Abstract
Aim
Corneal confocal microscopy (CCM) is a non-invasive ophthalmic imaging technique for evaluating the corneal microstructure, particularly the subepithelial nerve plexus. This dense, dynamic plexus contains small nerve fibers from the ophthalmic branch of the trigeminal nerve. This study aimed to evaluate corneal nerve alterations in individuals with migraine, including its subtypes and visual hypersensitivity (measured with the Leiden visual sensitivity scale (L-VISS), a validated nine-item questionnaire).
Methods
In total, 55 migraine participants were included, including 26 with episodic migraine and 29 with chronic migraine, as well as 55 age- and sex-matched controls. All subjects underwent CCM, and automated corneal nerve fiber density (ACNFD), length (ACNFL) and branch density (ACNBD) were assessed using the Rostock Cornea Module (Heidelberg Retina Tomograph III). Data were compared with age- and sex-matched healthy volunteers.
Results
Reduced ACNFD, ACNFL and ACNBD were found in migraine participants compared to controls (all p < 0.001). No difference was found between episodic migraine and chronic migraine, and regression analysis showed no significant effect for (inter)ictal visual hypersensitivity.
Conclusions
These findings support the utility of CCM as a sensitive tool for detecting nerve fiber pathology in migraine and highlight its potential in further understanding migraine pathophysiology. The observed nerve changes, present irrespective of migraine frequency status or visual hypersensitivity, suggest a broader role for peripheral nerve dysfunction in migraine beyond sensory hypersensitivity symptoms.
This is a visual representation of the abstract.
Keywords
Introduction
Corneal confocal microscopy (CCM) is a non-invasive imaging and diagnostic technique used to investigate the corneal microstructure, allowing high-resolution evaluation and quantitative analysis of the ocular surface, particularly to assess the corneal nerve plexus. This subepithelial, densely innervated and highly dynamic plexus comprises small nerve fibers that are the distal ends of the ophthalmic branch of the trigeminal nerve. Abnormalities in corneal nerve parameters have been observed in patients with peripheral neuropathies (1–3). These nerve densities can also change in response to treatment (4,5). Given the trigeminovascular system's central role in migraine, quantifying this plexus may offer insights into migraine-related nerve dysfunction. The pathogenesis of migraine has a strong genetic component and involves activation of the trigeminovascular pain pathway (6,7). While the majority of patients have episodic migraine (EM), approximately 3% of them convert annually from EM to chronic migraine (CM), characterized by experiencing at least 15 headache days per month, of which at least eight are migraine days, a transition often referred to as migraine chronification (1,6).
Until now, only five studies, some of them with small sample sizes, have reported original CCM data from migraine patients (8–12). All studies included the output measurements corneal nerve fiber density (CNFD), corneal nerve fiber length (CNFL) and corneal nerve branch density (CNBD). Three studies also reported total branch density (CTBD), total nerve fiber area (CNFA) and corneal nerve fiber width (CNFW). Importantly, these CCM studies showed conflicting results, likely due to small sample size and/or limited phenotyping of migraine. Therefore, larger and more detailed studies are needed, taking into account migraine frequency (EM versus CM) and (inter)ictal visual sensory hypersensitivity. The latter can be assessed using the Leiden visual sensitivity scale (L-VISS), a validated nine-item questionnaire, because L-VISS scores differ not only between migraine patients versus controls, but also among migraine subtypes (13).
The aim of our study was to compare the corneal subepithelial nerve plexus with CCM in individuals with migraine versus healthy controls, including its subtypes (EM versus CM) and (inter)ictal visual hypersensitivity measured with validated e-questionnaires and e-headache diaries.
Methods
Ethical approval
The study protocols were approved by the local and national Ethics Committee (Leiden University Medical Center, Leiden, the Netherlands and Medical Ethics Committee Leiden-Den Haag-Delft (METC-LDD), The Netherlands). All study procedures were conducted according to GCP guidelines and adhered to the tenets of the Declaration of Helsinki. The patients provided their verbal and written informed consent before the start of the study.
