Abstract
Background
Headache following traumatic brain injury (TBI) is a common, yet disabling, disorder with diverse mechanisms and treatment needs that remain poorly defined. Pharmacological regimens are the primary source of remedies for individuals with post-traumatic headaches (PTH). The main objective of this review is to describe the efficacy of pharmacological medications for the treatment of PTH with a specific focus on the effect of these medications on headache characteristics and headache-related quality of life (QoL).
Methods
This systematic review (CRD42024537719) followed PRISMA and SWiM guidelines. PubMed, CINAHL, Scopus, PsycINFO and the Cochrane Library were searched in April 2024 for peer-reviewed articles published in English between 2009 and 2024. Eligible studies included randomized controlled trials, controlled cohort studies, and systematic reviews or meta-analyses evaluating pharmacological treatments for PTH in adults. Studies were excluded if they did not assess outcomes related to PTH pain, only included pediatric populations, used animal models, investigated only non-pharmacological interventions, were case reports, narrative reviews, editorials or conference abstracts, or did not involve human participants with TBI-related headache. Risk of bias was assessed using RoB-2 for Randomized controlled trials (RCTs) and ROBINS-I for the non-randomized studies of the effects of interventions.
Results
Sixteen studies were included in the final review, comprising retrospective observational (n = 7), non-randomized prospective (n = 4) and randomized controlled trials (n = 5). Most studies reported some improvements in headache frequency and intensity following pharmacological treatment, although findings for headache-related QoL were inconsistent. Erenumab showed potential benefits for persistent PTH in small, uncontrolled studies of civilian samples. However, findings on its impact on headache-related QoL should be interpreted with caution, given the high discontinuation rate observed. Prazosin demonstrated similar benefits in military populations, with minimal side effects. In the acute care setting, metoclopramide, co-administered with diphenhydramine to minimize side effects, was associated with short-term relief of headache symptoms. Of the RCTs, only two had a low risk of bias, of which only one specifically focused on PTH.
Conclusions
Pharmacological treatments for PTH may provide improvements in headache frequency and intensity; however, evidence for their efficacy is limited and inconsistent. Given the limited high-quality evidence overall, no specific clinical recommendations can be made at this time. Future research should prioritize rigorous, controlled studies, particularly comparative effectiveness trials, and explore holistic, personalized approaches that incorporate treatment of psychiatric comorbidities and consider patient context.
Keywords
Post-traumatic headache (PTH) is a common secondary headache disorder.1–6 By definition, headache onset begins within seven days of traumatic brain injury (TBI), whiplash or similar physical injuries. 7 Acute PTH resolves within 3 months, and PTH that continues beyond 3 months is considered persistent (PPTH). 8 About 60% of patients report headache within the first few weeks post-concussion, and roughly 30% continue to experience persistent headache at one year.9,10 The persistence of PTH is influenced by a number of factors, including the specific population studied (e.g. athletes, military personnel or general population), injury severity, age and sex differences.11–13 In US military populations, headache is the most common symptom after sustaining a TBI, 14 with estimates suggesting that up to half may develop persistent headaches.15,16
The clinical management of PTH is challenging and is associated with significant occupational and functional consequences.17–19 Clinically, PTH often presents with overlapping features of primary headache, including migraine-like hallmarks (e.g. photophobia and nausea) and/or tension-type qualities (e.g. pressure/steady pain, bilateral distribution).20–29 Current treatment guidelines from the AAN/AHS 30 and VA/DoD 31 recommend phenotype-targeted pharmacological interventions, but these are largely repurposed from primary headache disorders.32–34 Preventative (e.g. topiramate, amitriptyline) and abortive (e.g. non-steroidal anti-inflammatory drugs (NSAIDs)) medications are used, yet their efficacy in PTH is inconsistent.34,35
This reliance on borrowed therapies stems in part from the application of the neurogenic inflammation model, one of several proposed mechanisms in migraine pathophysiology.36,37 This model posits that neuropeptides like calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase-activating polypeptide-38 (i.e. PACAP-38) drive pain through activation of the trigeminovascular system and peripheral neurogenic inflammation.38,39 Although PTH shares clinical features with primary headache disorders (including migraine), mechanisms may vary temporally. Indeed, by definition, the onset of PTH is dependent on trauma-based triggering, which is considered to initiate migraine-like biology in early phases, although, notably, may also be more likely to give way to broader central sensitization40,41 and is strongly influenced by other psychosocial factors that are not captured by the neurogenic inflammation model. Accordingly, while this framework has informed the implementation of promising treatments for PTH (including CGRP monoclonal antibodies and small-molecule CGRP receptor antagonists, commonly referred to as gepants), the intersection of neurobiological and psychological processes is central. Specifically, affective distress is highly prevalent in TBI populations, which can prolong and exacerbate pain.41–43 In military service members, conditions like depression and PTSD are common and have been directly linked to greater headache-related disability in PPTH.44–47 Assuming that co-occurring psychological factors actively influence the underlying biology, PTH may be more accurately understood through models that emphasize interactions between neurobiological and psychological mechanisms (i.e. a biopsychosocial model).
Given the lack of therapies specifically developed for PTH, and the yet incompletely understood pathophysiology, we hypothesize that available pharmacologic treatments, largely repurposed from primary headache disorders, may incompletely address the therapeutic needs of the condition and that the associated evidence risks being fragmented and/or methodologically heterogeneous. Thus, the aim of this review is to synthesize and critically appraise the existing evidence on pharmacologic treatments for acute and persistent PTH in adults, with a specific focus on their effects on headache frequency, pain intensity and headache-related quality of life, aiming to provide clinicians with a clear synthesis of current evidence and identify priorities for future mechanism-driven research for this unique patient population.
