Abstract
Aim
Atogepant is a novel oral calcitonin gene-related peptide (CGRP) receptor antagonist approved for migraine prevention. This study primarily evaluated its effectiveness and safety in real-world clinical practice, focusing on patients with treatment-resistant migraine, defined according to the European Headache Federation (EHF) criteria as failure of at least three classes of preventive medications, including onabotulinumtoxinA or anti-CGRP monoclonal antibodies (mAbs).
Methods
This prospective multicentre study was conducted across 15 tertiary Headache Units in Spain. Demographic and clinical data, prior preventives, monthly headache days (MHD), monthly migraine days (MMD), and adverse events (AEs) were systematically collected at baseline, 3 months (primary endpoint), and/or 6 months (secondary endpoint).
Results
A total of 513 patients were enrolled (mean age 48 years; 88% women). The 3-month analysis included 455 patients, with median MHD decreasing from 21 (IQR 15-30) to 14 (IQR 6-30) and MMD from 14 (IQR 10-20) to 8 (IQR 3-15) (both p < 0.0001). A ≥ 50% reduction was achieved by 34% (MHD) and 29% (MMD), with ≥75% responses in 16% and 13%. Adverse events were mostly mild, mainly constipation (30%) and nausea (18%), and the 3-month discontinuation rate was 11.8%. Responders had shorter migraine chronicity, less analgesic overuse, and fewer prior preventive failures. Although prior inadequate response to anti-CGRP mAbs reduced the likelihood of improvement, it did not prevent meaningful benefit. At analysis, 151 patients had reached the 6-month visit, showing further improvement (MHD 10 [IQR 5-20]; MMD 6 [IQR 4-12]) and fewer adverse events.
Conclusions
Atogepant showed robust real-world effectiveness and good tolerability in a large, treatment-resistant migraine cohort, with clinically meaningful improvement at 3 months and incremental benefit in the subgroup evaluated at 6 months. Lower migraine chronicity and fewer prior preventive failures were associated with better outcomes, supporting the earlier introduction of anti-CGRP therapies in clinical practice.
Trial Registration
Clinical Trials.gov NCT06241313
This is a visual representation of the abstract.
Introduction
Migraine pathophysiology involves activation of the trigeminovascular system and release of calcitonin gene-related peptide (CGRP), a key neuropeptide mediating vasodilation, neurogenic inflammation, and nociceptive sensitization.1,2 Elevated CGRP levels during spontaneous or provoked attacks, and the induction of migraine-like pain after intravenous CGRP infusion, confirm this pathway as a major therapeutic target. 1
The first CGRP-targeted preventive therapies were monoclonal antibodies (mAbs), erenumab, which binds the CGRP receptor, and fremanezumab, galcanezumab, and eptinezumab, which bind the ligand itself. 3 These mAbs, with long half-lives and high receptor specificity, have demonstrated consistent effectiveness and tolerability across randomised controlled trials (RCTs) and real-world studies, becoming established preventive options for high-frequency episodic and chronic migraine.
By contrast, gepants are small-molecule CGRP receptor antagonists (< 500 Da) suitable for oral administration. Their pharmacological profile enables use in both acute and preventive settings, offering a non-peptidic alternative to injectable mAbs. While first-generation agents such as telcagepant demonstrated clinical effectiveness, their development was discontinued because of hepatotoxicity. 4 The newer second-generation gepants, including ubrogepant, rimegepant (oral, approved for both acute treatment and preventive treatment of episodic migraine), and zavegepant (intranasal), retain efficacy without hepatic concerns and are approved for the acute treatment of migraine. 5
Atogepant, another second-generation gepant, is the first in its class approved for the preventive treatment of migraine and is under investigation for acute use (ECLIPSE trial, NCT06241313). Its efficacy and safety have been demonstrated across multiple phase 3 trials. In the pivotal ADVANCE study, atogepant significantly reduced monthly migraine days (MMDs) in episodic migraine over 12 weeks versus placebo. 6 The PROGRESS trial extended these findings to chronic migraine, confirming efficacy of both 30 mg twice-daily and 60 mg once-daily regimens. 7 Complementary studies further reported meaningful improvements in migraine-specific quality of life and functional outcomes 8 and benefit in patients with multiple prior preventive failures. 9 Consistent results have also been observed in Japanese populations. 10
Despite these robust clinical data, real-world evidence on atogepant remains limited. A recent prospective, multicentre Italian cohort by Barbanti et al., 2025, 11 confirmed and extended RCT findings in both high-frequency episodic and chronic migraine. However, most observational studies to date have included substantially smaller samples, ranging from 31 to 183 participants. 11 − 15
In Spain, atogepant has been approved and publicly reimbursed for preventive treatment since early 2024. Reimbursement criteria specify use in high-frequency episodic migraine after failure of at least three oral preventive agents, and in chronic migraine after failure of two oral preventives and onabotulinumtoxinA. Consequently, the population eligible for atogepant in real-world practice is typically highly resistant, underscoring the clinical relevance of evaluating treatment effectiveness in this setting.
Recent evidence-based guidelines from the International Headache Society 16 and their accompanying summary 17 have emphasized the importance of real-world data to inform preventive strategies. In this context, the GEMA (GEpants in MigrAine—Atogepant) Project represents the largest prospective, multicentre, real-world analysis of atogepant conducted to date. It aims to provide comprehensive data on its effectiveness, safety, and tolerability in clinical practice, offering insights into its performance beyond the controlled environment of randomised trials.
Methods
Study design
The GEMA Project is an investigator-initiated, multicentre, real-world collaborative study conducted across 15 tertiary Headache Units in Spain. It prospectively registers the use of gepants as preventive migraine treatments in routine clinical practice. Within this framework, we performed a prospective cohort study including adults with migraine who initiated oral atogepant for prevention. Eligible patients were consecutively enrolled from June 2024 to March 2025. Analyses included patients with available data at 3 and/or 6 months to evaluate short- and mid-term outcomes in routine clinical practice. Analyses were conducted using a complete-case approach and the study was not designed as a strict intention-to-treat analysis. All patients who initiated treatment were included in the 3-month analysis. At each follow-up visit, response rates were calculated exclusively among evaluable patients. Patients who discontinued treatment were considered non-responders at the time point of discontinuation. At the 6-month visit, only patients with available clinical data were analysed. No imputation of missing outcome data was performed.
The study followed STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) guidelines for observational research. As this was a real-world prospective cohort including all eligible patients initiating atogepant during the recruitment period, no formal sample size calculation was performed a priori.
Study objectives and outcomes
The primary objective was to assess the clinical response to atogepant at 3 months, measured as the change in monthly headache days (MHD) and monthly migraine days (MMD) from baseline, and by responder rates of ≥ 30%, ≥ 50%, ≥ 75%, and 100% (complete remission).