Population
Patients with migraine at the Leiden Headache Center, LUMC, in the Netherlands, were included. Patients were diagnosed with migraine according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) by one of the investigators and headache specialist GMT (7). Other inclusion criteria were: (i) Aage between 18 and 75 years and (ii) able to provide written informed consent. Potential subjects who met any of the following criteria were excluded from participation in this study: (i) neurological conditions, such as peripheral neuropathy or epilepsy, other than the specific types described in the group specific inclusion criteria; (ii) any (chronic) pain condition of moderate to severe intensity, or requiring pain medication, other than the headache types described in the group specific inclusion criteria; (iii) psychiatric disease, other than mild to moderate depression and anxiety, which in the opinion of the investigators may interfere with the study; (iv) other medical disorders such as pulmonary, renal, liver, cardiac, gastrointestinal, vascular disease, eye disorders or auto-immune disease (including DM type 1), which in the opinion of the investigators may interfere with this study; (v) regular use of non-triptan or non-analgesic acute anti-headache medication (e.g. ergots, high-dose opioids, barbiturates) or high-dose benzodiazepines; (vi) change in use of tricyclic antidepressants, serotonin–norepinephrine reuptake inhibitors or calcium channel inhibitors in the past three months; (vii) current or history of abuse of alcohol, soft/hard drugs which in the opinion of the investigators may interfere with the study; and (ix) pregnancy or lactation.
Validated e-headache questionnaires and e-headache diary
All patients were asked to fill out a screening and additional headache e-questionnaire consisting of questions about headache-specific characteristics: age of onset of migraine and migraine-specific characteristics (14,15).
To assess the clinical treatment response, all patients completed a validated daily e-headache diary (15,16). This diary contained questions on headache presence, headache characteristics, accompanying symptoms, and the use of acute headache-specific and non-specific pain medication. When a headache was present, an automated and validated algorithm following the ICHD-3 criteria determined whether it was a migraine day. Additionally, days on which a triptan was taken and a reported occurrence of an aura were also counted as migraine days (15,16). One month is defined as 28 days (four weeks).
L-VISS
The L-VISS is a questionnaire developed to quantify self-reported visual sensitivity to light and patterns and was validated in patients with migraine (13). It contains nine items, all answered on a five-point Likert scale (0–4, total range 0–36). Patients completed the questionnaire both regarding (hyper)sensitivity outside migraine attacks (L-VISS: interictal), and regarding (hyper)sensitivity during migraine attacks (L-VISS: ictal).
CCM
Automated cornea nerve fiber density (ACNFD), automated cornea nerve fiber length (ACNFL) and automated cornea nerve branch density (ACNBD) were determined using the Rostock Cornea Module with the Heidelberg Retina Tomograph III (Heidelberg, Germany) (17). Briefly, after topical anesthesia of the eyes, the microscope was placed at the surface of the cornea apex, as determined by the characteristic orientation of the nerve fibers in a superior–inferior direction. Confocal images were acquired with a field of view of 400 µm × 400 µm and automatically quantified using the ACCMetrics software (MA. Dabbah, Imaging Science, University of Manchester, Manchester, UK) (18–20).
Three to eight representative, high-quality images were selected from a total of at least 50 images per eye, according to the recommendations (3,21). ACNFD, ACNFL and ACNBD were quantified by one of the investigators MvV who was blinded for subject demographics and disease state. Data from the present study were compared with automatically quantified reference values of age- and sex-matched healthy volunteers provided by one of the investigators (MT). Healthy control subjects were eligible for inclusion if they were aged 17–75 years, had no neurological signs or symptoms including a clinical diagnosis of migraine, and no medical conditions known to cause neuropathy, including diabetes mellitus, impaired glucose tolerance, metabolic syndrome, vitamin B12 deficiency or other known causes of peripheral neuropathy. Individuals with a history of corneal dystrophies, rigid gas permeable (hard contact lens) use or previous refractive surgery were excluded. All control subjects underwent a comprehensive medical, neurological and ophthalmic examination to confirm eligibility. This study was conducted in accordance with the tenets of the Declaration of Helsinki, and ethical approval was obtained from the local research ethics committee. Written informed consent was obtained from each participant prior to enrolment.
