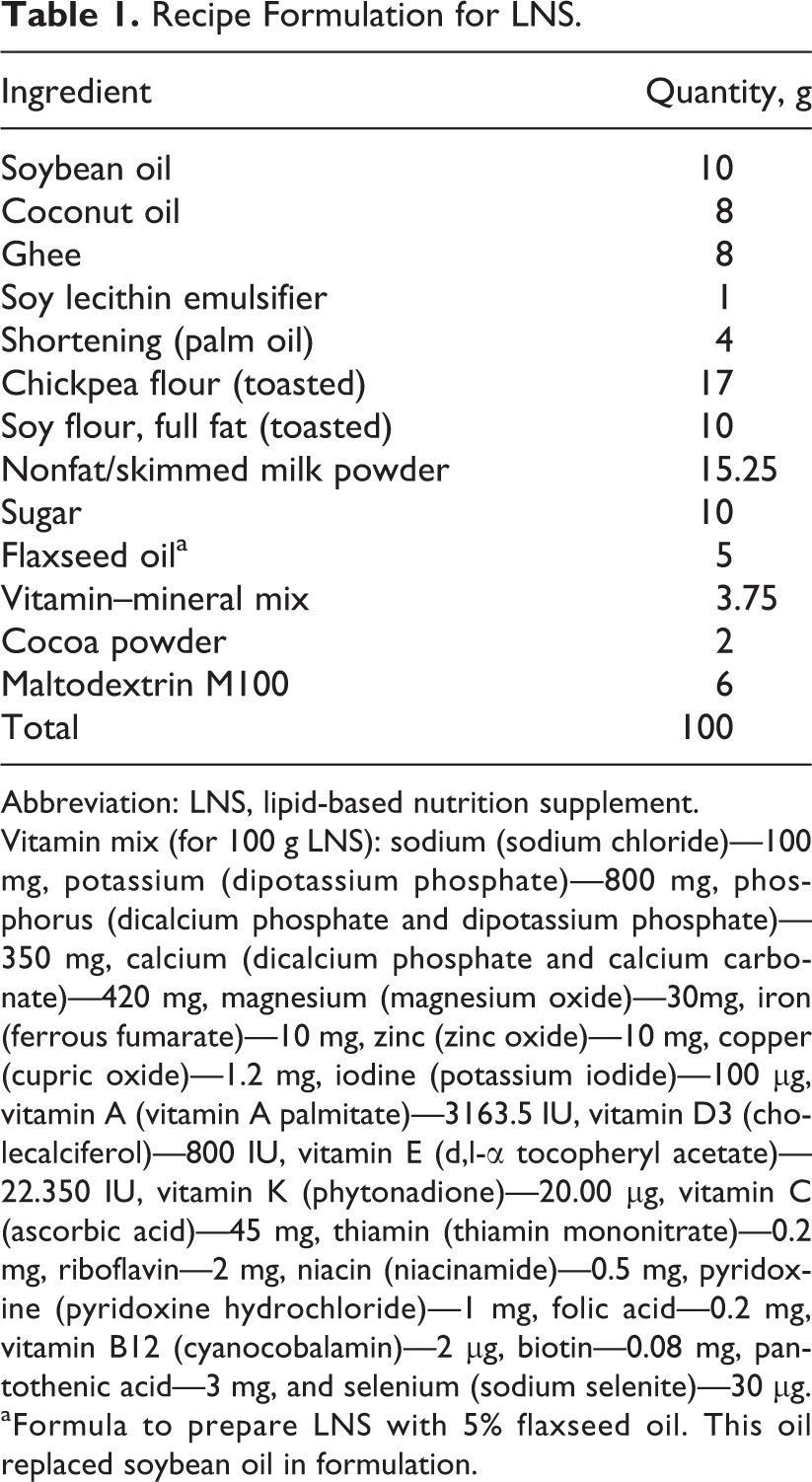
Abbreviation: LNS, lipid-based nutrition supplement.
Vitamin mix (for 100 g LNS): sodium (sodium chloride)—100 mg, potassium (dipotassium phosphate)—800 mg, phosphorus (dicalcium phosphate and dipotassium phosphate)—350 mg, calcium (dicalcium phosphate and calcium carbonate)—420 mg, magnesium (magnesium oxide)—30mg, iron (ferrous fumarate)—10 mg, zinc (zinc oxide)—10 mg, copper (cupric oxide)—1.2 mg, iodine (potassium iodide)—100 µg, vitamin A (vitamin A palmitate)—3163.5 IU, vitamin D3 (cholecalciferol)—800 IU, vitamin E (d, l-α tocopheryl acetate)—22.350 IU, vitamin K (phytonadione)—20.00 µg, vitamin C (ascorbic acid)—45 mg, thiamin (thiamin mononitrate)—0.2 mg, riboflavin—2 mg, niacin (niacinamide)—0.5 mg, pyridoxine (pyridoxine hydrochloride)—1 mg, folic acid—0.2 mg, vitamin B12 (cyanocobalamin)—2 µg, biotin—0.08 mg, pantothenic acid—3 mg, and selenium (sodium selenite)—30 µg.
aFormula to prepare LNS with 5% flaxseed oil. This oil replaced soybean oil in formulation.
General Lipid-Based Nutrition Supplement Preparation
Figure 1 illustrates the preparation of LNS. Weighed flours were toasted at 110°C for 120 minutes in a forced-convection hot-air oven (Mechanical Oven; Precision Scientific, Fisher Scientific, Marietta, OH). Flours were added to other dry ingredients and mixed (2 minutes); this constituted the dry mix. Variations in the content of antioxidant and emulsifier were made at this point. The dry mix was added to a wet grinder (PG503, Premier Wonder), and contents were mixed (3 minutes) to ensure homogenous mixing and size reduction. The fats and oils were brought to room temperature (23°C ± 2°C) and mixed in a glass bowl for 2 minutes. Variations in the amount of FO were made at this point. The lipid mixture was added to dry contents in a wet grinder and blended for 5 minutes.