Methods
Search strategy
This systematic review was registered in PROSPERO with the registration number (CRD42024537719), where the protocol can be accessed. Searches were conducted in April 2024 in five bibliographic databases: PubMed, CINAHL, Scopus, PsycINFO and Cochrane Library. The search strategy was developed in coordination with a librarian and is detailed in the Supplementary material (Doc. S1). The search string combined keywords and subject headings related to PTH treatments. This systematic review followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 48 and the Synthesis Without Meta-analysis extension (SWiM). 49 The search was subjected to PRESS peer review using a modified version of the PRESS document 50 and was then translated into all included databases.
Two investigators (SS and ES) initially screened articles by title and abstract, followed by a comprehensive full-text review to determine eligibility for inclusion. Web-based Rayyan 51 was used to screen and select studies that met the inclusion criteria. Two additional investigators (DDM and MJP) reassessed all selected articles. By consensus among the four investigators, article selection was finalized. Key methodological details, sample characteristics (including headache persistence and TBI severity), pharmacological agents and outcome measures were reported.
Eligibility criteria
We included peer-reviewed randomized controlled trials (RCTs), controlled cohort studies, observational studies and systematic reviews or meta-analyses published in English between 2009 and 2024 to capture contemporary evidence on pharmacological treatments for PTH in adults (≥ 18 years). Eligible studies had to report at least one outcome related to headache frequency, headache intensity or headache-related quality of life (QoL) in participants with TBI-related headache.
We excluded studies that: (i) did not assess outcomes related to PTH pain; (ii) included only pediatric populations; (iii) used animal models; (iv) investigated only non-pharmacological interventions; (v) were case reports, narrative reviews, editorials or conference abstracts; or (vi) did not involve human participants with TBI-related headache.
Synthesis methods and effect estimates
SWiM (Synthesis Without Meta-analysis) guideline 49 and the Effect Direction Plot method52,53 were used to summarize and group each study into conceptual outcome domains (e.g. headache frequency, intensity or headache-related QoL). Effect Direction plots are particularly useful in reviews that incorporate non-randomized studies, such as the present study, due to the variety of study designs and outcomes often reported in these trials. 52 Following recommended procedures, 52 related outcomes within each study were grouped into domains, and the overall effect direction was determined by vote counting. Where multiple outcomes within a study all pointed in the same direction, that direction was recorded. Where outcomes varied, we applied the SWiM guideline convention of requiring ≥ 70% concordance to assign a direction as recommended by the 2019 Cochrane Handbook; if < 70% of outcomes agreed, the result was classified as mixed/unclear. 52 To avoid dominance of studies reporting multiple outcomes or timepoints, each study contributed only one ‘vote’ per domain.