Secondary objectives included (a) identifying predictors of response and the impact of prior anti-CGRP monoclonal antibody exposure, (b) evaluating safety and tolerability, and (c) exploring the persistence of effectiveness at 6 months in patients with available follow-up data.
Response categories were predefined as mild (≥ 30 – < 50%), good (≥ 50 – < 75%), and excellent (≥ 75%), based on conventional thresholds used in real-world migraine studies evaluating preventive therapies. 17 Adverse events (AEs), particularly those leading to treatment discontinuation, were also recorded as secondary outcomes.
Participants
Eligible participants were adults (≥ 18 years) diagnosed with migraine according to ICHD-3 criteria and with a disease duration of at least one year, who had initiated preventive treatment with atogepant in routine clinical practice. All patients were required to have been on stable doses of any concomitant preventive therapy for at least three months before starting atogepant. Patients with other active primary or secondary headache disorders were excluded, except those with MO headache or infrequent tension-type headache.
Treatment resistance was defined according to the European Headache Federation consensus 18 as the failure of at least three classes of preventive medications, whereas refractory migraine was defined as the failure of all available preventive options, including onabotulinumtoxinA and CGRP-targeted therapies. These criteria were fully aligned with the national reimbursement policy for atogepant in Spain, requiring prior failure of ≥ 3 oral preventives in episodic migraine or ≥ 2 oral preventives plus onabotulinumtoxinA in chronic migraine and all included patients met these conditions.
Data collection and variables
Initial and follow-up visits were documented using standardized electronic case report forms specifically designed for this study within the REDCap platform. 19 Data were collected by the investigators through structured interviews and medical record review, ensuring consistency and data quality.
Collected variables included sociodemographic characteristics, number of prior preventive treatments, monthly headache days (MHD), monthly migraine days (MMD), analgesic overuse, and treatment-emergent adverse events (AEs). A month was defined as a standardized 30-day reference period for calculating MHD and MMD. Clinical follow-up assessments were scheduled at 3 and/or 6 months following atogepant initiation.
Medication overuse (MO) was defined according to ICHD-3 criteria as the use of acute medications on ≥ 10 days/month for triptans, ergot derivatives, or combination analgesics, or ≥ 15 days/month for simple analgesics or NSAIDs, assessed from patient diaries. MO was recorded for both chronic and episodic migraine cases.
In addition to clinical parameters, patient-reported outcomes were evaluated in a subset of participants from centres where these scales were routinely collected. The Headache Impact Test (HIT-6) was used to assess the functional impact of headache on daily activities and headache-related disability; the Hospital Anxiety and Depression Scale (HADS) was employed to evaluate symptoms of anxiety and depression through its two subscales (HADS-A and HADS-D); and the Insomnia Severity Index (ISI) was applied to measure the severity of sleep disturbance. Changes in total and subscale scores were analysed over time, and their association with treatment response was examined.
Statistical analysis
Quantitative variables were expressed as mean and standard deviation (SD) or median and interquartile range (IQR) for the variables with non-normal distribution. For qualitative variables, frequency and proportions were used. Normality was evaluated using the D’Agostino-Pearson test. To compare variables across groups, statistical differences were assessed using Kruskal-Wallis tests for quantitative variables and Fisher's exact test or Pearson's χ2 test for categorical variables.
MHD and MMD were analysed using Generalized Estimating Equations (GEE), with visits nested within each patient. Models were fitted using the “geepack” package in R. 20 Changes in MHD and MMD over time were assessed using GEE, and response was defined as a ≥ 30%, ≥ 50%, or ≥ 75% reduction in MHD/MMD from baseline. Dependent variables were fitted to a normal distribution according to Akaike's Information Criteria. 21 First, univariate analyses were performed to determine which demographic and clinical variables were associated with response to treatment. Next, multivariate models were built to better understand the dependence of MHD and MMD on the set of significant variables identified in the univariate analyses (p < 0.10), following the backward method. Longitudinal changes across visits were interpreted exclusively using the GEE framework, which appropriately models within-patient evolution across all available follow-up data without requiring identical patient populations at each time point. In order to identify the variables associated with treatment response at 3 months and at 6 months we fitted multivariate ordered logistic regression models with the same strategy as in the previous models (R package “ordinal”). Statistical analyses and figures were conducted using R version 4.4.0 (R Core Team (2024). R Foundation for Statistical Computing, Vienna, Austria. <https://www.R-project.org/>.).
Ethical considerations
The study protocol was approved by the Drug Research Ethics Committee of Hospital Universitario de la Princesa (Comité de Ética de la Investigación con medicamentos, CEIm; registration number 5600, 6th June 2024). All participants provided written informed consent electronically prior to enrolment. The study was conducted in accordance with the principles of the Declaration of Helsinki and complied with applicable data protection regulations.
Results
Study population
A total of 513 patients were enrolled at baseline. Of these, 455 (88.7%) attended the 3-month visit and constituted the main analytical cohort. According to local follow-up schedules, an additional 58 patients were assessed directly at 6 months without an intermediate 3-month evaluation.
Within the first 3 months, 57 patients discontinued atogepant due to lack of effectiveness or adverse events. At the time of analysis, 151 patients had completed the 6-month follow-up, providing additional longitudinal data on treatment persistence and sustained effectiveness.
A detailed flow of patient disposition is presented in Figure 1.
