Abstract
Background/Aim
Post-traumatic headache often resembles migraine or tension-type headache, but distinct phenotype and clinical characteristics necessitate further delineation. We aimed to characterize the clinical phenotype, headache patterns, associated features and comorbidities, medication patterns and functional impact of post-traumatic headache in an adult population following mild traumatic brain injury.
Methods
This is a cross-sectional analysis of a cohort of adults with post-traumatic headache after mild traumatic brain injury, by any mechanism, evaluated by a neurologist at an outpatient specialized concussion and headache center in Ontario, Canada between February 2021 and October 2023. Data were collected through standardized pre- and during-visit questionnaires. Descriptive statistics are presented.
Results
Among 405 patients assessed by a neurologist for post-traumatic headache, median time since injury was 37 days (IQR: 13–126). Most patients reported headache 26 + days per month (292, 72.1%). Headache was continuous in 114 (28.1%), whereas in 215 (53.1%) it lasted hours to days. Headache location was unilateral in 174 (43.0%) and bilateral in 159 (39.3%). Headache quality was described as pulsating/throbbing in 260 (64.2%). The median severity was 7/10 (IQR 5–8). Aggravation by routine physical activity was reported in 287 (70.9%), nausea/vomiting in 279 (69.0%), photophobia in 358 (88.4%) and phonophobia in 337 (83.2%). There was no positional preference for 147 patients (36.3%), while 216 (53.3%) preferred lying down/reclined. Acute medication use frequency was reported as 3 + days per week in 218 (53.8%) and daily in 143 (35.3%). Within this cohort, 201 (49.6%) endorsed one or more psychiatric comorbidities. Only 66 (16.3%) had returned to full work/school attendance, while 169 (41.7%) were completely off usual occupational activities post-injury. One hundred seventy-eight (44.0%) reported pending litigation or insurance claims related to their injury, and/or having a work-related injury. Among the 183 (45.2%) who had undergone neuroimaging, 160 (87.9%) studies were reportedly normal, while there were 13 (7.1%) incidental findings and eight (4.3%) injury-related.
Discussion
While select migraine features such as photophobia, phonophobia and worsening with routine physical activity are common in post-traumatic headache, there are also distinct features, including daily or near daily headache of long duration. The latter may suggest early sensitization in post-traumatic headache. There is an associated high risk of medication overuse headache, given frequent administration of acute medications, as well as high rates of psychiatric comorbidities and functional impairment. Future studies should aim to further delineate the longitudinal clinical, pathophysiological, and treatment response differences between post-traumatic headache and primary migraine.
This is a visual representation of the abstract.
Introduction
Post-traumatic headache (PTH) is the most common and disabling symptom following concussion, with a prevalence estimated at 60% at two weeks after mild traumatic brain injury (mTBI) (1,2). Persistent PTH at 12 months is experienced by nearly 30% of those with acute PTH (1). The psychosocial and economic impact of PTH is substantial (1,3,4). For instance, the median headache-related disability on the Migraine Disability Assessment Test (MIDAS) among patients with persistent PTH was found to be 48, suggesting severe disability (5).
This secondary headache disorder is described in the International Classification of Headache Disorders 3rd Edition (ICHD-3) as “headache attributed to trauma or injury of the head and/or neck” (6). It is diagnosed when a new headache occurs within seven days of injury; subclassifications include acute (if duration <3 months) or persistent (>3 months) and mild versus moderate-to-severe brain injury (6). Headache attributed to whiplash or to craniotomy are additional subcategories (6). Despite its impact, the specific features of PTH have not been well-defined, and there is a dearth of randomized controlled trials (RCTs) to guide treatment (2,7,8). In the absence of its own phenotypic description or its own evidence-based treatments, experts have generally recommended ascribing a phenotype according to the primary headache disorder that best fits, and mirroring treatment paradigms for that phenotype (9,10). In this regard, migraine-like phenotype has been reported as most common, although some studies have suggested tension-type-like is more frequent (6,9,10). This discrepancy may be due to variability in how symptoms are ascribed—whether they are attributed to the concussion spectrum, or to the headache disorder. Concussion symptoms may include headache, light and sound sensitivity, nausea/vomiting, symptoms worsening with physical activity, cognitive and emotional symptoms, autonomic or sleep dysfunction (11,12), all of which may also be reported in migraine (6,10). Moreover, inter-ictal symptoms are common in migraine: for instance, patients may experience phono- and photophobia between migraine headache attacks (13). These overlapping features underscore challenges in distinguishing post-traumatic headache from primary migraine clinically and point to a need for refined phenotypic criteria.
