Abstract
Background
Candesartan and topiramate are both recommended for migraine prevention, but direct comparative data remain limited.
Aim
To evaluate and compare the tolerability and effectiveness of candesartan versus topiramate for migraine prevention using systematically collected real-world data.
Methods
This longitudinal cohort study included patients treated at the Leiden Headache Center (LHC), with data collected using our validated E-headache diary. The 28 days preceding treatment initiation served as the baseline period. The primary endpoint was the proportion of participants that discontinued candesartan or topiramate within the 6-month follow-up. Secondary endpoints included ≥50%-response rate, change in monthly migraine days (MMD), headache days (MHD), acute medication days (MAMD), and HIT-6 from baseline to the last 28-day period with the highest achieved dosage of candesartan (ranging 4–32 mg daily) or topiramate (25–100 mg daily). Patients were assigned a propensity score based on baseline MHD, MMD, HADS, and number of failed preventive treatments. The primary analysis looked at discontinuation rates of candesartan and topiramate by using a Kaplan-Meier curve with Cox regression, adjusting for group differences by including the propensity score as a covariate. Secondary endpoint regressions were adjusted for propensity score and time to reach the highest dosage month. The primary and secondary outcomes were also investigated in a sensitivity analysis by applying optimal matching based on the propensity score.
Results
In total 661 migraine participants were included. In the candesartan group less participants discontinued medication within 6-months follow-up compared to the topiramate group (29.7% versus 67.3%; HR 2.5 [95% CI: 1.9–3.3], p < 0.001). Candesartan had higher ≥50% response rates than topiramate (47% versus 29%, OR 0.6 [95% CI: 0.4–0.8], p = 0.004) and greater reduction in MHD (difference −1.1 days [95% CI: −2.2 to −0.01], p = 0.04). No differences were observed between treatments in reduction of MMD, MAMD, or HIT-6 scores after adjustment. These findings were confirmed in the sensitivity analysis using optimal matching.
Conclusions
Candesartan demonstrates a favorable tolerability and effectiveness profile compared to topiramate. Therefore, (inter)national treatment guidelines for migraine prevention should be revised to spare patients from being prescribed medications that have a low tolerability profile, as demonstrated by this study.
This is a visual representation of the abstract.
Introduction
The aim of preventive treatment in migraine is to reduce the frequency and severity of attacks. It is usually indicated for patients who suffer from at least two migraine days per month. Preventive medication can be classified as first-, second- or third-line options. Regulatory restrictions limit CGRP-medication use to patients who have failed first-line medications in most countries. 1 This hierarchy can lead to a compulsory trial-and-error trajectory of a multitude of preventive medications. In migraine prevention, therapeutic success largely depends on adherence, which is often compromised by poor tolerability due to adverse effects. 2
Topiramate is regarded as a first-line option due to its strong evidence supporting its use in migraine prevention. Several placebo-controlled trials have demonstrated its efficacy, and one trial with the active comparator propranolol showed similar effectiveness.2,3 Importantly, the use of topiramate is relatively contra-indicated in some countries in young, fertile women due to the teratogenic risk. In the Netherlands, however, although debate on this issue is beginning to emerge, topiramate remains a compulsory preventive medication in the work-up towards CGRP-mAbs. The adverse effects of topiramate most commonly include paresthesia, fatigue, anorexia, and nausea. 4 Another first-line option is candesartan, an angiotensin-II-receptor antagonist. 1 A recent randomized, triple-blind, placebo-controlled, phase 2 trial from Norway has shown that candesartan 16 mg daily performed significantly better than placebo, and that even candesartan 8 mg once daily was similarly effective. 5 Additionally, small cross-over trials also show candesartan's efficacy is superior to placebo and non-inferior to propranolol in episodic migraine.6–8 Its low cost, minimal side effects, and few drug interactions make it an attractive first-line option.6–8 Two real-world retrospective studies investigated candesartan in a study population that included chronic and treatment-resistant migraine. Both studies showed beneficial effects, with dizziness and hypotension as the most reported adverse events.9,10
This large real-world cohort study aimed to directly compare candesartan to topiramate, building on a prior finding that the CGRP monoclonal antibody erenumab has fewer side effects than topiramate. 2 The Dutch reimbursement system requires the trial of both candesartan and topiramate, among others, before anti-CGRP therapy can be tried. To help guide clinicians in selecting a first-line migraine preventive treatment, we assessed treatment failure, measured by medication discontinuation, of candesartan and topiramate over six months, hypothesizing that candesartan offers comparable effectiveness but better tolerability due to fewer adverse events and better adherence. Our aim is to revise guidelines to prioritize candesartan, without the requirement to also try topiramate, before anti-CGRP monoclonal antibodies can be tried.