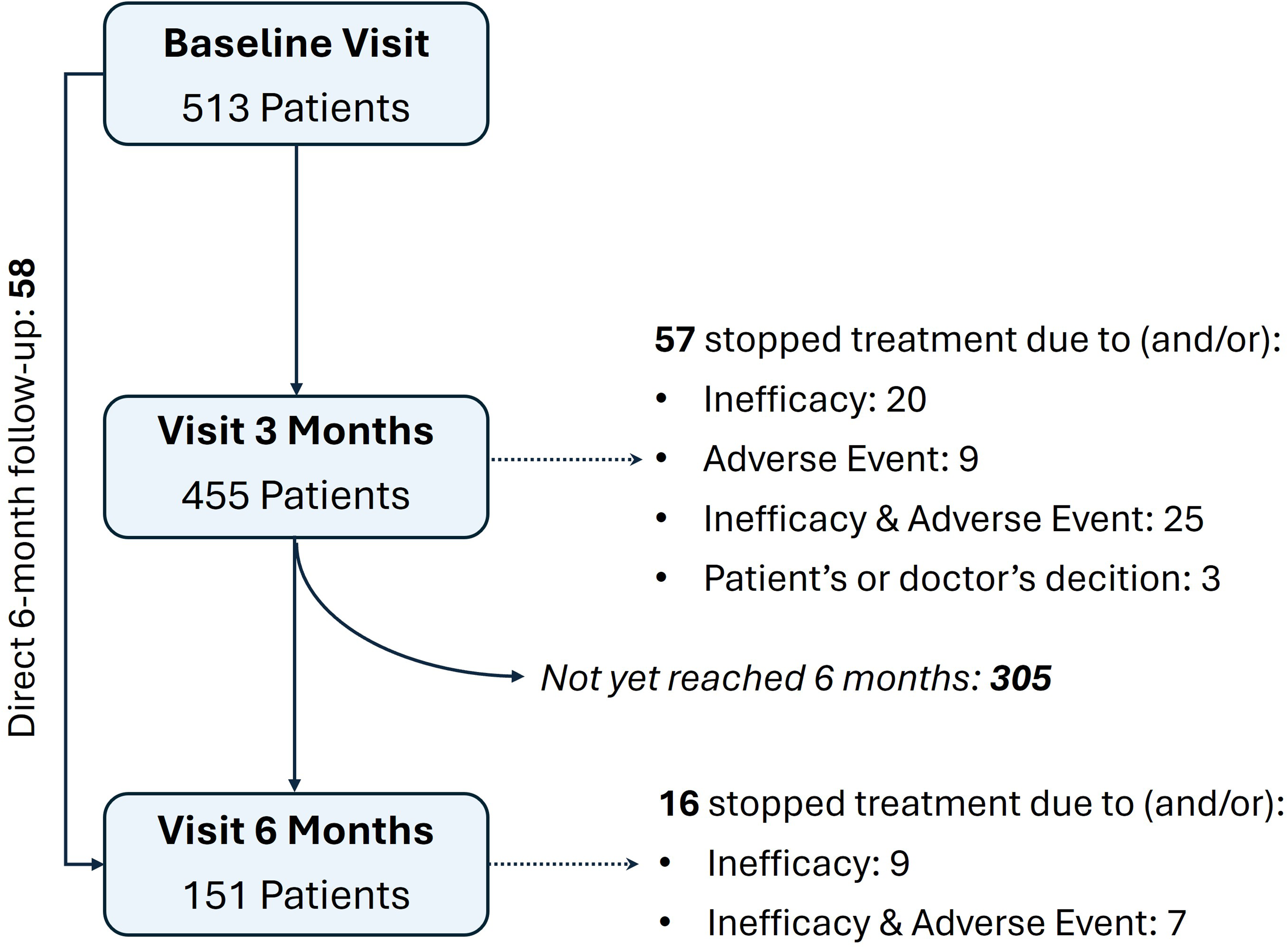
Flowchart of patient follow-up throughout the study period.
Seventy-four percent of patients were diagnosed with chronic migraine, and the median duration of chronic headache was 48 months (IQR 24-120). Patients had failed a median of six (IQR 5-9) preventive treatments prior to study entry. Eighty-four percent had failed prior treatment with onabotulinumtoxinA, and 54% had failed an anti-CGRP monoclonal antibody, indicating a highly treatment-resistant population. Phenotypic characteristics and prior preventive treatments are summarized in Table 1.
Baseline demographic and clinical characteristics of the study cohort (N = 513). Data are expressed as number and percentage [n (%)] for categorical variables and median (IQR) for continuous variables.

Real-world effectiveness of atogepant reduction in headache and migraine days and clinical response at 3 months (primary outcome)
Atogepant led to a significant reduction in monthly headache and migraine days at 3 months compared with baseline in the primary analytical cohort (n = 455). The analysis showed a mean decrease of −5.29 monthly headache days (MHD) (95% CI: −4.12 to −6.47; p < 0.0001). Median MHD decreased from 21 (IQR: 15-30) at baseline to 14 (IQR: 6-30) at 3 months (p < 0.001) (Figure 2A). Overall, headache frequency was reduced by approximately 30% at 3 months, despite the highly treatment-resistant profile of the cohort.

Changes in monthly headache days (MHD, panel A) and monthly migraine days (MMD, panel B) from baseline to 3 months of atogepant treatment in the primary analytical cohort (n = 455). Box plots represent the median and interquartile range (IQR); individual data points are shown as dots, and black lines indicate the overall trend. Statistical comparisons were performed using Generalized Estimating Equations (GEE). p < 0.05 was considered statistically significant.
Monthly migraine days (MMD) also significantly decreased within the first 3 months (Figure 2B), with a mean change of −4.83 days (95% CI: −3.78 to −5.89; p < 0.001), corresponding to a 43% reduction versus baseline. Median MMD decreased from 14 (IQR: 10-20) to 8 (IQR: 3-15) (p < 0.001).
To further characterize treatment effectiveness, patients were stratified according to their degree of response: mild (≥ 30% to < 50%), good (≥ 50% to < 75%), excellent (≥ 75%), and full remission (100%). At 3 months, 34% of patients achieved a ≥ 50% reduction in MHD and 29% in MMD. Excellent responses (≥ 75%) were observed in 16% and 13% of patients, respectively, and 12 patients (2.6%) achieved complete remission (Table 2).
Distribution of treatment response categories to atogepant at 3 months (n = 455). Response was defined as the percentage reduction from baseline in monthly headache days (MHD) or monthly migraine days (MMD), categorized as mild (≥ 30% to < 50%), good (≥ 50% to < 75%), excellent (≥ 75%), and full remission (100%). Values are expressed as percentages of the patients evaluated at this time point, with absolute numbers shown in parentheses.

These individual-level trends in response were further supported by the low rate of treatment discontinuation due to lack of effectiveness. During the first 3 months, 45 patients (9.9% of the 455 evaluated) discontinued therapy for this reason. This relatively small proportion highlights the consistent effectiveness of atogepant in this difficult-to-treat population.
Headache-related functional impact and comorbidities at 3 months
A subgroup of 338 patients completed the Headache Impact Test (HIT-6) both at baseline and at 3 months. A significant improvement in quality of life was observed, with median HIT-6 scores decreasing from 68 (IQR: 65-72) to 64 (IQR: 54-70) (p < 0.001).
In addition, a subset of patients (n = 127 for HADS and n = 89 for ISI) had available data for comorbid anxiety, depression, and insomnia. Scores for HADS-A and HADS-D showed significant reductions at 3 months (p < 0.05), whereas ISI exhibited a non-significant trend toward improvement (Figure 3). To assess whether these changes were associated with clinical response to atogepant, the cohort was stratified into responders (≥ 50% reduction in monthly headache days or monthly migraine days) and non-responders (< 50%). Reductions in HADS-A and HADS-D were significantly greater among responders, irrespective of whether response was defined by headache days or migraine days. In contrast, ISI improvement reached significance only in responders based on monthly migraine day reduction. For detailed statistical results, see Supplementary Material (Supplementary Table 1).

Evolution of comorbid anxiety, depression, and insomnia during the first 3 months of atogepant treatment. HADS-A (anxiety) and HADS-D (depression) scores (n = 127) and ISI (insomnia) scores (n = 89) are shown at baseline and after 3 months. Box plots represent the median and interquartile range (IQR); dots indicate individual patient data, and black lines indicate the median. Statistical comparisons were performed using the Wilcoxon test. p < 0.05 was considered statistically significant.