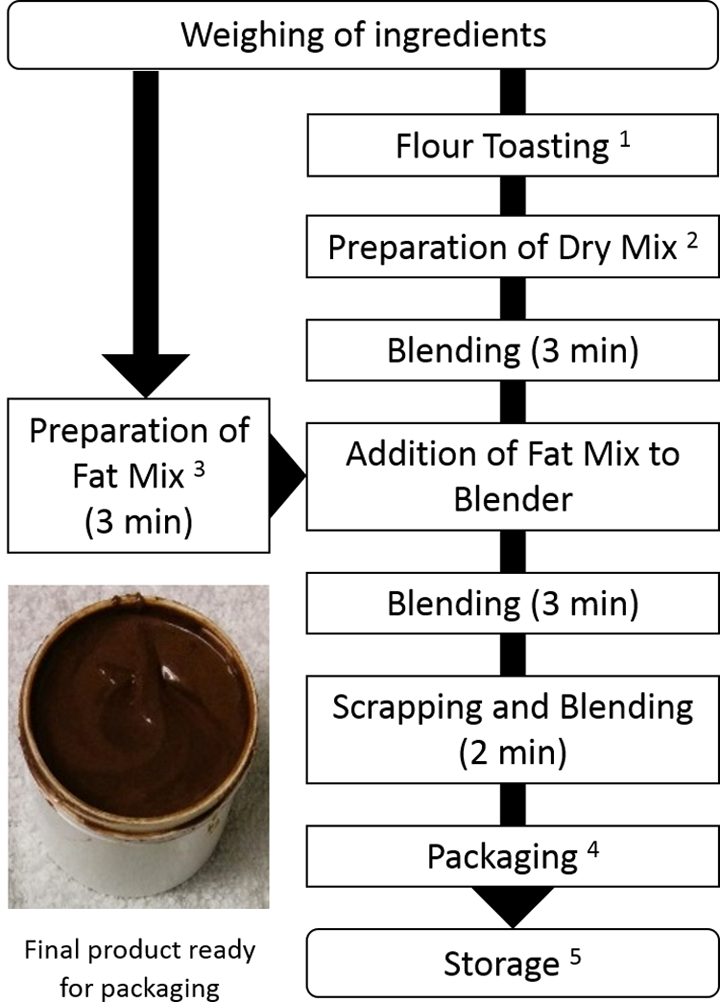
Process flowchart for the preparation of Indian lipid-based nutrition supplement (LNS). (1) Hot air–forced convection toasting of flours (chickpea and soy flour) at 110°C for 120 minutes. (2) Dry mix contains toasted chickpea flour, toasted soy flour, nonfat/skimmed milk powder, sugar, cocoa powder, maltodextrin, and vitamin–mineral mix. (3) Lipid mix contains soybean oil, coconut oil, ghee, soy lecithin emulsifier, shortening (palm oil), and flaxseed oil. (4) Packaged in 3-layer minipouches (PAKVF4), heat sealed under nitrogen (99.99% purity) using equipment MVS 31—XP at conditions vacuum 50%, gas ratio 20%, and time 2.5 seconds. (5) Pouches were stored in hot-air incubators (Precision by Thermo Scientific) held at 40°C ± 2°C for 6 months.
Packaging and Storage
The product was filled into 3-layer minipouches and heat-sealed under nitrogen (99.99% pure) using a Minipack America Sealer (MVS 31–XP) with the following settings vacuum 50%, gas ratio 20%, and time 2.5 seconds. Sealed pouches were held at 40°C ± 2°C for 6 months for accelerated shelf life testing equivalent to 12 months at room temperature (∼30°C). 22,23 Samples were evaluated at 0, 3, and 6 months.
Water Activity
Sample water activity (Aw) was measured using an Aqualab water activity meter (Aqualab 4TE, Decagon Devices Inc, Pullman, WA). 24 Sample (1 g) was weighed into 15 mL disposable sample cups and inserted into the instrument.
Peroxide Value
Peroxide value of samples was measured in triplicate using a modified method from AOCS, as described by Crowe and White. 25
Moisture Content
Modified oven drying method (sand pan technique) was used for the estimation of moisture content. 26 Aluminium metal dishes with lids containing 10 g of sand were dried along with a glass rod for 1 hour. The dish was weighed to the nearest 0.0001 g after cooling in a desiccator. Sample (5 g) was added to the dish and thoroughly mixed with sand using the glass rod. Samples were dried in a hot-air oven for 5 hours at 95°C.
Lipid Separation
Lipid separation was measured in triplicate using a modified method of Hinds et al and Perlman. 27,28 Pouches were thoroughly and uniformly massaged for 2 minutes before opening. A 30-g sample was carefully transferred to a 50-mL centrifuge tube. To simulate natural separation, tubes were centrifuged at a low speed (300 ×g) at 22°C (Sorvall ST 16R with TX400 swinging bucket rotor, Thermo Scientific) for 3 minutes. A digital Vernier calliper was used to determine the height of lipid layer separated at the top of the sample.
Texture Analysis
Sample hardness was measured using a texture analyzer (TA-XT2; Texture Technologies Corp, Scarsdale, New York) following a method adapted from Ahmed and Ali. 29 Pouches extracted from incubator were uniformly massaged for 2 minutes, and 50 g sample was placed in Petri dishes (15 cm × by 1.5 cm height) in triplicates. Samples were left undisturbed for 1 hour, and temperature was monitored using an infrared thermometer. During analysis, each sample dish was secured to the base. The probe (50 mm diameter, flat surface) was moved at a crosshead speed of 0.5 mm/s with 4 mm penetration. Hardness was measured as the peak force (N) of the first compression cycle.
Vitamin C Determination
The method of Tarrago-Trani et al with modifications was used for extraction and measurement of vitamin C. 30 The sample size used was 0.2 g, and the extraction buffer was 1 mmol/L EDTA/4% meta-phosphoric acid. Separation and quantification of vitamin C were carried out by reverse phase separation and UV detection (photodiode array at 265 nm) using a waters high-performance liquid chromatography (HPLC) with an isocratic pump system. An ISIS C-18 column (150 × 4.6 mm, 3 μm particle size; Supelco-Sigma, St Louis, Missouri) was equilibrated with mobile phase consisting of 25 mmol/L sodium phosphate buffer, pH 3.0, and pumped at 0.4 mL/min. Vitamin C was quantified using HPLC grade external standards. Results were expressed as mg of vitamin C per gram samples.