Statistical analysis
CM was defined as at least 15 headache days per month with at least eight migraine days per month according to the ICHD-3 criteria (7) Baseline characteristics were summarized using means and standard deviations or frequencies and proportions. Baseline scores of the questionnaires (L-VISS: interictal and L-VISS: ictal) were summarized as median and interquartile range. Descriptives are reported as the mean ± SD or numbers with proportions, and differences between groups were tested with independent sample t-tests and chi-squared tests.
Linear regression models were utilized to compare migraine patients and the control group. Comparisons between the EM and CM groups and controls were evaluated using analysis of variance (Anova), as well as Tukey’s honestly significant difference post-hoc tests for multiple comparisons, to test for differences between the groups, including sex and age as co-variates in the model. The visual hypersensitivity scores (L-VISS: interictal and L-VISS ictal) were compared between EM and CM patients in linear regression models including sex and age as co-variates. Additionally, a subgroup analysis of migraine without aura (MO) versus migraine with aura (MA) was performed. p < 0.05 was considered statistically significant. Statistical analyses were performed using R, version 4.0.5 (R Foundation, Vienna, Austria).
Results
In total, 55 migraine participants were included, comprising 26 with EM and 29 with CM, as presented in Table 1. The mean age was higher in the EM group compared to the CM group (44.0 ± 10.9 vs. 37.3 ± 13.4 years, p < 0.05). The prevalence of migraine with aura did not significantly differ between groups, as reported in 38.5% of EM and 58.6% of CM patients. As expected, clinical headache burden was greater in the CM group, as assessed with the validated daily e-headache diary, including monthly migraine days (13.9 ± 4.3 vs. 3.7 ± 1.9), monthly headache days (17.8 ± 4.2 vs. 5.2 ± 2.8) and monthly acute medication days (12.7 ± 3.8 vs. 4.1 ± 1.9), all with p < 0.001. CM patients also reported higher intake of simple analgesics and triptans (both p < 0.01). Medication overuse was present in 89.7% of CM patients but none in the EM group (p < 0.001). Interestingly, a higher proportion of EM patients had a history of prophylactic treatment (61.5% vs. 24.1%, p < 0.01). No significant differences were observed in overall L-VISS scores, nor in the interictal and ictal subcomponents, between groups.
Baseline characteristics of the included subjects

EM = episodic migraine; CM = chronic Migraine; MMD = monthly migraine days; MHD = monthly headache days; MAMD = monthly acute medication days; L-VISS = Leiden Visual Sensitivity Scale (range 0–36); IQR = interquartile range; NS = non-significant. Medication overuse was defined as at least 15 days of simple analgesics/non-steroidal anti-inflammatory drugs (NSAIDs) or at least 10 days of triptans or combination of analgesics/NSAIDS/triptans.
A representative example of CCM images from a migraine patient is shown in Figure 1.