The direction and strength of treatment effects across all studies evaluating pharmacologic interventions for PTH were summarized and categorized by outcome domains (headache frequency, intensity, headache-related QoL) and study design in Table 1.
Effect direction across studies (grouped by risk of bias).
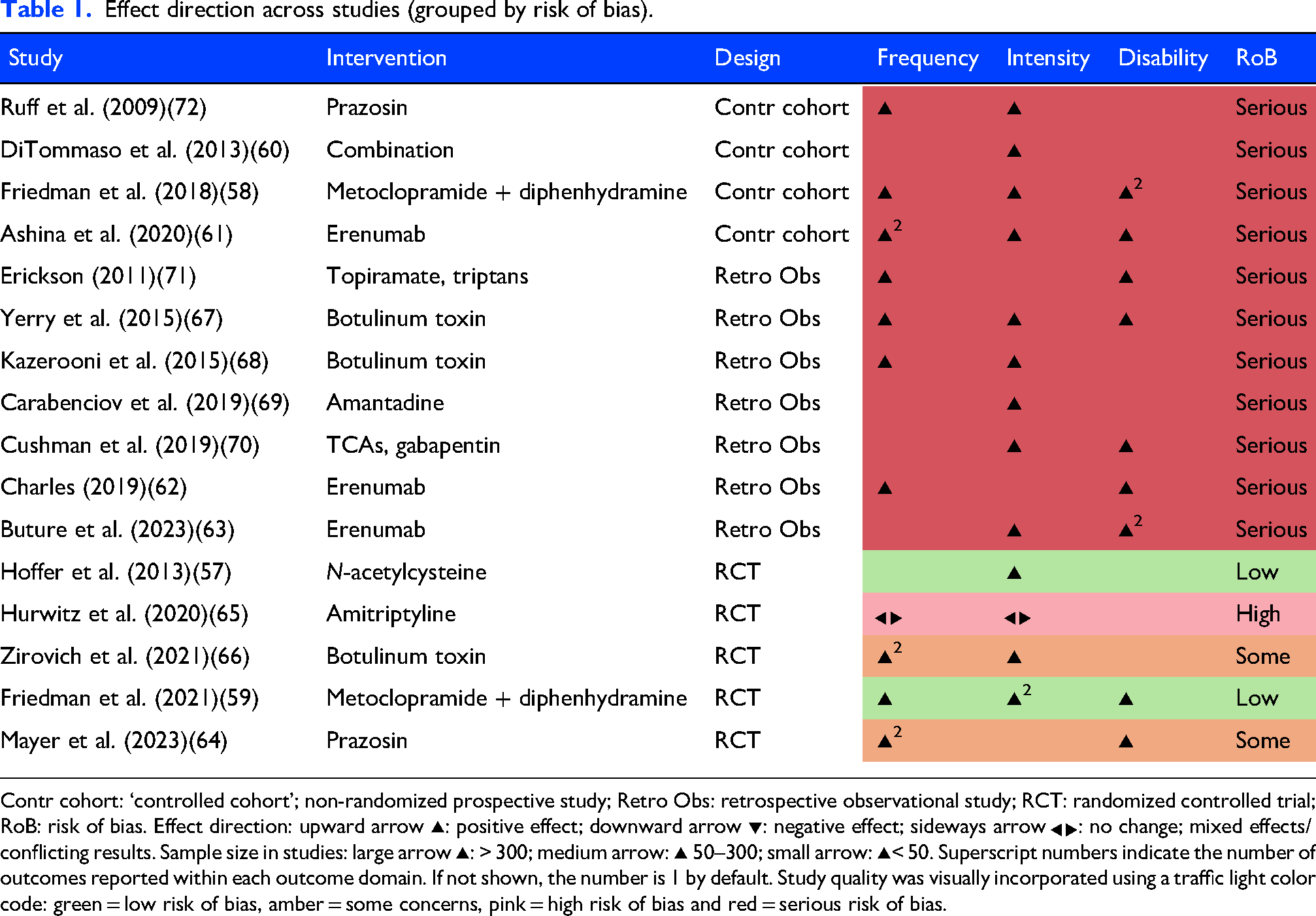
Contr cohort: ‘controlled cohort’; non-randomized prospective study; Retro Obs: retrospective observational study; RCT: randomized controlled trial; RoB: risk of bias. Effect direction: upward arrow ▴: positive effect; downward arrow ▾: negative effect; sideways arrow ◀▶: no change; mixed effects/conflicting results. Sample size in studies: large arrow ▴: > 300; medium arrow: ▴ 50–300; small arrow: ▴< 50. Superscript numbers indicate the number of outcomes reported within each outcome domain. If not shown, the number is 1 by default. Study quality was visually incorporated using a traffic light color code: green = low risk of bias, amber = some concerns, pink = high risk of bias and red = serious risk of bias.
Quality and risk of bias assessment
Version 2 of the Cochrane Risk-of-Bias tool for RCTs (RoB-2) 54 was used to assess RCTs across five domains: randomization process, deviations from the intended interventions, missing outcome data, outcome measurement and selection of the reported result. 55 For non-randomized (non-RCTs) studies of interventions, including both controlled cohorts and retrospective observational designs, the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool 56 was applied. ROBINS-I evaluates seven domains: confounding, participant selection, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes and selection of the reported result. 56 Overall risk-of-bias judgments in RCTs (low, some concerns or high) were assigned based on the highest level of bias identified across domains. Similarly, the overall risk of bias for non-RCTs was determined following the ROBINS-I guidance (low, moderate, serious or critical). Studies rated as ‘serious’ or ‘high’ in any domain were classified accordingly. All outcomes reported in the included studies were extracted and incorporated into the synthesis. For simplicity, the overall risk-of-bias judgments for both RCTs and non-RCTs are visually represented as color codes in the Effect Direction table. Detailed domain-level risk-of-bias assessments are provided in Tables 2 and 3.
Judgment of risk of bias by domain for selected non-randomized studies of intervention based on ROBINS-I.
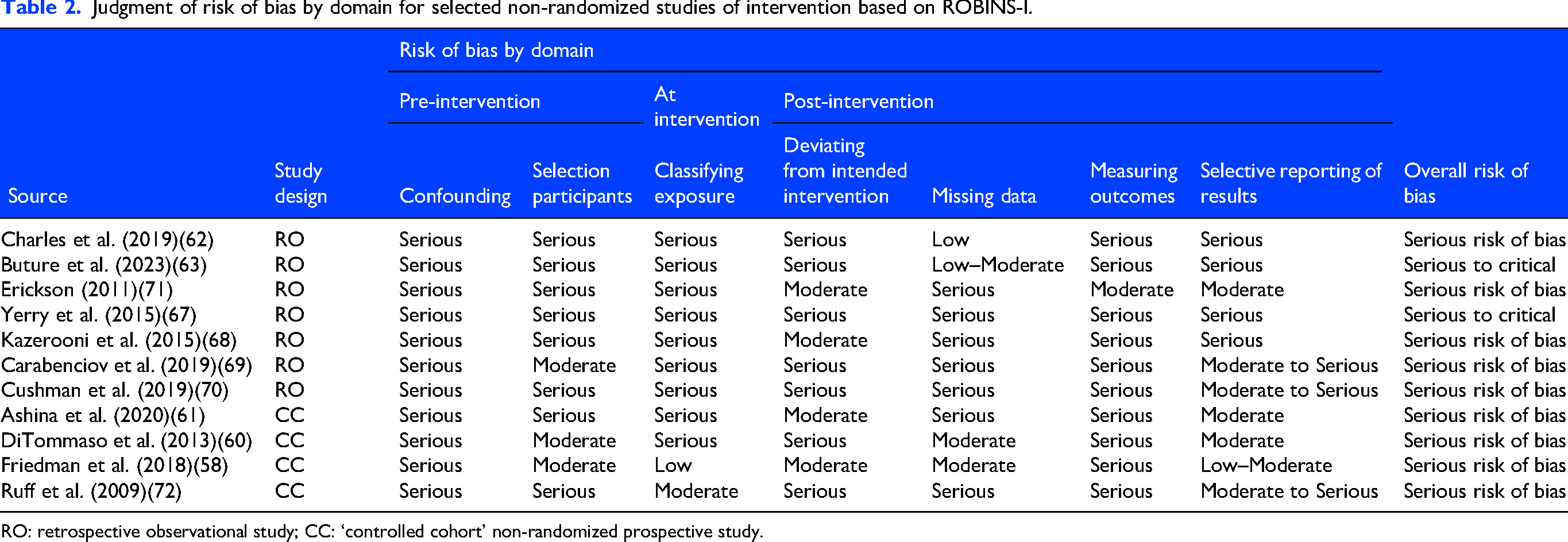
RO: retrospective observational study; CC: ‘controlled cohort’ non-randomized prospective study.