Predictors of response to atogepant in real-world clinical practice at 3 months
Several clinical and treatment-related factors were significantly associated with treatment effectiveness in the univariate analyses (p < 0.05); these variables are summarized in Supplementary Table 2.
In multivariable analyses (p < 0.05), variables associated with less reduction in MHD (more days at 3 months) included higher migraine chronicity (β 0.01, 95% CI 0.002-0.02, p = 0.0049), more analgesic MO (β 10.44, 95% CI 9.02-11.86, p < 0.001), higher number of prior preventive treatment failures (β 0.28, 95% CI 0.08-0.49, p = 0.0066) and previous anti-CGRP exposure either with (β 4.76, 95% CI 0.28-9.25, p = 0.0495) or without prior onabotulinumtoxinA (β 2.48, 95% CI 0.89-4.07, p = 0.0079). For MMD, less reduction was associated with higher age at onset (β 0.07, 95% CI 0.01 - 0.14, p = 0.0375), more migraine chronicity (β 0.01, 95% CI 0.002-0.02, p = 0.0066), higher analgesic MO (β 10.53, 95% CI 9.13-11.93, p < 0.001), a higher number of prior preventive treatment failures (β 0.25, 95% CI 0.04-0.46, p = 0.0167) and previous anti-CGRP with prior botulinum toxin therapy (β 4.76, 95% CI 0.28-9.25, p = 0.0495).
For ≥ 30% clinical response, univariate predictors reflected similar patterns. In multivariable analyses, lower probability of achieving a ≥ 30% MHD response was associated with a higher number of prior preventive failures (β = −2.23, 95% CI −3.76 to −0.71, p = 0.004) and prior anti-CGRP exposure, either with (β = −18.52, 95% CI −28.89 to −8.15, p < 0.001) or without (β = −14.89, 95% CI −24.90 to −4.88, p = 0.004) prior onabotulinumtoxinA. For MMD, the variables that remained significantly associated with a lower probability of achieving a ≥ 30% response, were prior anti-CGRP exposure, either with (β = −20.87; 95% CI: −39.70 to −2.04; p = 0.030) or without (β = −22.59; 95% CI: −42.02 to −3.17; p = 0.023) onabotulinumtoxinA.
Outcomes at 6 months (secondary analyses)
At the time of analysis, 151 patients had completed the 6-month follow-up, providing extended longitudinal data. Using all available observations, the GEE model showed that the mean reduction in MHD from baseline was −7.69 days (95% CI: −6.01 to −9.38; p < 0.0001), and −6.39 days for MMD (95% CI: −4.88 to −7.91; p < 0.001). Median MHD decreased to 10 (IQR: 5-20) and MMD to 6 (IQR: 4-12). The additional change from 3 to 6 months was −2.40 days for MHD (95% CI: −0.69 to −4.11; p = 0.0040) and −1.56 days for MMD (95% CI: −0.02 to −3.10; p = 0.023) (Figure 4 A and B).

Panels A (MHD) and B (MMD) show the model-based analysis using the GEE model, including all available observations (n = 513, 455, and 151 at baseline, 3 months, and 6 months, respectively). Across both analyses, a significant and progressive reduction in MHD and MMD was observed from baseline to 3 and 6 months. Panels C (MHD) and D (MMD) display the complete-case analysis restricted to patients with available data at 6 months. Box plots represent the median and interquartile range (IQR), dots correspond to individual patient data, and black lines indicate the overall temporal trend. Statistical comparisons in panels A and B were performed using GEE models. p value < 0.05 was considered statistically significant. MHD: Monthly headache days. MMD: Monthly migraine days. GEE: Generalized Estimating Equations.
In parallel, a complete-case analysis was conducted. Only patients with available clinical data at the 6-month visit were included in the corresponding analysis. Among these complete cases, median MHD decreased from 20 (IQR: 14–30) at baseline to 12 (IQR: 5–20) at 3 months, with a further reduction to 10 (IQR: 5–20) at 6 months (Figure 4C). For MMD, the median dropped from 14 (IQR: 10–20) to 7 (IQR: 3–13) at 3 months and remained stable at 6 months (7; IQR: 4–12) (Figure 4D).
Following the same analytical approach used at 3 months, treatment effectiveness at 6 months was further characterized by stratifying patients according to their degree of response, as previously explained. Patients who discontinued treatment between months 3 and 6 due to lack of effectiveness or adverse events (n = 16) were included in the 6-month response analyses as non-responders. At 6 months, 49.7% of patients achieved a ≥ 50% reduction in MHD and 47.7% in MMD. Excellent responses (≥ 75%) were observed in 19.2% and 23.2% of patients, respectively, and 1 patient achieved complete remission (Table 3).
Distribution of treatment response categories to atogepant at 6 months (n = 151). Response was defined as the percentage reduction from baseline in monthly headache days (MHD) or monthly migraine days (MMD), categorized as mild (≥ 30% to < 50%), good (≥ 50% to < 75%), excellent (≥ 75%), and full remission (100%). Values are expressed as percentages of the patients evaluated at this time point, with absolute numbers shown in parentheses.

A subset of 56 patients also completed the HIT-6 at 6 months, showing further improvement (median 60 [IQR: 57-65]) compared with baseline (68 [IQR: 65-72]) (p < 0.0001) and 3 months (62 [IQR: 54-66]) (p < 0.0001).
Regarding predictive factors, univariate analysis at 6 months identified the same significant variables as at 3 months, except for HIT-6 score. In the multivariate model, MHD reduction remained significantly associated with number of concomitant preventive treatments (β −6.13, 95% CI −11.81 to −0.45, p = 0.034), prior anti-CGRP exposure (β −14.89, 95% CI −29.73 to −0.05, p = 0.049), and analgesic overuse (β - 18.75, 95% CI −32.97 to −4.53, p = 0.010). For MMD reduction, only analgesic overuse remained significant (β −18.33, 95% CI −32.79 to −3.86, p = 0.013). Overall, the pattern of predictors at 6 months was consistent with that observed at 3 months, with the same variables retaining significance across both time points.
Effectiveness of atogepant in patients with prior anti-CGRP therapy
To assess whether prior anti-CGRP monoclonal antibody use influenced atogepant effectiveness, patients were stratified by treatment history (Figure 5). At three months, antibody-naïve patients exhibited higher response rates, with 47% achieving at least a 50% reduction in monthly headache days (MHD) compared with 21% among previously treated individuals, while nearly 70% of pre-exposed patients remained below the 30% improvement threshold. Similar trends were observed for monthly migraine days (MMD), with 51% versus 29% achieving a ≥ 50% reduction, respectively.