(A) Representative corneal confocal microscopy images from the subbasal plexus, illustrative case of migraine patient. (B) Fully automated corneal nerve quantification. Green indicates the nodes. Blue and red represent identified fibers, with red indicating the main branches and blue the side branches
Since CCM data of the left and right eye were similar (data not shown), these data were averaged per patient. Median ACNFD, ACNFL and ACNBD values are given in Table 2 and are illustrated in Figure 2. CCM revealed significantly reduced corneal nerve parameters in migraine patients compared to healthy controls. Median ACNFD was lower in migraine patients (22.3 ± 7.1 number/mm2) compared to controls (33.3 ± 8.3 number/mm2; p < 0.001). Similarly, ACNFL was reduced in the migraine group (13.7 ± 4.0 vs. 18.9 ± 3.2 mm/mm2; p < 0.001), as was ACNBD (27.5 ± 18.5 vs. 44.9 ± 23.1 number/mm2; p < 0.001) (see supplementary material, Table S1). When stratified by migraine subtype, both EM and CM groups showed significant reductions in ACNFD, ACNFL and ACNBD compared to controls (all p < 0.001). No significant differences were found between EM and CM for ACNFD (23.6 ± 6.7 vs. 20.5 ± 7.8 number/mm2; p = 0.98), ACNFL (14.5 ± 4.2 vs. 13.1 ± 4.3 mm/mm2; p = 0.65) and ACNBD (30.9 ± 16.3 vs. 25.6 ± 17.5. number/mm2; p = 0.92) (see supplementary material, Table S2). These findings suggest corneal nerve fiber loss in individuals with migraine, independent of disease chronicity. No difference was found between migraine with and without aura (see supplementary material, Table S3).

Results of corneal confocal microscopy. Controls (n = 60), Migraine patients (n = 55): EM (n = 26) and CM (n = 29). Linear regression models were used to compare migraine to controls and analysis of variance with Tukey’s honestly significant difference post-hoc tests were used to compare EM, CM and controls. Age and sex were added as covariates. *p < 0.05 was considered statistically significant. EM = episodic migraine; CM = chronic migraine; ACNFD = automated corneal nerve fiber density; ACNFL = automated corneal nerve fiber length; ACNBD = automated corneal nerve branch density. Data are the median ± interquartile range.
Results of corneal confocal microscopy
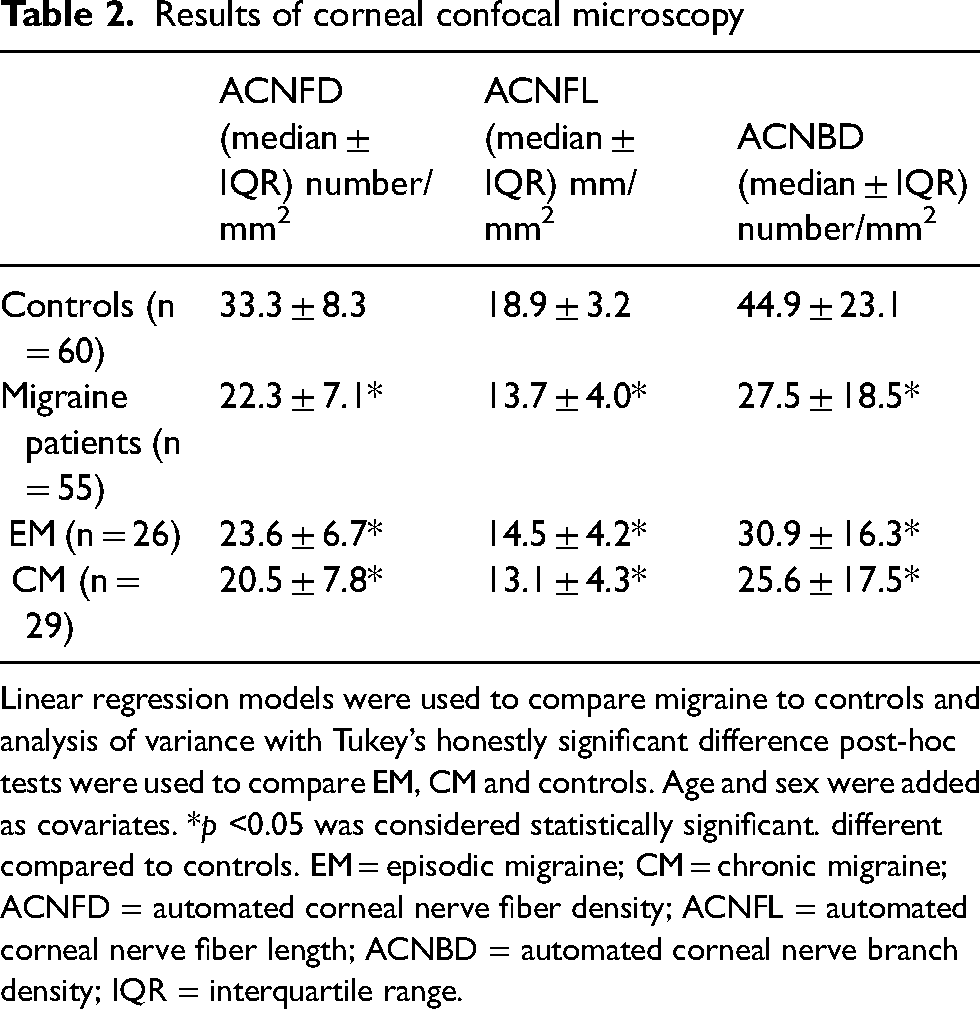
Linear regression models were used to compare migraine to controls and analysis of variance with Tukey’s honestly significant difference post-hoc tests were used to compare EM, CM and controls. Age and sex were added as covariates. *p <0.05 was considered statistically significant. different compared to controls. EM = episodic migraine; CM = chronic migraine; ACNFD = automated corneal nerve fiber density; ACNFL = automated corneal nerve fiber length; ACNBD = automated corneal nerve branch density; IQR = interquartile range.
