Abstract
The aim of this study was to assess the cost-effectiveness of topiramate vs. no preventive treatment in the UK. Model inputs included baseline migraine frequency, treatment discontinuation and response, preventive and acute medical cost per attack [2005 GBP (£)] and gain in health utility. Outcomes included monthly migraines averted, acute and preventive treatment costs and cost per quality-adjusted life year (QALY). Topiramate was associated with 1.8 fewer monthly migraines and a QALY gain of 0.0384. The incremental cost of topiramate vs. no preventive treatment was about £10 per migraine averted and £5700 per QALY. Results are sensitive to baseline monthly migraine frequency, triptan use rate and the gain in utility. Incorporating savings from reduced work loss (about £36 per month) suggests that topiramate would be cost saving compared with no preventive treatment. This analysis suggests that topiramate is a cost-effective treatment for migraine prevention compared with no preventive treatment.
Introduction
Migraine is a common, chronic and potentially progressive neurological disorder characterized by severe, disabling headaches that are usually accompanied by nausea or vomiting, photophobia and phonophobia (1). Approximately 6 million people in the UK (8% of men and 18% of women) have experienced migraine, with peak prevalence occurring during prime working years (ages 25–55 years) (2). About 50% of migraineurs in the UK report one or more attacks per month and as many as 13% experience one or more attacks per week (2). Despite the development of significantly improved treatment options in recent years, migraine remains widely underdiagnosed and undertreated in the UK and the majority of patients do not obtain effective relief (3–8). Diagnosis and management are further complicated by broad and varied clinical characteristics of migraine and potential concomitancy with other neurological diseases.
The economic burden of migraine to patients, employers, health systems and society is substantial because migraine predominately affects young and otherwise productive individuals (9–11). The majority of this economic burden is borne by employers as costs attributable to absenteeism or reduced/lost workplace productivity (i.e. ‘presenteeism’) and comprises 75–90% of the total economic cost of migraine (5, 9, 12, 13).
Treatment strategies for migraine have been primarily directed towards managing acute attacks, with preventive therapy generally restricted to patients with frequent migraine attacks (14–16). An effective migraine preventive therapy will reduce migraine frequency, severity and associated disability. It will also reduce the need for acute medication and dose escalation/titration, which in turn decreases the potential for overuse reported for several acute pharmacotherapies, such as triptans (14). However, due to the limited number of effective treatments, additional preventive options are needed (14–16).
Recent studies have demonstrated that some neurostabilizers (i.e. antiepileptics) are effective monotherapies for migraine prevention. Topiramate (TPM), a neuromodulatory compound with stabilizing properties, has been demonstrated in several large, randomized, placebo-controlled trials to be an effective and generally well-tolerated migraine preventive therapy and is approved in the UK and many other countries for migraine prevention (17–19).
Cost-effectiveness analyses of new treatments are increasingly requested by stakeholders to evaluate the value of new treatment strategies. A decision analytic model has been developed to assess the cost effectiveness of TPM in the USA (20). In this study, the economic savings associated with reduced migraine frequency offset approximately two-thirds of the cost of preventive TPM therapy. However, these results are not necessarily generalizeable to the UK due to differences in input prices and the treatment patterns in the USA vs. the UK. This analysis adapted the previous US model to assess the cost effectiveness of TPM from the UK National Health Service (NHS) perspective.
Methods
Study design
The general description of the decision-analytic model has been described in detail elsewhere (20). Briefly, all patients in the model are assumed to be migraineurs with moderate to high frequency of migraine. Model results are presented as monthly estimates, although we used a time horizon of 12 months to allocate the costs of treatment discontinuation. Model outcomes include the expected numbers of migraines averted per month, expected monthly costs of therapy for treated and untreated patients, expected acute treatment costs and expected work loss due to migraine episodes. From these estimates, cost-effectiveness ratios and net economic benefit associated with TPM use vs. no preventive treatment were calculated.
Structure of the decision tree
The model population was assumed to be adults who are candidates for migraine prevention using TPM. The base-case monthly migraine frequency was assumed to be six per month, consistent with the mean rate in the TPM trials (17–19). Treatment guidelines recommend consideration of preventive therapy for patients with two or more migraines per month (15). The beginning of the decision tree (Fig. 1) sets out the options of TPM treatment or no preventive treatment. Those in the no preventive treatment branch are assumed to have no preventive treatment costs or clinical benefits but incur acute treatment costs. Patients in the TPM arm could either discontinue or continue treatment, and have one of three possible clinical responses: ≥75%, 50–75% or <50% reduction in migraine frequency. Each response category was also assigned a gain in utility based on the change in health state associated with that response category (i.e. higher utility for health states with fewer migraines) and that utility value was used to calculate the gain in quality-adjusted life years (QALYs) for TPM treatment.