Judgment of risk of bias by domain for selected randomized studies of intervention based on ROB-2.

RCT: randomized controlled trial; DB: double-blind; PC: placebo-controlled.
Results
The database search yielded 2280 search results, and two additional articles were identified through a manual search of the reference lists of relevant primary articles. After removing duplicates, 1693 articles were screened by title and abstract and 21 were retrieved for full-text assessment. Ultimately, 16 articles met the inclusion criteria and were reviewed in this report (Figure 1).
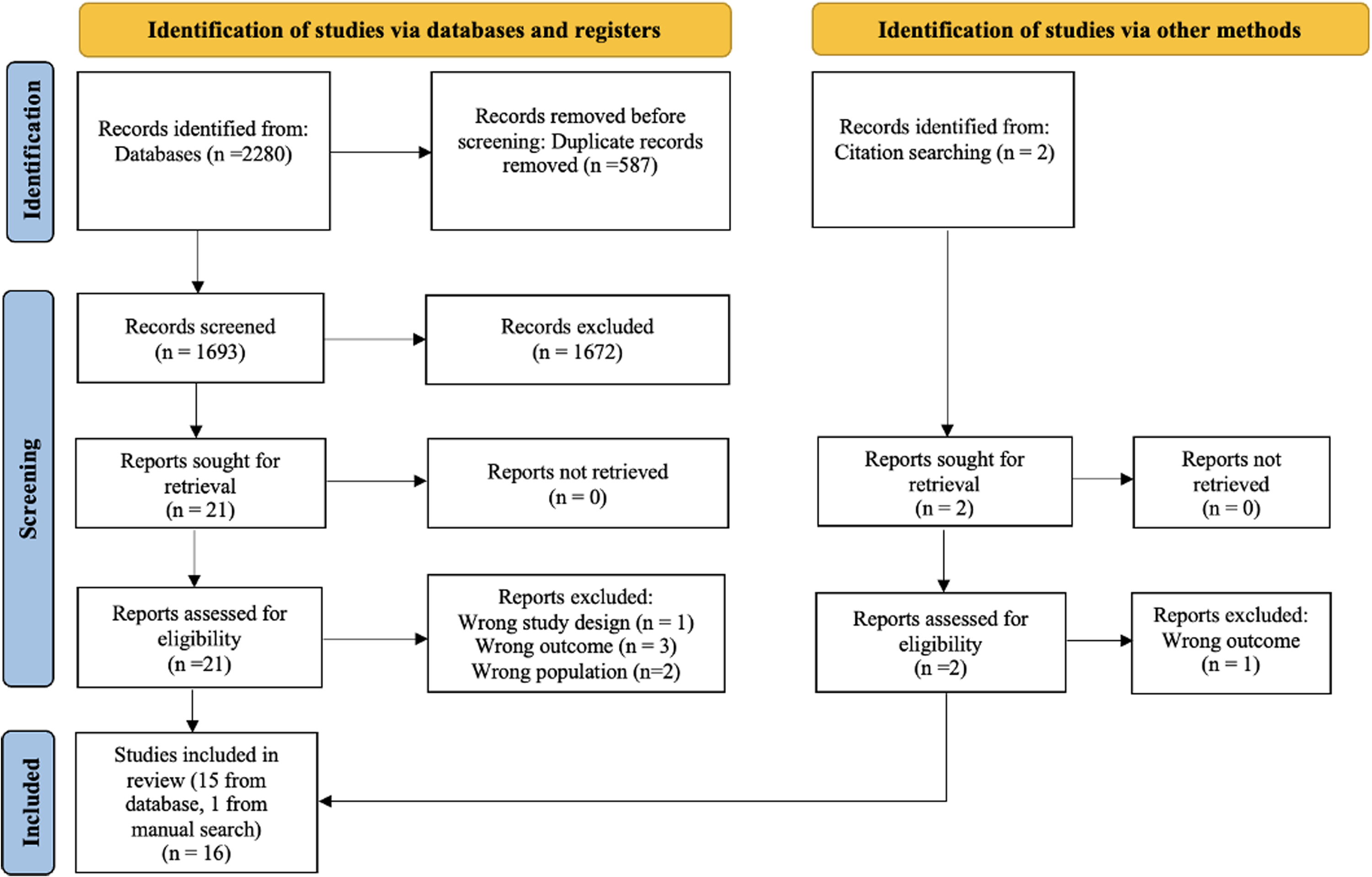
PRISMA flow chart depicting the selection process of studies for systematic review.
Of the 16 studies, four focused on abortive treatments57–60 and the other 12 either specifically evaluated preventive medications or combined pharmacotherapies.61–72 Study populations were highly heterogeneous: nine included civilians or athletes with acute or persistent PTH,58–63,65,69,70 while seven enrolled military service members and/or veteran populations.57,65,66–68,71,72 Designs included five RCTs,57,59,64–66 four controlled cohort58,60,61,72 and seven retrospective observational studies.62,63,67–71 Tables 4 and 5 summarize the study characteristics for all 16 included studies, categorized by non-RCTs and RCTs. To facilitate interpretation, a concise summary of findings is provided in Table 6, presenting key quantitative results for each study, including absolute and relative changes or responder rates where available, alongside a synthesis of reported adverse events.
