Abstract
Background
The role of central pulsatile hemodynamics in the pathogenesis of white matter hyperintensities in migraine patients has not been clarified.
Methods
Sixty patients with migraine (20–50 years old; women, 68%) without overt vascular risk factors and 30 demographically-matched healthy controls were recruited prospectively. Cerebral white matter hyperintensities volume was determined by T1-weighted magnetic resonance imaging with CUBE-fluid-attenuated-inversion-recovery sequences. Central systolic blood pressure, carotid-femoral pulse wave velocity, and carotid augmentation index were measured by applanation tonometry. Carotid pulsatility index was derived from Doppler ultrasound carotid artery flow analysis.
Results
Compared to the controls, the migraine patients had higher white matter hyperintensities frequency (odds ratio, 2.75; p = 0.04) and greater mean white matter hyperintensities volume (0.174 vs. 0.049, cm3, p = 0.04). Multivariable regression analysis showed that white matter hyperintensities volume in migraine patients was positively associated with central systolic blood pressure (p = 0.04) and carotid-femoral pulse wave velocity (p < 0.001), but negatively associated with carotid pulsatility index (p = 0.04) after controlling for potential confounding factors. The interaction effects observed indicated that the influence of carotid-femoral pulse wave velocity (p = 0.004) and central systolic blood pressure (p = 0.03) on white matter hyperintensities formation was greater for the lower-carotid pulsatility index subgroup of migraine patients. White matter hyperintensities volume in migraine patients increased with decreasing carotid pulsatility index and with increasing central systolic blood pressure or carotid-femoral pulse wave velocity.
Conclusions
White matter hyperintensities are more common in patients with migraine than in healthy controls. Increased aortic stiffness or central systolic blood pressure in the presence of low intracranial artery resistance may predispose patients with migraine to white matter hyperintensities formation.
Keywords
Introduction
Regions of white matter with signal intensities exceeding that of normal white matter in T2-weighted and fluid-attenuated inversion recovery sequence (FLAIR) processed brain magnetic resonance imaging (MRI) scans are known as cerebral white matter hyperintensities (WMHs) (1). WMHs have been associated with increased risk of stroke, dementia, major cardiovascular diseases, and morbidity (1,2). A wide range of WMHs frequencies (4–71%) have been reported for migraine patients, as well as for the corresponding controls (1–69%) (3–5). Migraine patients have been reported to be more likely to have WMHs than controls without headache (1.87–2.6 greater odds), especially in females (4,6–8). However, results pertaining to this potential association have been inconsistent. Gaist et al. found no association between WMHs presence and migraine with aura (9). Meanwhile, data from the Atherosclerosis Risk in Communities brain MRI study indicate that migraine diagnosis was not associated with WMHs progression over time (6).
Increased central blood pressure (CBP), aortic stiffness, and intracranial vascular resistance have been associated with WMHs formation in non-migraine populations (10,11). Stiffening of the central aortic vasculature reduces the vessels’ ability to cushion pulsatile blood flow from the heart to the brain (12,13). In the community-based Age, Gene/Environment Susceptibility – Reykjavik study, it was hypothesized that excessive flow pulsatility caused by increased aortic stiffness may damage cerebral microcirculation, thereby leading to tissue damage reflected by WMHs (11). Indeed, higher aortic stiffness was shown be associated with measures of reduced white matter integrity in young healthy adults in the Framingham Heart Study (14).
The pathophysiology of WMHs development in migraine patients remains unclear. Increased aortic stiffness in patients with migraine, relative to non-migraine controls, has been reported recently (15,16). However, the roles of aortic stiffness and intracranial vascular resistance in WMHs pathogenesis in migraine patients have never been explored. The primary aim of the present study was to investigate whether WMHs formation is associated with CBP or aortic stiffness (primary outcomes) in young migraine patients. Secondarily, we explored whether WMHs presence correlates with other potential vascular variables (secondary outcomes) in migraine patients, including carotid pulsatility index (CPI), which has been established as a useful index of intracranial arterial conditions (17) and demonstrated to be associated with cardiovascular disease (17–19). We hypothesize that greater WMHs volume may be associated with a more severe migraine profile and enhanced central pulsatile hemodynamics in patients with migraine.
Methods
Participants
We recruited patients with migraine with or without aura diagnosed by board-certified neurologists at the Headache Clinic at Taipei-Veterans General Hospital in accordance with the International Classification of Headache Disorders, 3rd edition, beta version (ICHD-3 beta) (20). Sex- and age-matched (±3 years) healthy controls were volunteers who had neither a personal or family history of migraine nor any headache attack within the past year.
Recruited patients underwent a detailed clinical interview and a neurological examination during their first visit to the clinic. Questionnaire-based interviews were carried out to collect demographic and clinical information, including disease duration, and medication use, as well as migraine frequency, duration, intensity, location, quality, triggers, and associated symptoms. Patients were asked to indicate the average intensity of their headaches over the past three months on a numeric rating scale ranging from 0 (no pain) to 10 (worst pain imaginable) (21).
