Abstract
Background
Migraine, especially with aura (MA), increases the risk of brain white matter hyperintensities (WMH) and ischemic stroke. Altered microvascular function has been proposed as a shared underlying mechanism, potentially involving endothelial dysfunction. Visually-induced hemodynamic response describes the process by which cerebral blood flow is locally increased to meet the metabolic demands of neuronal activity and may serve as a surrogate measure of cerebral endothelial function. We investigated the relationship between visually-induced hemodynamic responseand WMH volume in middle-aged women with ischemic stroke or MA.
Methods
We cross-sectionally measured hemodynamic response using blood-oxygen-level-dependent (BOLD) fMRI upon visual stimulation on 7T-MRI, and WMH volume on 3T-MRI, in three groups of women aged 40–60 with: (I)ischemic stroke, (II)MA, and (III)no history of stroke or migraine. We assessed the associations between BOLD parameters in the posterior circulation (amplitude, time-to-peak [TTP], and time-to-baseline [TTB]) and WMH volume using multivariable linear regression within each group.
Results
We included 87 women (mean age 51 years): (I)25 with ischemic stroke, (II)25 with MA, and (III)37 without stroke or migraine. Visually-induced hemodynamic response was similar across groups and not associated with WMH volume overall. However, lower amplitude was associated with higher deep WMH volume in those without stroke or migraine (adjusted-β= −0.28;95%CI = –0.53 to −0.02 mL), shorter TTB with greater periventricular WMH volume in women with MA (adjusted-β= −0.12;95%CI = –0.21 to −0.02 mL), and longer TTP with increased periventricular WMH volume in women with stroke (adjusted-β=0.22;95%CI = 0.06 to 0.40 mL).
Conclusions
No overall association was observed between visually-induced hemodynamic response and WMH volume. Exploratory findings suggest potential differences in hemodynamic response in the posterior circulation across groups, which require replication in larger datasets and confirmation in longitudinal studies to clarify their temporal or mechanistic relevance to WMH development.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine is three times more prevalent in women than in men and is a well-established risk factor for ischemic stroke, particularly in those with aura.1–3 In men, this risk is less certain.1–3 Women with migraine with aura also have an increased prevalence of structural brain changes, most notably white matter hyperintensities (WMH).4,5 WMH are frequently observed on MRI in the general population and increase with age, but they are also common in young and middle-aged women with ischemic stroke or migraine with aura.4–6
The mechanisms linking migraine to WMH and ischemic stroke are not fully understood. Population-based imaging studies suggest that women with migraine are particularly susceptible to deep WMH and silent infarcts, independent of traditional vascular risk factors or atherosclerosis.7–10 These findings point toward shared mechanisms between migraine and cerebrovascular disease.11,12 Cerebrovascular reactivity reflects the vascular response to vasoactive stimuli such as hypercapnia and, in previous studies of migraine with aura, has not been associated with WMH volume nor with increased stroke risk when compared with stroke patients or controls.13–15 In contrast, the hemodynamic response describes the process by which neuronal activation leads to a localized increase in cerebral blood flow to meet metabolic demand.16–18 Impairment of hemodynamic response may result in insufficient perfusion of local brain regions and could contribute to the development of WMH and ischemic stroke.16–18 Altered hemodynamic response may therefore help explain the increased vascular vulnerability observed in women with migraine and represents a potential pathway linking migraine to long-term brain changes.19–22 Blood-oxygen-level-dependent (BOLD) fMRI reflects mainly changes in blood oxygenation resulting from the interplay between cerebral blood flow, blood volume, and oxygen extraction and therefore provides an indirect measure of cerebrovascular–metabolic function rather than direct cerebral blood flow.
In this exploratory study, we first investigated whether hemodynamic response differs among middle-aged women with ischemic stroke, migraine with aura, and those without stroke or migraine in the posterior circulation. This response, which we will refer to as visually-induced hemodynamic response from now on, was measured by BOLD fMRI upon visual stimulation as a marker of visual cortex microvascular hemodynamics. Second, we investigated whether BOLD parameters are associated with WMH volume within these groups.