Methods
Study design
This longitudinal, prospective cohort study included patients treated at the Leiden Headache Center (LHC). The LHC functions not only as a secondary or tertiary headache referral center, but also accepts direct referrals from general practitioners in the Leiden region. The study design was fulfilling all important criteria as described by the RWD guideline of the International Headache Society (IHS). 11 A baseline period was defined as the 28 days preceding the start of treatment. Start of treatment was defined as the first diary entry when participants were either starting to use candesartan 4 mg or topiramate 25 mg. Monthly migraine days (MMD), headache days (MHD) and acute medication days (MAMD) were assessed at the baseline period using the validated Leiden Headache E-diary, and during the 6-month follow-up period.12,13 Change in HIT-6 scores from baseline were determined from a random, representative sub-set of participants. Data were collected prospectively using the validated Leiden Headache E-diary, which comprises a daily questionnaire on headache symptoms, acute and preventive medication use, and general well-being12,14 Medical records were obtained to determine preventive medication history. All participants provided informed consent, and the study was approved by the Leiden University Medical Center Medical Ethics Committee (G20.046, 25-3015).
Participants
The inclusion criteria were as follows: participants were ≥18 years old, had an active migraine diagnosis, and were proficient in the Dutch language. Upon entry in the Leiden Headache Center (LHC) all participants completed an electronic headache questionnaire and a daily electronic headache diary. 12 Patients fill out the E-diary for at least one month before their first appointment at the LHC, so all participants have a baseline period. Our validated electronic diary features a validated algorithm capable of distinguishing between MMD and MHD.12,13 Migraine diagnosis (Migraine without aura (MO) or Migraine with aura (MA)) was verified by a neurologist specialized in headache. Migraine was defined according to the International Classification of Headache Disorders (ICHD-3). 15 Both episodic (EM) and chronic (CM) migraine patients were included. Participants were excluded if they had comorbid cluster headache or medication overuse headache (MOH). Additional exclusion criteria were <80% diary adherence or use of candesartan or topiramate during the baseline month. Overall in our study, all participants had ≥80% diary adherence. Preventive medication was gradually up-titrated, based on effectiveness and side effects. For candesartan, the starting dose was 4 mg daily, with the dosage increased by 4 mg each month until reaching at least 16 mg per day (with the option to increase up to a maximum of 32 mg per day if preferred by the participant). Although some patients experience initial benefit at a 16 mg dose, the effect may fade over time, requiring an increase in dosage to as much as 32 mg in a small number of participants. For topiramate, the starting dose was 25 mg, with the dosage increased by 25 mg every two weeks until reaching 100 mg per day (50 mg twice daily). In accordance with our standard of care, patients were advised to maintain their current dose if it was effective, and to avoid further increases or reduce the dose only if side effects occurred at higher levels. All participants were naïve for specific anti-CGRP therapies. Drop-outs were defined as participants who stopped completing the E-diary within the 6-month follow-up.
Outcomes
The primary endpoint was the proportion of participants that discontinued candesartan or topiramate within the 6-month follow-up period. Additionally, we looked at the distribution of the dosages at discontinuation. Secondary effectiveness outcomes included ≥50% reduction rate in MMD from baseline, as well as change in MMD, MHD, MAMD and HIT-6 scores from baseline compared to the last 28-day period with the highest achieved dosage of candesartan (4–32 mg daily) or topiramate (25–100 mg daily). The sum of migraine, headache and acute medication days was calculated per 28-day period to determine MMD, MHD and MAMD. HIT-6 scores were assessed monthly.12,16 Secondary effectiveness measures were also determined for MO- and active MA-patients separately, as subgroup analyses. Patients were labelled as having active MA if they were diagnosed with MA and had at least one attack with aura during the follow-up period of the study. Only the active MA patients were included in the subgroup analysis of MA patients.