Regarding the L-VISS interictal and ictal scores, no significant covariates were identified in the linear regression model comparing the EM and CM groups for CCM. The L-VISS interictal score showed a trend toward a positive association with decreased ACNBD (p = 0.055), suggesting that higher visual hypersensitivity between attacks is associated with reduced corneal nerve parameters. In contrast, the L-VISS ictal score demonstrated a trend toward a negative association with decreased ACNBD and ACNFL (p = 0.058 and p = 0.076, respectively), indicating that higher ictal visual hypersensitivity may be linked to less reduction in these corneal nerve parameters. Age was significantly associated with lower ACNFD and ACNFL (p = 0.015 and p = 0.041, respectively).
Discussion
In the present study, we investigated clinical and corneal nerve characteristics in individuals with EM and CM. As expected, patients with CM exhibited a greater headache burden, higher medication intake and more frequent medication overuse compared to those with EM. Despite these clinical differences, both groups demonstrated significant reductions in corneal nerve parameters, ACNFD, ACNFL and ACNBD, compared to healthy controls, indicating peripheral nerve alterations independent of migraine chronicity. While no significant differences were observed in visual hypersensitivity scores between groups, exploratory regression analyses revealed opposing trends between interictal and ictal L-VISS scores and corneal nerve loss, suggesting that the temporal context of visual sensitivity may differentially relate to small fiber integrity.
Comparison with other studies
Our findings contribute to a growing but inconsistent body of literature on corneal nerve alterations in migraine. To date, five published CCM studies have reported varying results, likely due to small sample sizes, methodological heterogeneity and limited migraine phenotyping as highlighted in a recent systematic review (22). In the present study, a reduction in ACNFD was observed in migraine patients, consistent with findings from two earlier studies (8,10). Additionally, reductions in ACNFL and ACNBD were identified, aligning with results reported in three previous investigations (9–11). These findings suggest a robust pattern of small fiber loss in migraine. Notably, another study found no significant differences in corneal nerve metrics between EM and controls but instead identified altered corneal immune cell profiles (12). Our study adds value by using a relatively large and well-phenotyped migraine cohort and automated CCM analysis with a reliable control group, strengthening the evidence for corneal nerve loss in migraine across the spectrum of disease.
