Abstract
Introduction: Demographic and clinical variables were examined in a post hoc analysis of the PROlonged Migraine Prevention with Topiramate (PROMPT) study to determine potential contribution to relapse.
Methods: After a six-month open-label (OL) topiramate phase, patients were randomised to continue topiramate or switch to placebo in a six-month double-blind (DB) phase. ‘Relapse’ was investigated in terms of change in monthly migraine days after randomisation compared with the month before randomisation, and was analysed during the first (‘initial relapse’) and last month (‘sustained relapse’) of the DB phase. More than 40 potential predicting factors were entered into analyses of variance and covariance.
Results: For initial relapse, variable-by-treatment interactions were significant for the Headache Impact Test (HIT-6) at DB baseline, and decline in acute medication intake or reporting of ‘anxiety’ in the OL phase. For sustained relapse, no statistically significant interactions were observed.
Conclusion: Relapse after topiramate discontinuation in migraine prophylaxis appears to be unaffected by patient characteristics or baseline migraine frequency.
Introduction
People who experience regular migraine attacks have impaired quality of life and reduced ability to carry out normal daily activities (1,2). National and international guidelines recommend prophylactic therapy when the attacks are very frequent or particularly severe, with reassessment of treatment after 3–12 months (3–5). A recent study that investigated the outcome of discontinuing prophylactic therapy after six months—the PROlonged Migraine Prevention with Topiramate (PROMPT) study—found sustained benefit in terms of a permanently reduced migraine frequency level (6). There was, however, a small but sustained increase in migraine frequency after discontinuation compared with continuation of prophylactic therapy. In a post hoc analysis of the PROMPT data we examined demographic and clinical variables to determine their potential contribution to relapse.
Methods
Details of the PROMPT study have been published previously (6). In brief, migraine patients who experienced at least four migraine days per month (28 days), during a one- to two-month prospective baseline phase, entered a six-month open-label (OL) treatment phase in which they received topiramate (initial target dose: 100 mg/day; flexible dosing [50–200 mg/day] thereafter). At the end of the OL phase, patients were randomised in a six-month double-blind (DB) phase to receive either the same dose of topiramate or corresponding placebo (1 : 1). The primary end point was the change in number of migraine days during the last four weeks of the DB phase relative to the last four weeks of the OL phase. A migraine day was defined as a calendar day during which the patient had a migraine headache. Migraine headaches and intakes of acute medication (mainly triptans and analgesics) were recorded by patients using a headache diary. During pre-scheduled visits, health-related quality of life was assessed using the Short-Form 12 (SF-12) general health status questionnaire, the Migraine Disability Assessment (MIDAS) and the Headache Impact Test (HIT-6).
In the present analysis ‘relapse’ was investigated in terms of the change in the number of monthly migraine days after randomisation compared with the number of migraine days in the month before randomisation (‘DB baseline’; i.e. the last month of the OL phase). The analysis focused on relapse during the first month and during the last month (month 6) of the DB phase, referred to as ‘initial relapse’ and ‘sustained relapse’, respectively. Patients from both treatment arms (topiramate and placebo) were included in analyses of variance and covariance (
Potential predictive variables included: demographic parameters (sex, age, body mass index); disease-related parameters such as number of monthly migraine days, number of monthly acute medication intake days and quality-of-life parameters (SF-12, MIDAS, HIT-6 scores), assessed at prospective baseline or at DB baseline. Additional potential predictors included change in migraine days and change in acute medication intake days during the OL phase, assessed at DB baseline, and the reporting of ‘depression’ or ‘anxiety’ as an adverse event at any time before randomisation. Given the exploratory nature of the analyses, no corrections were made for multiple comparisons, and significance of treatment-by-variable interactions was defined at the P < .1 level.
Results
Initial relapse
For the change in migraine days over the first month after randomisation, the
During the first month of the DB phase the increase in migraine days with placebo was greater in those with a lower HIT-6 score (i.e. those less severely affected by their headaches), whereas the increase in migraine days in the topiramate group was consistent regardless of HIT-6 score, leading to a statistically significant interaction term (P < .05) (Figure 1). This can also be seen when HIT-6 scores are presented in the four standard categories with increasing impact on quality of life (Table 1); the increase in migraine days as a result of treatment withdrawal was greater in patients with a lower headache severity at DB baseline, and was also more extended in the placebo group than in the topiramate group.