Overview of the observational studies on pharmacological headache treatment

F: females; M: males; HA: headache; PTH: post-traumatic headache; PPTH: persistent post-traumatic headache; mTBI: mild traumatic brain injury; AE: adverse events; TTH: tension-type headache; FU: follow-up; HIT-6: headache impact test-6; MIDAS: migraine disability assessment; MSQ: migraine specific quality of life; MHDs: monthly headache days; NS: not specified; OIF: operation Iraqi freedom; OEF: operation enduring freedom; BMI: body composition index; TCAs: tricyclic antidepressants; MOCA: Montreal cognitive assessment; NRS: numeric rating scale; SY: symptom; ICD-9: International Classification of Diseases, 9th edition; BoNT/A: onabotulinum toxin A; FDA: Food and Drug Administration; FSFD: fixed site, fixed dose; CD: cervical dystonia; FTP: follow the pain; U: units; GEC: global evaluation of change; NSAIDs: non-steroidal anti-inflammatory drugs; PCS: post-concussion syndrome; ED: emergency department; IV: intravenous; PCSS: post-concussion symptom scale; SCAT: sport concussion assessment tool.
Overview of the clinical trials on pharmacological headache treatment.

RCT: randomized controlled trial; BoNT/A: botulinum toxin type; PTN: posttraumatic neuralgia; PGIC: Patient Global Impression of Change; NPSI: Neuropathic Pain Symptom Inventory; TMT: trail making test A & B; COWA: Controlled Oral Word Association; AN: animal naming; QTc: corrected QT interval.
Concise summary of findings on pharmacological treatments for post traumatic headache.

HA: headache; d/mo: days per month, d/wk: days per week, pts: points, QoL: quality of life; MIDAS: Migraine Disability Assessment; HIT-6: Headache Impact Test; MSQ: Migraine-Specific Quality of Life; PCSS: Post-Concussion Symptom Scale; LFT: liver function test; TCA: tricyclic antidepressant; AE: adverse event; Arrows (↓) indicate improvement/reduction, OR: odds ratio, CI: confidence interval, Tx: treatment
The narrative results below are stratified by phase (acute vs. persistent) and population (civilian vs. military/veteran) to highlight differences in treatment contexts and outcomes.
Acute phase treatments
Civilian populations
Anti-nausea (metoclopramide). Two related studies investigated the efficacy of nausea medication as a treatment for acute PTH rescue. Friedman et al. 58 conducted a prospective, open-label study of IV metoclopramide (plus diphenhydramine, M + D) for patients presenting to the emergency department (ED) with acute PTH. 71% of patients (n = 21) reported headache relief within 1 h of treatment with M + D, and effectiveness increased to 95% by 2 h. At 48 h, 55% of patients maintained sustained headache relief without needing rescue medication. By one week, headache recurrence was reported in >25% of patients, and 21% sought further medical care. Affective/mood changes were minimal.
Building on previous findings, Friedman et al. (2021) conducted a double-blind, placebo-controlled RCT to evaluate the efficacy of M + D for patients (n = 160) experiencing acute PTH following head trauma presented in the ED. 59 Similar to earlier results, M + D was associated with significant improvement in headache frequency and pain at one hour compared to placebo. There may have been a trend toward fewer mood-related symptoms in the treatment group compared to placebo, although these should be interpreted cautiously.
Analgesics, NSAIDs and triptans. A single study investigated the usage patterns of specific rescue agents as treatment for acute and persistent PTH. Ditommaso et al. 60 examined medication usage patterns for headache treatment 3, 6 or 12 months after mild TBI (mTBI) using a prospective observational design. Over 70% of patients used over-the-counter analgesics such as acetaminophen and NSAIDs for headache control, with mixed results. Triptans were underutilized in this sample (8%); no conclusions can be drawn regarding their effectiveness in PTH.
Military populations
Glutathione-restoring antioxidant
A single study investigated the efficacy of a glutathione-restoring antioxidant as a treatment for acute PTH following mTBI. Hoffer et al. 57 implemented an RCT to evaluate the effect of N-acetylcysteine (NAC) versus placebo, both administered in addition to standard mTBI care, on the symptoms associated with blast exposure mTBI in a combat setting (n = 81), of whom 53 reported headache symptoms. NAC, especially when initiated early (within 24 h), was associated with faster resolution of all mTBI-related symptoms by day seven post-blast. Participants who received early NAC had substantially higher odds of being headache-free at day seven compared to those who received placebo after 24 h. NAC also improved neuropsychological performance and balance dysfunction.
Persistent phase treatments
Civilian populations
Monoclonal antibody treatments. Three studies investigated the efficacy of erenumab, a CGRP receptor monoclonal antibody, as a preventative treatment for PTH. Charles 62 conducted a retrospective case series (n = 7) to evaluate the effect of erenumab in the acute phase of PTH after sustaining mTBI. All patients had normal exam and imaging, were disabled by high-frequency/intense headaches, had failed or been intolerant of conventional migraine preventatives, and most had no prior history of migraine. 62 The medication was generally well-tolerated. Patients reported improvement in monthly headache days and headache-related disability within days to 4 weeks, with most requiring only one dose.
Buture et al. 63 assessed erenumab among patients (n = 82) with treatment-refractory headache conditions, including 15 individuals with PPTH following minor head trauma. Of these, nine reported subjective improvement with erenumab. Some 65% of patients discontinued treatment within 6–25 months.