Statistical analysis
We calculated that a total of 186 participants, n = 93 per group, would need to be included to achieve 80% power at a two-sided 5% significance level. This calculation was based on an estimated 50% discontinuation rate in the topiramate group and a minimal detectable difference of 20% between treatment groups, which was considered clinically relevant.
Data cleaning and analysis were conducted using R statistical software (R version 4.2.1; R Foundation for Statistical Computing, Vienna Austria, 2016; URL: https://www.R-project.org/). For baseline characteristics, descriptive statistics were used. Differences in baseline characteristics were analyzed with a Mann Whitney test for continuous variables, and with a Chi squared test for categorical variables. For the primary analysis, which included all participants until they were censored, a Kaplan-Meijer curve with Cox regression was used to compare discontinuation rates between candesartan and topiramate, as well as an adjusted hazard ratio for discontinuation between the groups in a time-to-event analysis. A Cox regression was chosen, as it takes both censoring and time leading up to the event into account, whereas a logistic regression would just look at the difference in proportion between discontinuation rates.
First, both treatment groups were described using a propensity score consisting of the variables MHD, MMD, HADS-score at baseline and preventive medication history. The selection of variables for inclusion in the propensity score was determined a priori, guided by clinical reasoning. Next we plotted the density of the propensity scores for both medication groups (Supplementary Figure 1A), which showed different density peaks with partial overlap for the candesartan and topiramate groups. The density of a propensity score containing only MHD, MMD and HADS-score at baseline, as well as a propensity score containing only MHD, MMD at baseline and preventive medication history were also plotted. These plots determined that the difference in propensity score density peaks between candesartan and topiramate was mainly due to the difference in prior number of preventive medication use, and partially due to differences in MMD. Adjustment for these differences occurred by adding propensity score as a covariate to our model. For the secondary outcomes, a logistic regression model was used for comparing the ≥50%-response rates, and linear regression models were used for the change in MMD, MAMD, MHD and HIT-6 scores, adjusted for propensity score and time until the last month with the highest dosage was achieved.
Moreover, a sensitivity analysis was performed, which looked at the primary and secondary outcomes in a subset that was optimally matched based on propensity scores. Optimal matching is a matching method, which minimizes the overall sum of pair-wise distances between treatment subjects and matched control subjects. 17 The density of the propensity scores after matching is shown in Supplementary Figure 1B, demonstrating substantially greater overlap between the groups, indicating comparability. Additionally, the primary and secondary analyses were performed in a subset that excluded the drop-outs. Then, subgroup analyses for the secondary effectiveness measures for MO- and (active) MA-patients separately were performed, as well as an additional sensitivity analysis based on optimal matching in these subgroups. Lastly, a subgroup analysis for MMD effect for patients that reached 16 mg candesartan target dose was performed.
Results
Baseline characteristics
A total of 661 participants were included in the study, with 462 receiving candesartan and 199 receiving topiramate (Figure 1). Groups were comparable for sex, age, and HADS-scores (Table 1). Active MA was present in 21.9% (101/462) of the candesartan group and in 31.2% (62/199) of the topiramate group (p = 0.014). Baseline MMD, MHD and MAMD were all slightly higher in the topiramate group. In the candesartan group 27.5% of participants experienced ≥15 MHD with ≥8 MMD in their baseline month, compared to 41.2% in the topiramate group (p = 0.001). HIT-6 score in the candesartan group was 63.7 ± 4.6 and 65.1 ± 3.1 in the topiramate group (p = 0.01). A total of 167 participants (25%), out of both groups, had never received migraine prevention prior to the start of this study. In the candesartan group most participants had used 0–1 prior preventives (65.4%) whereas in the topiramate group most had used 2–3 prior preventives (57.3%) (p < 0.001). This confirmed the rationale for our study design with propensity scoring of the groups for comparison of the primary and secondary outcomes. The subset of participants for whom HIT-6 scores were calculated was representative, as baseline characteristics did not differ from the total group of participants (data not shown).