To eliminate potentially confounding factors, the following exclusion criteria were applied: age <20 or >50 years; body mass index (BMI) <18 or >30 kg/m2; history of cardiovascular disease, hypertension (brachial systolic blood pressure (SBP) >140 mm Hg or diastolic blood pressure (DBP) >90 mm Hg), diabetes (diagnosed and treated with diet or drugs or both; or a fasting serum glucose >126 mg/dl), hyperlipidemia (cholesterol level >240 mg/dl, low density lipoprotein >150 mg/dl, triglyceride level >150 mg/dl, or history of hyperlipidemia), or stroke; heavy smoker (>20 cigarettes/day) (22); current pregnancy or lactation; regular use of vasoactive drugs (e.g. nitrates, β-blockers and Ca2+-channel blockers); MRI contraindication; infectious or inflammatory disease within a month of study commencement; major psychiatric disorder; or history of substance abuse. Patients with medication overuse headache (23), chronic migraine (24), or who use migraine preventive agents were also excluded. Additionally, any history of migraine was an exclusion criterion for controls; infrequent episodic tension-type headache (<1 headache day/month) was allowed. Eligible subjects were examined after at least a full day of being headache-free. If an attack developed within 24 hours after the examination, the participant was re-scheduled. Subjects were asked not to use analgesics, non-steroidal anti-inflammatory agents, ergots, or triptans for at least three days, and also to abstain from caffeine, alcoholic beverages, and smoking for at least six hours, before their examinations. No medications were allowed within the month before the examination. All examinations were completed within a six-week time interval.
Brain MRI
Imaging data were acquired at Taipei-Veterans General Hospital on a 3-T GE Discovery MR750 (GE Healthcare, Waukesha, WI) scanner. To diminish motion artifacts during scanning, each participant’s head was immobilized with cushions inside the coil. An anatomical image was acquired with an axial three-dimensional (3D) T1-weighted, inversion-recovery-prepared gradient echo sequence [BRAVO, BRAin VOlumetry, GE Healthcare; repetition time (TR)/echo time (TE)/inversion time (TI)/flip angle (FA): 9.2 ms/3.7 ms/450 ms/12°] with the following parameters: matrix, 256 × 256 × 256; field of view, 256 × 256 mm2; slice thickness, 1 mm. All imaging was reviewed and individuals with structural abnormalities revealed on T1-weighted images were excluded. To quantify the total volume of white matter lesions, a sequence of iso-centric sagittal images produced with a 3D CUBE-FLAIR (25) (TR/TE/TI: 6000 ms/maximum/50 ms) was applied with the above-mentioned matrix, field of view, and slice thickness parameters.
MRI data processing
Total WMHs volumes (in cm3) were calculated automatically with the Lesion Segmentation Tool (http://www.applied-statistics.de/lst.html) in the Computational Anatomy Toolbox (CAT12, updated version of VBM8; http://dbm.neuro.uni-jena.de/vbm) running under SPM12 software (http://www.fil.ion.ucl.ac.uk/spm) with all default software settings (26,27). The volumes were calculated based on both anatomical and FLAIR images. Detected WMHs that were within 10 mm distance from the ventricular system were considered to be periventricular; all others were classified as deep WMHs (4,6).
Hemodynamic measures
Vascular measurements were performed in the afternoon in a quiet, temperature-controlled (24 ± 1℃) room. All subjects were scanned while lying in a supine position. They were asked to fast for ≥4 hours before and during the examination. We employed a previously described hemodynamic protocol (28). A cardiovascular monitoring system (VP-2000; Colin, Komaki, Japan) was employed with four pressure cuffs and two tonometric probes for collection of physiological data, including electrocardiogram, phonocardiogram, oscillometric signals from the arms and ankles, as well as tonometric signals from the right common carotid and right femoral artery (28). After resting for 10 minutes, brachial SBP, brachial DBP, heart rate (HR), pulse volume recording waveforms, and pressure waveforms of the right common carotid and femoral arteries were recorded simultaneously (28). All signals were digitized simultaneously (sampling frequency, 250 Hz) for off-line analysis (29). Brachial pulse pressure (PP) was determined by calculating the difference between SBP and DBP, and mean arterial blood pressure (MBP) was calculated as: DBP + 1/3 PP. Bilateral common carotid artery images and flows were assessed after a 10-minute quiet rest while subjects were in a head-straight, flat-supine position with an ultrasound system and 7 MHz linear transducer (iU22; Philips Medical Systems, Andover, MA). To optimize accuracy, three separate measures were obtained on both sides of the neck, with probe removal and relocation between the measures.