Schematic diagram of the model.
Monthly migraine frequency, cost of preventive therapy and health utilities were calculated for all patients based on their initial treatment choice and outcome (Fig. 1). Subsequently, the monthly migraine frequency for each group was used to calculate the direct and indirect costs of migraine-related treatment and lost productivity, as well as estimates of the number of migraine-induced hours of disability for both TPM-treated and no preventive treatment patients. Within each response category every migraine attack was categorized as occurring on a work day or a non-work day. Furthermore, there was an associated probability that each attack was treated with a triptan medication or ‘usual care’ [i.e. non-triptan treatment, such as over-the-counter medications or other prescription acute migraine treatments (e.g. dihydroergotamine)]. Model outcomes are combined to calculate the cost per migraine averted and the cost per QALY.
Clinical parameter estimates
All clinical parameter estimates are described in Table 1. Estimates from the TPM clinical trials are based on the TPM 100 mg/day group, matching the recommended dosage in the UK market (17–19, 21).
Clinical and healthcare utilization model parameters, with baseline values
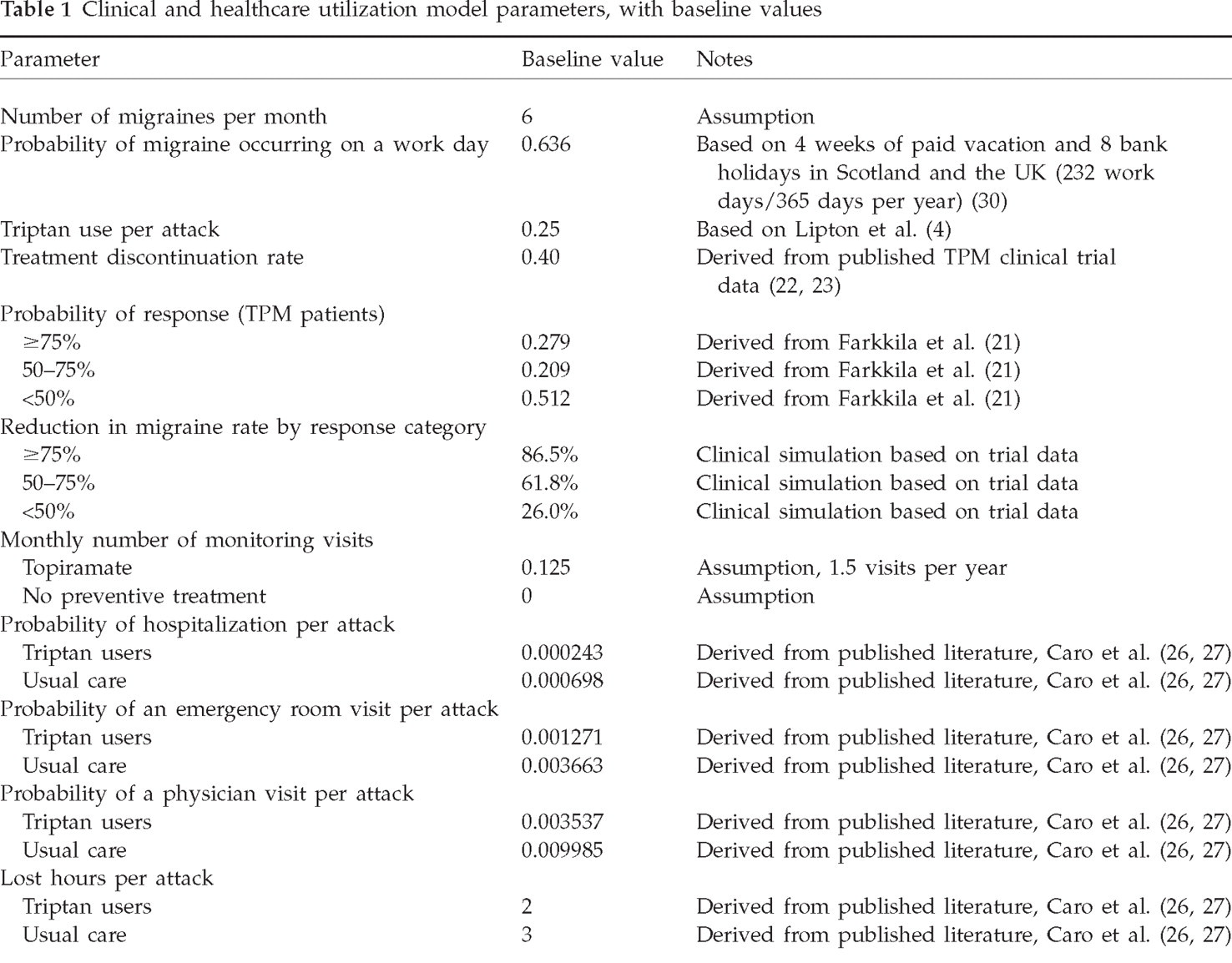
Treatment discontinuation
Pooled clinical trial data for the 100 mg/day TPM arm from three randomized, double-blinded, placebo-controlled studies of TPM in migraine prevention reported an overall discontinuation rate of approximately 40% (22, 23). This rate includes discontinuation for any cause (100 mg/day group), including adverse events (25%), subject choice (4%) and other causes such as lack of efficacy and loss to follow-up (7%) (22). The most common adverse events in the three pivotal trials of TPM in migraine prevention were paraesthesia, fatigue, anorexia, nausea and cognitive impairment. Treatment drop-outs were assumed conservatively to have discontinued treatment during the first month of treatment.
