Abstract
Background:
To compare the cost-effectiveness of nutrition programs, the anticipated nutritional benefits of each intervention must be expressed using a common metric.
Objective:
We present the methodology for estimating the benefits of vitamin A (VA)-related interventions among women and children in Cameroon.
Methods:
We estimated “reach” (proportion of the population that receives a program), “coverage” (proportion that is deficient and receives a program), and “effective coverage” (proportion that “converts” from inadequate to adequate VA intake following an intervention) using dietary data collected during a national survey in 3 macro-regions of Cameroon (North, South, and Yaoundé/Douala). Effective coverage of programs such as (bio)fortification and micronutrient powders was estimated by adding the dietary VA contributed by the intervention to baseline VA intakes, including the contribution of increased maternal VA intake to infant VA intake through increases in breast milk VA. For interventions that provide VA-related benefits through other pathways (eg, periodic high-dose VA supplements and deworming), we developed alternative methods of estimating “daily VA intake equivalents.”
Results:
Baseline VA intakes and intervention reach varied by geographic macro-region. On average, estimates of program reach were greater than the effective coverage estimates by ∼50%. Effective coverage varied by intervention package and macro-region, ranging from <20 000 (deworming, Yaoundé/Douala) to >400 000 (micronutrient powder or VA supplement, North) children effectively covered per year.
Conclusion:
These estimates of effective coverage, along with macro-region-specific information on the costs of each intervention package, serve as inputs into an economic optimization model to identify the most cost-effective package of VA interventions for each macro-region of Cameroon.
Introduction
Deficiencies in micronutrients, such as vitamin A (VA) and zinc, contribute to excessive morbidity and mortality of young children globally. 1 Although efficacious interventions are available to reduce this burden, 2 national program managers and policy makers lack a clear framework for prioritizing and coordinating micronutrient intervention programs. “Bio-economic” optimization modeling is a tool that can be used to support the development of a cohesive national micronutrient strategy. The approach uses a mathematical model to identify the most cost-effective portfolio of nutrition interventions to achieve a specific set of objectives in a particular setting, based on the estimated effectiveness (benefits) and the costs of each intervention or combination of interventions, and subject to selected constraints (financial or otherwise). The preceding paper in this series (Brown et al) 3 describes the rationale for this approach and summarizes the efforts of our research group in developing the model and applying it to the case of VA deficiency control programs for women of reproductive age and young children in Cameroon.
To compare the cost-effectiveness of nutrition programs, the anticipated nutritional benefits of each intervention must be expressed using a common metric. In this article, we present the methodology for estimating the benefits of VA-related interventions among women and young children in 3 geopolitical “macro-regions” of Cameroon. As described in the accompanying article (Brown et al, this volume), this example was selected based on the availability of data and partner organizations and known spatial heterogeneity of VA status and program costs in Cameroon. This article focuses on the estimation of “effective coverage,” which we defined as the proportion of a target population that has inadequate VA intake but which achieves adequate VA intake as a result of an intervention or package of interventions. However, for comparison, we also include selected results for “reach” (the proportion of individuals exposed to an intervention, regardless of their nutritional status) and “coverage” (the proportion of individuals who are deficient [or who have inadequate intake] and are exposed to an intervention). The effective coverage estimates presented in this article are combined with information on the costs of the respective interventions (as summarized by Kagin et al) 4 to conduct the bio-economic optimization modeling described in the fourth article of this series (Vosti et al, this volume). 5
Methods
Target Groups
The target population groups selected for this analysis were breast-feeding and non-breast-feeding children 6 to 59 months and women 15 to 49 years (women of reproductive age). For each of these population groups, we calculated the
Vitamin A-Related Interventions Considered for Modeling Effective Coverage and Target Population Groups Affected.a
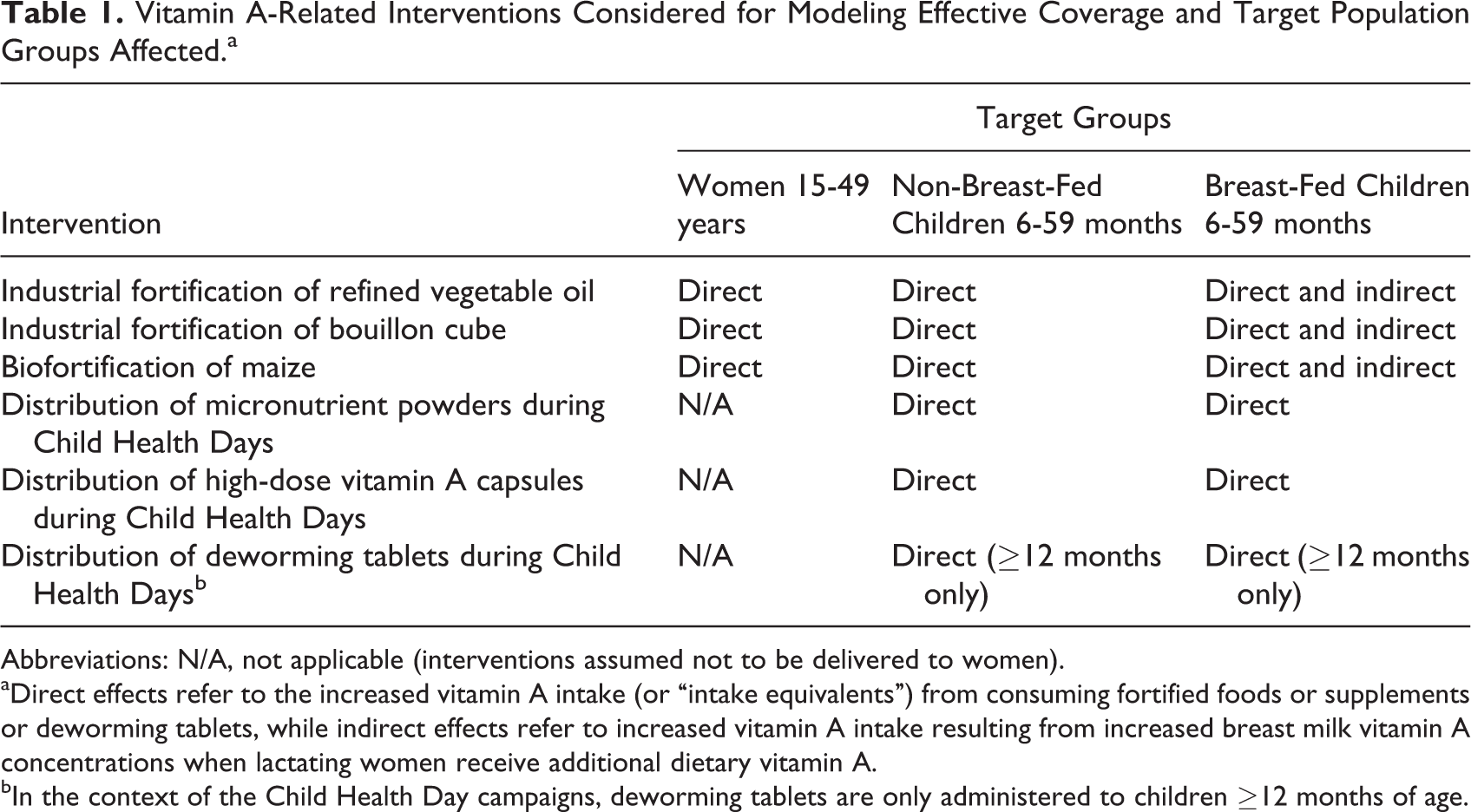
Abbreviations: N/A, not applicable (interventions assumed not to be delivered to women).
aDirect effects refer to the increased vitamin A intake (or “intake equivalents”) from consuming fortified foods or supplements or deworming tablets, while indirect effects refer to increased vitamin A intake resulting from increased breast milk vitamin A concentrations when lactating women receive additional dietary vitamin A.
bIn the context of the Child Health Day campaigns, deworming tablets are only administered to children ≥12 months of age.
Interventions Considered
We selected 6 interventions that are currently being implemented nationally (periodic, high-dose VAS, DW, and refined oil fortification), implemented at a smaller scale (bouillon cube fortification and MNP distribution) or have the potential to be implemented (biofortified maize) in Cameroon (Table 1). We then calculated effective coverage for all possible combinations of interventions (each intervention alone, all combinations of 2, 3, 4, or 5 interventions, and all 6 interventions together) in each macro-region.
Data Sources and Methods of Calculation for Effective Coverage Estimates
Dietary data collection and analysis
The dietary intake data used in these analyses were collected by the 24-hour dietary recall method during a national survey of micronutrient status and dietary intake among children 12 to 59 months and women 15 to 49 years in Cameroon in 2009, 6,7 and results related to biochemical indicators of VA status have been published separately. 8 The survey employed a multistage, cluster sampling design with 3 strata (geographic macro-regions), 30 clusters per stratum, and 10 households per cluster. 7,9 The 24-hour recall interview process was repeated on a nonconsecutive day in a ∼10% subset of households to permit estimation of the distribution of usual VA intakes 10 using the National Cancer Institute method. 11,12 Additional adjustments to the method 13 were employed to account for the stratified cluster sampling design of the 2009 survey, as described elsewhere. 9 Because the data are representative of usual intakes for each stratum of the study (rather than at the individual level), all analyses are presented at the stratum (macro-regional) level (North, South, and Yaoundé/Douala).
The 2009 dietary intake survey included breast-feeding and non-breast-feeding children 12 to 59 months of age. Breast milk intake was not measured in this survey. Thus, to obtain estimates of total VA intake among breast-feeding children, we estimated the VA intake from breast milk using (1) published values for breast milk intake among partially breast-fed children of different age-groups in developing countries 14 and (2) macro-region-specific data on milk VA concentration from the Cameroon 2009 study (Table 2). 8 Because the breast milk VA concentration in this survey was measured using casual milk samples, which tend to have a higher fat content than the full milk samples (ie, when all the milk from 1 breast is collected), we adjusted the fat content (and thus the VA content) to represent the daily average milk fat content. 15,16 Among children whose caregivers reported that the child was still receiving breast milk, we then added the estimated VA intake from breast milk to the calculated dietary intake from all non-breast milk foods (from the 24-hour recalls) to estimate total dietary VA intake from all sources.
Estimates of Breast Milk Intake by Partially Breast-Fed Infants and Vitamin A Intake From Milk, by Survey Stratum.

aThe value of 549 g/d represents the average breast milk intake by partially breast-fed children 12 to 23 months in developing countries. 14
bAssuming 34 g fat/L (average of published values from full milk samples 15,16 ) and specific gravity of milk = 1.03 g/mL.