Fatty Acid Analysis
Fatty acid composition of LNS samples was determined using Bligh and Dyer 31 method. Briefly, total lipid was extracted with hexane from a sample (500 mg) and then derivatized using methanolic HCl (4% HCl; 90°C, 40 minutes) to prepare fatty acid methyl esters. Compounds were analyzed by gas chromatography using a Hewlett Packard 5890 II system equipped with a 75-m, narrow-bore 0.18 mm internal diameter SP 2560 capillary column (0.14-µm film) and a flame ionization detector. The gas chromatograph was programmed from 100°C (5 minutes) to 250°C at 4°C/min and held for 80 minutes, with an injection and detector temperature of 250°C. A standard fatty acid methyl ester mixture (Supelco 37, Bellefonte, Pennsylvania) was used to identify sample peaks, and the fatty acid composition was reported as area percentage.
Microbiological Analysis
Microbial analysis was conducted for samples (25 g) at 0 and 6 months for total viable count (ISO 4833-1:2013) and Salmonella (ISO 6579). Coagulase test was done for Staphylococcus aureus in 10 g sample.
Response Surface Methodology for Formula Optimization
Response surface methodology (RSM), which includes the design of experiments, selection of levels of variables, fitting mathematical models, and optimization of variables, was applied in this study. Face-centered central composite design for 4 independent variables (ie, antioxidant, emulsifier, storage time, and FO content) at 3 levels was used for the experimental plan, resulting in 30 combined experiments. Responses associated with moisture content, Aw, peroxide value, lipid separation, hardness, and vitamin C content were recorded. For each response, its relationship with another variable was determined by fitting data to the second-order reaction using Equation 1:
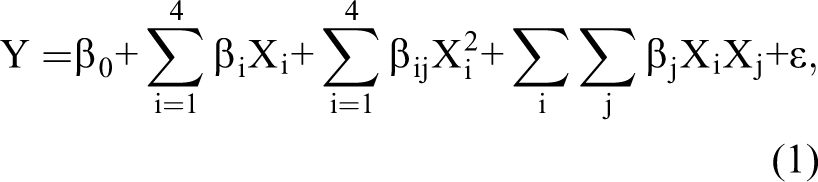
where Y is the predicted response, β0 is a constant, βi is the linear coefficient, βij is the quadratic coefficient, βij is the interaction coefficient of variables i and j, and Xi and Xj are the independent variables.
The statistical adequacy of the model was tested using the Fisher test value (F ratio), the coefficient of determination (R2), and Lack-of-Fit (LOF) test. To visualize the relationship between the response and the experimental levels for each factor and to deduce the optimum conditions, 2D and 3D response surface plots were generated. Supplementary Table S1 shows the respective low and high levels for the factors coded.
Consumer Acceptability
Research protocols using humanss were approved by the Institutional Review Board at the University of Illinois, Urbana–Champaign. The study was conducted in collaboration with the Mansinhbhai Institute of Dairy and Food Technology (MIDFT), Mehsana, India. Participants rated the characteristics of samples presented on a 9-point hedonic scale (ISO 11136:2014) from dislike extremely to like extremely. Further, participants ranked the samples from 1 to 4, where 1 was the top-ranked sample. Eighty-six students (18-24 years) and 25 women (18-45 years) with young children (1-6 years) at home, otherwise healthy (no diarrhoea, cold, or fever) were randomly recruited from MIDFT and within a 20-km radius from MIDFT, respectively. Written consents were obtained from patients. All the documents presented to women were translated into the local language (Gujarati). The participants were trained and instructed in local language by the team. Samples evaluated were LNS without FO (LNS without FO), LNS with FO (10% FO; LNS with FO), LNS A-20 and Sattu Maavu (a commercially available nutrition supplement in India). The responses from students were recorded on a visual analog scale using Qualtrics online (Online Survey Software and Insight Platform) and from women on paper-based forms. For students, the products were evaluated based on taste, color, sweetness, smell, stickiness, texture, appearance, and overall acceptability; whereas for women, the characteristics were taste, color, smell, mouthfeel, appearance, and overall acceptability.
The LNS samples were prepared based on ingredients shown in Table 1. The LNS with FO was prepared by replacing soybean oil with FO at a level of 10% of the final weight. The LNS-A20 was prepared based on the recipe developed by Briggs et al
22
using maltodextrin, soybean oil, confectionary sugar, nonfat milk powder, whey protein concentrate, cream powder, lecithin, pea protein isolate, vitamin/ mineral premix, salt, and antioxidants. Sattu Maavu was obtained locally and contains millet (finger and pearl), corn, wheat, barley, mung bean, groundnut, cashews, sago, and cardamom. Samples were prepared in 1-kg batches at the Food and Dairy Technology Laboratory in MIDFT. Coded samples (2 ± 0.5 g) maintained at 25°C ± 2°C were presented at random to the participants in clear disposable sensory cups under well-lit conditions. Water (∼25°C ± 2°C), and crackers were provided for rinsing between samples. All testing sessions were scheduled between 11:00
Statistical Analyses
Design Expert version 7.0 was used for experimental design, generation of RSM plots, and optimization. The variables were analyzed using the General Linear Model (GLM) procedure, and means were compared using Tukey Studentized Range Test and considered statistically different at P ≤ .05. A full factorial completely randomized design was used to study the effect of independent variables using GLM procedure. The normality assumption was tested by the Shapiro-Wilk test. Data from consumer acceptability test with women were analyzed with Kruskal-Wallis test using npar1way function in SAS and the Dunn test for post hoc mean analyses. All statistical analyses were performed using SAS version 9.4. The Friedman rank-sum test was performed to classify samples based on ratings. 32 The results are presented as mean (SD).