Corneal nerve and trigeminal nerve in migraine
The trigeminovascular hypothesis of migraine emphasizes the central role of the trigeminal nerve and its axonal projections containing vasoactive neuropeptides, particularly calcitonin gene-related peptide (CGRP), in innervating the meninges and associated blood vessels (6). This model underscores the significance of neuropeptide release and its downstream effects following trigeminal activation. Trigeminal innervation is often considered the final common pathway for upstream headache triggers and serves as a foundational concept for developing new therapeutic strategies (23). Corneal dendritic cells and the trigeminal nerve maintain close, bidirectional communication: dendritic cells can activate nerve fibers to initiate protective reflexes, while nerve fibers modulate dendritic cell function (12,24). Given that the cornea is densely innervated by the ophthalmic branch of the trigeminal nerve, it may provide a peripheral biomarker for trigeminal nerve integrity. One may hypothesize that migraine-related disruption of trigeminal function may impair axonal maintenance, regeneration, and turnover in the cornea, leading to measurable loss of nerve fibers. Dendritic cells also express type 1 CGRP receptors, implicating them in migraine pathophysiology through their sensitivity to CGRP signaling (25). Alterations in dendritic cell activity may disrupt the neuroimmune balance necessary for preserving corneal nerve structure. These mechanisms could contribute to the reduced corneal nerve parameters observed in both episodic and chronic migraine, independent of disease duration or severity.
Visual hypersensitivity
One of the previous studies on CCM in migraine aimed to incorporate ictal photophobia based on seven questions of an eight-part photophobia questionnaire (11,26). Participants who responded “yes” to any of the seven questions were classified as having photophobia (26). We estimated that the L-VISS, a validated nine-item questionnaire, provides more insight into visual hypersensitivity because L-VISS scores generally differ not only between migraine patients and controls, but also between patients with different migraine subtypes (13). The L-VISS questionnaire employs a linear scale, enabling straightforward comparisons across various groups, in contrast to other instruments that utilize binary or qualitative scales. In the present, we found no significant differences in L-VISS scores, including both interictal and ictal subcomponents, between EM and CM. While no strong covariates were identified in the regression models, the observed trends indicate that higher visual hypersensitivity, particularly during interictal periods, may be associated with more reduction in corneal nerve parameters, although the ictal hypersensitivity pointed to an opposite direction of association. These preliminary trends point toward a complex, context-dependent relationship between corneal nerve integrity and visual sensitivity, consistent with the broader interplay between migraine pathophysiology, trigeminal function and sensory processing.
Strengths and limitations
Apart from using a validated hypersensitivity tool, another strength of the present study is the inclusion of well-characterized migraine participants, defined using standardized criteria and confirmed through our e-headache diaries. This approach minimizes misclassification of migraine days, which is important as patients with at least eight monthly migraine days often overestimate their migraine frequency, potentially blurring the distinction between EM and CM as noted in our previous research (15). The study also benefits from a large sample size of migraine patients and direct comparisons between EM and CM, with full reporting of clinical and corneal nerve findings. However, limitations include the potential influence of medication overuse (headache) (MOH) that cannot be fully excluded, and longitudinal changes, such as nerve recovery after successful migraine and MOH treatment, remain to be explored. It is unclear whether the observed nerve fiber reductions will be reversible, as suggested by studies in other conditions such as sarcoidosis or diabetic peripheral neuropathy (4,5).
Translational implications and future directions
The present study supports the presence of corneal nerve fiber loss in both EM and CM, independent of migraine frequency, and highlights potential links between visual hypersensitivity and small fiber integrity. Although no differences with L-VISS scores were found, exploratory trends suggest that interictal and ictal hypersensitivity may reflect distinct peripheral mechanisms. Moreover, the consistent nerve fiber changes observed, regardless of migraine frequency or presence of visual hypersensitivity, suggest a broader role for peripheral nerve dysfunction in migraine pathophysiology. These findings support the utility of CCM as a sensitive, non-invasive tool for detecting nerve fiber pathology and advancing our understanding of migraine mechanisms. Given its accessibility and ability to quantify subtle nerve changes, CCM may also serve as a biomarker for peripheral nervous system involvement and a means to monitor response to therapy. Taken together, our study indicates that small fiber pathology may represent a core feature of migraine, potentially independent of sensory symptoms, and could serve as a peripheral biomarker of disease.