Methods
Study design and participants
We included women aged 40–60 years from two cross-sectional studies conducted at the Leiden University Medical Center (LUMC) between January 2019 and March 2022 that followed identical protocols: the Cardiovascular RiskprofilE in Women—MIcrovascular STatus (CREW-MIST) study and the WHIte matter lesions in young to middle-aged women with Stroke, PreEclampsia or Migraine (WHISPER) study.23–25 Participants were categorized into three groups: (I) women with a history of ischemic stroke only, (II) women with migraine with aura only, and (III) women without a history of ischemic stroke or migraine.
The age range was chosen to create a relatively homogeneous population at risk for stroke, while minimizing confounding by cardiovascular risk factors and atherosclerosis that increase with age. We included only patients with migraine with aura, as this subtype is associated with higher WMH volume and increased stroke risk compared with migraine without aura.
Group I participants (stroke) were derived from the CREW-MIST study, which included women with prior ischemic stroke and no history of migraine. Groups II and III were derived from the WHISPER study and consisted of women with migraine with aura (no history of stroke) and women without a history of either ischemic stroke or migraine, respectively.
Detailed recruitment procedures, inclusion and exclusion criteria, and diagnostic classifications (including TOAST criteria for ischemic stroke and ICHD-3 criteria for migraine with aura) are provided in the Supplementary Material.
During the research visit, participants completed a questionnaire on demographics, medical history, vascular risk factors, and medication use. Blood samples were collected, and MRI scans were performed using both 3 Tesla (3 T) and 7 Tesla (7 T) scanners. Visits were rescheduled if participants experienced a migraine attack on that day. The study protocols of WHISPER (P18.130) and CREW-MIST (P15.384) were approved by the Medical Ethics Committee Leiden–Den Haag–Delft (METC-LDD), and all participants provided written informed consent prior to participation. This manuscript follows the STROBE reporting guideline.
MRI data acquisition
Hemodynamic response in the posterior circulation was assessed using 7 T functional MRI (7T-fMRI), measuring visually stimulated BOLD amplitude, time to peak (TTP), and time to baseline (TTB). WMH quantification was performed on 3T-MRI. Detailed information on the 7T-fMRI and 3T-MRI scanning protocols is provided in the Supplemental Material.
BOLD analysis
7T-fMRI data were analysed to quantify hemodynamic response within the primary visual cortex (V1). Preprocessing and analysis were performed using the FMRIB Software Library (FSL) and custom MATLAB scripts. The V1 region of interest (ROI) was defined anatomically using the Juelich Histological Atlas.
For each participant, mean BOLD responses were derived from the V1 ROI and fitted to a double Gaussian function to determine three outcome measures: BOLD amplitude (reflecting vasodilation), TTP (time from stimulus onset to maximum vasodilation), and TTB (time from stimulus end to return to baseline). All responses were visually inspected, and datasets with artifacts were excluded.
For details see the Supplementary Material.
WMH volume
A semi-automated segmentation procedure was used to identify WMH, which were subsequently verified through visual inspection. Two trained raters (AEW and NvdW) independently assessed lesion volumes under guidance of a senior neuroradiologist (MCK, >20 years of experience). Detailed information can be found in the Supplemental Material.
Statistical analysis
Descriptive statistics were used to calculate frequency, medians, and means of baseline characteristics. Our primary endpoint, differences between the three groups in BOLD parameters (amplitude, TTP and TTB), was assessed with ANCOVA (jmv package, version 2.3.4). Further, our secondary endpoint, associations between BOLD parameters with WMH volume, was assessed by linear regression analysis for all participants combined and for the three groups separately. Analyses were adjusted for age, hypertension, hypercholesterolemia, smoking (ever), and menopause status. Because all participants with hypercholesterolemia used a statin no additional corrections were made for statin use. Amplitude was log-transformed, and WMH volumes were square-root transformed to get a normal distribution.