Results
Development of LNS Product
Each 100 g of LNS provided 452 kcal, 13.2 g of protein, 30 g of fat, and 53 g of carbohydrates. Flaxseed oil incorporation at 5% and 10% level reduced the omega-6–omega-3 (LA/ALA) fatty acid ratio to 2.6 and 1.4 compared to 5.8 in control LNS with 0% FO (Table S2). All formulas of the LNS showed a higher proportion of saturated fatty acids and a lower omega-6–omega-3 ratio than the LNS-A20 counterpart (Table S2).
Responses to RSM
Results for the following response variables: Aw, lipid separation, peroxide value, hardness, moisture content, and vitamin C are presented in Table 2. The estimated regression coefficients of the fitted model are given in Table 3. Analysis of variance indicated that responses Y2, Y3, Y4, and Y6 were significant at P ≤ .001, and model Y1 was significant at P ≤ .05. Also, all the coefficients of determination (R2) were >.73, except for moisture. Thus, more than 73% of the total variations provoked by the experimental conditions could be explained by the models fitted to the regression curve.
Results of Face-Centered Central Composite Design for Optimization.a
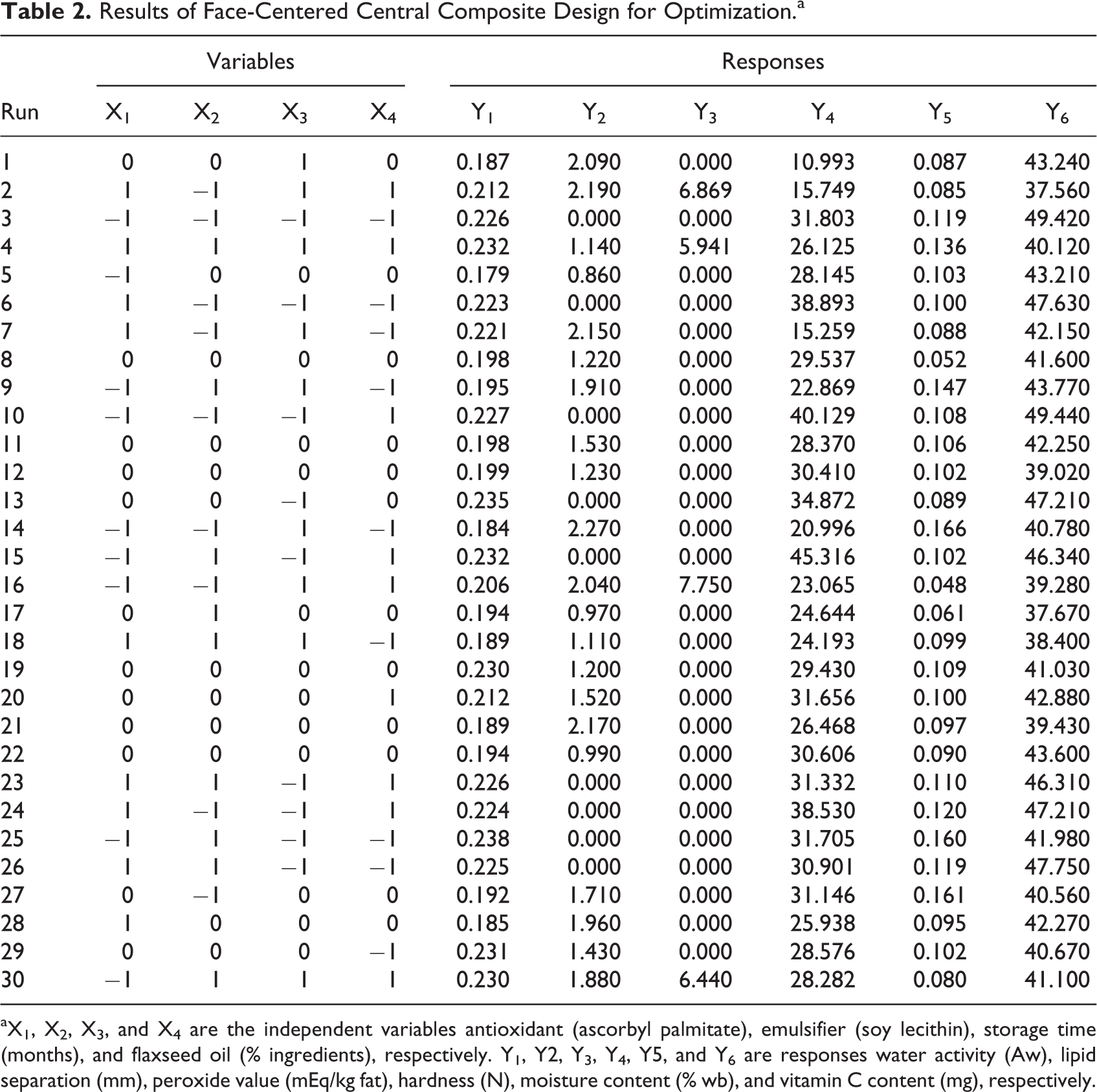
aX1, X2, X3, and X4 are the independent variables antioxidant (ascorbyl palmitate), emulsifier (soy lecithin), storage time (months), and flaxseed oil (% ingredients), respectively. Y1, Y2, Y3, Y4, Y5, and Y6 are responses water activity (Aw), lipid separation (mm), peroxide value (mEq/kg fat), hardness (N), moisture content (% wb), and vitamin C content (mg), respectively.
Estimated Coefficients of the Fitted Quadratic Equation for Different Responses Based on t Statistic.