Change in number of migraine days during the first month of the double-blind (DB) phase according to HIT-6 score at DB baseline (P < .05 for treatment-by-HIT-6 score interaction). HIT-6 = Headache Impact Test. Change in number of migraine days during the first month in the DB phase, per treatment group, for variables with a statistically significant treatment-by-variable interaction DB = double-blind. SD = standard deviation. HIT-6 = Headache Impact Test. OL = open-label.
P < .05; **P < .001 vs. DB baseline.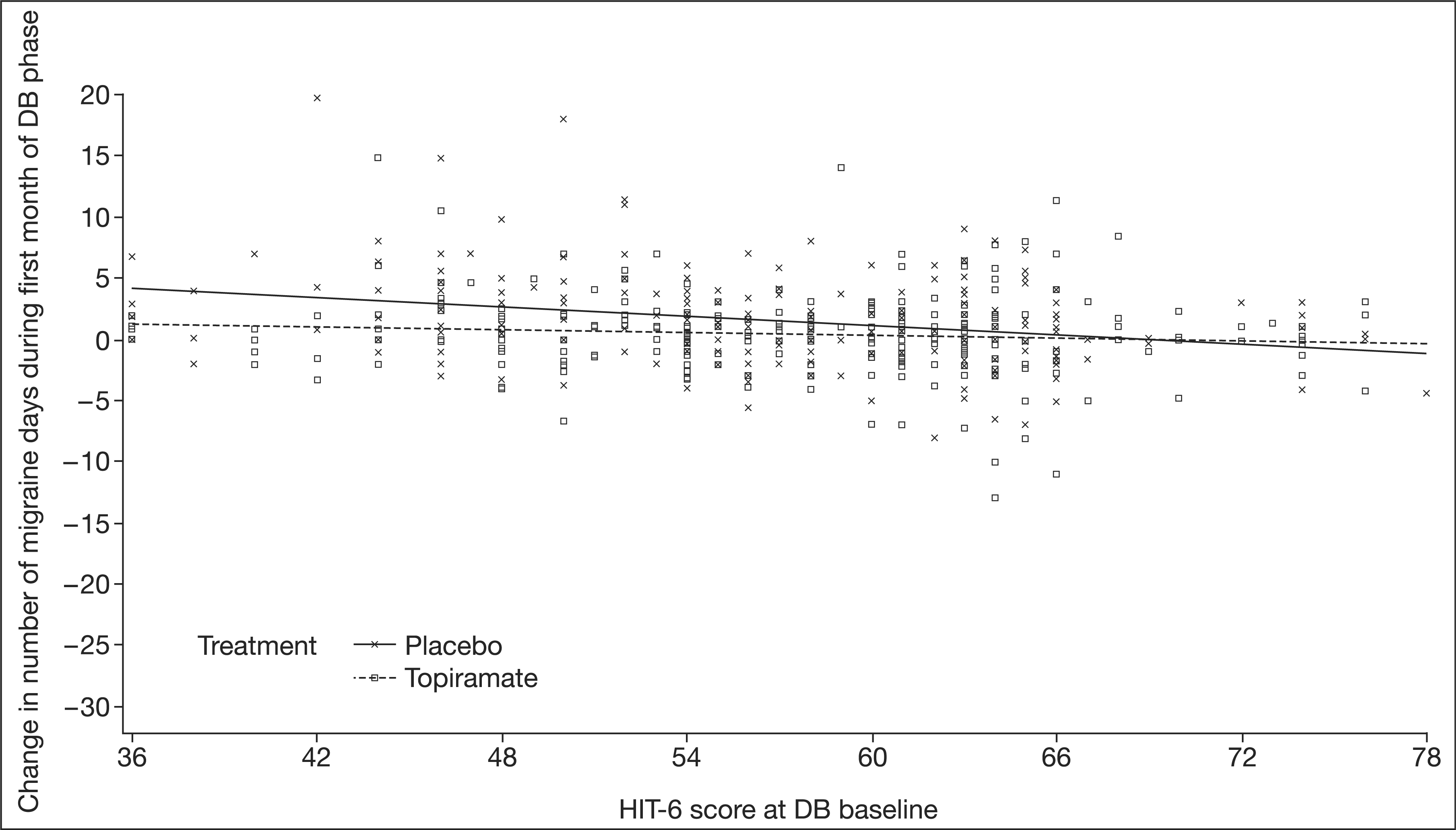

Patients who generally used medication for the acute treatment of migraines before randomisation showed a larger increase in migraine days immediately after randomisation if switched to placebo than if they continued topiramate (Table 1). The reverse was true in patients who did not receive acute medication in the OL phase, although it should be noted that there were very few patients without acute medication in the study.