Ashina et al. 61 conducted an open-label study evaluating the effectiveness of erenumab for PPTH following mTBI (n = 100). A ≥ 50% reduction in monthly headache days of moderate to severe intensity was observed in 28% of participants, while the ≥ 50% responder rate for headache days of any intensity was 13% during weeks 9–12. Erenumab was generally well tolerated, but placebo-controlled RCTs are needed to confirm efficacy and safety.
Antidepressants. Two studies investigated the efficacy of antidepressant agents as preventatives for PPTH. In an RCT, Hurwitz et al. 65 evaluated the efficacy and safety of amitriptyline in preventing the development of PPTH after mTBI (n = 50). Participants were recruited from ED acute care admissions, outpatient clinics and community brain injury events. This study could not determine the efficacy of amitriptyline for preventing PPTH because of recruitment and adherence problems.
Using a retrospective cohort design, Cushman et al. 70 evaluated the association of gabapentin and tricyclic antidepressants (TCAs) with headache symptoms over time after mTBI. Headache and symptom scores, which comprised 22 post-concussion related symptoms, improved over time for all patients (n = 277), regardless of medication status, but piecewise regression analysis suggested that symptoms may have improved more rapidly with gabapentin or TCAs compared to the no-med group.
Antidyskinetics. A single study investigated the efficacy of amantadine in the treatment of post-concussion syndromes, including headaches. Using a retrospective observational design, Carabenciov et al. 69 evaluated the effect of amantadine on post-concussion symptoms, particularly PPTH; 33 patients were reported by physician documentation to have PPTH. One-third (11/33) discontinued because of adverse effects (dizziness, nausea, worsened headaches) and no symptom improvement was recorded in those who discontinued. Of those who completed a full trial, 80% showed improvement in headache symptoms.
Military populations
Antihypertensives
Two studies investigated the efficacy of prazosin, an antihypertensive agent, for PPTH. In a prospective observational cohort, Ruff et al. 72 evaluated whether treating impaired sleep using prazosin was associated with PPTH headache frequency and severity among operation Iraqi freedom/operation enduring freedom veterans with blast-exposure mTBI (n = 74). The study was conducted as part of quality-assurance monitoring during routine care, with no control group or blinding. Of the 71 participants diagnosed with PTSD, 69 reported poor sleep. Veterans who completed prazosin showed large improvements in headache frequency (from 12.4 to 4.8) and pain intensity (from 7.3 to 4.1) at 9 weeks. Restful sleep rose from 2% at baseline to 100% by 6 months with a significant decrease in daytime sleepiness. A positive result was also significant in cognitive performance.
In an RCT, Mayer et al. 64 evaluated the efficacy of prazosin for the prophylactic treatment of PPTH in active-duty service members and veterans with blast or blunt mTBI (n = 48). PTSD was present in 66% of the prazosin group and 81% of the placebo group. The prazosin group was associated with a significantly decreased incidence of headaches. Self-reported disability related to headaches was also significantly lower in the prazosin group relative to placebo. Overall, this RCT was assessed to have some concerns of bias.
Botulinum toxins
Three studies investigated the efficacy of botulinum toxins as a treatment for PTH. In a retrospective, uncontrolled design, Yerry et al. 67 provided preliminary data on the use of onabotulinumtoxinA (BoNT/A) for PPTH in military service members (n = 64). All participants had mTBI; 56.3% of these injuries were blast-related. 64.1% reported being ‘better’ on the global evaluation of change (GEC); 28.5% unchanged; 3.2% worse. Slightly more than half of the cohort were able to maintain or return to active military service.
In an RCT, Zirovich et al. 66 evaluated the efficacy of a single dose of BoNT/A versus saline placebo in treating PPTH patients (n = 40) reporting mixed complicated and uncomplicated mTBI. BoNT/A treatment was associated with a significant reduction in headache burden, with a cumulative decrease of 2.24 headache days per week over 16 weeks, compared to a slight increase in the placebo group; the between-group difference in headache days was statistically significant. There was also a small but significant reduction in pain severity with BoNT/A versus a non-significant increase with placebo.
Kazerooni et al. 68 designed a retrospective case series to support the off-label use of incobotulinumtoxinA in reducing headache frequency and intensity among veterans with chronic migraine (n = 21), including a subset with PPTH (n = 6) following TBI. Among these individuals with PPTH, five reported clinically meaningful improvement, with a reduction in headache days per month from a mean of 19.5 to 6.7.
Topiramate, TCAs and triptans
A single retrospective study examined treatment patterns and outcomes for topiramate, tricyclic antidepressants and triptans in service members with PPTH. Erickson et al. 71 conducted a retrospective observational study with follow-up to evaluate treatment outcomes in US Army soldiers (n = 100) with PPTH attributable to mTBI. At baseline, 52% screened positive for PTSD and 38% for depression. Triptans were often effective for aborting acute headache attacks, including both blast-related and non-blast-related PPTH. Topiramate was associated with a significant reduction in headache frequency. In contrast, low-dose TCAs did not show a significant impact on headache frequency. This study supported that a comprehensive headache management plan that addresses PTSD and sleep issues was associated with lower headache-related disability.
Discussion
Overview and key findings
This systematic review highlights the emerging landscape of pharmacological interventions for headaches following TBI. Beyond evaluating overall efficacy, we aimed to assess the extent to which included studies addressed adverse effects, headache frequency, pain intensity and headache-related QoL. Although the effect direction summary indicated predominantly positive effects, the overall strength of the evidence was limited. Most reviewed studies were small in size (all but two of them had n ≤ 100) and often lacked a priori power analyses to justify sample size. In addition, few employed rigorous randomized or controlled designs, and almost all studies suffered from moderate to serious risk of bias. Taken together, these findings support our hypothesis that current pharmacologic treatments for PTH, largely adapted from primary headache disorders, are supported by a fragmented and methodologically heterogeneous evidence base and may not fully meet the therapeutic needs of this population.