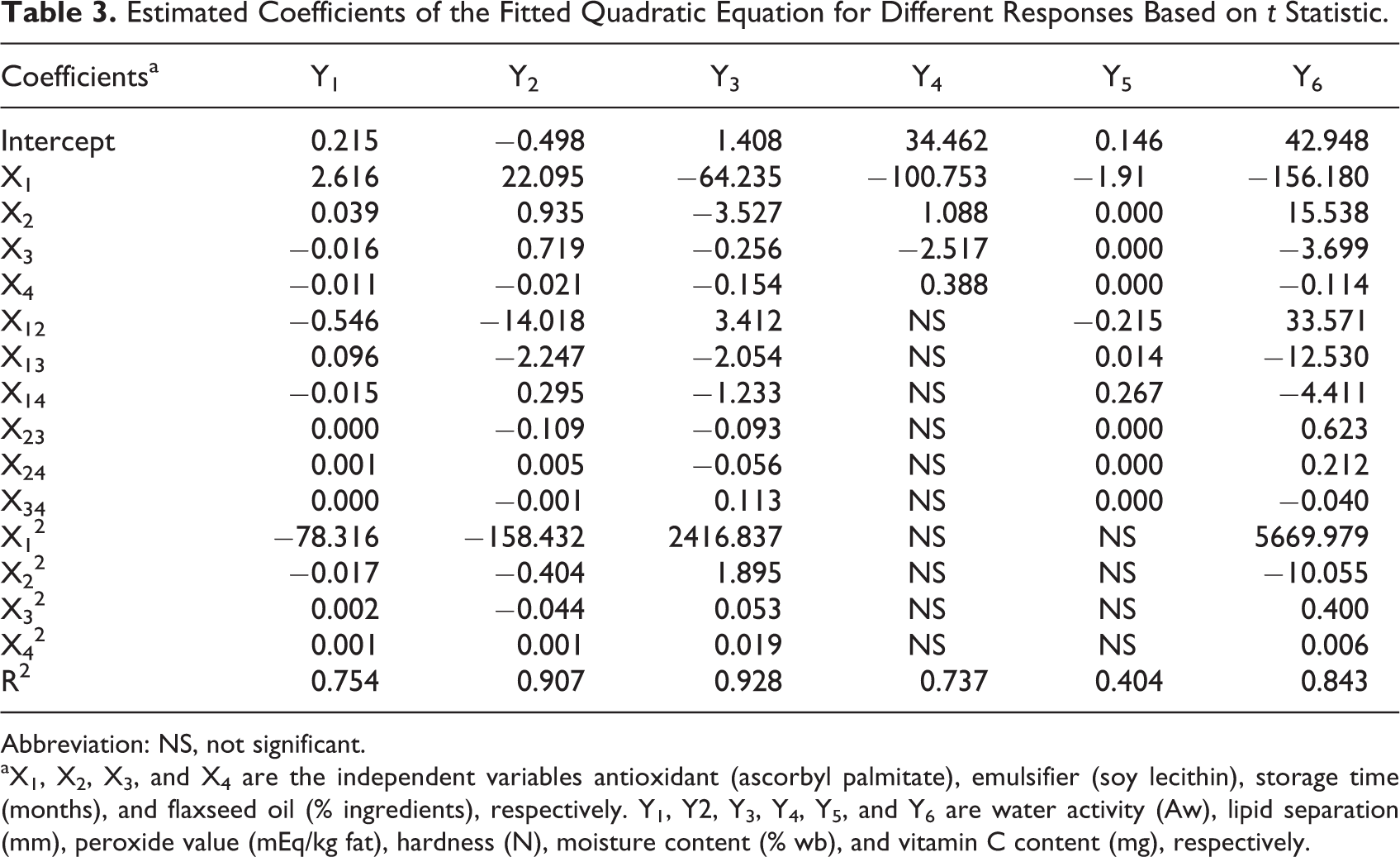
Abbreviation: NS, not significant.
aX1, X2, X3, and X4 are the independent variables antioxidant (ascorbyl palmitate), emulsifier (soy lecithin), storage time (months), and flaxseed oil (% ingredients), respectively. Y1, Y2, Y3, Y4, Y5, and Y6 are water activity (Aw), lipid separation (mm), peroxide value (mEq/kg fat), hardness (N), moisture content (% wb), and vitamin C content (mg), respectively.
Only significant terms in second-order polynomials were used to generate 3-dimensional RSM plots and 2-dimensional line plots. Peroxide values ranged from 0 to 7.75 mEq/kg fat, and no oxidation was determined in samples at 0 and 3 months (Figure 2A). Slight oxidation was detected in the samples containing 5% and 10% FO at 6 months. Storage time and FO content had a significant factor and interaction effect. Lipid separation ranged from 0 to 2.27 mm and was affected by storage time and emulsifier content (Figure 2B). An interaction was observed between lipid separation and these 2 variables. Fresh samples did not show oil separation. The Aw decreased in samples from 0 to 3 months (Figure 3A). No further changes in Aw were observed after 3 months. Moisture content remained the same throughout the study, ranging from 0.0476% to 0.1657% (Figure 3B). Only storage time had an effect on hardness (N), in which the force (ranging from 10.93 to 45.31 N) required to penetrate the samples decreased during storage (Figure 3C). Vitamin C content declined in samples from 0 to 3 months in storage, but it remained the same after 3 months (Figure 3D).