With placebo, patients who had reduced their acute medication intake in the OL phase appeared to have a higher rate of initial relapse in the DB phase than those who had used the same or even more acute medications in the OL phase compared with prospective baseline. With topiramate, however, initial relapse in the DB phase appeared to be independent of the change in acute medication intake over the OL phase (Figure 2).
Change in number of migraine days during the first month of the double-blind (DB) phase according to the change in intake of acute medication over the open-label (OL) phase (P < .05 for treatment-by-intake of acute medication interaction).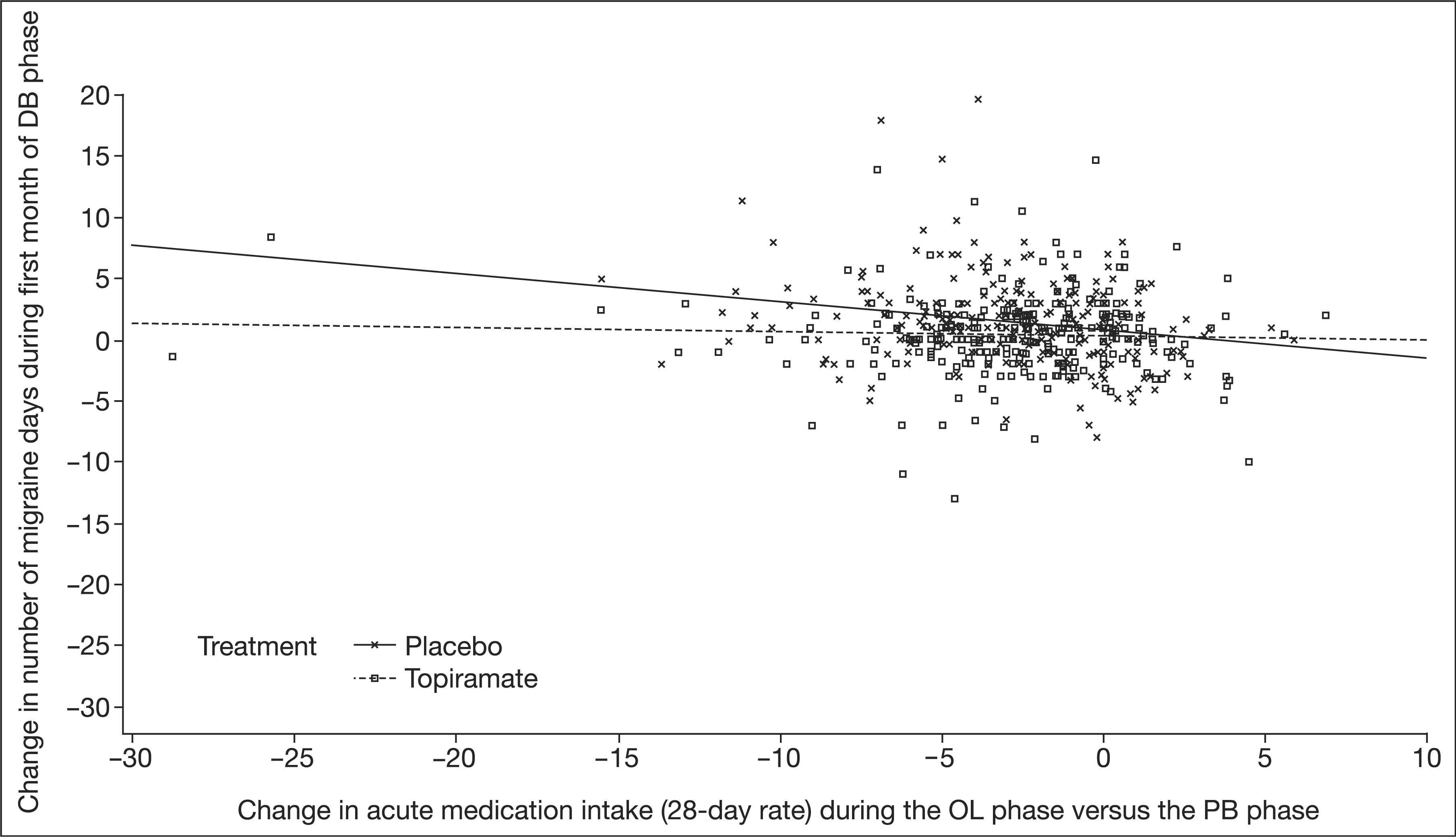
Patients who were switched to placebo had a higher likelihood of initial relapse if they had reported ‘anxiety’ earlier in the study, compared with those who had not. This was not true for those randomised to continue topiramate (Table 1), although the number of patients in the topiramate group with previously reported anxiety was small.