Reduction in migraine frequency
The clinical response estimates were based on the reduction in migraine frequency reported in the pooled trial results for the 100 mg/day TPM arm (21). Data from the pooled analyses were used to assign each patient to a response category (≥75%, 50–75% and <50% reduction in migraine frequency) which represents a distinct health state based on migraine frequency. The probability of being assigned to each response category was as follows: 0.279 for the ≥75% group, 0.209 for the 50–75% group and 0.512 for the <50% group (21). We then simulated (n = 10 000 trials) the average reduction in migraine frequency for each response category (Table 1). For example, from this simulation, we estimated that patients in the highest response category (≥75% group) would have an average reduction in migraine frequency of 86.5%. The simulation was conducted using Crystal Ball 2000.2 Standard Edition (Decision Engineering, Denver, CO, USA). Treatment drop-outs were assumed conservatively to receive no clinical benefit, as were those receiving no treatment. Table 1 includes details of all estimates of clinical parameters.
Quality-adjusted life years
Changes in quality of life associated with TPM treatment were estimated through societal valuations (i.e. ‘utilities’) derived from transformations of Short Form-36 (SF-36) Health Survey data collected as part of the TPM clinical trials (24). Utilities quantify the subjective preferences of individuals for particular health states and typically range from perfect health (utility = 1.0) to dead (utility = 0.0). The utility calculations for this study have been described previously and values are presented in Table 2 (20). Patients not receiving treatment, or those discontinuing TPM treatment, were assumed to have no utility gain.
Cost and utility estimates
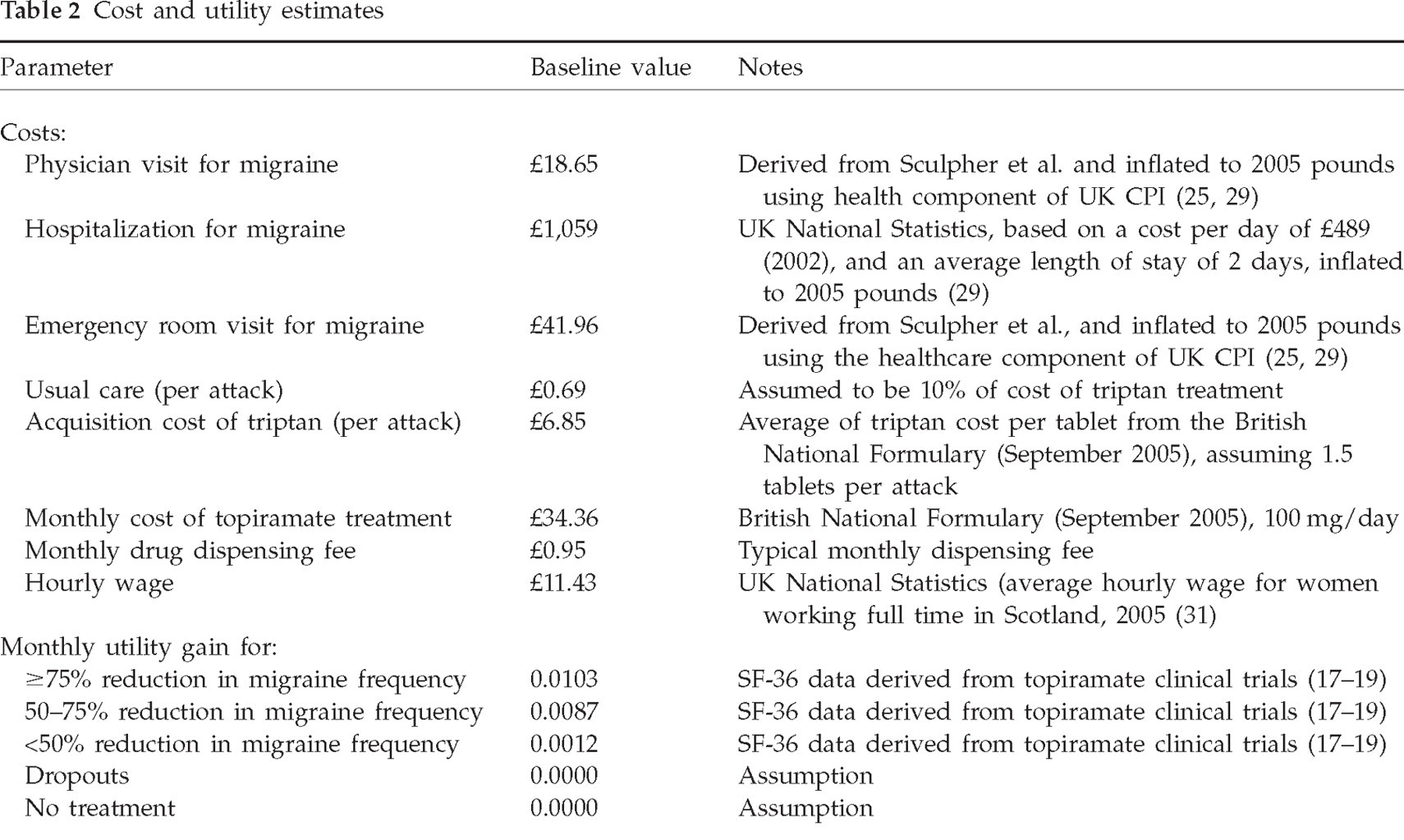
Cost parameter estimates
Direct preventive treatment costs