Currently, there is insufficient evidence to support the use of erenumab in both acute and persistent PTH in the civilian population. Across the three identified studies, two were uncontrolled and underpowered62,63 and one was an open-label, single-arm design without a placebo control. 61 While a reduction in headache frequency and intensity was observed, these findings should be viewed as hypothesis-generating rather than evidence for ereumab efficacy. Controlled randomized trials are needed to determine whether erenumab has a clinically meaningful effect in PPTH.
Among veterans and military populations, prazosin has been examined primarily in small and methodologically limited studies. Prazosin has previously been associated with improvements in headache pain in individuals with severe trauma-related sleep issues, 73 but larger studies are needed to determine its efficacy for persistent PTH. Thus, the findings should be interpreted cautiously and require validation in larger, controlled trials. Dose-finding studies also help determine whether a lower dose could retain efficacy while decreasing the frequency of adverse events.
Metoclopramide, a dopamine receptor antagonist with analgesic properties, is often administered off-label as a first-line treatment in the ED setting due to its effectiveness in relieving both headache pain and associated nausea in acute migraine management.74,75 Diphenhydramine is often co-administered in EDs to reduce the side effects of metoclopramide. In the included open-label study, patients experienced headache relief within one hour of M + D administration; however, the lack of a comparison group, small sample size and diminishing effects by one week limit the conclusions. 58 Subsequent RCT confirmed early benefits when greater pain reduction was observed at one hour in the M + D group compared to placebo. 59 Although the trial was assessed as low risk of bias, its generalizability is limited by its urban ED setting, and no long-term follow-up was reported.
Antidepressants are considered to offer therapeutic benefits for headache symptoms, 76 potentially reducing some of the comorbid affective burden. Mechanistically, it is considered that antidepressants may help reduce the frequency and severity of headaches by influencing serotonin and other neurotransmitters.77,78 However, evidence for their use in preventing PPTH remains limited and inconclusive. In the RCT evaluating amitriptyline, 65 the risk of bias was assessed as high due to recruitment difficulties, poor medication adherence and incomplete daily diary records. Amitriptyline may be effective for migraine prevention, 78 but this trial did not demonstrate efficacy for PPTH. A large retrospective cohort examining gabapentin and TCAs reported symptom improvement across all groups, 70 but findings were constrained by the retrospective design, substantial loss to follow-up and absence of a control group.
Evidence supporting the use of botulinum toxin for PTH remains limited and methodologically weak. Mechanistically, BoNT/A is considered to exert its effects through presynaptic inhibition of nociceptive neurotransmitters and neuropeptide release (e.g. glutamate, substance P and CGRP), which may lead to muscle relaxation.68,79 Although the blocking mechanism could be relevant to PTH, the retrospective and open-label study designs included in this review limit the strength of inference. IncobotulinumtoxinA was evaluated in one small retrospective case series and was associated with clinically meaningful improvement in a subset of patients with PPTH. Larger, controlled trials are needed to determine whether either formulation provides consistent benefit for PPTH.
Preliminary evidence suggests that N-acetylcysteine
Generalizability and other studies
These findings align with prior literature indicating that most pharmacological approaches for PTH have been extrapolated from treatments for primary headache disorders such as migraine and tension-type headache. Erenumab and other CGRP-targeting monoclonal antibodies, for example, have been widely used in migraine prophylaxis since 2018 by blocking CGRP signaling(80). An obvious next step was the evaluation of these therapies for PTH treatment. Individuals with PPTH demonstrate hypersensitivity to CGRP; CGRP infusions reliably induce migraine-like headaches in this population.81,82 Preclinical models indicated that early and continuous blockade of CGRP using MaB-derived therapies after mTBI prevents both acute PTH and the establishment of a sensitized state that leads to PPTH. 83 Although these biological parallels make CGRP antagonists such as erenumab attractive as mechanism-based treatments, the clinical evidence remains too limited to support any definitive conclusions.
Treatment approaches for military populations must account for common comorbidities, including PTSD, which affects almost 30% of veterans and service members, 45 as well as high rates of anxiety, depression and medication overuse. 84 Among individuals with comorbid PPTH and affective distress, some pharmacologic interventions that target psychiatric and/or sleep-related conditions, such as prazosin, appear to be effective.73,85 A multidisciplinary care model that combines headache management medication with behavioral interventions may be especially effective. 41
Headache remains a common complaint among the 1.4 million patients who visit US EDs annually after head trauma, 86 and future research should explore optimal dosing and duration of rescue medications, such as metoclopramide, and whether early intervention can improve longer-term outcomes, including comorbid symptoms like depression, sleep disturbances, and anxiety, which often affect individuals with PTH.
Pharmacological treatments remain the most accessible pain management option for many individuals with PTH, particularly those with limited resources or time. This review provides an updated synthesis of available evidence to support clinical decision-making, although consistent with prior reviews,34,87 our conclusions are limited by the scarcity of high-quality, powered trials across both abortive and preventive therapies. Nevertheless, this work adds value by reassessing the literature and interpreting the data through the most recent lens. Unlike previous reviews that included multiple treatment types and mixed populations, 34 this review focuses solely on pharmacological interventions in adults to deliver a precise assessment of treatment effectiveness.
Future research direction
There is a clear need for powered, randomized, placebo-controlled clinical trials to rigorously evaluate the efficacy and safety of both abortive and preventative treatments for PTH. Current evidence is largely observational or based on expert opinion, with few RCTs. Further research is warranted to evaluate newer treatment options or re-evaluate old treatment paradigms across diverse populations and headache phenotypes.