Carotid-femoral pulse wave velocity (cf-PWV)
Large artery stiffness was indexed by measurement of cf-PWV, an independent predictor of all-cause and cardiovascular mortality (28). cf-PWV was calculated as the distance between two arterial recording sites divided by the pulse transit time between the right carotid and right femoral arteries (29). Simultaneously digitized tonometric waveforms were analyzed with customized software to identify the foot of each cardiac-cycle waveform (29).
Central blood pressure (CBP) and augmentation index
CBP has been shown to be more important than peripheral blood pressure in predicting all-cause mortality, cardiovascular mortality, and cognitive impairment (11,30). Central SBP (cSBP) and central DBP (cDBP) were derived by an ensemble averaged carotid artery pressure waveform calibrated to brachial DBP and MBP (28). Central pulse pressure (cPP) and central MBP (cMBP) were determined from cSBP and cDBP according to the relationships defined above. The inflection point of the wave reflection on the upstroke or down-stroke of the waveform was identified by finding the zero-crossing timings of the fourth derivative of the pressure waveform (31). Augmentation index (AI) and augmented pressure (AP), which are wave reflection measures that are important determinants of cardiovascular, cerebrovascular risk and all-cause mortality (29,32), were then calculated as described previously (29).
Carotid pulsatility index (CPI)
We included CPI as an exploratory variable because it has been used to assess intracranial arterial condition and shown to correlate with aortic stiffness and the Framingham 10-year cardiovascular risk (17–19). It is a measure of downstream resistance to blood flow based on Doppler waveform analysis (33). Downstream arterial vasodilatation can result in a low pulsatility index due to reduced distal vascular resistance (34). We computed CPI for each carotid artery by dividing flow pulse amplitude (peak flow minus flow at the onset of the systolic up-stroke) by mean flow, using values derived from carotid ultrasonography (17). Right and left CPI values were averaged for each subject.
Both the investigator who conducted the MRI quantifications and the technician who performed the vascular tests were blinded to the diagnosis of the participants.
Statistical analysis
Based on our preliminary results and under the assumption that WMHs frequency in migraine patients would be 50%, a total sample size of 48 patients (24 with and 24 without WMHs) was calculated to have 80% power to detect significant differences in mean CBP by an independent t-test with a two-tailed 0.05 significance level. Accounting for a potential attrition rate of 10%, we determined that we would need a total sample size of at least 56 (28 patients in each group). We elected to recruit 60 patients with migraine in our study. In addition, for comparisons, we also recruited 30 age- and sex-matched healthy controls.
All statistical analyses were performed in SPSS version 18.0 (SPSS, Chicago, IL). Continuous variables were compared between groups by t-tests or one-way analyses of variance (ANOVAs). Categorical variables were compared between groups with the chi-square test. Pearson correlation coefficients were calculated to detect associations between WMHs volume, clinical characteristics, and arterial parameters. The primary outcomes of the study were associations of WMH formation with CBP or aortic stiffness; the secondary outcomes were correlations or interactions between WMH formation and other vascular variables. Univariate and multivariable regression analyses were used to detect associations between arterial parameters and WMHs load after controlling for possible confounding factors. Bonferroni’s correction for multiple comparisons was applied in the univariate analyses. One-way ANOVA followed by Scheffé’s post-hoc analysis was used to identify differences in vascular parameters between WMH-volume distribution tertile subgroups of migraine patients. Two-way ANOVAs were conducted to examine the interaction effects of CBP and aortic stiffness on WMHs formation between low-CPI and high-CPI migraine patient groups (divided at median CPI value). Because we did not establish an a priori strategy for stratifying the groups of patients, the results of this part of the analysis are considered hypothesis generating. Statistical significance was defined as p < 0.05, or as adjusted for the multiple-comparison Bonferroni correction. All tests were two-tailed. Mean values are reported with standard deviations (SDs). Odds ratios (ORs) are reported with 95% confidence intervals (CIs).
Standard protocol approvals, registrations, and patient consents
The study protocol was approved by investigators’ hospital’s Institutional Review Board. Written informed consent was obtained from each participant before entering the study.
Results
Clinic characteristics
A flow diagram of the study participants is provided in Figure 1. The final cohort consisted of 60 migraine patients (mean (SD) age, 31.4 (7.5) years; 68% women) and 30 age- and sex-matched healthy controls (mean (SD) age, 31.7 (6.3) years; 68% women). The two groups were similar in terms of BMI, lipid profile, fasting glucose, and smoking habits (Table 1).
Flow diagram of study participants. Clinical characteristics of study participants. All patients with migraine with aura also had attacks of migraine without aura. Some patients used more than one medication type. Abbreviations: BMI: body mass index; NRS: numeric rating scale.

WMHs volume and arterial parameters between migraine patients and controls
Comparisons of WMH volume, blood pressure measures, heart rate, and vascular parameters between migraine patients and healthy controls.