Sustained relapse
In the last month of the DB phase, no statistically significant interactions were observed for any of the investigated factors listed above. Plotting the increase in migraine days during the DB phase (last month DB vs. DB baseline) against the reduction in migraine days during the OL phase (last month OL vs. prospective baseline) resulted in parallel linear regression lines for placebo and topiramate (Figure 3). The slopes of the lines indicate higher relapse probability with higher initial benefit (i.e. the more pronounced the reduction in migraine frequency during six months of OL treatment with topiramate, the higher the likelihood of relapse after randomisation to continuation or discontinuation of topiramate treatment in the DB phase). The distance between the lines represents the maintained migraine-reducing effect of topiramate over placebo.
Change in number of migraine days during the last month of the double-blind (DB) phase according to the change in migraine days over the open-label (OL) phase (P = .9131 for treatment-by-change in OL phase interaction).
During the analyses no new safety issues were identified that would necessitate adjustment of the previously reported safety profile of topiramate in this study (6).
Discussion
The
In the first month after randomisation, an increase in migraine frequency was found in both treatment groups, but the increase was statistically significantly more pronounced in the placebo group than the topiramate group (6). After randomisation, patients in the topiramate group continued treatment at the same dose, so the initial relapse in this group cannot be explained by topiramate treatment, but should rather be ascribed to some sort of ‘inverse placebo effect’: the expectation of being switched to placebo might have raised patients’ awareness of their migraines, leading to an increased reporting of migraines compared with just before randomisation.
Three factors positively predicted ‘initial relapse’—low HIT-6 score at randomisation, decrease in acute medication intake during topiramate treatment, and reports of ‘anxiety’ as an adverse event—although, given the large number of variables, the possibility of a chance finding cannot be ruled out.
HIT-6 is a measure of the impact of migraine headaches on the patient’s quality of life, and a low HIT-6 score reflects comparatively low impairment in daily functioning. Acute medication intake is needed only after a migraine headache occurs, so an overall reduction in acute medication intake can be expected with a reduction in the number of migraines. Indeed, the number of acute medication intake days paralleled the number of migraine days in both phases of the study (6). Therefore, these two predicting factors seem to represent patients who responded comparatively well to migraine preventive treatment with topiramate, similarly to those described above who presented with a more pronounced reduction in migraine frequency. These predicting factors can therefore also be explained, indirectly, by the same phenomenon: regression to the mean.
There is ample evidence for comorbidity between migraine and anxiety, as there is for migraine and depression, with some indication for a stronger relationship of the former compared with the latter (8). A retrospective analysis of the effectiveness of the antidepressant duloxetine in migraine prevention indicated that duloxetine was only minimally effective as a migraine prophylactic agent overall, but that it might have greater than average effectiveness in patients with anxiety (9). Conversely, no evidence of greater effectiveness was found in patients with depression. In the present analysis we found that anxiety, but not depression, increased the probability of relapse after discontinuation of migraine prevention. It is tempting to speculate that the fear of receiving placebo, as the apparent driving force for initial relapse in both treatment groups, is more pronounced in patients with coexisting anxiety, thus explaining why anxiety appeared to be a predicting factor for initial, but not sustained, relapse after discontinuation of migraine preventive therapy.
In conclusion, the probability of relapse after discontinuation of migraine prophylaxis with topiramate is not affected by patient characteristics or baseline migraine frequency, but appears to be highest in those who benefited most from the original therapy instalment.
Footnotes
Acknowledgements
The authors gratefully acknowledge recommendations on the statistical analyses, given by William H. Olson, PhD, Ortho-McNeil Janssen Scientific Affairs, Raritan, NJ, USA, and editorial assistance in the preparation of the manuscript, provided by Daniel Booth, Bioscript Stirling, London, UK.