Given the relatively small sample size, this study was considered exploratory. Therefore, all p-values are reported uncorrected, and no adjustment for multiple comparisons was applied, as many outcomes are interrelated. Borderline statistically significant findings were interpreted with caution. A p-value < 0.05 was considered statistically significant. All statistical analyses were performed in R version 4.2.1.
Results
We included a total of 87 women: 25 with a history of ischemic stroke (group I; mean age 51 years), 25 with migraine with aura (group II; mean age 52 years), and 37 without ischemic stroke or migraine (group III; mean age 53 years) (Table 1). Among the 25 women with ischemic stroke, 6 had large-artery atherosclerosis, 1 a cardioembolic source, 6 small-vessel disease, 6 other determined etiologies, and 6 strokes of undetermined origin. On average, women with ischemic stroke were enrolled 5 years (SD 5) after their event. Women with ischemic stroke more frequently had hypertension, hypercholesterolemia, and a history of smoking compared with women with migraine with aura or those without stroke or migraine. They were less often postmenopausal and had less frequent ever use of oral contraceptives.
Baseline characteristics.
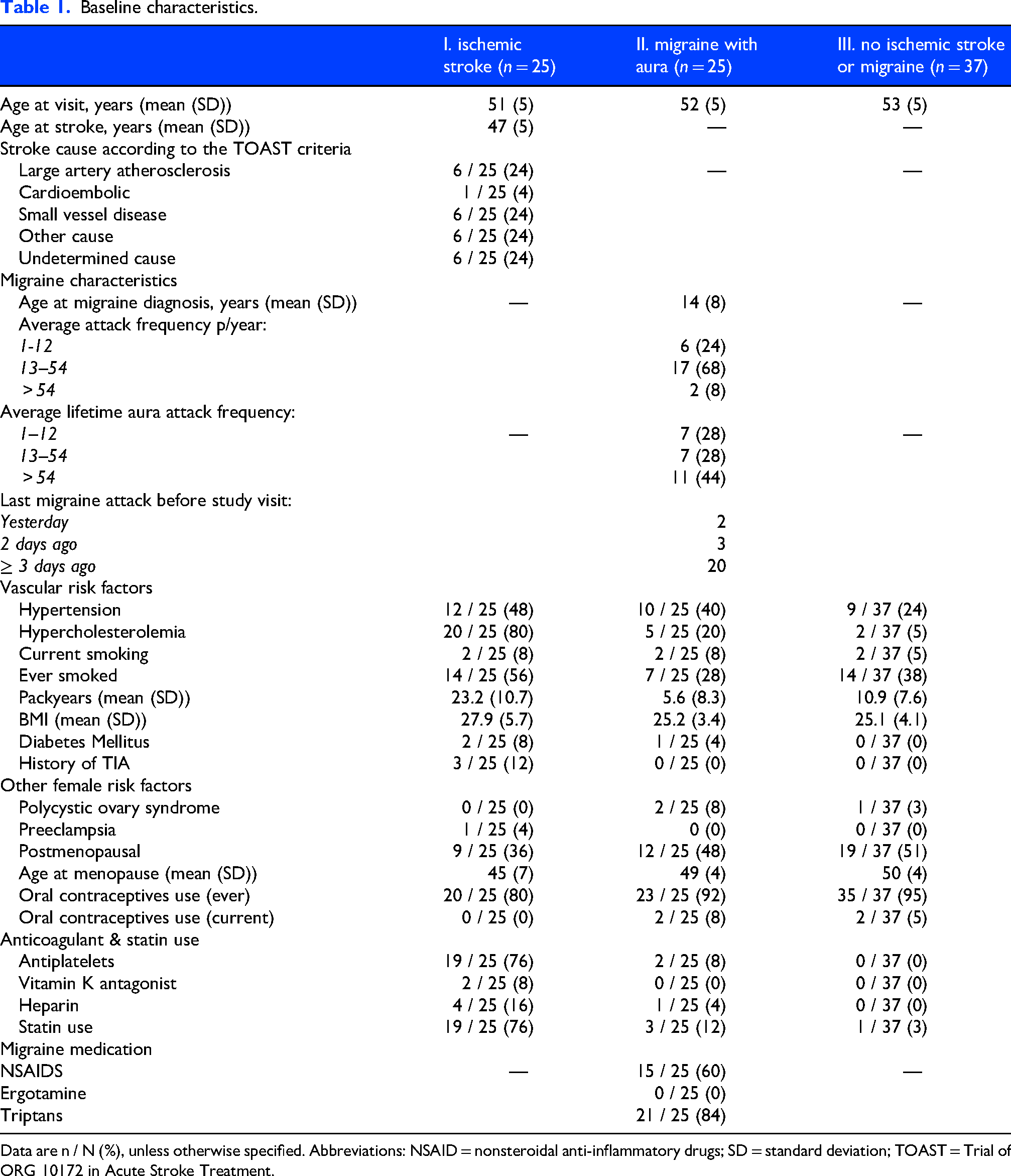
Data are n / N (%), unless otherwise specified. Abbreviations: NSAID = nonsteroidal anti-inflammatory drugs; SD = standard deviation; TOAST = Trial of ORG 10172 in Acute Stroke Treatment.