Flowchart of study.
Baseline characteristics of the study population.

MA: migraine with aura.
MMD: monthly migraine days.
MHD: monthly headache days.
MAMD: monthly acute medication days.
HADS: hospital anxiety and depression scale.
HIT-6: headache impact test.
Medication dosages
In the candesartan group, 44% (203/462) reached the target daily dose of at least 16 mg, while 52% (104/199) of the topiramate group reached the target daily dose of 100 mg, after six months of follow-up for each participant. Additionally, within the candesartan group, 20% reached 4 mg, 21% reached 8 mg, and 15% reached 12 mg. For topiramate, 21% reached 25 mg, 9% reached 50 mg, and 18% reached 75 mg.
Primary outcome
After six months of follow-up for each participant, 29.7% of patients in the candesartan group discontinued their medication compared to 67.3% in the topiramate group (p < 0.001). The adjusted hazard ratio (HR) for discontinuation was 2.5 (95% CI:1.9–3.3, p < 0.001). The unadjusted Kaplan-Meier curve is shown in Figure 2. Among participants who discontinued medication, most stopped at lower or intermediate doses, about 39% discontinued candesartan at 4–8 mg, and around 37% discontinued topiramate at 25–50 mg. Despite many stopping early, approximately 30% of candesartan users and 30% of topiramate users tried the target dosage before discontinuation (Figure 3).
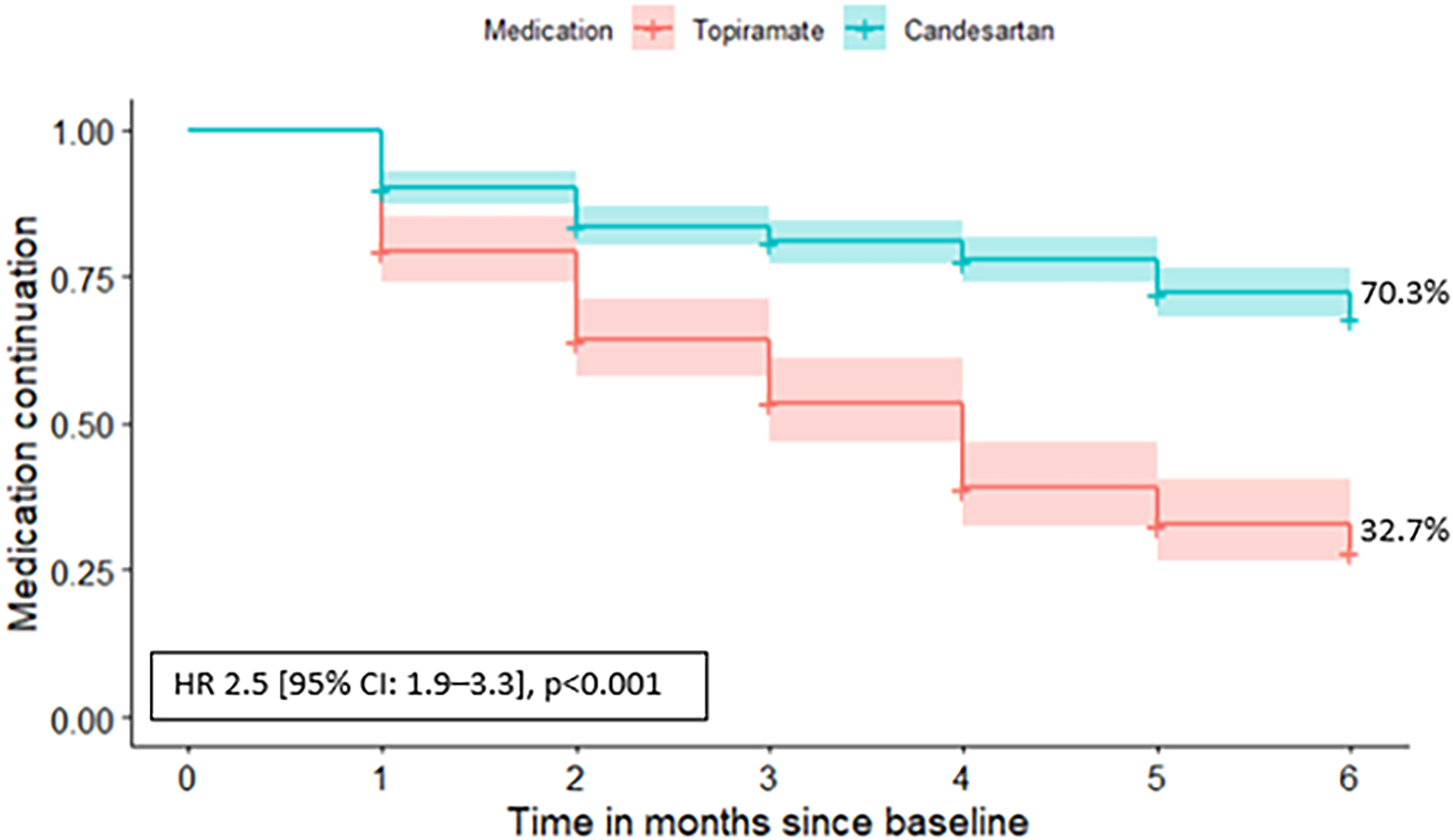
Kaplan-Meier curve of participants who discontinued medication and the calculated hazard ratio, n = 661.

Distribution of the dosages at which participants discontinued medication, within the group that stopped medication. 1 = candesartan 4 mg, topiramate 25 mg; 2 = candesartan 8 mg, topiramate 50 mg; 3 = candesartan 12 mg, topiramate 75 mg; 4 = candesartan 16–32 mg, topiramate 100 mg.
Secondary outcomes