A deeper dive into the clinical phenotypes that may distinguish PTH from primary headache disorders –or inform sub-classification of PTH– is required. This study aimed to describe the detailed clinical characteristics of PTH in an adult cohort.
Methods
Study population
This was a cross-sectional analysis of a cohort of patients with PTH after mTBI evaluated at a specialized outpatient concussion centre in Ontario, Canada, by a neurologist. This cohort was a convenience sample of all patients assessed between February 2021 and October 2023. Patients were referred either externally – by local emergency departments or family physicians- or internally, by other concussion specialist physicians within our network of clinics. Participants were included if a neurologist assessed for a chief complaint of PTH secondary to mTBI. The diagnosis of mTBI was made based on the 2023 American Congress of Rehabilitation Medicine (ACRM) criteria (14). All other causes of post-traumatic headache, including moderate to severe TBI, were not investigated in this study. Patients were excluded if their headache had resolved prior to initial neurology consultation or if headache was not a primary reason for evaluation at the time of consultation.
Study procedure
Demographics were recorded through standardized pre-visit patient questionnaires and during the consultations. Data elements collected as part of the TRANSCENDENT program of research included time since injury, age, sex, past medical history, past psychiatric history, active insurance/legal claims, and occupational status (15). The detailed TRANSCENDENT protocol is available elsewhere (15). Headache characteristics and associated features as well as past headache, family, and medication histories were collected during the neurology assessments, through patient interview. Significant baseline headache was considered as any past history of severe or disabling headache, or chronic headache disorders. Photophobia or phonophobia were recorded as dichotomous variables, defined as patient-reported discomfort with or avoidance of light and noise. For the following characteristics, participants could multi-select: duration, quality, associated symptoms, positional preference, exacerbating and palliating factors. Imaging data was extracted from the initial neurology consultation notes. The Electronic Health Record (EHR) that formed the basis of this data collection was Telus Health Collaborative Health Record (CHR). The Children's Hospital of Eastern Ontario Research Board approved the secondary use of data for this study (#24/99X). STROBE guidelines (16) were followed.
Statistical methods
Categorical data were summarized as frequency counts and percentages, while continuous data were presented as median and interquartile range (IQR). Missing data were assessed prior to analysis and variables with greater than 50% missingness were excluded from the analysis (17). All statistical analyses were performed using R version 4.1, and conducted by biostatisticians (KC, RJW). The inter-rater reliability between the trained research assistant who initially extracted the data (KJ) and the post-doctoral researcher (FD) who independently performed a second extraction was assessed using Cohen's kappa statistics. Ten percent of the charts were randomly selected for this evaluation. Kappa values were interpreted as follows: no agreement (≤0), slight (0.01–0.20), fair (0.21–0.40), moderate (0.41–0.60), substantial (0.61–0.80), and almost perfect agreement (0.81–1.00) (18).
Anonymized data not published within this article can be made available on request from any qualified investigator.
Results
Demographics
There were 457 charts extracted, of which 52 were excluded due to resolved or non-significant headache. Among the 405 patients included in the analysis, the median age was 38 (IQR: 27–50) and 73.6% identified as female. The median time since injury was 37 days (IQR: 13–126 days). Of the 405 patients, 68% were evaluated within three months of their index injury, while 21% were seen between three months and one year and 10% were evaluated more than one-year post-injury (Table 1). The mechanism of injury was motor vehicle collision in 127 (31.4%), fall in 110 (27.1%), playing sports in 58 (14.3%), workplace injury in 31 (7.7%), assault in 10 (2.5%), and other in 69 (17.0%).