The expected monthly cost of TPM treatment (UK£2005) is based on its acquisition and dispensing cost, as well as on expenses incurred from healthcare visits for drug titration and monitoring. The monthly TPM ingredient cost (£34.36) is based on the British National Formulary (September 2005) price for 60 50 mg tablets of TPM and a daily dosage of 100 mg. The model assumes 1.5 TPM-related physician-monitoring visits per year (0.125/month) at a cost of £18.65 (25). This estimate of 1.5 visits represents care that is related solely to topiramate treatment; it is assumed that most patients who are candidates for preventive migraine treatment are under the care of a physician and have multiple visits per year. The monthly dispensing fee is set at £0.95. Treatment drop-outs are assigned 1 month of the cost of TPM treatment.
Direct acute treatment costs
The use of acute medical services per attack was estimated using published literature (Table 1) (26, 27). For each migraine attack and acute treatment type (i.e. triptan or usual care), the probability of an emergency room visit, hospitalization and/or a physician visit was calculated and incorporated into the model. Attacks treated with triptan medications have a lower probability of requiring these additional healthcare services compared with attacks treated with usual care (26–28). It is estimated that 25% of all migraine attacks will be treated with a triptan (Table 1) (4).
Acute healthcare service costs (i.e. emergency room visit, hospitalization and/or a physician visit) were estimated based on published data and were adjusted to £2005 using the healthcare component of the UK consumer price index (Table 2) (25, 29). The cost of triptan treatment was based on the average cost of triptan tablets from the British National Formulary (September 2005) and an assumption of 1.5 doses per attack.
Indirect costs
Total disability hours (work days and non-work days) are based on the number of hours of disability associated with each migraine. Lost productivity costs are based on the number of hours of lost work associated with each migraine attack. Only attacks occurring on work days (assumed five work days per week, 20 vacation days and eight bank holidays per year) are included in the cost calculations (30). The cost of a lost hour of work was assumed to be £11.43, based on the average hourly wage in 2005 for women working full time in Scotland (31).