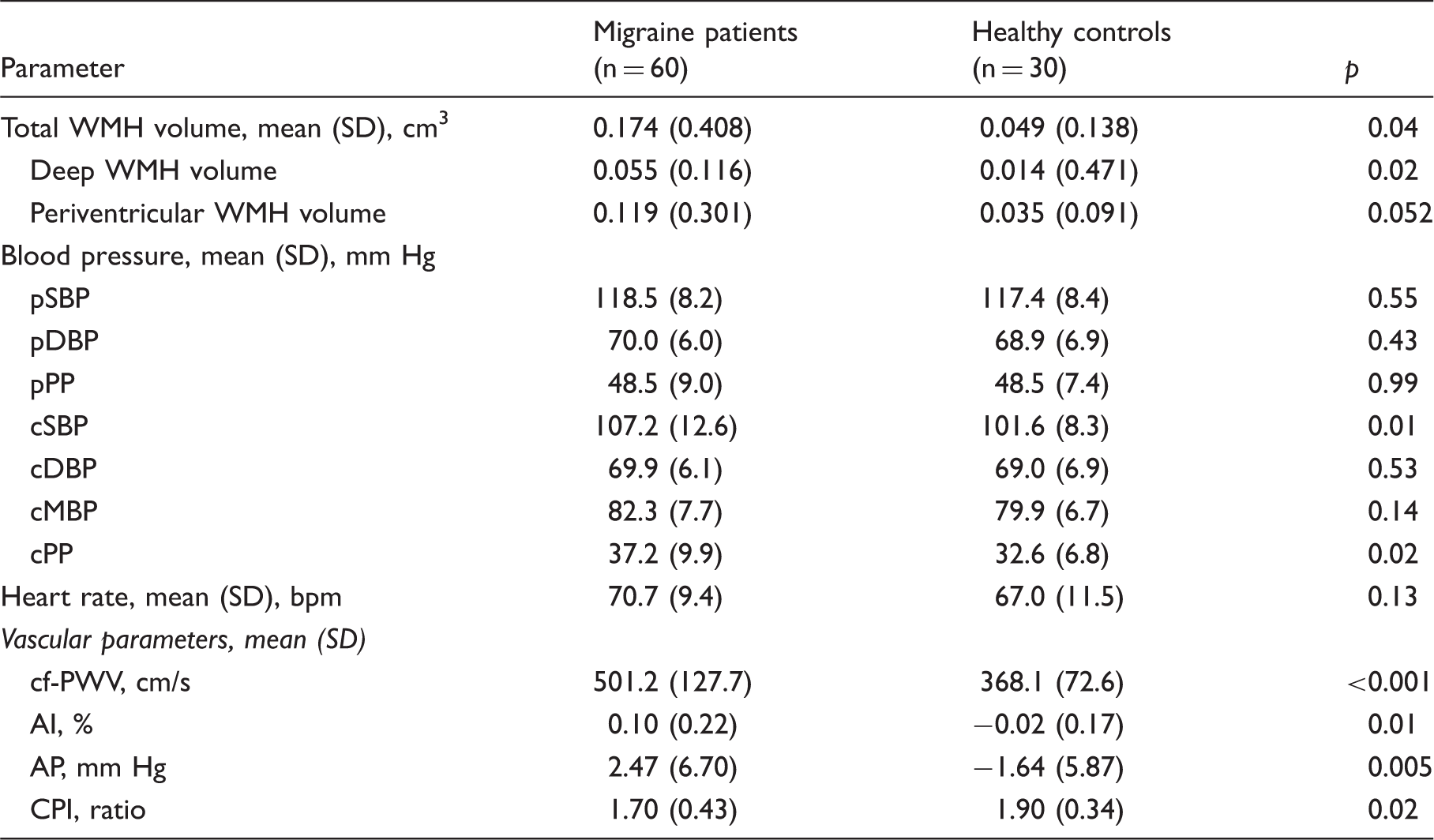
Abbreviations: WMH: white matter hyperintensity; CBP: central blood pressure; pSBP: peripheral (brachial) systolic blood pressure; pDBP: peripheral (brachial) diastolic blood pressure; pPP: peripheral (brachial) pulse pressure; cSBP: central systolic blood pressure; cDBP: central diastolic blood pressure; cMBP: central mean arterial pressure; cPP: central pulse pressure; bpm: beats per minute; cf-PWV: carotid-femoral pulse wave velocity; AI: augmentation index; AP: augmented pressure; CPI: carotid pulsatility index; PSV: peak systolic velocity; EDV: end diastolic velocity.
WMHs load, CBP, arterial stiffness, and CPI in migraine patients
Univariate and multivariable regression analyses between measures of arterial function and WMH load in migraine patients.

The significant variables in univariate analyses, after Bonferroni correction (p < 0.05/9 = 0.0055), were included in the multiple regression analysis with backward elimination. Potential confounding factors (age, sex, BMI, presence/absence of aura and smoking habit) were controlled in the multiple regression model. Instead of cPP, cSBP and cDBP were enrolled into regression model for possible multiple collinearity among CBP variables.