Visually-induced hemodynamic response
Comparison of BOLD parameters showed no statistically significant differences among the three groups in amplitude (p = 0.397), TTP (p = 0.163), or TTB (p = 0.119) (Table 2). Women with ischemic stroke (group I) had higher periventricular WMH volumes than both women with migraine with aura (group II; p = 0.02) and those without stroke or migraine (group III; p = 0.01). Deep WMH volume did not differ significantly among groups (p = 0.442).
Association between BOLD parameters and white matter hyperintensities volume.

Adjusted for age, hypertension, hypercholesterolemia, smoking (ever), and menopause status.
Abbreviations: A.U. = arbitrary units; WMH = white matter hyperintensities.
BOLD parameters are presented as mean (SD); WMH volumes as median (IQR).
*Tukey post hoc test: statistically significant differences between group I and III (p = 0.01) and between group I and group II (p = 0.02).
Visually-induced hemodynamic response and WMH volume
Overall, BOLD parameters were not associated with WMH volume across all participants. However, exploratory analyses suggested that lower amplitude was associated with higher deep WMH volume in women without stroke or migraine (group III; β = –0.28, 95% CI −0.53 to −0.02 mL), but not in women with ischemic stroke (group I; adjusted β = –0.06, 95% CI −0.24 to 0.12 mL) or migraine with aura (group II; β = 0.06, 95% CI −0.88 to 1.00 mL) (Table 3). In contrast, longer TTP was associated with greater periventricular WMH volume in women with ischemic stroke (group I; β = 0.23, 95% CI 0.07 to 0.39 mL), but not in those with migraine with aura (group II; β = 0.08, 95% CI −0.06 to 0.23 mL) or without stroke or migraine (group III; β = 0.06, 95% CI −0.05 to 0.16 mL). Finally, shorter TTB was associated with higher periventricular WMH volume in women with migraine with aura (group II; β = –0.12, 95% CI −0.21 to −0.02 mL), whereas no associations were found in women with ischemic stroke (group I; β = 0.05, 95% CI −0.04 to 0.15 mL) or without stroke or migraine (group III; β = 0.04, 95% CI −0.04 to 0.13 mL).
Association between BOLD parameters and white matter hyperintensities volume.

Note: β coefficients were calculated using linear regression models adjusted for age, hypertension, hypercholesterolemia, smoking (ever), and menopausal status.
“All” refers to all participants combined (n = 87); group I = ischemic stroke (n = 25); group II = migraine with aura (n = 25); group III = no ischemic stroke or migraine (n = 37).
Discussion
In this study, we found no overall differences in hemodynamic response among middle-aged women with a history of ischemic stroke, migraine with aura, or without either condition in response to a visual stimulus. Likewise, visually-induced hemodynamic response measures were not associated with WMH volumes overall. When analysing groups separately, lower BOLD amplitude was associated with higher WMH volume in women without stroke or migraine, longer TTP in women with ischemic stroke, and shorter TTB in women with migraine with aura. Given the modest subgroup sizes, these findings should be interpreted as exploratory.