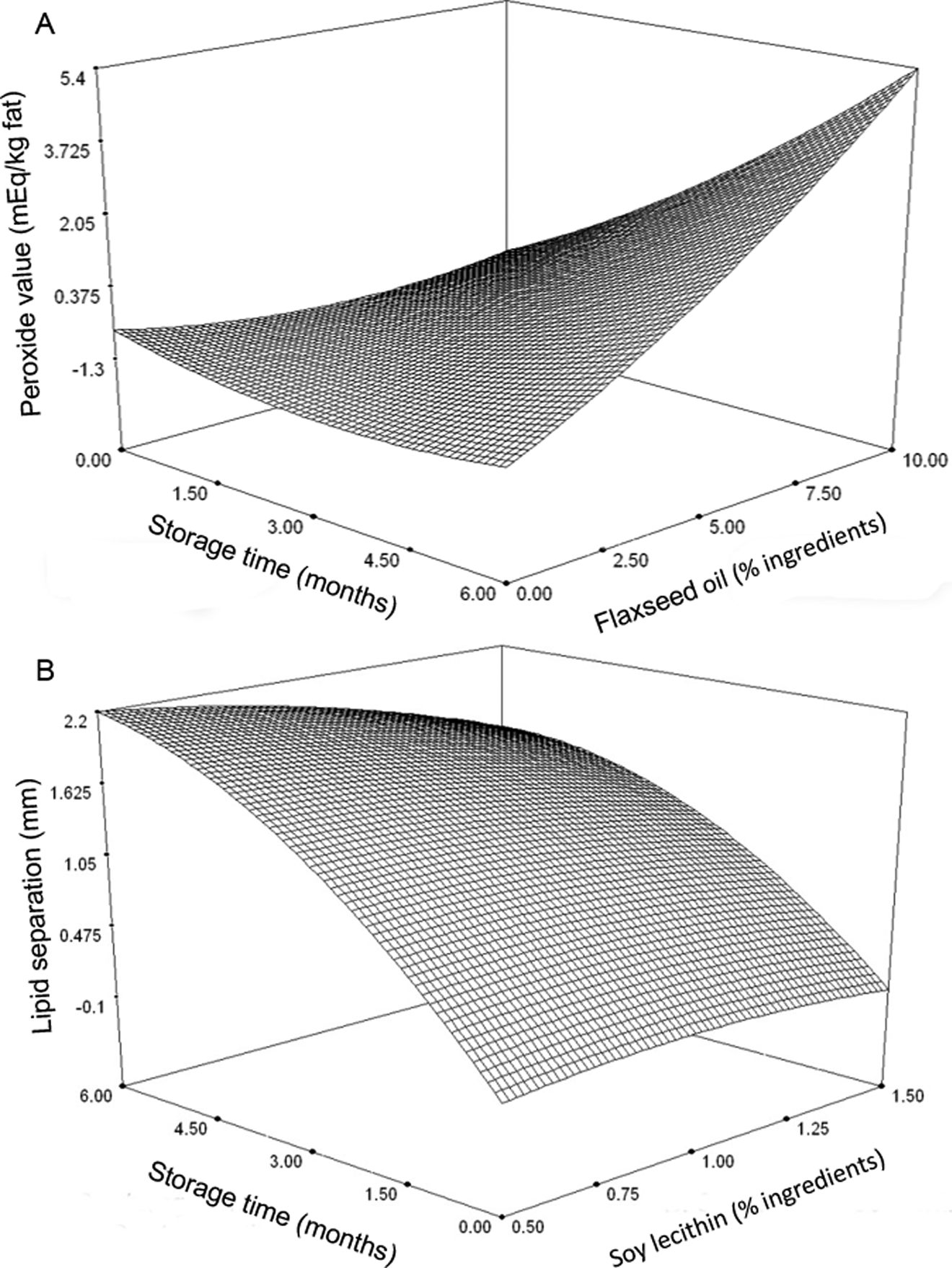
Response surface plots for peroxide value and lipid separation. A, Peroxide value (mEq/kg) of lipid-based nutrition supplement (LNS) showing interaction of storage time (months) and flaxseed oil (% ingredients), in which the following values were kept constant: antioxidant (0.01%) and emulsifier (1%). B, Lipid separation value (mm) of LNS showing interaction of storage time (months) and soy lecithin content (% ingredients), in which the following values were kept constant: antioxidant (0.01%) and flaxseed oil (5%).
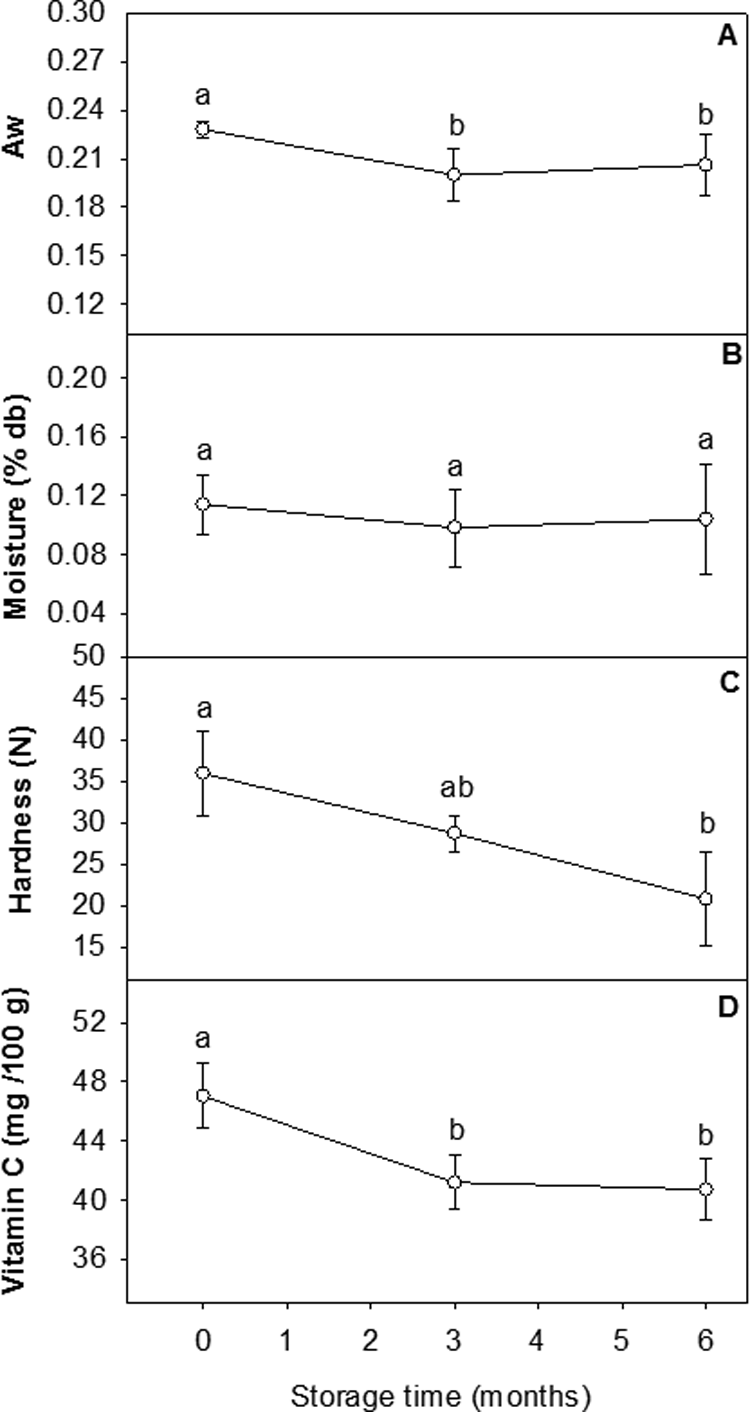
Effect of accelerated storage (40°C) on (A) water activity (Aw), (B) moisture content (% db), (C) hardness (N), and (D) vitamin C (mg/100 g lipid-based nutrition supplement [LNS]) of LNS. Data points with different letters indicate significant differences (P ≤ .05).