Abbreviations: pSBP: peripheral (brachial) systolic blood pressure; pDBP: peripheral (brachial) diastolic blood pressure; cSBP: central systolic blood pressure; cDBP: central diastolic blood pressure; cf-PWV: carotid-femoral pulse wave velocity; AI: augmentation index; CPI: carotid pulsatility index.
Relation of headache profile to WMHs volume and vascular parameters
Pearson correlation coefficients (r values) of WMH volume and vascular parameters with headache profile variables.
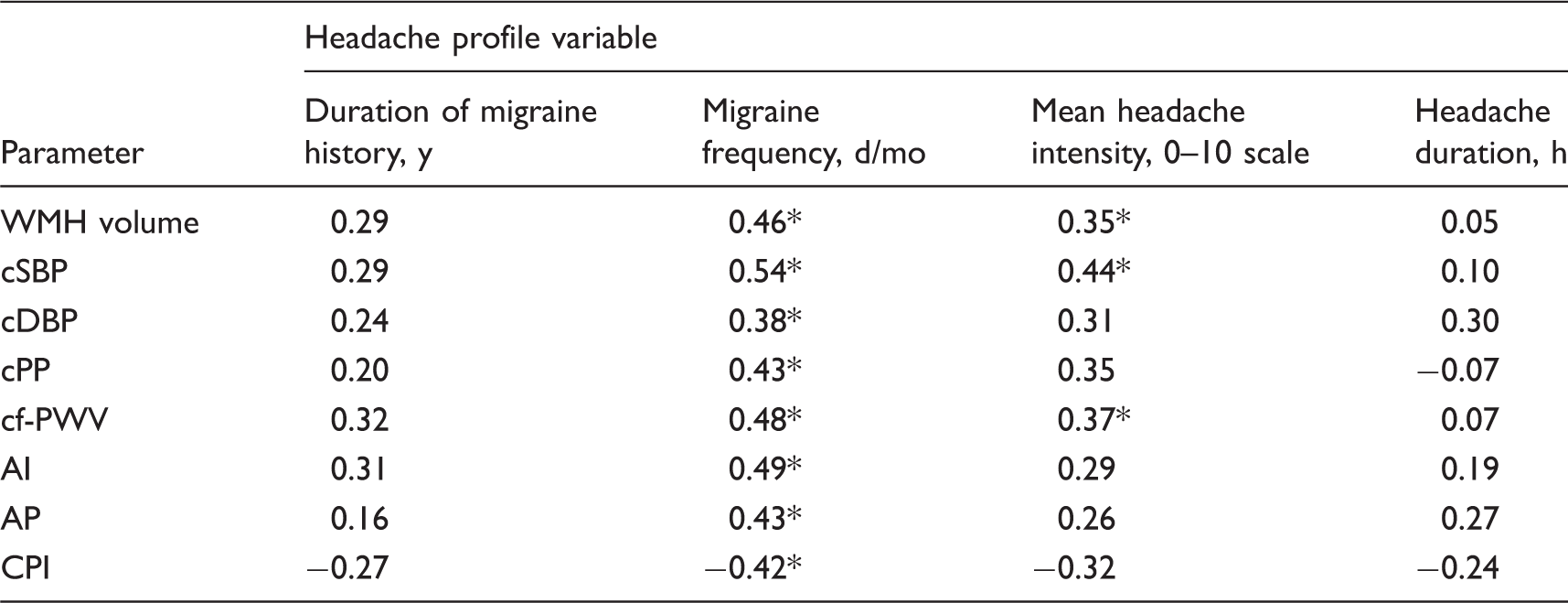
p < 0.0063 (0.05/8 = 0.0063) was considered significant with Bonferroni correction.
Abbreviations: WMH: white matter hyperintensity; cSBP: central systolic blood pressure; cDBP: central diastolic blood pressure; cPP: central pulse pressure; cf-PWV: carotid-femoral pulse wave velocity; AI: augmentation index; AP: augmented pressure; and CPI: carotid pulsatility index.