Response to atogepant according to prior anti-CGRP therapy. Stacked bar plots represent the percentage of patients categorized as non-responders (< 30%), mild (≥ 30 - < 50%), good (≥ 50 - < 75%), and excellent responders (≥ 75%) based on the reduction at 3 months (n = 455) and 6 months (n = 151) in monthly headache days (A, B) and monthly migraine days (C,D). Percentages are calculated only among patients with available data; individuals with missing values are excluded from the denominators, and therefore total counts may not sum to the cohort n. Responses are stratified by prior exposure to anti-CGRP monoclonal antibodies (“No” vs. “Yes”).
By six months, response rates improved in both groups, with absolute outcomes remaining higher in antibody-naïve patients but relative gains greater in pretreated ones, suggesting a delayed yet progressive benefit. The proportion of ≥ 50% responders increased from 47% to 61% in antibody-naïve patients and from 21% to 40% in those previously treated, while corresponding rates for MMD rose from 51% to 56% and from 29% to 42%.
When stratified by the number of prior antibody therapies received (0-4), baseline disease burden and prior preventive exposure increased progressively, with a median of 5 to 13 previous treatments (p < 0.001) (Table 4). Patients with up to two prior therapies showed meaningful reductions in MHD and MMD, whereas those with three or more therapies exhibited limited improvement. In these latter groups, MHD remained largely unchanged, and MMD decreased only modestly, from 30 to 22. HIT-6 scores worsened with increasing numbers of prior anti-CGRP treatments, and no improvement was observed in patients with four prior antibodies.
Baseline demographic and clinical characteristics of patient subgroups classified according to the number of previously tested anti-CGRP therapies. Data are expressed as number and percentage [n (%)] for categorical variables and as median (IQR) for continuous variables. MHD, monthly headache days; MMD, monthly migraine days; MO, medication overuse; BoNT-A, onabotulinumtoxinA; IQR, interquartile range.
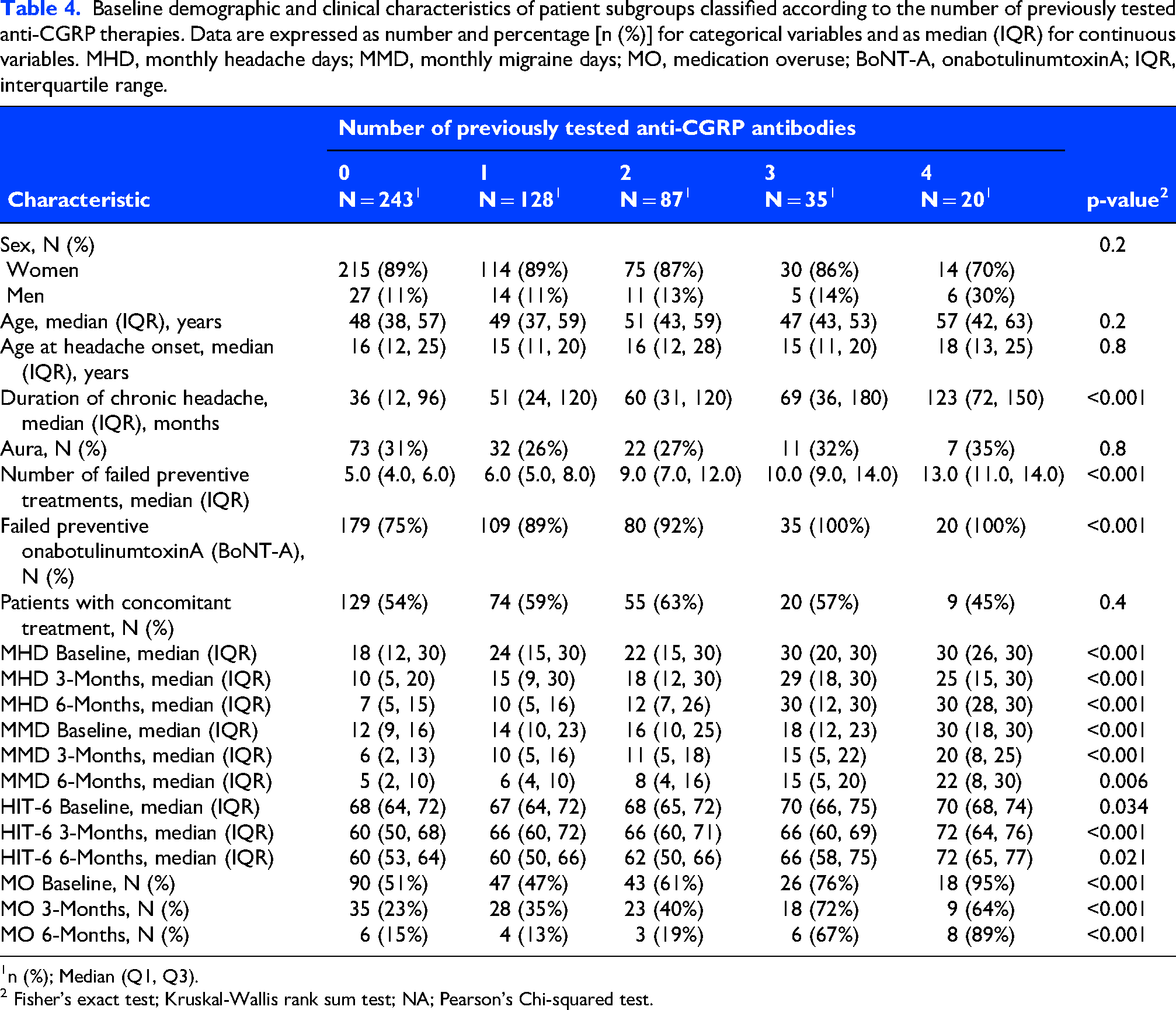
1n (%); Median (Q1, Q3).
2 Fisher's exact test; Kruskal-Wallis rank sum test; NA; Pearson's Chi-squared test.
Analgesic overuse declined markedly among patients without or with limited prior exposure, from 51-61% to 13-19%, but only slightly among those with extensive prior use, from 76-95% to 67-89%. Adverse event rates were comparable across subgroups, except for nausea and dizziness, which were more frequent among patients who had received four prior anti-CGRP therapies.
Considering the type of monoclonal antibody previously used, a subanalysis was conducted including only patients exposed to a single therapeutic target (CGRP receptor or ligand). Among the 270 individuals with prior anti-CGRP therapy, 173 (64%) met this criterion: 23 had received a receptor antibody and 150 a ligand antibody.
Baseline demographic and clinical characteristics were largely comparable between groups (Supplementary Table 3). Patients previously treated with a ligand antibody had failed a slightly higher number of preventive treatments (median 7 vs. 6; p = 0.033), suggesting marginally greater baseline refractoriness. Headache and migraine frequency, as well as changes in MHD and MMD at three and six months, were similar across groups. HIT-6 scores showed a transient difference at three months (p = 0.038), which normalised by month six. No other clinically meaningful differences in efficacy or tolerability were identified.