Conditions for Optimum Response
For optimization, multiple regression equations were solved to maximize FO content, storage time, and minimize oxidation, using a multiple response method called desirability. The solution to obtain a storability of 3.61 months was a product of mixing ingredients containing 0.02% antioxidant, 1.49% soy lecithin, and 4.87% FO.
Microbiological Testing
At 0 and 6 months, the total viable count of samples was 100 ± 25 CFU/g against the tolerance of 10 000 CFU/g. The LNS samples were negative for Salmonella and S aureus (coagulase test).
Consumer Acceptability
Demographic results showed that a large proportion (72%) of the women was involved in farming and cattle-rearing businesses. Almost all (92%) women provided unfortified homemade complementary foods to their children, which mostly consisted of grains, pulses, fats, and, in some occasions, fruits.
Consumer acceptability ratings from both patient groups and for all LNS samples were above 6.0 (“like slightly”) on the 9-point hedonic scale. Only Saatu Maavu received acceptability ratings below 6.0. Results from acceptability tests can be found in the Supplemental Figures S1A and S1B. Among sensory attributes, clear differences in acceptability ratings were observed for taste, mouthfeel, and appearance. For most sensory attributes, LNS samples were accepted above Sattu Maavu. Patients accepted LNS with FO to a lesser extent in terms of taste and smell than the other LNS samples. Both women and students consistently ranked LNS without FO and LNS-A20 as number 1 and 2 (data not shown) compared to the other products. Sattu Maavu was consistently ranked last (P ≤ .05).
Discussion
An omega-3-fortified LNS product was designed using staple ingredients and optimized using RSM with the objective to improve omega-3/omega-6 PUFA balance and to serve as a take-home ration or complement the meals provided at supplementary feeding centers (Anganwadi) in India. A 20-g serving size of this formula provides 90 kcals and can contribute a significant quota of micronutrients and essential fatty acids to children with MAM. Flaxseed oil, a vegetarian source of omega-3 PUFA, was selected for its high content of ALA, the precursor of long-chain omega-3 EPA and DHA, and for its additional benefits beyond energy, such as anti-inflammatory, antithrombotic, and hypolipidemic effects. 33 Other ingredients such as chickpea flour, milk powder, soybean oil, ghee, and coconut oil were selected due to their commercial availability, cost, and acceptability. The obtained lipid blend is high in saturated and medium chain fatty acids, which increases oxidative stability. Ascorbyl palmitate was the antioxidant of choice based on the recommendations from Nguyen and Bouzambou. 18 Finally, cocoa powder was used to improve flavor and aesthetic appeal.
Enteric infection, a trigger for inflammatory response, and intestinal inflammation are very common among malnourished children in India, resulting in nutrient malabsorption. 13 The omega-6–omega-3 ratio is an indicator for the balanced synthesis of eicosanoids in the body and a lower ratio is recommended for undernourished children not only for normal growth, cell functioning, and immune function but also for an adequate anti-inflammatory response. 8,11,34 In a recent study of children with SAM, a 2-month supplementation with RUTF high in oleic acid and with a 1:1 LA–ALA ratio increased circulating EPA and DHA and reduced arachidonic acid compared to standard RUTF with LA–ALA ratio of 53:1. 9 In another study, a peanut-based RUTF fortified with FO significantly increased plasma EPA and docosapentaenoic acid but not DHA among children having SAM. 10 A 4-week treatment with FO (14 g/d) decreased the levels of proinflammatory cytokines among adult patients. 35 In contrast, a lower dose (ie, 2 or 3.5 g/d) did not have this effect. 36,37
Most companies struggle with the addition of PUFAs into product formulation. Any encapsulation of PUFAs will impact product cost. In addition, plant sources of PUFAs are more suitable for the Indian culture. Although a better ratio is important in inflammation, it cannot be at the expense of product’s flavor and acceptance. The most difficult problems in formulation with PUFAS are oxidation within storage and their recognized flavors. In this study, we found that reducing the FO content in LNS to 5% will balance the essential fatty acid ratio while enhancing its stability during storage and consumer acceptability.
The RSM studies pinpointed adequate levels of FO, antioxidant, and lecithin that resulted in lower oxidation during storage. Studies also helped identify factor effects and their interactions and to minimize experimental runs and time. A similar optimization technique was used to evaluate physicochemical and sensory properties of peanut pastes. 38 The main changes associated with accelerated storage were increased oxidation (ie, peroxide value), decreased Aw, decreased product hardness, increased separation of fat and oil from the matrix, and increased vitamin C degradation.