Exploratory analyses of associations between WMHs volume and vascular parameters
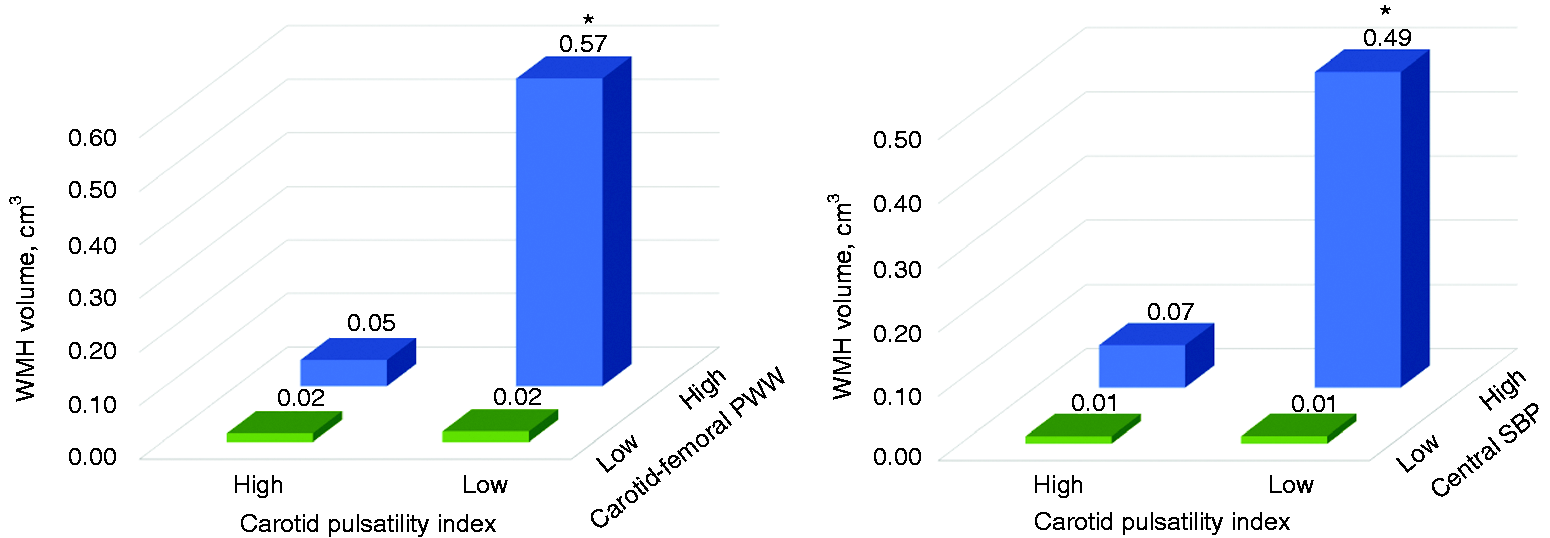
The highest WMH-volume tertile subgroup of migraine patients had a lower CPI (F2,57 = 6.89, p < 0.001) and had a higher AI (F2,57 = 43.52, p < 0.001), cf-PWV (F2,57 = 26.95, p < 0.001), and cSBP (F2,57 = 10.76, p < 0.001) than patients in the middle- and low-tertile subgroups (Figure 2). WMHs volume in migraine patients increased with decreasing CPI and with increasing cSBP or cf-PWV values (Figure 3). We observed significant interaction effects indicating that the influence of cf-PWV (F1,56 = 9.08, p = 0.004) and cSBP (F1,56 = 5.13, p = 0.03) on WMHs presence was greater for the low-CPI subgroup than for the high-CPI subgroup.
Comparisons of CPI, cSBP, AI, and cf-PWV values among WMH-volume tertile subgroups of migraine patients. Comparisons of WMH volume in migraine patients above versus below CPI, cf-PWV, and cSBP medians.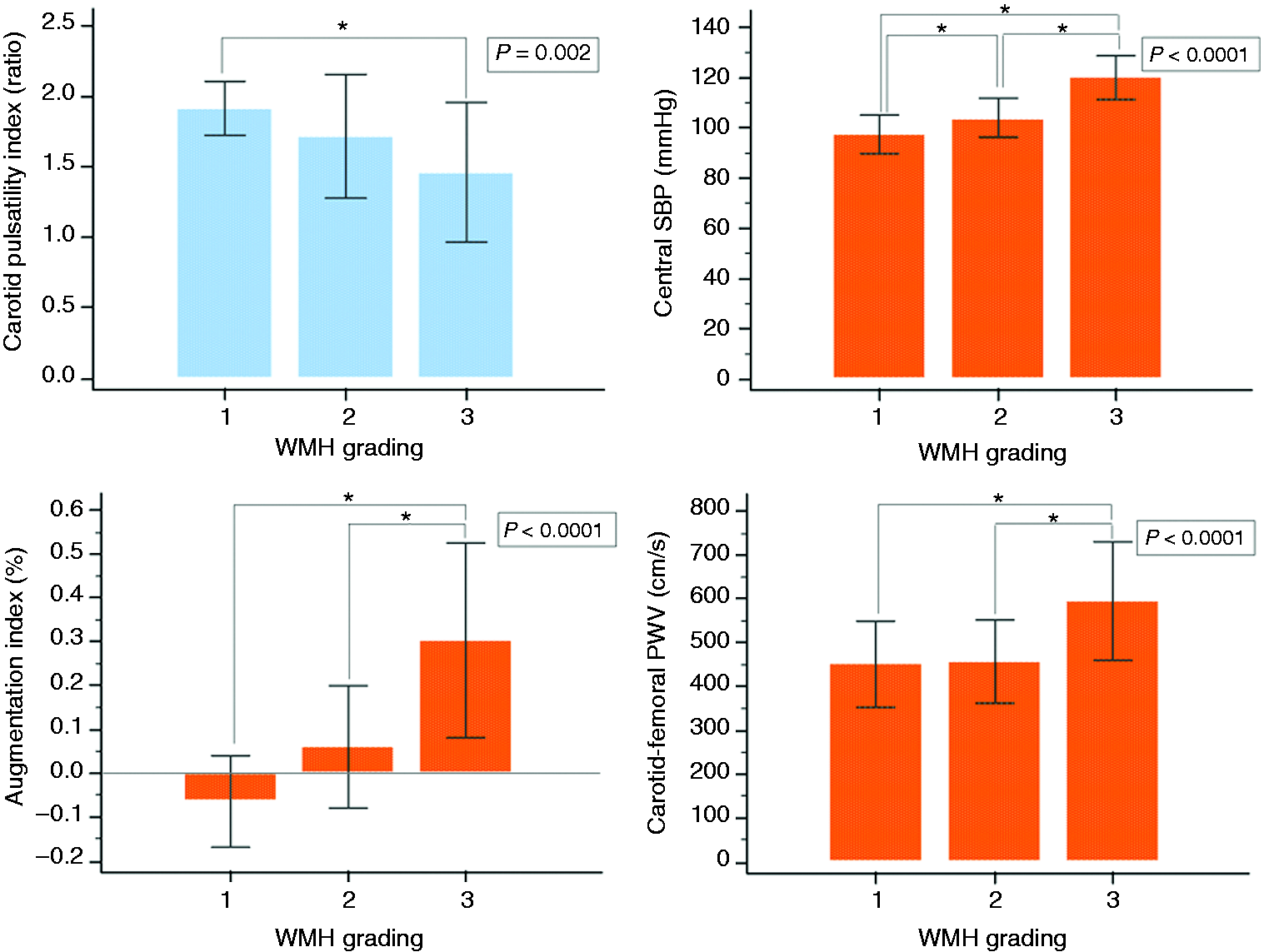
Discussion
The results of the present study demonstrated that, relative to healthy controls, migraine patients were more likely to have WMHs and had greater WMHs volumes. In patients with migraine, a large WMHs volume was associated with more frequent and severe migraine attacks. The presence of WMHs was associated with several hemodynamic parameters, including increased CBP, greater aortic stiffness, and decreased resistance in intracranial arteries. Our novel finding of an association between high WMHs load and low intracranial arterial resistance in migraine patients contrasts with one recent study reporting high intracranial arterial resistance in an elderly cohort with WMHs (35).
Our results are consistent with prior reports of a greater WMHs frequency in migraine patients and an association of high WMHs volume with migraine attack frequency and severity (4,7,8). The elevated cf-PWV, AI, and CBP values obtained for our migraine group are also consistent with prior reports (16). It is noteworthy that, relative to healthy controls, our migraine group