At least 50% reduction in MMD was achieved in 47% of participants in the candesartan group, compared to 29% in the topiramate group (adjusted OR 0.6 [95% CI:0.4–0.8], p = 0.004) (Table 2). Participants using candesartan showed a greater reduction in MHD compared to those on topiramate (−4.5 vs. −3.1, p = 0.04). Adjusted MMD, MAMD and HIT-6 changes were not significantly different between groups (MMD: MMD: −3.0 vs −2.1, p = 0.05, MAMD: −1.1 vs −1.2, p = 0.15, HIT-6: −3.4 vs −1.7, p = 0.68) (Table 2).
Secondary effectiveness outcomes from baseline compared to the month with the highest achieved dosage for all cases, n = 661.

Adjusted OR: odds ratio adjusted for covariates, logistic regression.
Adjusted difference: difference adjusted for covariates, linear regression.
Crude OR: unadjusted odds ratio, Chi-squared test.
Crude difference: unadjusted difference, t-test.
MMD: monthly migraine days.
MHD: monthly headache days.
MAMD: monthly acute medication days.
HIT-6: headache impact test.
*Statistically significant.
Sensitivity analysis based on propensity score matching
Matching was applied as a sensitivity analysis since groups were different at baseline, to be able to replicate the main findings in groups that were more comparable. Matching was based on a propensity score including baseline MMD, MHD, HADS, and preventive medication history. The propensity score density plot after matching is shown in Supplementary Figure 1B, which shows substantial overlap for both groups. In total 37% (72/193) of participants discontinued candesartan versus 67% (129/193) of participants with topiramate, HR 2.4 [95% CI:1.8–3.2] p < 0.001, as seen in Figure 4. All matched secondary outcomes were comparable to the primary analysis that included all participants (Supplementary Table 1).

Kaplan-Meier curve of matched participants who discontinued medication with hazard ratio, n = 386.