It is important to recognize that, while the neurogenic inflammation model informs our understanding of the pathophysiology of primary headaches and has effectively provided key targets for pharmacotherapies, it may be an incomplete model to guide the development of treatments for secondary headaches. Injury mechanisms leading to PTH, such as motor vehicle collisions, combat-related blast exposures and physical assaults, are often accompanied by significant psychological stressors that can influence symptom expression, treatment response and recovery trajectories. Investigators who explore pathophysiology and treatment models that account for both biological and psychological dimensions of PTH may develop more consistently effective and personalized therapies.
Strengths and limitations
This systematic review provides a comprehensive synthesis of pharmacological treatments for PTH in adults, incorporating both RCTs and observational studies across military and civilian populations. Key strengths include a registered protocol (PROSPERO), a librarian-assisted and PRESS-reviewed search strategy, adherence to PRISMA and SWiM guidelines, and rigorous risk-of-bias assessments using validated tools (RoB-2 and ROBINS-I). The review offers clinically relevant insights by focusing on adult populations and well-defined headache outcomes. However, the findings are limited by methodological heterogeneity among controlled studies, and the rest were retrospective or observational. Additionally, the exclusion of non-English and unpublished literature may have introduced bias. Despite these limitations, the review identifies important gaps and offers direction for future high-quality research. Finally, while we acknowledge that meta-analyses may often provide added insight, the substantial clinical and methodological heterogeneity across studies was assessed to be more likely to introduce additional bias than to answer questions, and so a meta-analysis was not conducted.
Conclusions
Pharmacological therapies are a key component of guideline-based, multimodal treatment for PTH, yet none are specifically developed, nor approved, for this condition. Among military population with PPTH and comorbid psychiatric distress, prazosin has demonstrated preliminary benefits; among civilians without such comorbidities, erenumab may warrant further study as a potential mechanism-based therapy. Metoclopramide plus diphenhydramine appears effective for short-term relief in emergency settings. However, these findings are largely derived from small or uncontrolled studies, underscoring the urgent need for adequately powered randomized trials. Future research should aim to clarify the underlying mechanisms distinguishing PTH from primary headache disorders and to develop targeted, evidence-based pharmacologic strategies for both acute and persistent PTH.
Public health relevance
Pharmacological therapies remain the first-line and most accessible treatment for PTH, with behavioral interventions serving as adjuncts rather than replacements.
Early studies of CGRP monoclonal antibodies (e.g. erenumab) suggest possible benefit, but current data are small, uncontrolled, or retrospective.
Prazosin may help some patients with co-occurring PTSD and sleep disturbance, although findings come from limited and heterogeneous studies.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261441563 - Supplemental material for Efficacy and outcomes of pharmacological treatments for headaches after traumatic brain injury: A systematic review
Supplemental material, sj-docx-1-cep-10.1177_03331024261441563 for Efficacy and outcomes of pharmacological treatments for headaches after traumatic brain injury: A systematic review by Shirin Saleh, Elizabeth M Sanford, Donald D McGeary, Melissa M Cortez, Matt Hayward, Erin D Bouldin, Jacob Kean and Mary Jo V Pugh in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024261441563 - Supplemental material for Efficacy and outcomes of pharmacological treatments for headaches after traumatic brain injury: A systematic review
Supplemental material, sj-docx-2-cep-10.1177_03331024261441563 for Efficacy and outcomes of pharmacological treatments for headaches after traumatic brain injury: A systematic review by Shirin Saleh, Elizabeth M Sanford, Donald D McGeary, Melissa M Cortez, Matt Hayward, Erin D Bouldin, Jacob Kean and Mary Jo V Pugh in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024261441563 - Supplemental material for Efficacy and outcomes of pharmacological treatments for headaches after traumatic brain injury: A systematic review
Supplemental material, sj-docx-3-cep-10.1177_03331024261441563 for Efficacy and outcomes of pharmacological treatments for headaches after traumatic brain injury: A systematic review by Shirin Saleh, Elizabeth M Sanford, Donald D McGeary, Melissa M Cortez, Matt Hayward, Erin D Bouldin, Jacob Kean and Mary Jo V Pugh in Cephalalgia
Footnotes
Acknowledgements
Dr Pugh was also supported by a Research Career Scientist Award (IK6 HX002608; RCS 17-297).
Author contributions
Study concept and design: Shirin Saleh, Elizabeth M. Sanford, Donald D. McGeary and Mary Jo V. Pugh. Analytic planning and/or analyses: Shirin Saleh, Elizabeth M. Sanford, Donald D. McGeary and Mary Jo V. Pugh. Interpretation and discussion of results: Shirin Saleh, Elizabeth M. Sanford, Donald D. McGeary, Melissa M. Cortez, Matt Hayward, Erin D. Bouldin, Jacob Kean and Mary Jo V. Pugh. Drafting the manuscript: Shirin Saleh, Elizabeth M. Sanford, Donald D. McGeary, Melissa M. Cortez, Matt Hayward, Erin D. Bouldin, Jacob Kean and Mary Jo V. Pugh. Revisions for intellectual content and final approval: All authors were involved in the revision process and approval of the final manuscript submitted for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the US Department of Defense through the US Army Medical Research and Materiel Command, Congressionally Directed Medical Research Programs, Psychological Health and Traumatic Brain Injury Research Program award (E01 HT9425-23-1-1046) (PI: Donald D. McGeary, PhD).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
The data that supports the findings of this study are available in the supporting information of this article.
Supplemental material
Supplemental material for this article is available online.