Few studies have examined visually-induced hemodynamic response in migraine or ischemic stroke. Previous transcranial Doppler studies assessing hemodynamic response during visual stimulation in the posterior circulation have reported heterogeneous findings in migraine, with some studies suggesting enhanced responses, particularly in migraine with aura, while pooled analyses show no consistent overall difference compared with controls. 16 In addition, only one fMRI study has assessed visually-induced hemodynamic response during cognitive tasks, reporting region-specific alterations with reduced responses in parietal regions and increased coupling in occipital areas, indicating heterogeneous and task-dependent neurovascular changes. 26
In ischemic stroke, two previous fMRI studies reported alterations in BOLD responses, including delayed peak latency and reduced signal amplitude, which were attributed to impaired cerebral hemodynamics and autoregulation rather than neuronal dysfunction.17,18 However, these studies included both men and women, had very small sample sizes, and differed substantially in patient selection and stroke phase, limiting direct comparability with the present study.
Reduced BOLD-derived visually-induced hemodynamic response parameters have previously been associated with greater WMH volume in ischemic stroke. 27 Although we observed no difference in amplitude, longer TTP in our ischemic stroke group was related to higher periventricular WMH volume, a finding that warrants confirmation. We found a possible association between shorter TTB and greater periventricular WMH volume in women with migraine with aura, but this finding may be due to chance given the limited sample size.
Hemodynamic regulation differs between the anterior and posterior cerebral circulation, with the posterior circulation being more vulnerable to disturbances in cerebrovascular control. 16 In migraine with aura, previous studies have predominantly reported abnormalities in the posterior circulation, including altered cerebrovascular reactivity and hemodynamic response during visual stimulation, whereas findings in the anterior circulation are less consistent. This posterior vulnerability may be related to the high metabolic demand of the visual cortex and regional differences in vascular regulation. 16 Accordingly, the visual stimulation paradigm used in our study primarily probes hemodynamic response within the posterior cerebral artery territory, which is particularly relevant in migraine with aura. 16
A previous study showed impaired cerebrovascular reactivity as one of the primary pathophysiological mechanisms of WMH. 28 In contrast, a recent large prospective study assessed cerebral vasomotor reactivity using transcranial Doppler breath-holding index and found that vasomotor reactivity was preserved in the acute phase of non-disabling stroke/TIA and did not differ across stroke etiologic subtypes (large-artery atherosclerosis, cardioembolism, or small-vessel disease). 29 Vasomotor reactivity declined from the acute phase to 6-month follow-up, particularly in the middle cerebral artery, independent of stroke etiology, vascular risk factors, or initial stroke severity. These findings suggest that elevated vasomotor reactivity in the acute phase reflects transient hemodynamic compensation, likely mediated by collateral circulation, rather than stroke subtype–specific mechanisms.
Our results cautiously suggest that there might be different underlying features of visually-induced hemodynamic response for patients with ischemic stroke and patients with migraine with aura. The association between prolonged TTP and WMH in ischemic stroke could reflect delayed vascular response due to endothelial dysfunction. In contrast, the association between shorter TTB and WMH in migraine with aura may reflect altered vascular recovery dynamics rather than preserved vascular health. TTB represents the recovery phase of the hemodynamic response, and a shorter TTB may indicate accelerated or dysregulated microvascular relaxation and clearance of deoxygenated blood. In migraine with aura, repeated episodes of cortical spreading depolarization and transient vascular perturbations could induce maladaptive changes in the neurovascular unit, leading to abnormal vascular timing characteristics and chronic microvascular stress that may contribute to periventricular WMH development.11,30 Given the exploratory nature of these analyses and the small subgroup size, this interpretation should be considered hypothesis-generating and requires confirmation in larger longitudinal studies.