Subgroup analysis without drop-outs
Among the 661 participants, 16% (105/661) were lost to follow-up, 19% (86/462) for candesartan and 10% (19/199) for topiramate. This analysis included 425 complete cases, and difference in loss to follow-up was not significant. Medication was discontinued in 33% of the candesartan group compared to 75% of the topiramate group (adjusted HR 2.6 [95% CI: 1.9–3.6], p < 0.001) (Supplementary Figure 2). No secondary outcomes were significantly different between the two groups (Supplementary Table 2).
Subgroup analyses for MO and MA
For MO patients, candesartan gave higher ≥50%-response rates than topiramate (adjusted OR = 0.6 [95% CI: 0.4–0.9], p = 0.03) but not for MMD, MAMD and HIT-6 scores (Supplementary Table 3). For MA patients, candesartan provided a higher reduction in MHD than topiramate (−5.1 vs −2.4, p = 0.04), but results were similar for all other outcomes (Supplementary Table 4).
Moreover, a sensitivity analysis for MO patients separately, again based on propensity score matching, showed medication discontinuation in 39% of the candesartan group compared to 67% of the topiramate group (HR 2.4 [95% CI: 1.7–3.4, p < 0.001). The sensitivity analysis for MA patients separately resulted in discontinuation of medication in 32% of the candesartan group compared to 66% of the topiramate group (HR 2.7 [95% CI: 1.5–4.8, p < 0.001). Results for secondary outcomes were similar between groups (data not shown).
Subgroup analysis for target dose candesartan 16 mg
For patients that reached candesartan 16 mg, ≥50%-response rates, reduction in MMD, MHD and HIT-6 scores were similar to that of the topiramate group, but the reduction in MAMD was higher in the topiramate group (−0.36 vs −1.19, p = 0.005) (data not shown).
Discussion
In this large study candesartan outperformed topiramate. Those who took candesartan were much more likely to stick with the medication compared to those on topiramate; people on topiramate were 2.5 times more likely to discontinue medication. Candesartan treated patients were about twice as likely to see their migraine days cut in half compared to those on topiramate.
This study directly compared the use of candesartan and topiramate for migraine prevention in a stringent and systematic real-world setting. All results were similar for migraine with aura and migraine without aura separately. The primary analysis, which included all participants, yielded comparable results to the sensitivity analysis where optimal matching was applied, and to the analysis that excluded drop-outs.
Our study was inspired by the HER-MES trial, which compared the anti-CGRP-receptor antibody erenumab to topiramate for migraine prophylaxis in adults and found that discontinuation occurred more frequently in the topiramate group. 2 To compare efficacy outcomes the HER-MES trial assessed changes in MMD, ≥50%-response rates, and HIT-6 scores, while our study also included change in MHD and MAMD. In many countries, anti-CGRP treatments are not approved as first-line options for migraine prevention. Health authorities often prioritize cost-effectiveness, favoring the use of more affordable, established medications, such as topiramate, before newer, typically more expensive therapies like anti-CGRP monoclonal antibodies can be considered. As a result, patients are often required to try and subsequently fail 2–4 of these ‘conventional’ preventive treatments, due to lack of effectiveness or intolerable side effects, before physicians are permitted to prescribe anti-CGRP therapies. Reimbursement policies are also commonly tied to this stepwise approach, meaning that insurance coverage or public health funding for anti-CGRP treatments is often contingent upon documented failure of alternative options. This practice may delay access to more effective and better-tolerated treatments, potentially setting back patients by exposing them to preventive therapies that are either ineffective or associated with a high burden of side effects, further impacting their quality of life and long-term treatment adherence. Additionally, candesartan is highly affordable and generally well-tolerated, making it a cost-effective option too. 8 In contrast, topiramate is less well-tolerated and relatively contra-indicated in young, fertile females due to the teratogenic risk. All in all, this study indicates that treatment guidelines and reimbursement-protocols should be revised to prioritize trying candesartan, without the requirement to also try topiramate, before anti-CGRP monoclonal antibodies can be tried.