Higher peroxide values in LNS were associated with increased addition of FO (at a level of 10%). Essential fatty acids in FO are less stable and more susceptible to electrophilic attack and metal-catalyzed autoxidation. 39 Briggs et al found a similar trend in LNS-A20 samples containing a higher proportion of unsaturated oils. 22 Higher peroxide values were reported in FO-containing cookies (>30%) compared to controls stored at 45°C for 28 days. 40 Both storage time and temperature influenced sample oxidation similar to the observations of Riveros et al 41 from studies with peanut pastes and Sumainah et al 42 from studies with peanut-sesame soy blends. Although the peroxide values of LNS were higher after 3 months, these values remained well below the permitted level of 10 mEq/kg oil, a threshold for perception of rancid flavors in oils. 43
The Aw of LNS products is often low, which enhances their ability to last longer in harsh storage conditions. A decrease in Aw from 0 to 3 months could be a result of moisture equilibration in the samples, in which free water migrated from ingredients with high to low Aw, such as from toasted flours and sugar, thus reducing the vapor pressure. 44 The Aw remained below 0.6 after 3 months, which prevented the growth of microorganisms, as confirmed by microbial analysis, and resulted in a product safe for consumption at all time points. Despite the changes in Aw, the moisture content remained unchanged. This could be attributed to the packaging material, which prevented migration of moisture from the external environment. Briggs et al reported similar moisture results in LNS-A20 samples during a 6-month accelerated storage. 22
Most times oil ingredients in LNS tend to separate after several months in storage. Decreased hardness during high-temperature storage could be attributed to melting of fat crystals and oil penetration in the matrix during the quiescent conditions. Similar results were observed by Muego-Gnanasekharan and Resurreccion, 38 in which hardness of peanut paste samples stored at 40°C decreased by 43% after 12 months. These authors also reported a higher separation of fat and oil from the product matrix, an indication of lower product quality and that could result in higher oxidation rates. Lecithin content, storage time, and their interaction had an effect on oil separation. Oil separation increased with time because melted crystals of oils and lecithin rose to the surface after the collapse of the fat crystal network at a high temperature. Furthermore, differences in lipids’ specific gravity resulted in flocculation, then partial or complete coalescence with time. 45,46 UNICEF recommends minimum oil separation 20 ; however, it does not provide any indication as to how to measure separation or quality specifications.
Vitamin C reduction during storage might be attributed to autoxidation at high temperature and in the presence of metal catalysts such as iron and copper. Similar reductions were observed in peanut paste stored at 40°C for 3 months. 47 After 3 months, vitamin C levels remained unchanged probably due to the lower Aw as reported previously. 47 In another study, vitamin C losses (∼20%) were reported in LNS-A20 after 6 months of storage at 38°C. 22 Therefore, overage amounts will be needed to ensure vitamin C is present by the end of the shelf life of products.
Although most times overlooked in emergency relief products for supplementation in low-income settings, consumer acceptability tests are important to evaluate the likelihood of product adoption by end users and to address potential organoleptic issues based on patient’s feedback. Women were chosen instead of infants for this study, as they often taste the food before feeding it to their children, and their acceptability of a product is positively associated with both their children acceptability and the likelihood to use the product for such purposes. 48 Students were selected to determine acceptability among general consumers. Although most products evaluated scored above average on several attributes (ie, values ≥5), the LNS without FO was the most acceptable among women and students. The FO’s taste was the primary reason for low acceptance. Low consumer acceptability was reported by a semitrained sensory panel who evaluated cookies containing FO at a level of 40% or higher as a direct replacement for shortening. 40 Similarly, a lower acceptability in terms of crust color, flavor, mouthfeel, and overall acceptability was reported in bagels with increased FO content. 49 The FO’s PUFAs oxidize readily, which leads to the accumulation of undesirable flavors. 50 The brown color of the LNS was appealing among patients. Chocolate-based products are available in India, and thus participants could relate to the LNS products in terms of color, taste, and flavor. In Bangladesh, a study found that 60% of the women did not accept the taste of peanut-based LNS, 51 which led to reduced acceptance. Therefore, appropriate use of flavors in LNS products is critical for their successful adoption among end users.
Some of the potential limitations of this study may need to be addressed prior to the full adoption of this and similar LNS products. First, the activation energy to predict shelf life was not determined due to lack of at least 1 additional temperature point. 23 Second, the sample size for acceptability tests was small and did not include children or adult men. This reduces the extrapolation of results. Due to the significant ethnic diversity in India, it is important that sensory tests be conducted with similar formulations before production and delivery of products to other states and regions.
Conclusion
An LNS with reduced omega-6–omega-3 PUFA ratio was successfully formulated and optimized for ingredients and stability using RSM; this resulted in recommendations for specific amounts of antioxidant, emulsifier, and FO to obtain an increased storage time with lower oxidation. Flaxseed oil addition was the most significant factor in reducing storage stability. Due to vitamin C reduction during the first 3 months, adjustments in the micronutrient mix are needed (ie, overage). The LNS with higher FO content (10%) had lower acceptability than both LNS without FO and LNS-A20, which may be attributed to its characteristic off-flavor. Based on the importance of essential fatty acids in growth and development and their role in inflammation, the next generation of LNS products should aim at maximizing the amount of omega-3 fatty acids and their stability in storage and ultimate consumption compliance while minimizing their oxidation.
Footnotes
Authors’ Note
Shashank Gaur contributed to conception, design, acquisition, analysis, interpretation, drafted manuscript, and provided final approval of the manuscript. Juan E. Andrade contributed to conception and design. Elizabeth M. Sloffer, Falguni Patra, Nicki J. Engeseth, Dharmendra Shukla, Pramthesh R. Patel, Ankur Ojha, and Juan E. Andrade contributed to acquisition, analysis, and interpretation. Elizabeth M. Sloffer, Falguni Patra, Nicki J. Engeseth, Dharmendra Shukla, Ankur Ojha, and Juan E. Andrade critically revised the manuscript. Elizabeth M. Sloffer, Falguni Patra, Nicki J. Engeseth, Pramthesh R. Patel, Ankur Ojha, and Juan E. Andrade are involved in the final approval of the manuscript.
Acknowledgments
The authors thank Dr Rufino Perez (DCHA/FPP, USAID) for providing A-20 samples and for contributing to the discussion about the formulation of LNS products. The authors also thank Watson Inc for providing LNS micronutrient premix.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by Office of International Programs at the College of ACES, UIUC and the Indian Council for Agricultural Research Fellowship program.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