We defined dietary inadequacy as the proportion of the population with usual VA intakes below the estimated average requirement (EAR) for each age- and sex-specific group (ie, the EAR cut-point method 17 ). We defined the prevalence of inadequate intakes in women of reproductive age as the proportion of women with usual VA intakes <500 µg retinol activity equivalents (RAEs)/d (the EAR in nonpregnant, nonlactating women 19-50 years 18 ). For children, we defined the prevalence of inadequate intakes as the proportion of children with usual VA intakes <210 µg RAE/d (the EAR for 1-3 years). 18 Because consumption of very high levels of preformed VA (ie, retinol) can lead to adverse health effects, we also calculated (but do not report here) the prevalence of excessive VA intakes defined as the proportion of preformed retinol intakes above the tolerable upper intake level for each group. 18
We calculated dietary inadequacy separately for breast-fed and non-breast-fed children by including them as subgroups in the estimation of dietary adequacy using the National Cancer Institute method and weighted the results (prevalence of inadequate intake, by macro-region and breast-feeding status) by the prevalence of breast-feeding by age-group and region (from the 2011 Demographic and Health Survey 19 ) to extrapolate data from the 12 to 59 months age-group to the target 6 to 59 months age-group within each macro-region.
Calculation of reach and coverage
Program reach was calculated as the proportion of children who were exposed to an intervention. For VAS, DW, and MNP, reach was calculated as receipt of VAS within the past 6 months (as a proxy for access to Child Health Day [CHD] campaigns). Thus, the reach of each of these 3 interventions is the same, except that the DW results are adjusted for the fact that children 6 to 11 months do not receive DW tablets. For refined oil, bouillon cube, and maize, reach was defined as consumption of the food in the past 24 hours.
Coverage was calculated in the same way as reach, except that individuals were considered covered if they were both reached by the program and had biochemically defined VA deficiency (inflammation-adjusted plasma retinol-binding protein < 0.83 µmol/L).
20
We did not use inadequate intake to define “deficiency” because the dietary data available (one or two 24-hour recalls) are not representative of an
Calculating effective coverage
Effective coverage was calculated by (1) computing the prevalence of inadequate VA intake at “baseline” (including only VA intake from diet, without any interventions in place), (2) simulating the effects of different interventions and intervention packages on VA intake, and (3) recalculating the prevalence of inadequate VA intake. We expressed effective coverage as the proportion of the population that has inadequate intake at baseline and then “achieves sufficiency” as a result of a given intervention or set of interventions. That is, if the baseline prevalence of inadequacy is 40%, and the prevalence of inadequacy after a given intervention is in place is 30%, the effective coverage is 10% (40%-30%). We then translated the effective coverage (expressed as a percentage) into the number of individuals effectively covered using 2011 population projections from the Lives Saved Tool. 21 Estimates of deaths averted calculated by linking the effective coverage of VA interventions with the Lives Saved Tool will be reported separately.
Calculation of the contribution of VA-related interventions to VA intake
Industrial fortification of refined vegetable oil and bouillon cube
To calculate the direct contribution of industrially fortified foods to daily VA intake, we used data from the national dietary survey to calculate daily intake of “fortifiable food equivalents.” 7 For oil, we included only oils that were industrially refined and excluded oils that were produced and consumed at the household or community level and would not be part of the national fortification program (eg, red palm oil and groundnut oil). This information on fortifiable food intake, in addition to the expected fortification levels of each food, was used to simulate the impact of added VA on total VA intakes by women and young children. Specifically, we calculated the additional VA intake from the fortified food as the amount of food consumed (grams/day) multiplied by the fortification level (the food vehicles considered contain negligible intrinsic VA). This additional VA from fortification was added to the baseline VA intake and the distribution of usual VA intake was then recalculated using the National Cancer Institute method.
Fortification of refined vegetable oil was introduced in Cameroon in 2011, with a target fortification level of 12 mg/kg. However, in a survey of urban areas in 2012, we found that only ∼44% of refined, “fortifiable” oil actually contained detectable VA. 22 Thus, we modeled 2 scenarios for oil fortification: (1) an “optimal” scenario in which all fortifiable oil was fortified at the target level and (2) a scenario more likely to reflect the impact of the current program, in which an average fortification level of 5.28 mg/kg (ie, 44% of 12 mg/kg) was applied. For bouillon cube, we assumed a fortification level of 48 mg/kg, the approximate fortification level of VA-containing bouillon cubes which have been introduced by 1 company in Cameroon on a voluntary basis. Because there are only 3 brands of bouillon cubes that cover most of the market in Cameroon, we assumed that 100% of the product would be fortified if bouillon cube fortification was introduced as a mandatory program. A detailed analysis of the predicted impact of different industrial fortification scenarios on VA intake in this population is presented elsewhere. 9
Biofortification of maize
The additional VA intake provided by biofortification of maize was calculated as the amount of maize consumed multiplied by the target fortification levels (baseline VA intake from the white and yellow maize already being consumed was negligible). We assumed that biofortified maize would contain 15 µg/g β-carotene (raw, dry weight 23,24 ) and applied a 12:1 conversion factor for β-carotene to VA to express the target VA content in µg RAE. 18
We explored 2 scenarios for biofortified maize. First, we modeled the potential impact of a “maximum scenario,” in which biofortified varieties completely replace all other varieties of a given crop. Although this scenario is highly unlikely, it allows for rapid assessment of whether the maximum potential impact of a program would be sufficient to cause the economic optimization model to choose this intervention (ie, if the intervention is not cost-effective when it replaces 100% of the traditional variety of the crop, then this intervention would surely not be selected under a more realistic scenario). To examine the importance of the assumption regarding substitution rates, we also modeled a second scenario with a lower substitution rate (50%). This method assumes that there is no change in patterns of consumption of maize and that the biofortified variety simply replaces the conventional, low-VA varieties. Theoretically, increases in total maize consumption (eg, resulting from a campaign to promote the biofortified product) could also be modeled.
We did not include other biofortified crops, such as yellow cassava or orange sweet potato. However, the methods for estimating the impact of these crops on VA intake would be the same as described here for maize.
Indirect impact of fortification and biofortification on VA intake by breast-feeding children
As mentioned earlier, to estimate effective coverage, we included the “indirect” effects of interventions that increase maternal VA intake (ie, fortification and biofortification) on the VA intake by the breast-feeding child through an increase in breast milk VA concentrations. For breast-feeding children only, we estimated VA intake in the fortification and biofortification scenarios by summing (1) baseline intakes from complementary foods, (2) the estimated baseline VA intake from breast milk, (3) the direct contribution of fortified foods to VA intake (food vehicle intake × fortification level), and (4) the indirect contribution of these interventions through increases in breast milk VA content (increase in maternal VA intake × change in breast milk VA for each 100 µg increase in maternal VA intake × estimated breast milk intake).
To estimate the relationship between increases in maternal VA intake and breast milk VA, we reviewed studies that examined the effect of increased daily maternal intake of preformed VA on breast milk VA (Table 3). We excluded studies of maternal supplementation with high-dose VAS, daily supplements with pro-VA carotenoids or mixed carotenoid and preformed retinol supplements, and studies of supplementation during pregnancy. For each of the 4 studies retained, we calculated the difference in breast milk VA between women in the supplemented and control groups at the final study time point (with the exception of the observational study in Guatemala, in which the value represents breast milk VA of different women in the same communities before and after sugar fortification) and divided this value by the estimated additional daily VA intake by the women. By applying an unweighted average of this value from the 4 studies (the last column in Table 3), we estimated an increase of 18 µg RAE/kg breast milk or 0.018 µg RAE/g (0.019/100 mL, assuming specific gravity = 1.03 g/mL) milk for every additional 100 µg RAE maternal intake (Table 3). Assuming daily intake of 549 g breast milk per day, the child’s additional daily VA from milk would be 0.018 µg/g × 549 g = 10 µg/d for each additional 100 µg RAE of maternal intake. Thus, the additional VA from breast milk would be 10 µg/d × (maternal food intake × fortification level/100) or 0.1 × maternal food intake × fortification level.
Summary of Studies Examining the Effects of Consumption of Small, Daily Doses of Vitamin A, Including Vitamin A-Fortified Foods, During Lactation on Breast Milk Vitamin A Concentrations.
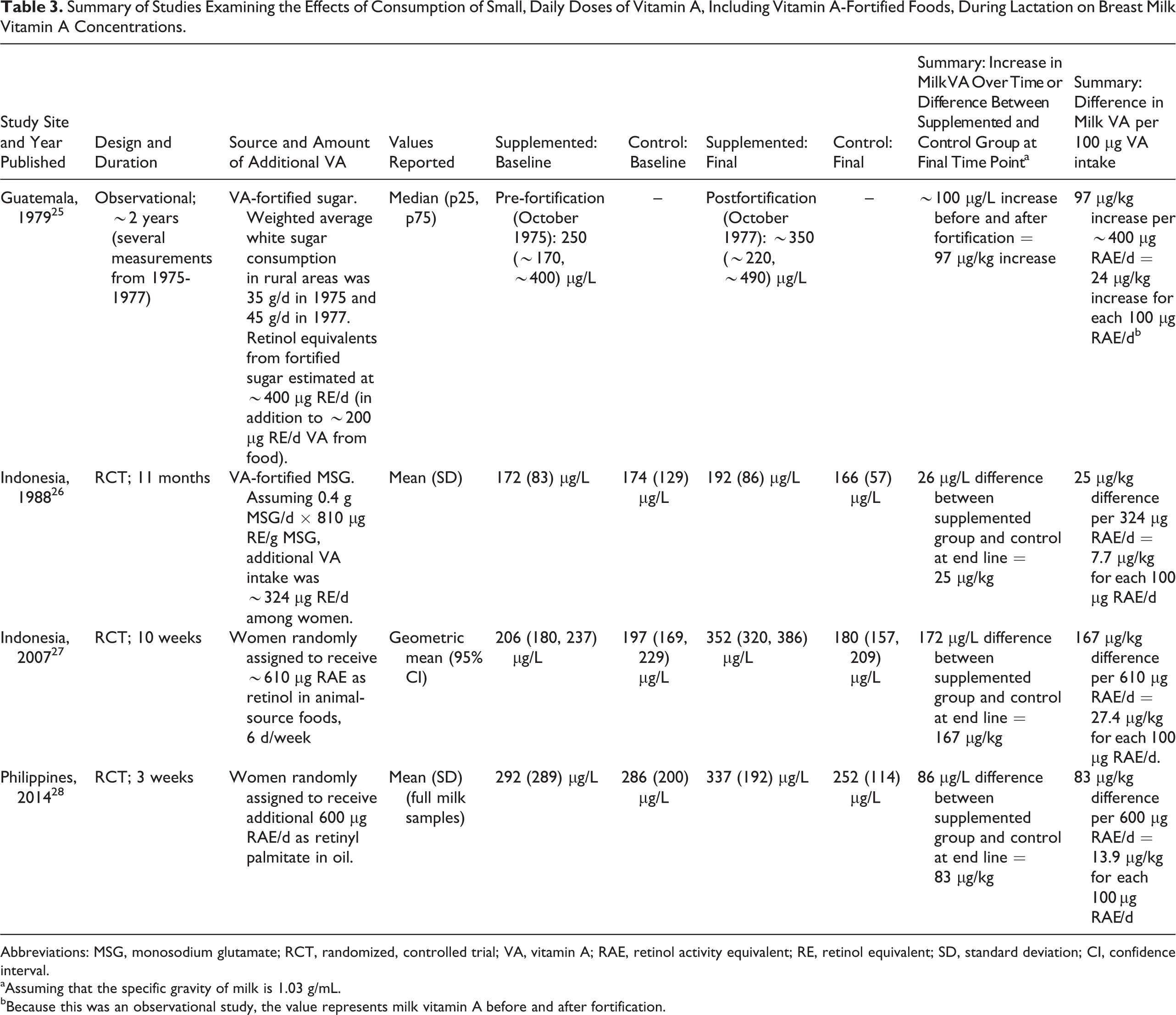
Abbreviations: MSG, monosodium glutamate; RCT, randomized, controlled trial; VA, vitamin A; RAE, retinol activity equivalent; RE, retinol equivalent; SD, standard deviation; CI, confidence interval.