Our study has several limitations. First, the sample sizes in each group were small, which limits statistical power and makes the subgroup analyses exploratory. Given this exploratory design, we did not apply correction for multiple testing. 31 It is therefore possible that the study was underpowered to detect true associations, and the significant subgroup findings, based on borderline p-values, should be interpreted with caution and confirmed in larger cohorts. Despite these limitations, the study provides substantial added value due to the unique inclusion of well-characterized middle-aged women with ischemic stroke and migraine with aura, combined with high-quality fMRI to assess visually-induced BOLD responses. This unique combination of participant groups and detailed hemodynamic imaging has not been previously reported and offers novel mechanistic insights that warrant further investigation in larger cohorts. Second, the BOLD response was measured only in the visual cortex, primarily reflecting posterior circulation hemodynamic response, whereas WMH volume was quantified across the whole brain. This may limit spatial correspondence between the measures. However, BOLD fMRI in the visual cortex is a highly reproducible and well-validated method for assessing visually-induced hemodynamic response. Third, research visits were rescheduled if participants experienced a migraine attack on the day of scanning. Most participants had their last migraine attack ≥2 days before the research visit; however, two participants were scanned within 1–2 days of an attack. Information on prodromal and postdromal migraine phases was not systematically collected, and hemodynamic response may have been altered in these participants, potentially attenuating the observed associations. Fourth, no formal washout period for acute migraine medications, including triptans, was applied prior to scanning. Given the vasoconstrictive properties of triptans, recent use could theoretically influence BOLD-derived visually-induced hemodynamic response measures. However, visits were rescheduled in case of an active migraine on the day of scanning, most participants were scanned ≥2 days after their last attack, and triptans have relatively short-lasting pharmacological effects.
Our study also has several strengths. First, we used a reliable visual stimulus—a flickering checkerboard—that induces a robust and reproducible BOLD response and has demonstrated good performance in small vessel disease research.27,28,32 Second, we adjusted for vascular risk factors and menopause status, which are often not accounted for in similar studies, and we used a homogeneous population of middle-aged women to reduce confounding. Thirdly, we determined WMH volume on 3 T MRI, maintaining the relevance to WMH seen clinically. WMH seen on 7 T MRI are not yet well explored and their clinical interpretation remains unknown.
Conclusions
This exploratory study did not demonstrate meaningful differences in visually-induced hemodynamic response, measured with 7 T BOLD-MRI, among middle-aged women with ischemic stroke, migraine with aura, or neither condition. We also found no overall association between visually-induced hemodynamic response and WMH volume. However, our subgroup findings suggest that the relationship between visually-induced hemodynamic response and WMH development may differ between women with ischemic stroke and those with migraine but this requires confirmation in larger studies.
Clinical implications
Visually-induced hemodynamic response did not differ between middle-aged women with ischemic stroke, migraine with aura, and patients with neither condition.
No overall association was found between visually-induced hemodynamic response and WMH volume.
Subgroup analyses suggest different visually-induced hemodynamic response across groups; longitudinal studies are needed to clarify temporal or mechanistic links to WMH development.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261439256 - Supplemental material for Visually-induced hemodynamic response and white matter hyperintensities in middle-aged women with ischemic stroke or migraine with aura
Supplemental material, sj-docx-1-cep-10.1177_03331024261439256 for Visually-induced hemodynamic response and white matter hyperintensities in middle-aged women with ischemic stroke or migraine with aura by Nelleke van der Weerd, Mariam Ali, Thijs W. van Harten, Ay-Min Ma, Ghislaine Holswilder, Annelise E. Wilms, Erik W. van Zwet, Mark C. Kruit, Arn M.J.M. van den Maagdenberg, Gisela M. Terwindt, Matthias J.P. van Osch, Hendrikus J.A. van Os and Marieke J. H. Wermer in Cephalalgia
Footnotes
Acknowledgements
We thank Jaap L. Kappelle (University Medical Center Utrecht) for referring patients and Bart Zick and Katie M. Linstra (LUMC) for their assistance with patient inclusion and MRI scanning.