Future longitudinal studies should assess the reversibility of corneal nerve loss with effective treatments, particularly in patients with medication overuse or high sensory burden. In parallel, mechanistic studies exploring the interplay between corneal dendritic cells, CGRP signaling, and small fiber integrity may deepen our understanding of migraine as a systemic neuroimmune disorder. By linking clinical phenotypes with objective biomarkers, such work could ultimately support the development of personalized, mechanism-based treatment strategies for migraine.
Article highlights
CCM showed reduced corneal nerve fiber density, length and branching in migraine patients versus controls.
The observed nerve changes, present irrespective of frequency status (EM versus CM) or (inter)ictal visual hypersensitivity, suggest a broader role for peripheral nerve dysfunction in migraine beyond sensory hypersensitivity symptoms.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251414172 - Supplemental material for Corneal confocal microscopy reveals nerve fiber alterations in migraine irrespective of subtype or visual hypersensitivity
Supplemental material, sj-docx-1-cep-10.1177_03331024251414172 for Corneal confocal microscopy reveals nerve fiber alterations in migraine irrespective of subtype or visual hypersensitivity by Rein F. van Welie, Floor C. van Welie, Albert Dahan, Mitra Tavakoli, Monique van Velzen and Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251414172 - Supplemental material for Corneal confocal microscopy reveals nerve fiber alterations in migraine irrespective of subtype or visual hypersensitivity
Supplemental material, sj-docx-2-cep-10.1177_03331024251414172 for Corneal confocal microscopy reveals nerve fiber alterations in migraine irrespective of subtype or visual hypersensitivity by Rein F. van Welie, Floor C. van Welie, Albert Dahan, Mitra Tavakoli, Monique van Velzen and Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024251414172 - Supplemental material for Corneal confocal microscopy reveals nerve fiber alterations in migraine irrespective of subtype or visual hypersensitivity
Supplemental material, sj-docx-3-cep-10.1177_03331024251414172 for Corneal confocal microscopy reveals nerve fiber alterations in migraine irrespective of subtype or visual hypersensitivity by Rein F. van Welie, Floor C. van Welie, Albert Dahan, Mitra Tavakoli, Monique van Velzen and Gisela M. Terwindt in Cephalalgia
Footnotes
Acknowledgments
We acknowledge the University of Manchester, the owner of ACCMetrics, for providing access to this software.
Ethical considerations
The study protocols were approved by the local and national Ethics Committee (Leiden University Medical Center, Leiden, The Netherlands and Medical Ethics Committee Leiden-Den Haag-Delft (METC-LDD), The Netherlands).
Consent to participate
Patients gave their verbal and written informed consent before the start of the study.
Consent for publishing
All authors agree to publish the paper in Cephalalgia.
Author contributions
Rein F. van Welie and Floor C. van Welie were responsible for writing the original draft. They also handled methodology, data curation, investigation, formal analysis, visualization, project administration, and writing, review and editing. Albert Dahan, Monique van Velzen, Mitra Tavakoli and Gisela M. Terwindt were responsible for conceptualization, methodology, and writing, review and editing. Monique van Velzen and Mitra Tavakoli additionally worked on software, data curation, investigation, validation, supervision and resources. Gisela M. Terwindt was also involved in validation, supervision and resources.
Funding
The authors disclosed there was no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: G. M. Terwindt reports consultancy or industry support from Abbie, Lilly, Lundbeck, Novartis, Organon, Pfizer, Teva and Interactive Studios, and independent support from the Dutch Research Council, the Dutch Brain and Hearth Foundations, Dioraphte, Clayco Foundation, and the European Community. A. Dahan received funding from the Dutch Research Council (NWO, The Hague, The Netherlands) and awards/grants from the US Food and Drug Administration. Dr Dahan received consultancy fees from Trevena Inc. and Enalare Therapeutics Inc. (USA).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