Characteristics of study population.

Definition of medical history; psychiatric conditions (depression, anxiety, bipolar disorder, post-traumatic stress disorder, or other); baseline pain disorder (fibromyalgia, chronic pain, somatoform disorder, or other).
Of those with headache history.
Eighty-five (21.0%) reported having a family history of migraine. A personal history of significant baseline pre-injury headache was endorsed in 150 (37.1%), with migraine being most common (138, 34.1%), followed by tension-type headache (12, 3.0%). One hundred and ninety (46.9%) reported no significant baseline headache disorders.
Concerning past medical history, psychiatric conditions (depression, anxiety, bipolar disorder, post-traumatic stress disorder, or other psychiatric diseases) were most common, with 201 (49.6%) endorsing one or more. Eighty-four (20.7%) reported persistent symptoms after a previous concussion, which had not resolved before the index injury. Thirty-six (8.9%) reported a pre-existing sleep disorder (insomnia, obstructive sleep apnea, or other). Twenty-six (6.4%) reported a baseline pain disorder (fibromyalgia, chronic pain, somatoform pain disorder, or other). Irritable bowel syndrome was comorbid in 17 (4.2%).
Only 66 (16.3%) of patients had returned to full attendance at work or school since the injury, and 169 (41.7%) were completely off their usual occupational activities since the injury. One hundred seventy-eight (44.0%) reported having pending litigation or insurance claims related to their injury, and/or having a work-related injury.
Headache characteristics and associated features
Post-injury headache frequency was most commonly in the 26 or more days per month category (292, 72.1%) (Table 2). Headache was continuous in 114 (28.1%), while the duration was hours to days in 215 (53.1%). The location of the headache was described as unilateral in 174 (43.0%), bilateral or holocephalic in 159 (39.3%), and unilateral or bilateral in 58 (14.3%). Headache quality was endorsed as pulsating/throbbing in 260 (64.2%), specifically not pulsating/throbbing in 70 (16%), steady in 66 (16.3%), pressure in 132 (32.6%), and other in 19 (4.7%). Median headache severity was 7.0/10.0 (IQR: 5.0–8.0). Worsening with routine physical activity was endorsed by 287 (70.9%), nausea/vomiting by 279 (68.9%), photophobia by 358 (88.4%), and phonophobia by 337 (83.2%). Criteria B-D for a diagnosis of migraine without aura in the ICHD-3 were met by 272 (67.2%).