aAssuming that the specific gravity of milk is 1.03 g/mL.
bBecause this was an observational study, the value represents milk vitamin A before and after fortification.
Distribution of MNPs that include VA
Distribution of MNPs has been piloted in several regions of Cameroon. We modeled the effects of scaling up MNP distribution using the CHD program as a delivery platform. Although we chose to focus on MNP, the procedure would be the same for estimating the impact of other daily supplements, such as lipid-based nutrient supplements, perhaps delivered using the same platform.
The proportion of children in each region who are reached by the CHD campaigns was estimated using data on receipt of VAS in the past 6 months from the 2009 survey. 7 We assumed that a child reached by the CHD campaign would receive a 60-day supply of sachets, each containing 400 µg RAE (an intended daily dose of 400 µg RAE/d). We further assumed that the entire 60-day supply would be consumed during the 6 months following the Child Health Day campaign and computed the average contribution to VA intake for 6 months as (60 × 400 µg RAE/d)/180d = 133 µg RAE/d. This additional VA from MNP was added to the daily VA intake for all children who reportedly received a VA capsule in the past 6 months. These assumptions were based on Home Fortification Technical Advisory Group recommendations 29 and are consistent with the MNP formulation currently being piloted in Cameroon; alternative scenarios with different amounts of VA per sachet or different numbers of sachets could be modeled using the same approach (eg, the World Health Organization [WHO] recommends a lower daily dose of 300 µg RAE 30 ). In addition, we chose to model MNP distribution through CHD because data on CHD exposure were available. The use of other distribution platforms, such as health centers or community distributors, could also be modeled if information is available on the likely reach of these strategies.
The WHO recommendation for MNP administration (to control anemia and iron deficiency) is specific to children 6 to 23 months of age, 30 but theoretically MNP could be distributed to children 6 to 59 months, the traditional target age range for VAS distribution in CHD. 29 Thus, we modeled separate scenarios for distribution of MNP to children 6 to 23 months and to children 6 to 59 months. Due to insufficient duplicate 24-hour recalls, we could not estimate dietary inadequacy using the National Cancer Institute method separately for the 2 age-groups. Thus, we used a single value for dietary adequacy with and without MNP (thus assuming that the risk of dietary inadequacy does not differ by age within this range) and applied the population estimates for the 2 age-groups to create the scenarios representing dietary adequacy if only children 6 to 23 months receive MNP via CHD, and dietary adequacy if all children 6 to 59 months receive MNP via CHD.
Periodic, high-dose VA capsules
To compare the effects of periodic high-dose VA capsules with interventions to increase daily VA intake, such as oil and bouillon cube fortification, we used data on reported VAS receipt from the 2009 Cameroon national survey. We then derived “daily intake equivalents” for VAS (ie, the amount of daily VA intake that would be equivalent to receipt of a single, high-dose VA capsule) using a simple kinetic model of body VA metabolism. 31 Briefly, the daily intake equivalent for VAS was estimated as the amount of daily VA intake that would be necessary to maintain adequate liver VA stores (≥20 µg/g 32 ) for the same number of days as a single high-dose VAS during the 6-month interval between CHD. Using this approach, we developed a “best estimate” for the average daily intake equivalent of VAS of 167 µg RAE/d (which is equivalent to exactly half of the total possible intake equivalent calculated as 60 mg/180 d = 333 µg RAE/d), as described below. Thus, to estimate the effect of high-dose VAS receipt on dietary adequacy (and effective coverage), 167 µg RAE was added to the daily intake by children who reportedly received VAS during the past 6 months.
The kinetic model of liver VA stores used in this exercise has been described by Allen and Haskell. 31 The model applies assumptions about liver weight (assumed to be a fixed percentage of body weight), proportion of total body VA stored in the liver, starting liver VA concentration, daily VA intake from all sources, and daily rate of VA catabolism (as a fixed proportion of total VA stores each day 33 ) to estimate the total body VA stores and liver VA concentration over time. For this modeling exercise, we used a starting age of 12.0 months and assumed that the VAS was delivered as a single 60-mg dose, given at 12.0 months. Daily VA loss from the total body VA pool was estimated to be 2.2% per day, the value measured among children 1 to 2 years of age in Peru. 33 Retention of VA from all sources (VAS and dietary) was assumed to be 50% 34 (although it should be noted that this is a conservative estimate, since higher values have been reported 35,36 ). To estimate the liver VA concentration, we assumed that 90% of body VA was stored in the liver and that liver weight was 267 g at 12 months and 306 g at 18 months (3% of 8.9 kg and 10.2 kg, respectively).
To explore the effects of baseline liver VA concentration, we constructed separate models using different assumed baseline liver VA concentrations (at 12 months of age) of 5, 10, 20, 30, and 40 µg/g (Table 4). For each value of baseline liver stores, the modeling was conducted for each of 2 levels of baseline daily intake from all sources: 120 µg/d (the mean VA intake among non-breast-feeding children 12 to 59 months in the North macro-region of Cameroon, where deficiency is common) and 290 µg/d (the mean VA intake among non-breast-feeding children 12 to 59 months in the Southern macro-region, in which VA intakes were greatest). For reference, using this model, daily intake of 120, 200, or 290 µg RAE/d from 6 to 12 months would yield liver stores of approximately 9.1, 15.1, and 21.9 µg/g at 12 months (depending on initial stores).
Summary of Modeling Results Used to Estimate Daily Intake Equivalents for a High-Dose VA Supplement Using a Kinetic Model of Liver VA Stores.a ,b
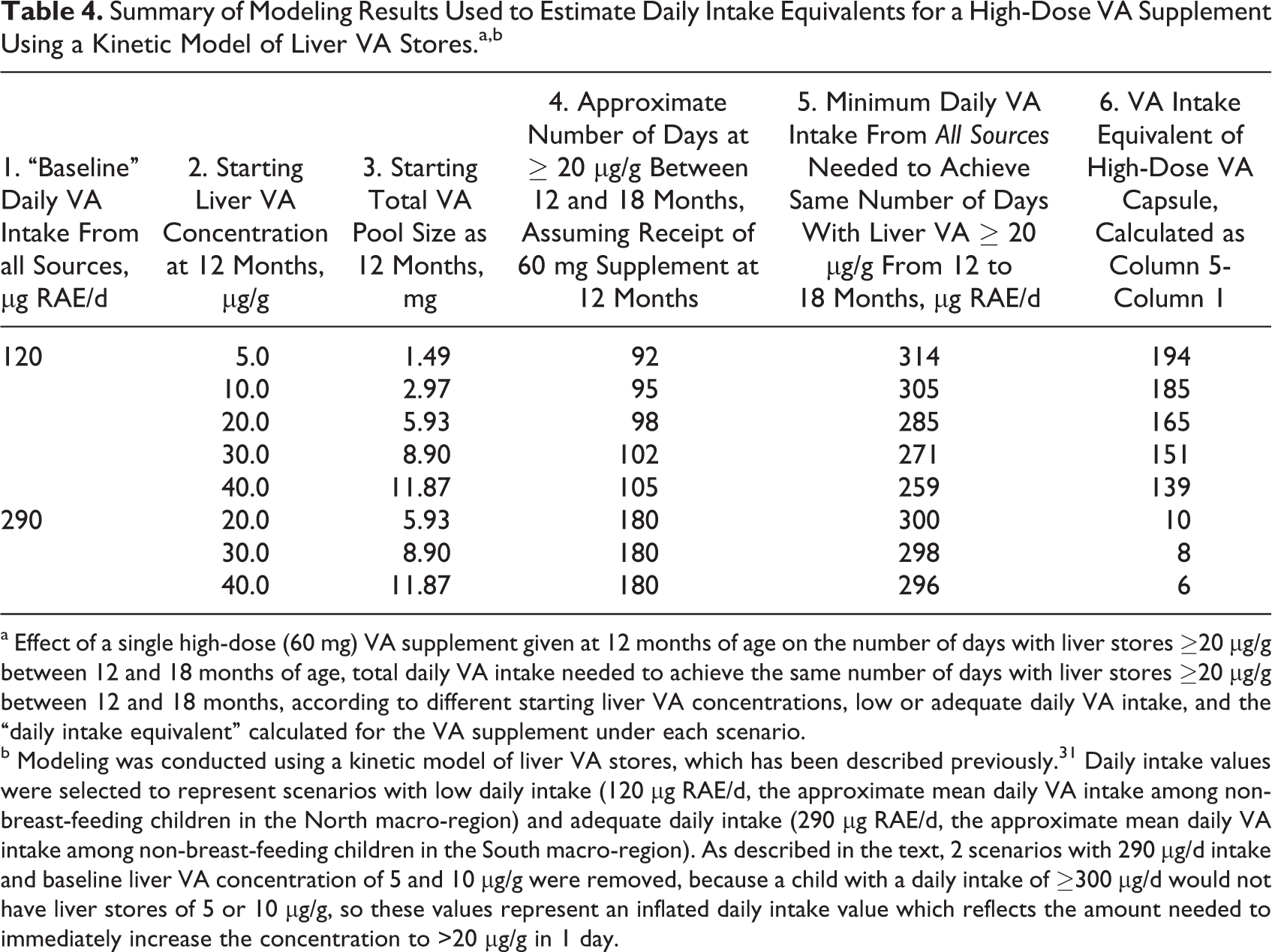
a Effect of a single high-dose (60 mg) VA supplement given at 12 months of age on the number of days with liver stores ≥20 µg/g between 12 and 18 months of age, total daily VA intake needed to achieve the same number of days with liver stores ≥20 µg/g between 12 and 18 months, according to different starting liver VA concentrations, low or adequate daily VA intake, and the “daily intake equivalent” calculated for the VA supplement under each scenario.
b Modeling was conducted using a kinetic model of liver VA stores, which has been described previously. 31 Daily intake values were selected to represent scenarios with low daily intake (120 µg RAE/d, the approximate mean daily VA intake among non-breast-feeding children in the North macro-region) and adequate daily intake (290 µg RAE/d, the approximate mean daily VA intake among non-breast-feeding children in the South macro-region). As described in the text, 2 scenarios with 290 µg/d intake and baseline liver VA concentration of 5 and 10 µg/g were removed, because a child with a daily intake of ≥300 µg/d would not have liver stores of 5 or 10 µg/g, so these values represent an inflated daily intake value which reflects the amount needed to immediately increase the concentration to >20 µg/g in 1 day.