Tolerability and side effects
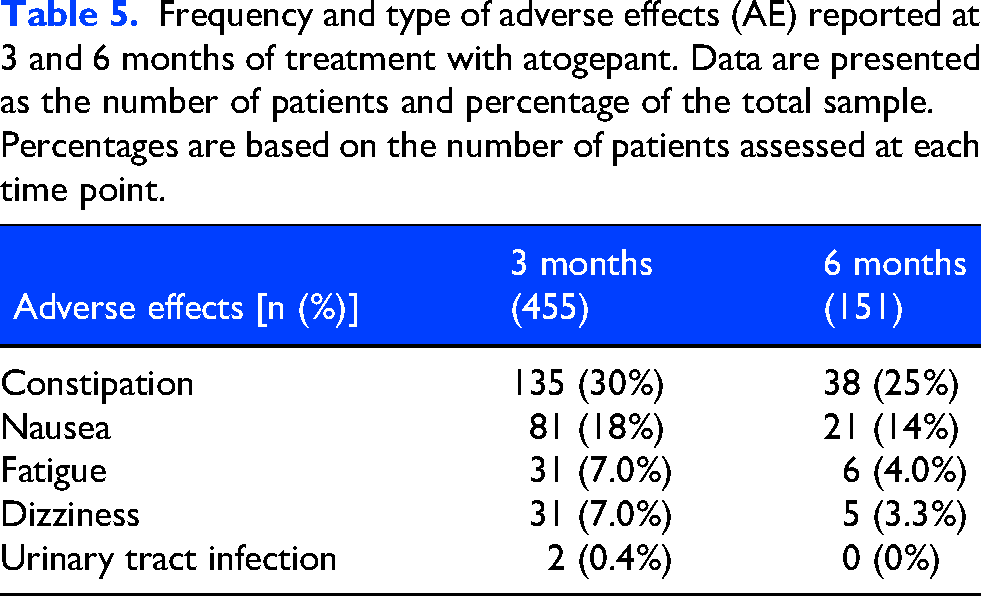
Atogepant demonstrated a favourable tolerability profile throughout the study period. Nearly half of participants reported no AEs. Among those who did, most AEs were mild and of limited clinical relevance, with constipation (135/455, 30%) and nausea (81/455, 18%) being the most frequent at 3 months (Table 4). A slight decrease in AE frequency was observed at month 6 (38/151, 25% for constipation and 21/151, 14% for nausea), consistent with progressive treatment adaptation over time. The overall incidence of AEs remained stable between 3 and 6 months, supporting a consistent safety and tolerability profile.
According to the flowchart (Figure 1), 34 out of 455 patients (7.5%) discontinued treatment during the first 3 months due to AEs, and an additional 7 out of 151 patients (7/151, 4.6%) discontinued at month 6. Among the early discontinuations, 9 patients stopped atogepant exclusively because of AEs, most commonly nausea, dizziness, and fatigue. The remaining cases resulted from a combination of AEs and insufficient therapeutic benefit. No new or unexpected safety signals were identified during follow-up (Table 5).
Frequency and type of adverse effects (AE) reported at 3 and 6 months of treatment with atogepant. Data are presented as the number of patients and percentage of the total sample. Percentages are based on the number of patients assessed at each time point.
Discussion
Our study provides robust real-world evidence supporting the clinical utility of atogepant in a population with highly treatment-resistant migraine. The vast majority of patients had chronic migraine and had accumulated a very high number of prior preventive failures (median 6), with more than half previously exposed to anti-CGRP monoclonal antibodies and over 80% treated with onabotulinumtoxinA. Despite this degree of refractoriness, atogepant achieved significant and sustained clinical reductions in both monthly headache days (MHD) and monthly migraine days (MMD).
At 3 months, in the main analytical cohort (n = 455), atogepant was associated with clinically meaningful reductions in both MHD and MMD, with mean decreases of −5.29 and −4.83 days, corresponding to approximately 30% and 40% reductions from baseline, respectively. Around one-third of patients achieved a ≥ 50% response, and approximately 15% reached an excellent response (≥ 75%), underscoring the clinical relevance of these findings in routine practice. Treatment effectiveness was further supported by the low rate of discontinuation due to lack of efficacy during the first three months and by parallel improvements in headache-related disability and patient-reported outcomes. Although response rates were slightly lower than those reported in the ADVANCE and PROGRESS trials,6,7 they fall within the expected range given the greater refractoriness and chronicity of the real-world population included in the GEMA project.
In the exploratory 6-month analyses, interpretation of treatment effects over time suggests a tendency toward additional clinical benefit with continued atogepant exposure. Across the overall analyses, reductions in headache and migraine frequency from baseline were greater at 6 months than at 3 months, and response rates increased over time, which may be consistent with a cumulative treatment effect. These findings should, however, be interpreted with caution, given the smaller number of patients with available 6-month follow-up. In analyses restricted to patients with complete data at all time points, progressive improvement was less evident, with only a non-significant trend observed for headache days and no clear progression for migraine days. Taken together, these observations suggest that sustained treatment may be associated with incremental benefit, particularly in headache frequency, especially in highly treatment-resistant populations such as the present cohort, in whom longer treatment exposure may be required to achieve meaningful clinical modulation, pending confirmation in larger real-world populations.
Patient-reported outcomes evolved in parallel with clinical improvements. HIT-6 scores decreased significantly at 3 months, with further reduction in the 6-month subgroup. Likewise, in subgroups with available assessments, anxiety, depression, and insomnia improved as early as month 3, with more pronounced reductions among responders. These findings suggest that the benefit of atogepant extends beyond headache-frequency control, favorably impacting relevant functional and comorbid domains.
Our findings extend the efficacy observed in clinical trials to a more complex and treatment-resistant real-world population that is frequently under-represented in randomised studies. Compared with phase 3 cohorts, patients included in the GEMA project exhibited higher levels of chronicity, a greater prevalence of medication overuse, and a substantially higher burden of prior preventive failures. Despite these challenges, the magnitude of clinical improvement observed underscores the therapeutic value of atogepant in difficult practice settings. These results are consistent with findings from the ELEVATE trial, which specifically enrolled individuals with multiple prior preventive failures, 9 as well as with real-world multicentre cohorts such as the Italian STAR and GIANT studies. Differences in baseline refractoriness and chronicity across populations likely account for the variability in reported response rates.11,15
Recent real-world evidence has further explored the effectiveness of atogepant in patients previously exposed to anti-CGRP monoclonal antibodies. In a retrospective cohort of highly selected patients with prior lack of response to anti-CGRP therapies, the RESCUE study 13 showed that atogepant may provide clinically meaningful benefit in a subset of refractory patients. Similarly, in a large multicentre cohort restricted to individuals with prior anti-CGRP monoclonal antibody failure, Muñoz-Vendrell et al. 22 reported that atogepant retains effectiveness in a substantial proportion of patients, although response rates appeared to be influenced by both the type and number of previously failed antibodies, with differential outcomes according to receptor- versus ligand-targeting therapies.