Analyses
We calculated the incremental differences (i.e. TPM treatment vs. no preventive migraine therapy) for all model outcomes. Primary model outcomes focus on direct medical costs and include the expected number of migraines averted, cost of preventive and acute treatment, and QALYs. We then calculated incremental cost-effectiveness ratios to determine the incremental direct medical cost per migraine averted and incremental cost per QALY gained for TPM treatment vs. no preventive migraine treatment. We performed sensitivity analyses to assess the robustness of model estimates with respect to changes in key model parameters. The impact of including indirect costs and disability hours was addressed in a separate ‘total cost’ evaluation. The model was simulated using TreeAge DATATM 4.0 (TreeAge Software, Inc., Williamstown, MA, USA).
Results
Direct medical cost assessment
We estimated that TPM-treated patients would experience approximately 4.2 migraines per month compared with six migraines per month in the no preventive treatment cohort (Table 3). The expected monthly cost of preventive therapy, including treatment failures and monitoring visits, was £24.19 for TPM-treated patients (Table 3). Due to the reduction in migraine frequency, the cost of acute treatment for TPM-treated patients (£12.94) was about £6 less than the cost estimate for no preventive treatment patients (£18.80). In total, the savings in acute medical services brought about by treatment with TPM would offset approximately 24% of the £24.19 monthly cost of drug therapy.
Base-case model results: effectiveness of prevention, expected monthly cost, and cost per migraine averted

Cost of TPM treatment (based on two 50-mg tablets per day) and 1.5 monitoring visits/year (0.125 visits per month). Treatment drop-outs accrue 1 month of drug costs.
Cost of acute medical services (i.e., acute medications, physician, emergency room and in-patient hospitalizations).
The total monthly medical cost for TPM patients was £18.33 more than that of the no preventive treatment group, yielding an incremental direct cost per migraine averted of £10.13 (Table 3). The total annual incremental direct cost of TPM treatment vs. no preventive treatment was estimated to be about £220. The estimated number of QALYs gained was 0.0384 (the equivalent of approximately 14 days of perfect health over 1 year) (Table 4), resulting in a cost-effectiveness ratio of £5728 per QALY (Table 4).
Incremental cost per QALY: direct medical and total cost analysis

Includes only medical cost offsets.
Includes medical cost and lost wage offsets.
Approximated based on SF-36 scores.
Treatment averts migraines and is cost saving.
Sensitivity analyses
The results of the sensitivity analyses are presented in Table 5. The cost per migraine averted is sensitive to the assumed baseline migraine frequency. A baseline rate of three migraines per month results in a cost per migraine averted of £23.36, compared with £3.44 for a baseline of 12 migraines per month. The model is also sensitive to the gain in health utilities. Decreasing the gain in utility 60% (from 0.0384 to 0.015) increases the direct cost per QALY to £14 320, whereas increasing the utility gained 60% (to 0.061) decreases the cost per QALY to £3580 (Table 5).
Sensitivity analyses

QALY, Quality-adjusted life year; Dominant, treatment averts migraines and is cost saving.
Includes only medical cost offsets.
Includes medical cost and lost wage offsets.
Base-case.
Total cost assessment
Incorporating the cost of lost wages substantially impacts the overall findings of the model. When lost wages are included in the model, the total cost of TPM treatment is estimated to be £17.91 lower than having no preventive therapy. These results suggest that from a societal perspective TPM is a dominant strategy (i.e. results in fewer migraines at a lower cost) compared with no preventive treatment (Tables 4 and 5).
Discussion
In this study, we adapted to the UK setting a previously published decision-analytic model of the use of TPM as preventive treatment for patients experiencing frequent migraine headaches. Our findings suggest that TPM treatment is associated with an average of 1.8 fewer migraines per month (assuming a baseline rate of six migraines per month). These savings translate into a reduction in acute treatment costs which yields a base-case incremental direct cost per migraine averted of £10.13 and a cost per QALY of about £5700. This cost-effectiveness ratio compares favourably with that of other generally accepted medical interventions and is below thresholds commonly used by agencies such as the UK National Institute for Clinical Excellence (NICE) (32). The findings were sensitive to the assumed baseline migraine frequency and gain in health utility from migraine prevention, although the cost per QALY remained below UK thresholds across all of the sensitivity analyses performed. Further, incorporating savings in lost wages results in TPM being a dominant strategy compared with no preventive treatment.
The three pivotal trials of TPM in migraine prevention reported reductions in migraine frequency of approximately 2.1 migraines per month from a baseline of about 5.6 and a median reduction of 48% for the intention-to-treat population (17–19, 21). The estimated reduction of 1.8 migraines per month generated by the model equates to a reduction in migraine frequency similar to the trials. The difference in outcomes is due to the conservative assumptions used in the model, most importantly, that those who discontinued treatment did so in the first month and generated no clinical benefit. The clinical trial data found that TPM led to a substantial reduction in monthly migraine frequency during the first month of treatment and that the effect increased over the course of the trials and continued during the open-label extension phase of the trials (23).