For each scenario, we first estimated the number of days during the 180-day supplementation period for which the index child would have liver stores >20 µg/g (Table 4 and Figure 1A). Second, we calculated the total daily VA intake that would be necessary to achieve the same number of days with liver stores >20 µg/g, assuming no high-dose VAS (Table 4 and Figure 1B). Finally, we calculated the additional daily intake equivalent for VAS as the total daily intake estimated in step 2 above minus the assumed baseline daily intake. In these scenarios, the
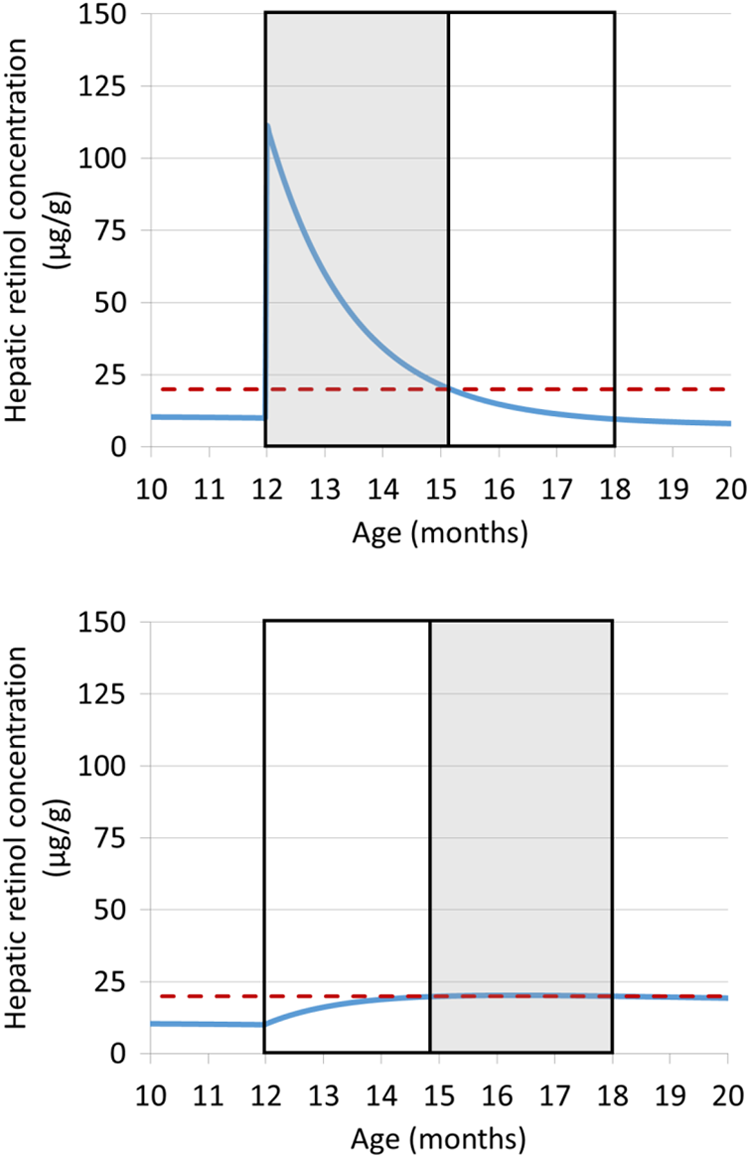
Change in estimated hepatic retinol concentration from 12 to 18 months estimated using a model of liver VA stores under 2 scenarios: (A) assuming baseline liver stores of 10 µg/g, daily intake of 120 µg RAE/d and receipt of a high-dose (60 mg) VA supplement at 12 months (liver stores remain above 20 µg/g for ∼95 days postdose) and (B) assuming baseline liver stores of 10 µg/g and daily intake of 305 µg/d selected to achieve ∼95 days with adequate liver VA stores. The horizontal dashed line represents the cutoff for inadequate liver stores (20 µg/g). The outer black box indicates the 6-month reference time period from 12 to 18 months, and the shaded area represents the portion of this time period during which liver stores are adequate (≥20 µg/g). VA indicates vitamin A; RAE, retinol activity equivalent.
Under a scenario of low baseline daily VA intake (120 µg RAE/d), the capsule dose would yield liver stores ≥20 µg/g for 92 to 105 days, depending on the baseline liver stores. The total daily VA intake required to maintain liver stores >20 µg/g for 92 to 105 days was 259 to 314 µg RAE/d. To calculate the daily intake equivalent for VAS, we subtracted the prior daily intake of 120 µg RAE/d from the new estimate of total daily intake, yielding an estimate of 139 to 194 µg RAE/d (depending on baseline liver VA).
Under a scenario of moderate/adequate daily intake (290 µg RAE/d, the mean in the South macro-region), daily intake was already very close to the minimum amount needed to maintain liver stores at 20 µg/g, so, with the VA capsule, children would have liver stores >20 µg/g for the entire 180-day period. At very low baseline liver stores (5 and 10 µg/g), children would need a very high daily VA intake to immediately increase their liver stores to 20 µg/g and maintain liver stores above 20 µg/g for 180 days. Thus, these cases have been excluded from the analysis, since liver stores would unlikely be this low if the daily intake was adequate, and the results for these 2 scenarios reflect the amount of VA intake needed to boost liver stores from 5 or 10 to 20 µg/g in a single day (>3000 µg), which is more than the theoretical maximum contribution of the 60-mg VA capsule to daily intake (60 mg/180 d = 333 µg RAE/d). However, if the baseline liver stores are 20 µg/g and above, only 296 to 300 µg RAE/d would be needed to maintain adequate liver stores, so the additional contribution of the VA capsule (above the prior intake of 290 µg/d) would be just 6 to 10 µg RAE/d.
In summary, the daily intake equivalents estimated by this method ranged from ∼195 µg RAE/d, among children with low liver stores and low intake, to ∼5 µg RAE/d among children with greater liver stores and daily intake. This is consistent with the concept that children will benefit more from the high-dose VAS when they have low VA intake and status. For these analyses, we chose to adopt the average daily intake equivalent from the models of low baseline VA intake (167 µg/d), with the following rationale: For children with low VA intake, the higher estimate of daily intake equivalents is appropriate because these children are likely to also have low liver stores and thus to receive a greater benefit from VAS. In contrast, among children with higher VA intake (eg, those with intake above the EAR of 210 µg RAE/d), the additional benefit of VAS may be much smaller. However, the aim of this modeling exercise is to estimate the proportion of children with VA intakes below the EAR. Thus, these children have already been identified as having adequate intake, so the value used for daily intake equivalents will not change their classification.
Modeling of the above assumptions using WinSAAM software (version 3.0.8, www.winsaam.org) yielded similar estimates of the daily intake equivalent (Janet Novotny, person communication). The primary purpose of this document is to describe the methodology and illustrate the variation in predicted nutritional impact by macro-region, intervention, and definition of impact. Further refinements using updated assumptions will be necessary, particularly as new data become available. For example, in the case of VA deficiency, less than 90% of the total body VA would be expected to remain in the liver, since a larger proportion of the total body VA would be mobilized for use in the body. As a sensitivity analysis, we examined the possibility of using separate values for daily intake equivalents for each macro-region, based on the mean estimated daily intakes by non-breast-feeding children in each macro-region. This approach gave similar effective coverage values for VAS in the North and in Yaoundé/Douala (compared to using a single value of 167 µg/d for all macro-regions). However, because using the mean intake in the South (290 µg/d) gave a daily intake equivalent of ∼10 µg/d (as shown in the lower half of Table 4), using macro-region-specific estimates underestimated effective coverage values in the South. We also conducted tests assuming 75% retention of VA from all sources 35,36 and found that this change would lower the daily intake equivalent for VAS (∼73 and 103 µg RAE/d, assuming starting liver retinol concentrations of 20 and 10 µg/g, respectively). We retained the higher estimate of daily intake equivalents for the reasons outlined above. However, the effective coverage results may overestimate the contribution of high-dose supplements to VA status if daily VA intake and/or retention of dietary VA are higher than the values used to calculate the daily intake equivalent of 167 µg RAE/d. Finally, we limited the simulations to children 1 to 2 years of age, because data on VA disposal rate were available for children in this age range, and repeating the calculations using an 18- to 24-month age range gave similar results.
Deworming
In the context of CHDs designed to control VAD, we considered the role of DW in the enhancement of VA absorption, although it is recognized that DW programs have other benefits not considered in the present analysis. We estimated the daily VA intake equivalents for DW as the amount of additional VA that would be absorbed by children who were infected with soil-transmitted helminths (STHs) and treated with a DW tablet.
The proportion of children who were infected and treated was estimated by multiplying the estimated prevalence of STH infection with the proportion of children who received VAS in each macro-region. This approach relies on the use of the proportion of children who received high-dose VAS (from the 2009 survey, as described above) as a proxy for receipt of a DW tablet and assumes that STH infection and receipt of DW tablet are independent. Because information on STH infection was not available from the 2009 survey, we reviewed the literature to obtain estimates of STH prevalence in each of these macro-regions.