In the GEMA cohort, prior exposure to anti-CGRP monoclonal antibodies was also associated with lower overall response rates. Nevertheless, among previously treated patients, longitudinal analyses suggested a more gradual pattern of benefit over time, consistent with a slower but clinically meaningful response trajectory in this subgroup. Furthermore, stratification by the number of prior monoclonal antibody failures supported an accumulative refractoriness effect, with patients who had failed one or two agents still achieving marked reductions in headache and migraine days, whereas those with three or more prior failures showed more modest improvements, despite higher baseline disease burden. Importantly, a clinically relevant minority of these highly treatment-resistant patients still attained meaningful responses. In contrast to some published data, we did not observe significant differences in effectiveness according to the specific target of prior antibody treatment (receptor versus ligand). This discrepancy may be explained, at least in part, by differences in study design and patient populations, as the GEMA project evaluated a broader real-world cohort of individuals with treatment-resistant migraine and included a relatively smaller proportion of patients previously treated with receptor-targeting antibodies. Taken together, these findings support the concept that lack of response to monoclonal antibodies does not necessarily imply cross-resistance to gepants. Pharmacokinetic and pharmacodynamic differences between antibody-based therapies and small-molecule CGRP antagonists may underlie this observation, supporting therapeutic switching within the anti-CGRP pathway and highlighting the need for further studies to better define predictors of response in this clinically challenging subgroup.
Regarding safety, atogepant demonstrated an overall favourable tolerability profile. Constipation (30%) and nausea (18%) were the most common adverse events, with most events being mild and showing a slight decrease over time. As expected, these rates were somewhat higher than those reported in pivotal trials, where constipation and nausea typically ranged from 7-10% and 4-10%, respectively,6,7 reflecting the greater clinical complexity and less standardized monitoring inherent to real-world practice. When compared with other observational cohorts, our findings align with the STAR and RESCUE studies, which also reported moderate gastrointestinal adverse event rates and very low discontinuation frequencies, although the slightly higher rates in our cohort likely mirror its increased refractoriness.13,14 In contrast, the GIANT study documented a substantially lower overall incidence of adverse events (5.5%), highlighting variability across real-world populations while consistently confirming the predominantly mild nature of these events. 11 Long-term extension data have similarly shown a stable safety profile with sustained exposure up to one year. 23 Collectively, these findings support good tolerability and the feasibility of maintaining atogepant in routine clinical care without emerging safety concerns.
Discontinuation rates were low for both adverse events and lack of efficacy, with 11.8% of patients discontinuing treatment by month 3 and 10.5% by month 6; these low withdrawal rates, together with the progressive improvement observed through six months, support the feasibility and long-term sustainability of atogepant therapy under real-world conditions.
In line with our predictor analyses, treatment outcomes were largely shaped by markers of accumulated refractoriness. The main negative predictors, listed according to their relative contribution in our study, were analgesic overuse, previous exposure to anti-CGRP monoclonal antibodies or onabotulinumtoxinA, a higher number of prior preventive failures, and longer migraine chronicity, all of which were consistently associated with reduced improvement. This coherent pattern suggests that initiating atogepant earlier in the disease course, before extensive preventive failure or marked chronification, may enhance the likelihood of achieving meaningful benefit.
Some limitations should be acknowledged. The heterogeneity in follow-up timing and the smaller number of patients with 6-month data may have introduced bias, although the use of mixed-effects models likely mitigated this effect. Data were obtained from routine clinical practice and patient-reported headache diaries, which may introduce variability in reporting accuracy. Psychometric assessments such as HADS, ISI, and HIT-6 were not available for all participants, reflecting the heterogeneity inherent to real-world studies, where such scales are not systematically administered to every patient. Finally, because participants were recruited from tertiary headache centres, results may not be fully generalizable to broader or less treatment-resistant migraine populations.
Conclusion
The GEMA project provides robust real-world evidence supporting the effectiveness and tolerability of atogepant in patients with highly treatment-resistant migraine. Clinically meaningful improvements in both monthly headache days and monthly migraine days were evident by three months, accompanied by reductions in headache-related disability and improvements in relevant comorbid symptoms. These benefits were observed despite a substantial burden of prior preventive treatment failures, including anti-CGRP monoclonal antibodies and onabotulinumtoxinA, highlighting the clinical value of atogepant in difficult-to-treat populations. In this context, exploratory analyses in patients with longer follow-up were compatible with sustained benefit over time.
Atogepant demonstrated a favourable tolerability profile in routine clinical practice, with predominantly mild adverse events and low discontinuation rates, supporting its feasibility as a preventive option in real-world settings.
Overall, our results support atogepant as a practical and valuable preventive therapy for treatment-resistant migraine in clinical practice. In this large prospective multicentre real-world cohort, factors associated with better response consistently pointed toward lower disease chronicity and fewer prior preventive failures, reinforcing the importance of earlier intervention within the anti-CGRP pathway.
Clinical implications
Atogepant was effective in a large real-world, treatment-resistant migraine cohort.
At 3 months, about one third achieved ≥ 50% response despite a high proportion with prior anti-CGRP mAb exposure.
Continued treatment for six months allowed additional clinical improvement.
Adverse events were mostly mild, with few discontinuations.