exhibited elevated cSBP but similar brachial SBP. CBP in central arteries, such as the ascending aorta and common carotid arteries, is directly related to cardiac load. Because branching of and impedance changes in the arterial system can lead to amplified blood pressure changes in peripheral arteries, peripheral blood pressure is not necessarily an accurate reflection of CBP. Indeed, CBP has been shown to relate more strongly to vascular hypertrophy, extent of atherosclerosis, and cardiovascular events than to peripheral blood pressure (36,37). Although there is evidence suggesting that intracerebral microcirculation changes secondary to aortic stiffening and elevated brachial SBP may lead to WMH formation (11,38), the absolute hemodynamic values obtained for our migraine patients were well within normal limits. It remains to be clarified how slightly elevated hemodynamics in young migraine patients, still within clinically normal ranges, may contribute to WMH formation.
The brain, like the kidney, has the unusual physiological characteristic of continuous, passive high-volume perfusion with very low vascular resistance (39), which makes the brain vasculature highly susceptible to pressure and flow fluctuations in the presence of aortic stiffness (13). Our exploratory finding of a lower CPI in the migraine group than in controls may be related to reduced distal vascular resistance caused by intracranial arterial vasodilatation (34). Intracranial vasodilatation during spontaneous migraine attacks has been observed previously (40) and levels of calcitonin gene-related peptide (a vasodilator) have been reported to be increased during migraine attacks (41) as well as in the interictal period (42). Moreover, reduced CPI of the intracerebral artery has been observed during migraine attacks (43). The presently observed associations of a low CPI with a higher attack frequency and longer migraine history support the notion that low intracranial artery resistance during migraine-free periods might reflect a sustained vasodilatation condition.