Although we do not know of any other migraine cost-effectiveness models in the UK, it is interesting to compare these results with our USA-based study. As expected, the reduction in migraine frequency in the UK model was similar to the US model findings because the relevant clinical inputs were similar (the current model uses pooled data from all three pivotal trials whereas the US model used pooled data from the two USA-based trials). In addition, even with substantially different unit cost estimates, both models found that savings in direct costs offset approximately one-quarter of the cost of TPM treatment. In the US model, savings in direct and indirect costs offset about two-thirds of the cost of TPM therapy, but these savings completely offset the cost of TPM in the UK model.
Even though many healthcare payers (e.g. the NHS) are especially interested in the direct costs of treatment, the significant impact that migraine has on work productivity makes our results of particular interest to employers and occupational health professionals seeking to determine which interventions are most likely to result in improved performance for their organizations. Patients with migraine are much more likely to miss work and be less productive than the general population, with the average patient losing the equivalent of eight to 12 work days each year due to absence or lost productivity (5, 33). Results from our study indicate that preventive treatment with TPM saves approximately 3.2 work hours per month and about £36 in lost wages. Over the course of 1 year, these savings amount to approximately five work days (assuming an average 8-h work day) and over £400 in lost wages.
Our study has several limitations, including using assumptions for some parameter estimates when data were lacking, our method for estimating QALYs, and the use of the human capital approach to value lost work time. Appropriate data were not available for all parameters in the model, necessitating assumptions for some parameters and inclusion of estimates based on publications that were not specific to the UK setting. Most notably, the rate of resource use per migraine attack was based on Canadian clinical trial data and the rate of topiramate-specific physician visits and triptan use per migraine attack were assumed.
In the field of health economics, quality-of-life information is increasingly viewed as an important factor in healthcare decision making, and transforming quality-of-life measures into QALYs has become standard practice in pharmacoeconomic modelling. It is well established that migraine is associated with significant disability, but a review of published literature reveals that its toll on patients' quality of life as measured by health utilities has not been examined extensively (11, 34, 35). The current version of our model accounts for the improvement in functioning and well-being that patients derive from migraine prevention through use of utilities calculated from SF-36 Health Survey data. The SF-36 was designed as a psychometric tool and not, originally, as a utility instrument for economic evaluation. Although utility weights have been created for the instrument using respondents to a standard gamble survey in the UK (36) – a strong methodological approach – use of these derived utilities in our model is less than ideal; directly measured utilities would be preferred. However, data from a migraine-specific health utility study illustrate that migraine frequency is inversely associated with health utility values (37, 38). In addition, there is also recent evidence that TPM treatment improves quality of life based on the Migraine-Specific Questionnaire (39).
Another limitation of the study can be found in our use of the human capital approach to quantify patients' disability time in economic terms. The human capital approach is based on the economic theory that the time patients lose because of a medical condition or its treatment can be assigned a value corresponding to patients' hourly wage rates, although, in practice, the opportunity cost of lost productivity may be less than gross earnings (40). On the other hand, the overestimation may be offset by the fact that we conservatively estimated lost work time per attack at 2–3 h and did not include costs associated with patients' travel time for acute healthcare visits or the costs of care provided by friends and family members, even though such healthcare resources are often included in cost-effectiveness analyses (40). Also, we assumed that the value of leisure time gained through migraine prevention has no monetary value.
The emergence of new preventive migraine treatments has prompted interest in assessing not only the best choices for patients' needs, but consideration of the cost effectiveness of the new drugs. Payers are increasingly demanding detailed clinical and economic data regarding the impact of new therapies. In addition, employers are increasingly interested in quantifying the economic impact of migraine drugs on their employees' productivity. Results of our study suggest that use of TPM vs. no preventive treatment may be a cost-effective way to prevent occurrence of migraine headache in the UK.
Footnotes
Acknowledgements
We thank Steve Migausky for his help in adapting, testing and reviewing the model and revising the manuscript. The authors also express their appreciation to Mason Russell for his thoughtful comments. Financial support for this study was provided by Johnson & Johnson (J&J) Pharmaceutical Services, LLC, Raritan, NJ, USA and Janssen-Cilag Ltd, High Wycombe, UK.