37
–40
Although there was large regional heterogeneity in the reported prevalence of STH, the available studies included different types of parasites, and data for many areas were missing, outdated, included only 1 or 2 types of STH (
To estimate the daily VA intake equivalent for DW, that is, the additional VA that would be available to the body each day, following the treatment of STH infection, we reviewed studies that measured VA absorption in infected and uninfected individuals. We identified 1 study that measured VA absorption using radiolabeled VA among children with diarrhea,
42
2 studies of individuals with ascariasis in which absorption was measured as the rise in plasma retinol levels 3 to 5 hours after an oral dose of VA (although this method may be affected by alterations in VA metabolism during inflammation),
43,44
and 1 study of children infected with
In summary, we randomly selected a 25% subsample of children in the 2009 survey database to be designated as “infected.” Among the infected children who received VAS (and thus, we assume, DW tablets), we randomly selected a 70% subset to remain uninfected over the 6-month period between CHD. Among this subset, we applied an estimated 39% increase in VA intake (ie, by dividing total VA intake by 0.72) to reflect the estimated increase in VA absorption subsequent to treatment of STH infection.
Calculations of effective coverage for combinations of interventions
When calculated as described above, the effects on effective coverage of combined interventions are not necessarily additive. For example, if a given individual achieves adequate VA intake by consuming either fortified refined oil or fortified bouillon cube, then the marginal contribution of a second food to effective coverage would be 0, given that the other fortified food has already allowed this individual to achieve sufficient dietary VA intake. Thus, to avoid “double counting,” we estimated the effects of combinations of interventions using the individual intake values and then recalculated the prevalence of dietary inadequacy under each scenario (all possible combinations of 2, 3, or more interventions in each macro-region).
Results
At baseline, the prevalence of inadequate VA intakes among children 6 to 59 months was 33% in the South, 71% in the North, and 48% in Yaoundé/Douala. Inadequate VA intakes were less common among breast-feeding children (1% South, 21% North, and 1% Yaoundé/Douala) compared with non-breast-feeding children (41% South, 89% North, and 56% Yaoundé/Douala) because of the estimated contribution of milk VA to total VA intake. Among women of reproductive age, inadequate intakes were present in 15% of women in the South, 100% in the North, and 58% in Yaoundé/Douala. Applying the population estimates to the measures of dietary intake adequacy revealed that slightly over 1.5 million children are predicted to have inadequate intake in the absence of any VA intervention programs.
The proportion of women and children reached by various intervention programs, and the estimated amount of additional VA delivered to those women and children reached by the program, varied by macro-region and by intervention (Tables 5 and 6). Fortification and biofortification programs differed in terms of the proportion of women or children reached by the intervention and the amount of additional VA intake provided by the intervention, reflecting differences in consumption patterns of the foods examined. Interventions delivered via CHD would presumably each reach the same number of children, but the amount of VA delivered would differ.
Estimated Direct Effects of Vitamin A-Related Interventions Among Children 6 to 59 months Expressed as the Proportion of Children Reached and the Amount of Vitamin A Delivered by Each Intervention Among Those Reached.a
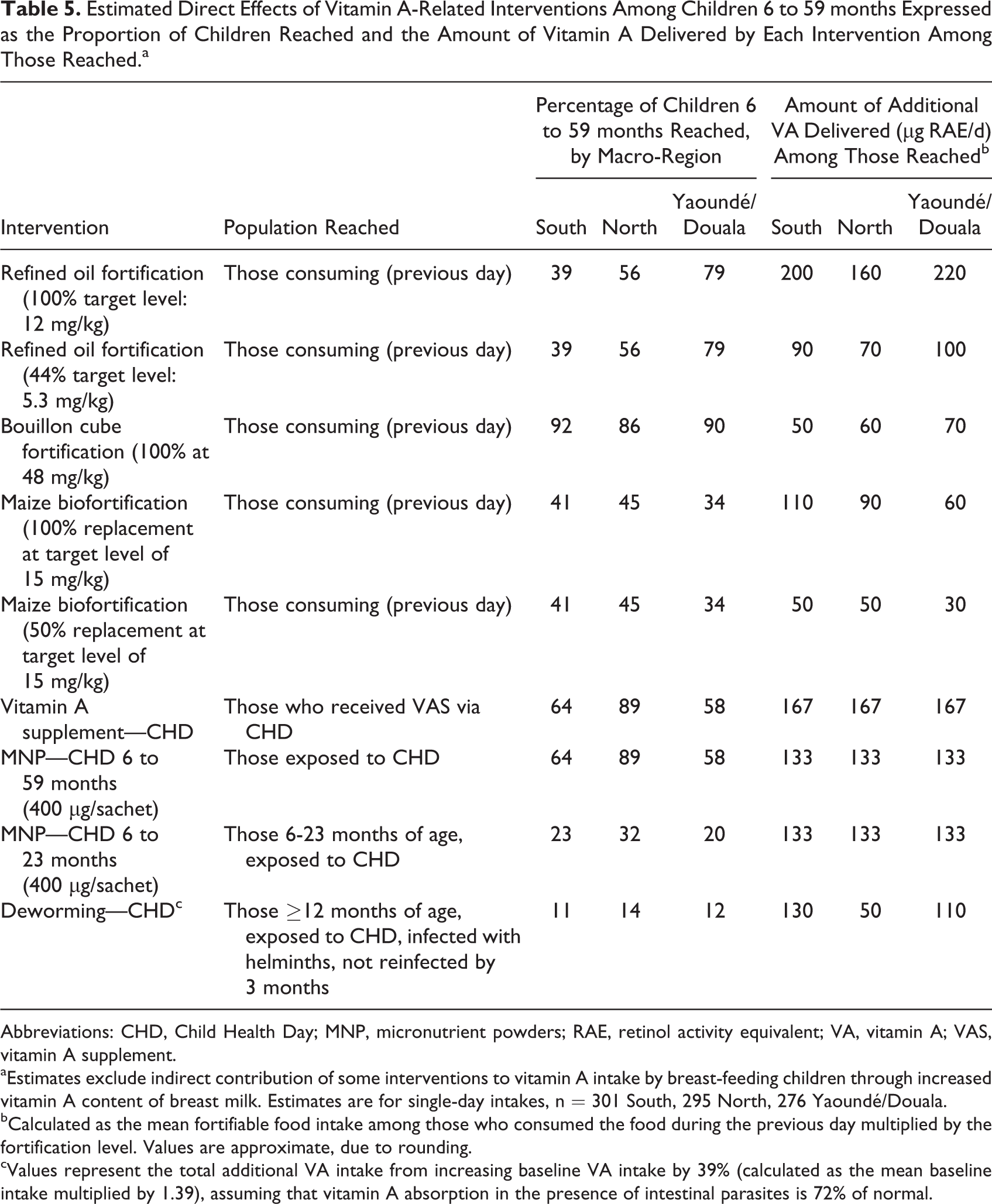
Abbreviations: CHD, Child Health Day; MNP, micronutrient powders; RAE, retinol activity equivalent; VA, vitamin A; VAS, vitamin A supplement.
aEstimates exclude indirect contribution of some interventions to vitamin A intake by breast-feeding children through increased vitamin A content of breast milk. Estimates are for single-day intakes, n = 301 South, 295 North, 276 Yaoundé/Douala.
bCalculated as the mean fortifiable food intake among those who consumed the food during the previous day multiplied by the fortification level. Values are approximate, due to rounding.
cValues represent the total additional VA intake from increasing baseline VA intake by 39% (calculated as the mean baseline intake multiplied by 1.39), assuming that vitamin A absorption in the presence of intestinal parasites is 72% of normal.
Estimated Direct Effects of Fortification and Biofortification Interventions Among Women.a
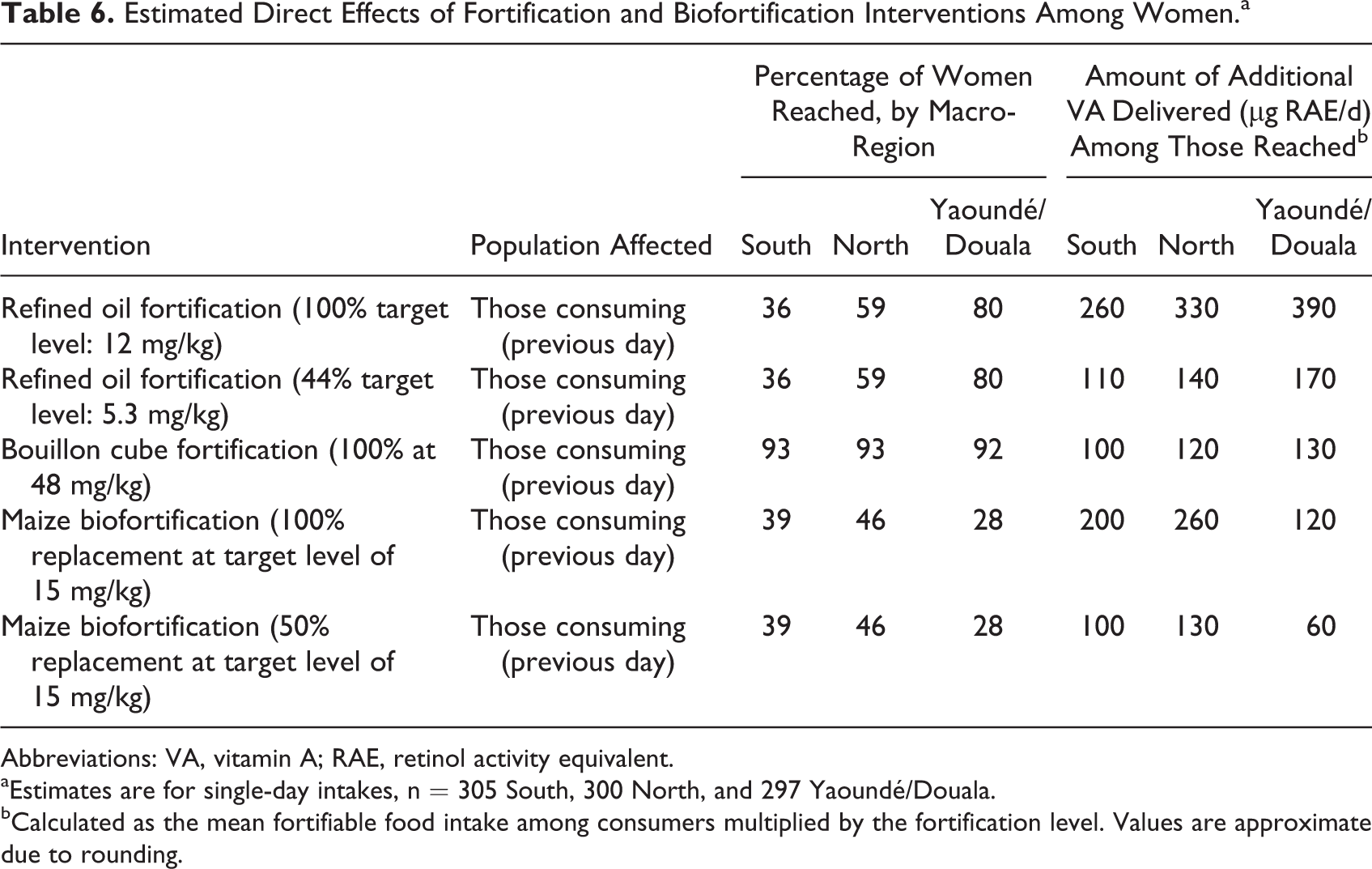
Abbreviations: VA, vitamin A; RAE, retinol activity equivalent.
aEstimates are for single-day intakes, n = 305 South, 300 North, and 297 Yaoundé/Douala.
bCalculated as the mean fortifiable food intake among consumers multiplied by the fortification level. Values are approximate due to rounding.