Higher chronicity and multiple preventive failures were associated with poorer outcomes.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261431337 - Supplemental material for Atogepant for migraine in real-world clinical practice: Insights from a large multicentre study in a treatment-resistant population (GEMA project)
Supplemental material, sj-docx-1-cep-10.1177_03331024261431337 for Atogepant for migraine in real-world clinical practice: Insights from a large multicentre study in a treatment-resistant population (GEMA project) by Ana Beatriz Gago-Veiga, Ana Belen Lopez-Rodriguez, Marina Sanchez Jimenez, Alvaro Iglesias Rubio, Nuria Montes, Javier Camiña Muñiz, Marta Dominguez Gallego, Carlos Calle de Miguel, Germán Latorre, Jaime Rodriguez-Vico, Alex Jaimes, Andrea Gomez Garcia, Sara Urtiaga, Marta Gonzalez Salaices, Michele Dileone, Nuria Gonzalez-García, Jesús Porta-Etessam, María-Luz Cuadrado, Alejandro Herrero San-Martin, Ángel-Luis Guerrero-Peral, Yesica Gonzalez-Osorio, Javier Casas-Limón, Antonio Sánchez-Soblechero, Alberto Lozano-Ros, Javier Díaz-De-Terán, Leonardo Portocarrero-Sánchez, Francisco-José Molina-Martínez, Sonia Santos-Lasaosa, Guillermo Martín-Ávila, Elena Riva and Iris Fernández-Lázaro in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024261431337 - Supplemental material for Atogepant for migraine in real-world clinical practice: Insights from a large multicentre study in a treatment-resistant population (GEMA project)
Supplemental material, sj-docx-2-cep-10.1177_03331024261431337 for Atogepant for migraine in real-world clinical practice: Insights from a large multicentre study in a treatment-resistant population (GEMA project) by Ana Beatriz Gago-Veiga, Ana Belen Lopez-Rodriguez, Marina Sanchez Jimenez, Alvaro Iglesias Rubio, Nuria Montes, Javier Camiña Muñiz, Marta Dominguez Gallego, Carlos Calle de Miguel, Germán Latorre, Jaime Rodriguez-Vico, Alex Jaimes, Andrea Gomez Garcia, Sara Urtiaga, Marta Gonzalez Salaices, Michele Dileone, Nuria Gonzalez-García, Jesús Porta-Etessam, María-Luz Cuadrado, Alejandro Herrero San-Martin, Ángel-Luis Guerrero-Peral, Yesica Gonzalez-Osorio, Javier Casas-Limón, Antonio Sánchez-Soblechero, Alberto Lozano-Ros, Javier Díaz-De-Terán, Leonardo Portocarrero-Sánchez, Francisco-José Molina-Martínez, Sonia Santos-Lasaosa, Guillermo Martín-Ávila, Elena Riva and Iris Fernández-Lázaro in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024261431337 - Supplemental material for Atogepant for migraine in real-world clinical practice: Insights from a large multicentre study in a treatment-resistant population (GEMA project)
Supplemental material, sj-docx-3-cep-10.1177_03331024261431337 for Atogepant for migraine in real-world clinical practice: Insights from a large multicentre study in a treatment-resistant population (GEMA project) by Ana Beatriz Gago-Veiga, Ana Belen Lopez-Rodriguez, Marina Sanchez Jimenez, Alvaro Iglesias Rubio, Nuria Montes, Javier Camiña Muñiz, Marta Dominguez Gallego, Carlos Calle de Miguel, Germán Latorre, Jaime Rodriguez-Vico, Alex Jaimes, Andrea Gomez Garcia, Sara Urtiaga, Marta Gonzalez Salaices, Michele Dileone, Nuria Gonzalez-García, Jesús Porta-Etessam, María-Luz Cuadrado, Alejandro Herrero San-Martin, Ángel-Luis Guerrero-Peral, Yesica Gonzalez-Osorio, Javier Casas-Limón, Antonio Sánchez-Soblechero, Alberto Lozano-Ros, Javier Díaz-De-Terán, Leonardo Portocarrero-Sánchez, Francisco-José Molina-Martínez, Sonia Santos-Lasaosa, Guillermo Martín-Ávila, Elena Riva and Iris Fernández-Lázaro in Cephalalgia
Footnotes
List of abbreviation
Acknowledgements
We thank the patients and researchers for their dedication.
ORCID iDs
Ethical considerations
The study was approved by the Drug Research Ethics Committee of Hospital Universitario de la Princesa (CEIm; registration number 5600, 6 June 2024). The study complied with the Declaration of Helsinki and applicable data protection regulations
Consent to participate
All participants provided written informed consent.
Consent for publishing
The authors agree to publish with Cephalalgia.
Author contributions
ABGV and IFL designed the study. ABGV, ABLR and IFL drafted the manuscript and figures; ABGV, ABLR, NM and IFL carried out data analysis; ABGV, MSJ, AIR, JCM, MDG, CC, GL, JRV, AJ, AGG, SU, MGS, MD, NGG, JPT, AHS, ALGP, YGO, JCL, ASS, ALR, JDD, LP, FJMM, SSL, GMA and ERA were responsible for acquisition of dal of the authors revised the manuscript and approved the final version of the manuscript submitted for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Instituto de Salud Carlos III (ISCIII) and Fondo Europeo de Desarrollo Regional (FEDER), through IMPaCT project PMP22/00158, and co-funded by the European Union through the Recovery, Transformation and Resilience Plan - Next Generation EU to A.B.G.V and I.F.L.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ABG-V has received speaker honoraria and/or served as a clinical advisor for Novartis, Lilly, Organon, TEVA, Exeltis, Chiesi, AbbVie, Pfizer, Dr. Reddy's, and Lundbeck. She is the coordinator and principal investigator of a research IMPaCT project, grant number PMP22/00158. JCM has received speaker honoraria and/or served as a clinical advisor for Novartis, Lilly, Organon, TEVA, Exeltis, Chiesi, AbbVie, Pfizer, and Lundbeck. CCDM has received speaker honoraria for TEVA, Allergan-AbbVie and Lundbeck. GLT has received speaker honoraria and/or served as a clinical advisor for Novartis, Lilly, Organon, TEVA, AbbVie, Pfizer, and Lundbeck. JRV has received speaker honoraria and/or served as a clinical advisor for Novartis, Lilly, Organon, TEVA, Exeltis, Chiesi, AbbVie, Pfizer, Dr. Reddy's, and Lundbeck. AJ has received speaker honoraria and/or served as a clinical advisor for Lilly, TEVA, Organon, Allergan-AbbVie and Lundbeck. NG-G has received honoraria from Novartis, Lilly, TEVA, Organon, Allergan-AbbVie and Lundbeck. JPE has received honoraria from Novartis, Lilly, TEVA, Organon, Allergan-AbbVie and Lundbeck. M-LC has received honoraria as a consultant or lecturer for Novartis, AbbVie, Lundbeck and Teva. AHS has received speaker honoraria for AbbVie and Pfizer. ALG has participated in advisory and speaker boards for AbbVie, Dr. Reddy's, Eli Lilly, Lundbeck, Novartis, Organon, Pfizer and Teva. JC-L has received honoraria as a consultant or speaker for: AbbVie, Almirall, Chiesi, Eli Lilly, Lundbeck, Novartis, Organon, Pfizer and Teva. AS-S has received honoraria from TEVA, Lundbeck, Organon, AbbVie, Almirall, and Pfizer. AL-R has received honoraria as a consultant and speaker for: AbbVie-Allergan, Dr. Reddy's, Teva, Pfizer and Lundbeck. JDDT has received speaker honoraria and/or served as a clinical advisor for Novartis, Lilly, Organon, TEVA, Exeltis, Chiesi, AbbVie, Pfizer, Dr. Reddy's, and Lundbeck. FJMM has received speaker honoraria and/or served as a clinical advisor for Novartis, Lilly, TEVA, AbbVie, Pfizer, and Lundbeck. SSL has received speaker honoraria and/or served as a clinical advisor for Novartis, Lilly, TEVA, AbbVie, Pfizer, and Lundbeck. GMA has received speaker honoraria for Organon, and Lundbeck. ER has received honoraria as a consultant and speaker for: Organon and Dr. Reddy's. The remaining authors declare that they have no conflicts of interest.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Open practices
Not applicable.
Supplemental material
Supplemental material for this article is available online.