The findings of our study are consistent with two small crossover trials that compared candesartan to placebo and propranolol,6,7 as well as with two retrospective cohort studies conducted in patients with chronic migraine (CM).9,10 Another relevant study, the CandeSpartan trial, included patients with CM and medication overuse headache (MOH), thus representing a more severely affected population. 8 This study reported a slightly lower discontinuation rate for candesartan than our study, but it did not include an active comparator. Our results regarding the ≥50% response rate for candesartan are also in line with those from the recent candesartan phase 2 trial, which compared candesartan 8 mg, candesartan 16 mg, and placebo. 5 In this study, approximately half of the participants in the candesartan group achieved ≥50% reduction in MMD, closely matching the 47% response rate observed in our study. However, important differences should be noted: the trial excluded participants with CM, did not use dose titration, and relied on self-reported migraine days.
Our study's strengths include the use of a real-world cohort with a validated E-diary that can objectively distinguish between (non-)headache and migraine days. 13 While an RCT may ensure a minimal influence of confounding factors, our study reflects actual clinical conditions, which contributes to the external validity of our study. Additionally, our six-month follow-up period for each participant provides valuable insight into the medium- to long-term effects of both medications. This duration also allows for slow and gradual dose escalation, which can help reduce the occurrence of side effects. This slow up-titration allows both candesartan and topiramate to be optimally tried and not be dismissed too early due to side effects. Additionally, a small subset of participants escalated their candesartan dose, with 11 individuals increasing to 24 mg and two reaching the maximum dose of 32 mg. Furthermore, our study gives valuable insights into the tolerability of both medications, which potentially influences the relevant discussion if topiramate should be regarded as a first-line prophylactic treatment at all. Lastly, in addition to the primary analysis, we performed a sensitivity analysis with matched data, which produced similar discontinuation rates, showing the robustness of our findings.
A potential limitation of our study is the baseline differences between the two groups, which we accounted for by adding a propensity score to our models based on baseline MMD, MHD, HADS and number of prior preventives. The density of the candesartan curve peaks at the lower end of the propensity scores, which can be explained by the fact that we looked at prior number of prevention use in our propensity score, and most patients in our cohort will have used candesartan before topiramate due to the Dutch national treatment guideline. In our cohort, 164/199 topiramate participants had tried candesartan before topiramate, at other hospitals or at the general practitioner before visiting our outpatient clinic. However, both candesartan and topiramate remain first-line options in the Dutch treatment guidelines, and have to be tried in addition to all other standard of care preventive options, before CGRP-mAbs can be tried. Moreover, this study was performed when there was no official reimbursement available for CGRP-mAbs yet. Thus, eagerness to try CGRP-mAbs and therefore reporting lower tolerability is believed to not have played a role in either group. Additionally, more patients in the topiramate group had CM and higher MAMD compared to the candesartan group, and it should be noted that certain unmeasured factors such as pain catastrophizing are more common in CM patients. However, we accounted for this difference in number of CM patients and higher MAMD by including MMD and MHD in our propensity score. Then, we also matched our patients based on propensity score; leaving only comparably affected participants in both groups. This produced similar results as when we adjusted for propensity scores. Moreover, in a trial setting only treatment-naïve participants would be included, but we believe that this real world study more accurately reflects everyday clinical practice. Another limitation might be the unequal group sizes, with the candesartan group being approximately twice as large as the topiramate group, however the matched analysis mitigates this issue as well by ensuring equal group sizes. Anotherther limitation that needs to be addressed is the relatively small number of participants in the MA sub-analysis, although results remained stable across all analyses. Also, timelines for dose escalation were different for candesartan and topiramate. Additionally, the ≥50%-response rate in the topiramate group was 29%, which is lower than in previous studies about the efficacy of topiramate. 18 This could be due to a higher number of CM patients and a higher number of prior preventives in the topiramate group. Adjustment for these differences occurred via propensity score matching. Lastly, we did not ask our participants to specify the reason for failure of medication, which could be due to insufficient response or non-tolerability of treatment. However, our study focused on practical applicability and aimed to provide real-world outcomes on candesartan and topiramate tolerability and effectiveness, regardless of the underlying reason for discontinuation, since from a clinical perspective the specific reason matters less than the outcome, which is treatment discontinuation. Moreover, while the exact reason for failure of either medication was not recorded, nonetheless, we may conclude that candesartan outperforms topiramate in this study.