Comparison of Estimates of Reach, Coverage, and Effective Coverage
To illustrate the differences in estimates of reach, coverage, and effective coverage, we present macro-regional results for 6 interventions (Figure 2). For many programs, the program reach (ie, the proportion of all children exposed to the program) is quite high. However, coverage (defined as the proportion of children who are deficient and are exposed to each program) is much lower. Finally, considering whether the intervention provides sufficient additional VA to allow individuals with inadequate intake to achieve adequate VA intake yields an even lower estimate of effective coverage. Because effective coverage is arguably the most likely to predict an intervention’s public health impact (reduction in morbidity and mortality), we present effective coverage in the following results.
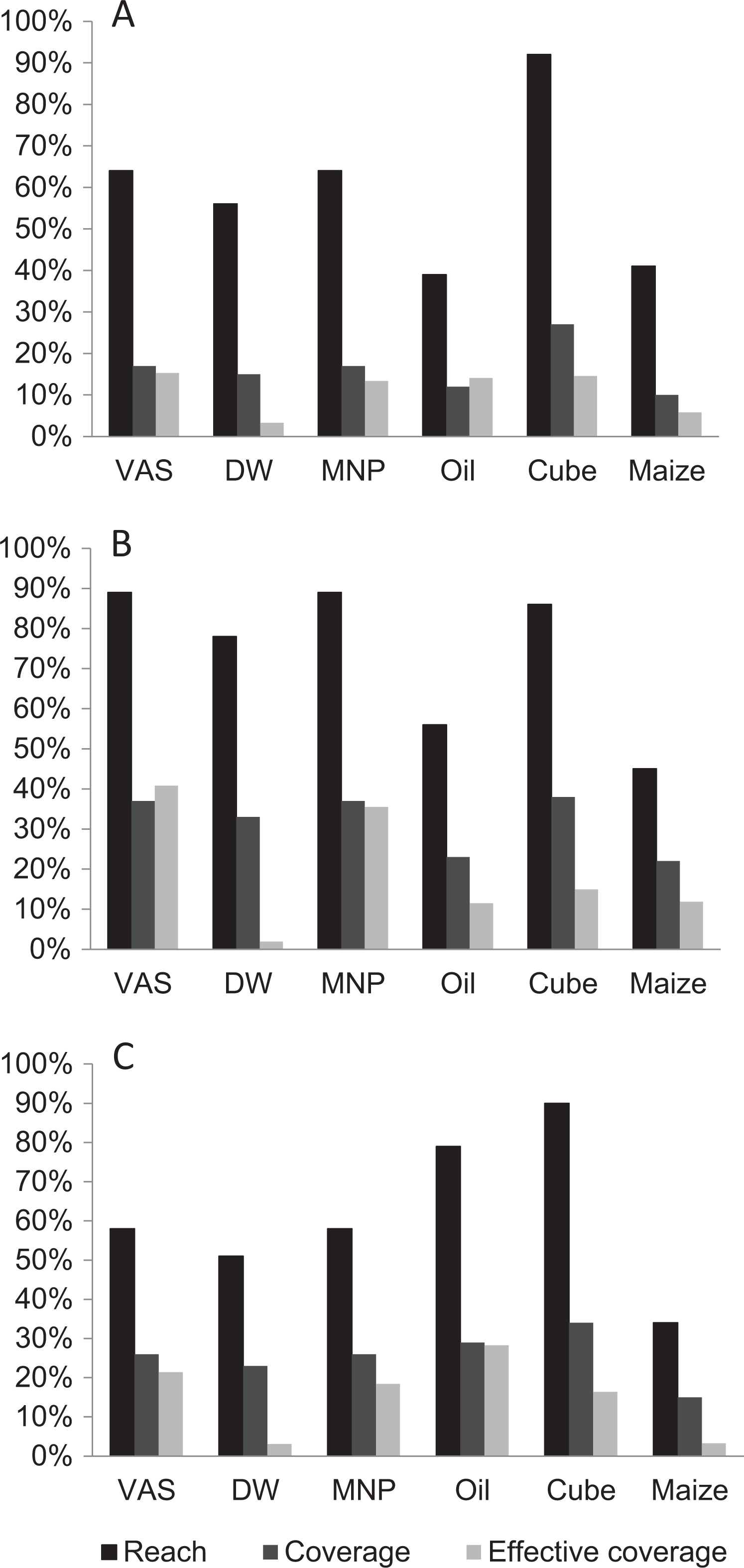
Comparison of program reach (percentage of children who receive the program), coverage (percentage of children who are deficient and receive the program), and effective coverage (percentage of children who have inadequate intake and achieve dietary adequacy as a result of the program) among children 6 to 59 months for selected vitamin A-related interventions in Cameroon, in the South (A), North (B), and Yaoundé/Douala (C). Effective coverage estimates assume that 44% of refined oil is fortified and an adoption rate of 100% for biofortified maize. DW indicates deworming; MNP, micronutrient powder; VAS, vitamin A supplement.
Children
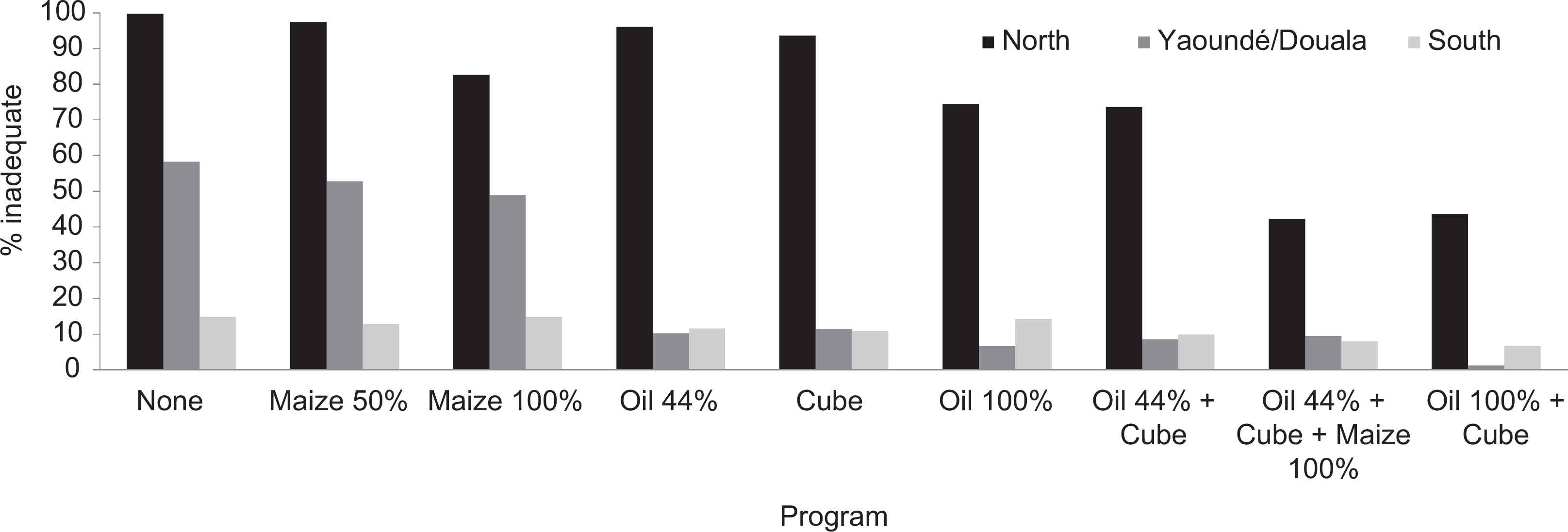
Considering the effects of each intervention alone, programs such as DW would have the smallest effect on the prevalence of inadequate intakes, while VAS, MNP, and oil fortified at 100% of the target level would have the largest effect (Figure 3). Spatial variation in effective coverage was apparent for some programs; for example, oil fortified at 44% of the target level would resolve over half of the cases of inadequate intake in Yaoundé/Douala, but only ∼15% of cases of inadequate intake in the North macro-region. Conversely, distribution of MNP to children 6 to 59 months of age would decrease the prevalence of inadequate intakes in the North macro-region by approximately half, but the decrease in inadequate intakes in the South and Yaoundé/Douala was smaller. We also examined the effects of selected combinations of VA programs to explore the extent to which implementing complementary programs would achieve further decreases in the prevalence of inadequate intake (Figure 3B). Under a scenario with VAS, DW, and oil fortification at 44% of target (the current portfolio in Cameroon), the prevalence of inadequate intakes among children 6 to 59 months was 8% in the South, 16% in the North, and 8% in Yaoundé/Douala compared with 33%, 71%, and 48%, respectively, without any programs in place (Figure 3B).