We found that, relative to migraine patients with a lower WMHs load, patients with a higher WMHs load had a lower CPI, which is indicative of low intracranial artery resistance (44,45). We observed significant interaction effects indicating that the influence of aortic stiffness on WMHs formation was greater for the low-CPI subgroup. It is possible that these features may predispose young migraine patients to WMHs formation; however, the results still need confirmation in future research. The contrast between the present observation of low intracranial arterial resistance being associated with WMHs formation in our migraine patients versus prior observations relating WMHs changes to high intracranial artery resistance in non-migraine subjects (35,46) suggests that WMHs formation in migraine patients may differ pathophysiologically from aging-related white matter changes.
The present cohort of patients differs from cohorts in prior studies (6,8) in that our patients were younger (range, 20–50 years) and free of clinically significant vascular risk factors. Our strict participant selection criteria enabled us to assemble a homogeneous cohort, with all participants being assessed in a headache-free period without other confounding factors. We employed a previously validated hemodynamic assessment technique (29) and 3D CUBE-FLAIR sequence that is superior to conventional 2D FLAIR for WMH detection owing to its higher contrast-to-noise ratio (25).
Our study has clinical implications. The present data suggest that WMHs load may reflect migraine headache severity and could serve as a marker for cerebral damage resulting from an interaction of elevated central pulsatile hemodynamics with low intracranial resistance. The presence of increased central pulsatile hemodynamics in young patients with migraine implies that the migraine condition may be fundamentally a systemic vascular disease involving intracranial and central aortic vasculature. Our results expand on the findings from a meta-analysis indicating that migraine-susceptible loci show enrichment of genes expressed in vascular tissues, consistent with a vasculature-based etiology of migraine (47). Such a pathophysiology may explain, at least in part, why migraine patients are vulnerable to future cerebrovascular and cardiovascular morbidity (48). Measurements of central pulsatile hemodynamics are useful for cardiovascular risk assessment and prevention because they enable patients with premature vascular aging to be identified (49). Our study expands the utility of such assessments by providing information about the hemodynamic characteristics of young migraine patients without remarkable vascular aging. CBP and vascular parameters may become important markers for cardio- or cerebrovascular risk stratification.
This study had several study limitations. First, because we studied a relatively homogenous group of healthy adults with episodic migraine without known cardiovascular risk factors, it is not known how well these findings would generalize to patients with comorbidities or cardiovascular risk factors, older patients, or patients with chronic migraine or medication-overuse. Second, because we used a 3D CUBE-FLAIR MRI sequence, we were able to detect WMHs changes with a higher sensitivity than can be detected with other methods. Nevertheless, our main findings were based on total WMHs volume, or WMHs load, rather than merely on WMHs frequency. Further investigation is needed to clarify whether the compliance of internal or external carotid systems, which might be altered during migraine attacks, contributes to low CPI in migraine patients. Third, we did not compile systematic data on the patients’ history of oral contraceptive/hormone use, which is a vascular risk factor. Fourth, because of the relatively small sample number of men and of patients with aura, our study did not analyze gender or aura as possible risk factors for WMHs load. Finally, our study employed a cross-sectional design in which WMHs prevalence, rather than incidence, was determined. Therefore, additional work is warranted to ascertain whether the associations observed in this study represent a causal relationship.
In conclusion, the present results indicate that WMHs are more common in patients with migraine than in healthy controls and that a greater WMHs load is associated with a more severe disease profile. Increased aortic stiffness or CBP in the presence of low intracranial artery resistance may predispose migraine patients to WMHs formation; this suggested pathophysiology differs from that observed in non-migraine elderly subjects. Elucidating the relationships between cerebral structural alterations and central pulsatile hemodynamics may lead to a better understanding of the mechanism underlying the elevated WMHs load in migraine patients.
Clinical implications
Greater total WMHs volume was associated with increased CBP (but not brachial blood pressure), aortic stiffness, and decreased resistance in intracranial arteries (decreased CPI). A novel present finding of WMHs being associated with low, rather than high, intracranial artery resistance suggests that WMH formation in migraine patients may have a distinct pathophysiology relative to that in small vessel diseases. Our findings highlight the potential utility of CBP and vascular parameters related to arterial stiffness and vascular pulsatility for cardio- or cerebrovascular risk stratification in migraine patients.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from Taipei Veterans General Hospital [VGHUST105-G7-1-1, V105C-127, V105E9-001-MY2-1, VTA105-V1-1-1], the Ministry of Science and Technology of Taiwan [MOST 104-2314-B-010-015-MY2, MOST 103-2628-B-075-001-MY3 and MOST 103-2321-B-010-017-], the Ministry of Science and Technology support for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan [MOST 103-2911-I-008-001], Academia Sinica [Grant No. IBMS-CRC103-P04], and the Brain Research Center at National Yang-Ming University, Ministry of Health and Welfare, Taiwan [MOHW 103-TDU-B-211-113-003, MOHW 104-TDU-B-211-113-003, MOHW 105-TDU-B-211-113-003], as well as a grant from the Ministry of Education, Aim for the Top University Plan. The opinions, results, and conclusions reported in this article are those of the authors and were developed independent of the funding sources.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