Conclusion
Candesartan demonstrates a favorable tolerability and effectiveness profile compared to topiramate over a 6-month follow-up period: subjects on topiramate were 2.5 times more likely to stop taking it. Candesartan treated patients were about twice as likely to have a 50% reduction of their migraine days compared to those on topiramate. Based on these findings, (inter)national treatment guidelines for migraine prevention should be revised to spare patients from being prescribed medications with a low tolerability profile, due to either ineffectiveness or adverse side effects, as demonstrated by this study.
Key Findings
In this real-world data comparative cohort study, candesartan demonstrated favorable tolerability and effectiveness compared to topiramate for migraine prevention over a 6-month period.
(Inter)national treatment guidelines for migraine prevention should be revised to spare patients from being prescribed medications with a low tolerability profile, as demonstrated by this study.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261426952 - Supplemental material for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study
Supplemental material, sj-docx-1-cep-10.1177_03331024261426952 for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study by Annemijn S.J.C. Oosterlee, Britt W.H. van der Arend, Nancy van Veelen, Irene de Boer, Anique de Groot, Benthe Geuns, Erik van Zwet, Nadine Pelzer and Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-png-2-cep-10.1177_03331024261426952 - Supplemental material for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study
Supplemental material, sj-png-2-cep-10.1177_03331024261426952 for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study by Annemijn S.J.C. Oosterlee, Britt W.H. van der Arend, Nancy van Veelen, Irene de Boer, Anique de Groot, Benthe Geuns, Erik van Zwet, Nadine Pelzer and Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-png-3-cep-10.1177_03331024261426952 - Supplemental material for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study
Supplemental material, sj-png-3-cep-10.1177_03331024261426952 for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study by Annemijn S.J.C. Oosterlee, Britt W.H. van der Arend, Nancy van Veelen, Irene de Boer, Anique de Groot, Benthe Geuns, Erik van Zwet, Nadine Pelzer and Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-png-4-cep-10.1177_03331024261426952 - Supplemental material for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study
Supplemental material, sj-png-4-cep-10.1177_03331024261426952 for Superior outcomes of candesartan over topiramate in the treatment of migraine: A comparative cohort study by Annemijn S.J.C. Oosterlee, Britt W.H. van der Arend, Nancy van Veelen, Irene de Boer, Anique de Groot, Benthe Geuns, Erik van Zwet, Nadine Pelzer and Gisela M. Terwindt in Cephalalgia
Footnotes
List of abbreviations
Acknowledgements
Not applicable.
ORCID iDs
Ethical considerations
The study is performed in accordance with the declaration of Helsinki Ethical Principles and Good Clinical Practices and was approved by the local ethics committee. STROBE guidelines for observational studies were followed.
Consent to participate
All participants provided written informed consent.
Consent for publishing
All authors read and approved the final manuscript.
Author contributions
ASJCO, BWHvdA, NvV, IdB, NP and GMT contributed to the study design. ASJCO and EvZ are responsible for analyzing the data. ASJCO, AdG and BG are responsible for data collection. ASJCO is responsible for drafting the manuscript. GMT is the principal investigator and supervised this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.S.J.C. Oosterlee has no conflicts of interest to report. B.W.H. van der Arend reports independent support from the Netherlands Organization for Health Research and the Dutch Brain Foundation, and support from Organon. N. van Veelen has no conflicts of interest to report. I. de Boer reports independent support from the European Community, Dutch Heart Foundation, Dioprahte and the Clayco Foundation. A. de Groot, B. Geuns and E. van Zwet have no conflicts of interest to report. N. Pelzer reports consultancy or industry support from AbbVie and Lundbeck and independent support from the Dutch Research Council. G.M. Terwindt reports consultancy support from AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Teva and independent support from the Dutch Research Council, European Community, Dutch Heart Foundation, Dutch Brain Foundation, Dioraphte, and the Clayco Foundation.
Data availability statement
The data that support the findings of this study are available on reasonable request to the corresponding author.
Open practices
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