Ethical considerations
The Medical Ethics Committee Leiden–Den Haag–Delft approved the study protocols for CREW-MIST (P15.384) and WHISPER (P18.130).
Consent to participate
Written informed consent was obtained from all participants.
Consent for publishing
The authors agree to publish with Cephalalgia.
Author contributions
N. van der Weerd: Methodology; Data curation; Formal analysis; Writing – original draft; Writing – review & editing. M. Ali: Data curation; Writing – original draft; Writing – review & editing. T.W. van Harten: Methodology; Investigation; Data curation; Formal analysis; Writing – review & editing. A. Ma: Data curation; Formal analysis. G. Holswilder: Methodology; Investigation; Data curation; Project administration; Writing – review & editing. A.E. Wilms: Investigation; Data curation. E.W. van Zwet: Formal analysis; Writing – review & editing. M.C. Kruit: Methodology; Investigation; Validation; Data curation; Resources; Supervision; Writing – review & editing. A.M.J.M. van den Maagdenberg: Supervision; Writing – review & editing. G.M. Terwindt: Conceptualization; Methodology; Investigation; Data curation; Validation; Visualization; Resources; Funding acquisition; Project administration; Supervision; Writing – original draft; Writing – review & editing. M.J.P. van Osch: Methodology; Interpretation; Writing – review & editing. H.J.A. van Os: Conceptualization; Methodology; Data curation; Writing – review & editing. M.J.H. Wermer: Conceptualization; Methodology; Investigation; Funding acquisition; Supervision; Writing – review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The CREW project was funded by the Dutch Heart Foundation (2013T083). The WHISPER project received funding through a personal grant to M.J.H.W. (VIDI 91717337, ZonMw/NWO). The funders had no role in the design, data collection, analysis, or reporting of this study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.M.T. reports independent support from the Dutch Research Council, Dutch Heart Foundation, Dutch Brain Foundation, Dioraphte, Clayco Foundation, and consultancy for Novartis, Lilly, Teva, AbbVie, Organon, Pfizer, and Lundbeck. A.E.W. and G.M.T. report grant support from the European Community (101070917), Stichting Dioraphte (20010407), and Clayco Foundation. A.M.V.D.B. received honoraria and research/travel grants from Allergan/AbbVie, Amgen/Novartis, Eli Lilly, Manistee, Pfizer, Satsuma, Teva, and Tonix, as well as independent support from the Dutch Research Council and ZonMw. M.J.P. van Osch reports support from a NWO-VICI grant (016.160.351), a NWO-Human Measurement Models 2.0 grant (18969), and additional support from the Dutch Research Council, European Community, Dutch Heart Foundation, and Dutch Brain Foundation. M.J.P. van Osch and T.W. van Harten also received support from the Dutch Heart Foundation and the Netherlands Organisation for Scientific Research (NWO) as part of their joint strategic research programme Earlier recognition of cardiovascular diseases.
Data availability statement
The data supporting the findings of this study are not publicly available due to privacy regulations protecting research participants. Further information about the dataset is available from the corresponding author upon reasonable request.
Open practices
Not applicable.
The CREW consortium (in alphabetical order)
Yolande Appelman, Sara Baart, Laura Benschop, Eric Boersma, Laura Brouwers, Ricardo Budde, Suzanne Cannegieter, Veerle Dam, Rene Eijkemans, Bart Fauser, Michel Ferrari, Arie Franx, Christianne de Groot, Marlise Gunning, Annemieke Hoek, Erik Koffijberg, Wendy Koster, Mark C. Kruit, Giske Lagerweij, Nils Lambalk, Joop Laven, Katie Linstra, Aad van der Lugt, Angela Maas, Antoinette Maassen van den Brink, Cindy Meun, Saskia Middeldorp, Karel GM Moons, Bas van Rijn, Jeanine Roeters van Lennep, Jolien Roos-Hesselink, Luuk Scheres, Yvonne T. van der Schouw, Eric Steegers, Regine Steegers, Gisela Terwindt, Birgitta Velthuis, Marieke Wermer, Bart Zick, Gerbrand Zoet
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