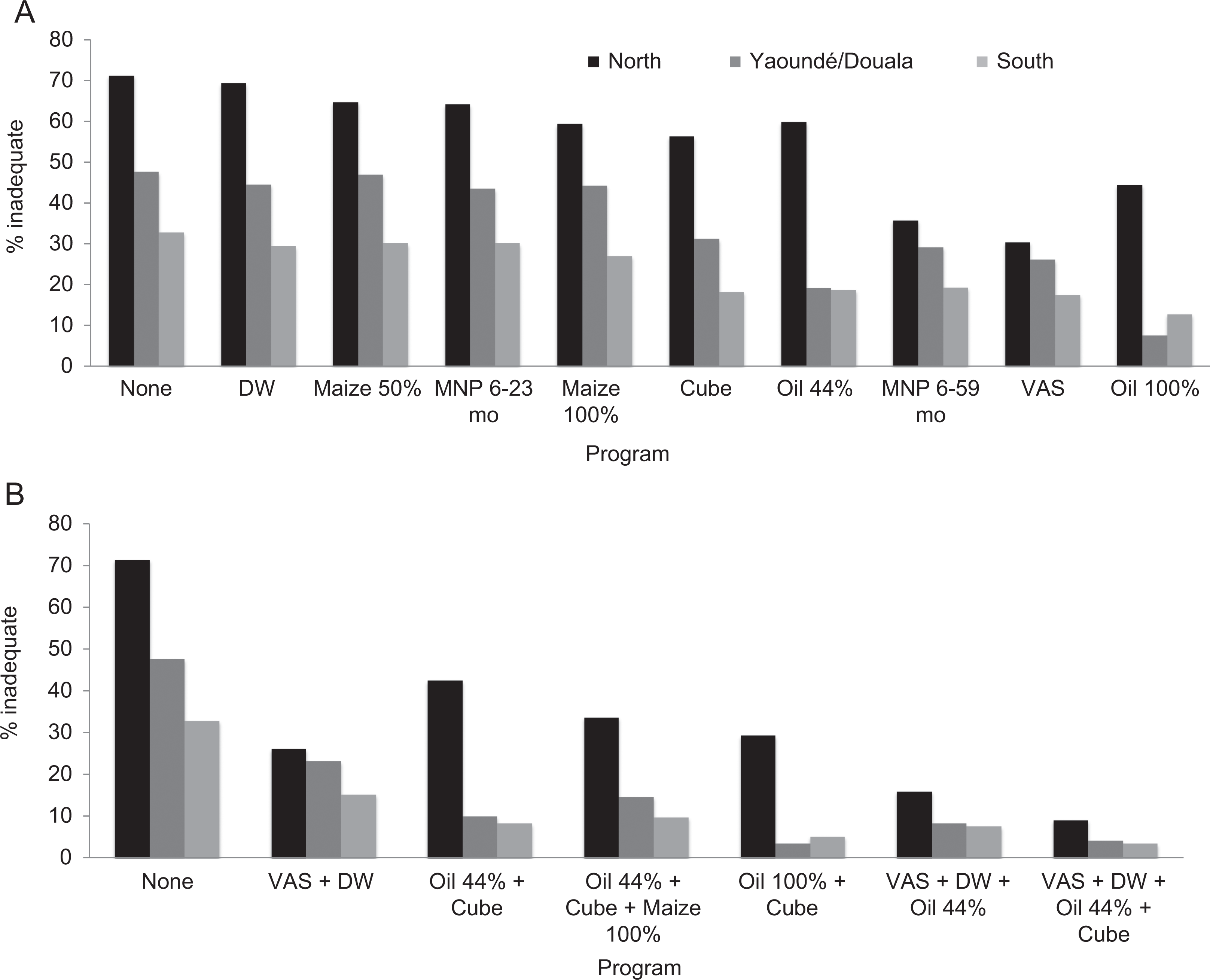
Proportion of children 6 to 59 months of age with inadequate intake in the absence of vitamin A intervention programs and with (A) individual vitamin A programs or (B) combinations of programs. VAS indicates vitamin A supplement.
The number of cases of inadequate intake resolved per macro-region (effective coverage) also varied by intervention, depending on the baseline prevalence of inadequate intake, reach of the program, and the population of the macro-region (Figure 4). VAS administration via CHDs would effectively cover the most children in the North macro-region due to the large numbers of children with inadequate intake and relatively high reach of VAS in this macro-region. Oil fortification at the target level would yield a similar number of cases of inadequate intake resolved, while other interventions would result in lower values, ranging from ∼80 000 for DW to ∼700 000 for MNP distribution in children 6 to 59 months of age. Compared to VAS alone (the individual program with the greatest number of cases resolved), implementing combinations of programs could increase the number of children who convert from inadequate to adequate intake by ∼100 000 children (oil + cube) to ∼550 000 children (VAS + DW + oil + cube; Figure 4B).
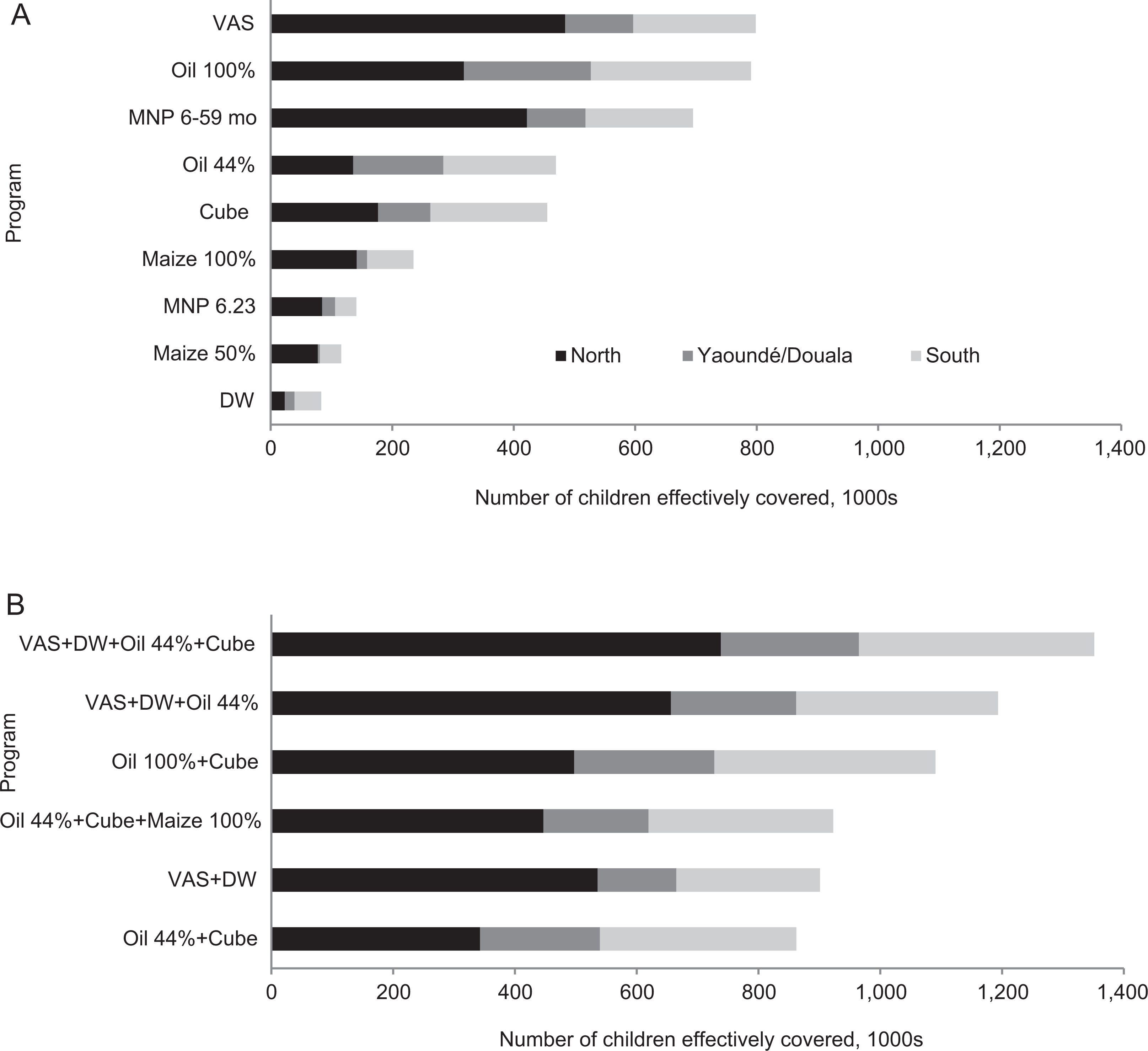
Number of children effectively covered in simulations representing (A) different individual vitamin A programs or (B) combinations of programs by macro-region. Population estimates are from 2011. DW indicates deworming; MNP, micronutrient powder; VAS, vitamin A supplement.
Women
The effect of fortification and biofortification programs on the prevalence of inadequate intakes among women also varied spatially: most programs yielded a fairly large decrease in inadequate intakes in Yaoundé/Douala (presumably because mean intake in this macro-region was close to the EAR; Figures 5 and 6). In contrast, smaller effects were observed in the South, where the prevalence of inadequate intakes was low in the first place, and in the North, where baseline intakes were fairly low (and thus greater amounts of VA are needed for individuals to achieve adequate intake). As for children, combining interventions would result in further decreases in the prevalence of inadequate intake.
Proportion of women with inadequate intake in the absence of vitamin A interventions and in simulations of different vitamin A-related programs or combinations of programs by macro-region.
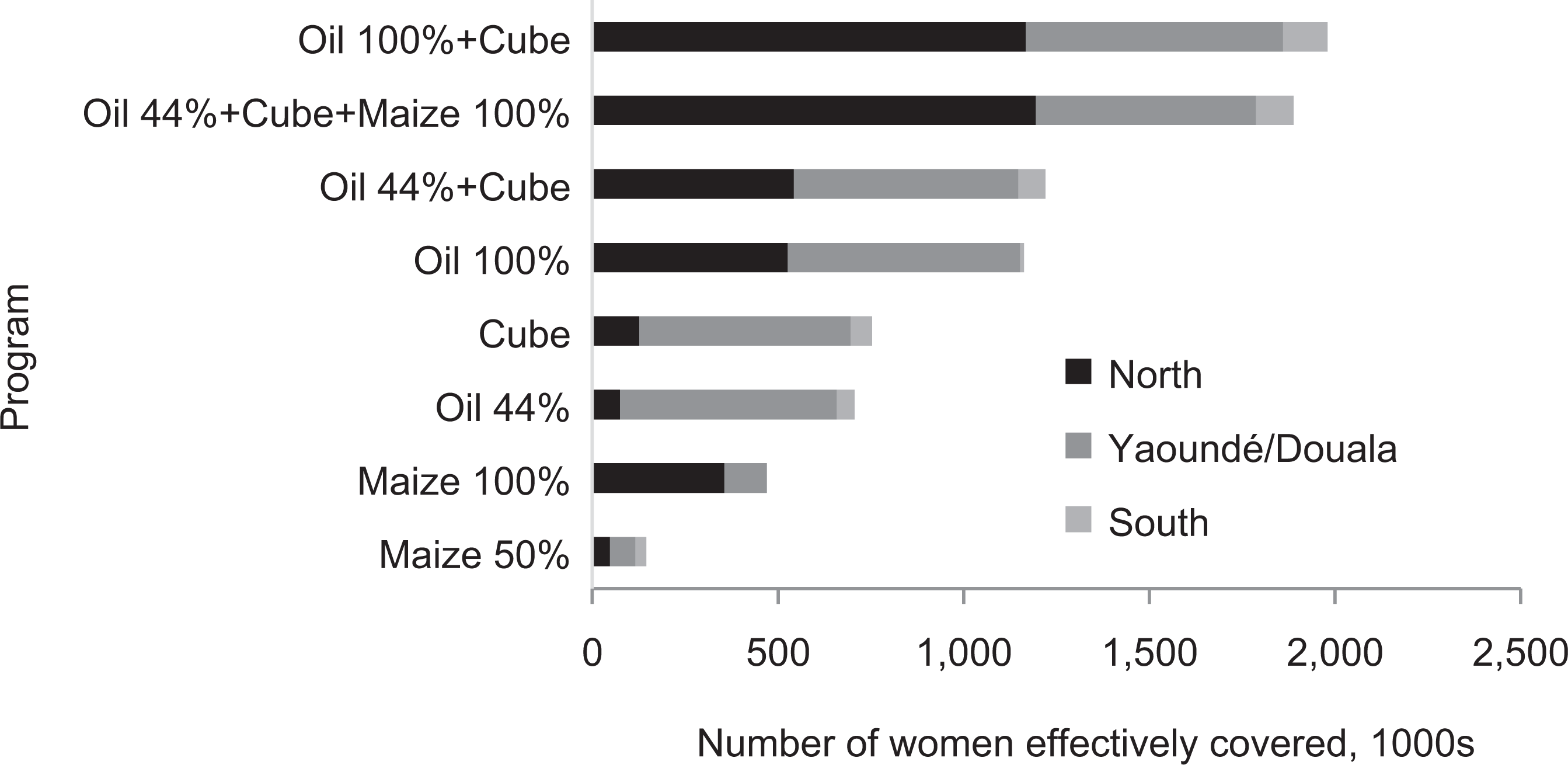
Number of women with inadequate intake who are predicted to achieve adequate intake with individual or combined vitamin A programs in place by macro-region. Population estimates are from 2011.
Discussion
In this article, we describe the methods used to estimate effective coverage of VA-related interventions expressed both as the proportion of the population and as the number of individuals who achieve adequate dietary VA intake (or “intake equivalents”) as a result of implementation of an intervention or package of interventions. The results indicate that effective coverage varies by type of intervention and by macro-region. In particular, in this setting, the potential to benefit (number and percentage of individuals with inadequate dietary intake at baseline) is greatest in the North macro-region, where dietary VA intakes are lowest, and the potential change in dietary adequacy is lowest in the South macro-region, where the baseline VA intakes are greater.
Notably, a similar change in dietary adequacy could be obtained through several programs (or combinations of programs), emphasizing the need to consider other information, such as estimates of program costs and any constraints to implementation, to inform program selection. These estimates of effective coverage, along with region-specific information on the costs of each intervention package, 4 will serve as inputs into an economic optimization model to identify the most cost-effective package of interventions for each macro-region. 5
Although the most cost-effective program portfolio may vary spatially, in practice, not all programs can be targeted spatially. For example, large-scale food fortification programs are typically implemented by industries that distribute products nationally. Thus, this type of program would be difficult to target regionally. In contrast, it would be feasible to implement spatially targeted distribution of VAS or MNP through campaign-type platforms such as CHDs. Indeed, selected components of the intervention package delivered during the twice-annual campaign in Cameroon are already targeted spatially (eg, polio vaccinations). Finally, other programs, like large-scale food fortification and biofortification, are by default targeted to those who currently consume the particular foods (eg, in this setting, the regional distribution of benefits differs for oil fortification compared to maize biofortification).
Limitations
These simulations rely on a number of assumptions and thus care should be taken in interpreting the results. We chose to express effective coverage in terms of dietary intake because of the availability of dietary intake data and the relative ease of expressing the effects of many interventions in terms of additional dietary intake, in comparison with the changes in biomarkers such as serum retinol, for example. However, dietary assessment by any method is subject to numerous sources of error. The 24-hour recalls in particular may under- or overestimate intake on the previous day due to omission of foods or incorrect estimation of portion sizes. 46 Although there are many challenges associated with collecting accurate and precise dietary intake data in large surveys, VA intake as measured in this survey was correlated with breast milk VA concentration 8 and with plasma retinol-binding protein concentrations, indicating that the dietary data capture spatial differences in the risk of biochemical VA deficiency.
Translation of the effects of high-dose VASs or DW capsules to daily intake equivalents requires some important assumptions regarding the pathways by which these interventions confer VA-related benefits to the individual. In particular, the effects of DW on VA status are uncertain due to the paucity of available data (in particular, recent data on the prevalence of infection among children in the target age-group in all regions are not available). In addition, translation of periodic VAS into daily intake equivalents is counterintuitive because a single VAS is likely to increase liver VA stores only temporarily. Thus, we do not suggest that VAS delivered every 6 months (eg, as part of CHD campaigns) will improve VA status. Indeed, studies indicate that the impact of VAS on indicators of status such as serum retinol disappears within ∼2 months, 47 which is consistent with the kinetic modeling results that suggest that liver VA concentrations return to baseline 2 to 4 months postdose (Figure 1). 31 Nevertheless, we have retained both VAS and DW in the analysis to (1) describe the methodological approach, (2) allow comparisons with other interventions (considering the range of plausible assumptions), and (3) indicate topics for which more research is needed.
Despite the limitations involved in translating VAS and DW into intake equivalents, this must be done if the purpose is to consider all interventions together and develop a cohesive national strategy for VAD control. The choice of dietary intake as a common metric for the effects of interventions is justified by the fact that the single and combined effects of most interventions (eg, fortification, biofortification, MNP/daily supplements, etc.) can be easily estimated using dietary intake. Moreover, the procedures used to translate intermittent high-dose VAS and DW into daily intake equivalents are based on careful examination of relevant research and articulation of all assumptions regarding the pathways by which these interventions are expected to affect VA status. Many of these assumptions can be tested empirically. Thus, this method has also helped to define a research agenda to improve the accuracy of these estimates and confirm their utility.
We estimated the change in breast milk VA concentration following increases in maternal VA intake using only studies that provided daily doses of preformed retinol. We did not include studies of maternal supplementation with pro-VA carotenoids in the review because absorption and conversion to retinol likely depend on a number of factors, including the food matrix and the status of the individual, and we did not feel that there was adequate information available to model these patterns. Increases in milk retinol following maternal supplementation with various sources of pro-VA carotenoids were observed in some studies but not others. 16,27,48,49 Any increase in milk retinol would be attenuated if the carotenoids are poorly absorbed and/or if bioconversion to retinol is inhibited because the mother has adequate VA status. Thus, our use of an estimate based on preformed retinol probably overestimates the change in breast milk VA that would be expected from increases in maternal carotenoid intake, such as through biofortification of maize.
However, this overestimation has little impact on the modeling results because (1) when estimated VA intake from milk was included in the diet, the VA intakes by breast-feeding children were mostly adequate, except in the North macro-region and (2) the contribution to VA intake through changes in BMVA was relatively small compared to the “direct” contribution of other interventions to children’s intake (eg, consumption of fortified oil). For example, in a sensitivity analysis in which we assumed no change in milk VA, effective coverage of maize alone decreased by less than 0.5% in all macro-regions, and this difference was even smaller when other interventions were assumed to be in place (eg, VAS + maize).
In addition to the biological assumptions made, some scenarios may not be programmatically feasible. In particular, even the assumption that 50% of maize consumed is replaced by the biofortified variety may be too optimistic for a medium-term (eg, 10 years) planning timeline. Nevertheless, these optimistic estimates are useful for assessing the maximum potential impact of biofortification (or large-scale fortification) in a specific setting to identify programs that merit further consideration. Additional simulations, including expected changes in adoption over time, will be needed to obtain a more realistic estimate of the impact of biofortification.
An additional caveat is that the effective coverage of a given program is likely to vary over time due to changes in dietary intake patterns (eg, micronutrient intake and consumption of fortifiable foods) and access to health services (platforms for program delivery). Accounting for these temporal changes is beyond the scope of the current analysis. However, temporal variation in population size due to migration and changes in fertility rates are considered in the accompanying description of the economic optimization model (Vosti et al, this volume). 5
Finally, comparisons of effective coverage by region are particularly instructive in Cameroon, where dietary patterns and program reach vary substantially over space. The extent to which these results apply to other micronutrients and other settings depends on the types of interventions available and the spatial heterogeneity of the country or region of interest. The value of the optimization modeling approach relies on heterogeneity of benefits and costs according to some “targetable” characteristic (geographic region, wealth group, etc.). However, these dietary intake simulations used alone could also be a useful tool to estimate the relative effects of different interventions (or for other purposes, such as setting fortification levels), even in relatively homogenous populations. Information on total dietary intake (using methods such as 24-hour dietary recalls or observed, weighed food records) collected during representative surveys is needed to use this modeling approach to inform nutrition policy.
Conclusions
In summary, the estimated effective coverage of VA-related interventions in Cameroon varies by intervention and over space. These results suggest that there may be cost saving from regionally targeting VA interventions. Effective coverage must be set alongside region-specific intervention costs (as described by Kagin et al, this volume) 4 to determine the most cost-effective strategy for achieving VA sufficiency objectives (Vosti et al, this volume). 5 This modeling approach serves as a decision-support tool to aid policy makers in developing a coherent national micronutrient deficiency control strategy.
Footnotes
Acknowledgments
We thank Marjorie Haskell (University of California, Davis) and Janet Novotny (United States Department of Agriculture) for guidance on the translation of high-dose VAS to daily intake equivalents and Janet Novoty for confirmatory simulations using WinSAAM. We also thank Janet Peerson (University of California, Davis) and Kevin Dodd (National Cancer Institute) for statistical advice, and Amanda Perkins, Ingrid Friburg, and Neff Walker (Johns Hopkins Bloomberg School of Public Health) for providing the population estimates. Finally, we thank Justin Kagin (Kagin’s Consulting), Ann Tarini (Independent Consultant), Georg Lietz (Newcastle University) and Joanne Arsenault, Erica Rettig, Ryan Murphy, and Robert Hijmans (University of California, Davis) for useful comments on the results.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Collection and analysis of the dietary data were supported by the Michael and Susan Dell Foundation, Sight and Life, UNICEF, and the Bill & Melinda Gates Foundation.