Headache characteristics of study population.
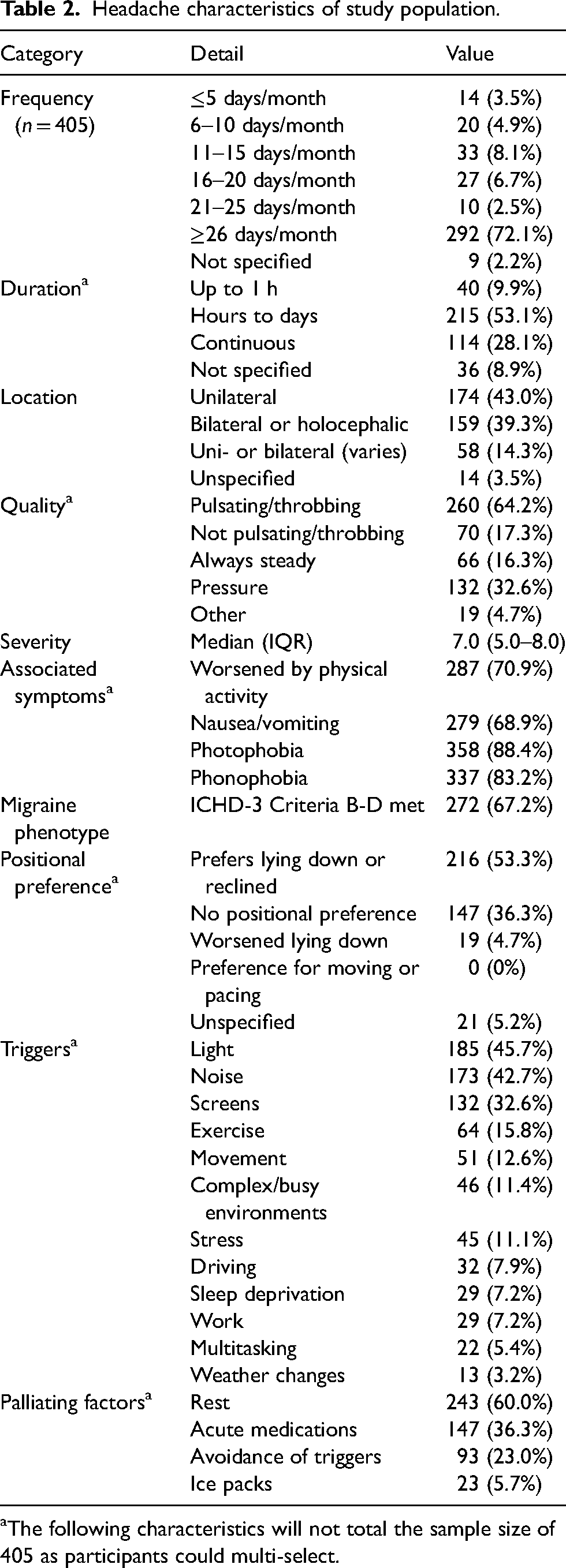
The following characteristics will not total the sample size of 405 as participants could multi-select.
With regards to positional preference with headache, 216 (53.3%) favored lying down or reclined. None (0%) in this cohort preferred moving or pacing.
The most common exacerbating factors identified by patients were light in 185 (45.7%), noise in 173 (42.7%), screens in 132 (32.6%), and exercise in 64 (15.8%). The most common palliating factors described were rest in 243 (60.0%), acute medications in 147 (36.3%), and avoidance of triggers in 93 (23.0%).
Medications
Acute
Acute medications that had been tried prior to the initial consultation with neurology included: acetaminophen (380, 93.8%), non-steroidal anti-inflammatories (355, 87.7%), triptans (116, 28.6%), and gepants (7, 1.7%) (Online Supplementary Table S2). The frequency of acute medication use was reported as fewer than three days per week in 167 (41.2%), three or more days per week in 218 (53.8%), and daily in 143 (35.3%).
Preventive
Prior to initial consultation, 201 (49.6%) had been tried on any preventive medications, most commonly tricyclic antidepressants (99, 24.4%). In our cohort, 1.5% and 0% of patients had tried at least two and at least five preventative medications respectively.
Neuroimaging
At the time of the neurology consultation, 183 (45.2%) reported having received brain imaging post-injury, specifically 136 (33.3%) computed tomography (CT) scans and 36 (8.9%) magnetic resonance imaging (MRI). (Online Supplementary Figure S1) An additional 11 (2.7%) had undergone both CT and MRI. The imaging was indicated as normal in 160 (87.9%). Among those with imaging findings recorded, there were 13 (7.1%) deemed incidental and eight (4.3%) injury-related (Online Supplementary Figure S2). The specific findings can be accessed in Online Supplementary Table S3.
Inter-rater reliability
For the subset of 40 charts randomly selected to undergo independent data extraction by a second researcher, agreement between reviewers was strong, with a mean kappa of 0.94 and individual values ranging from 0.82 to 1.00 (Online Supplementary Table S1).
Discussion
In our large adult cohort with mTBI, the phenotype and clinical characteristics of PTH demonstrate a high prevalence of some, but not all migraine features. Specifically, most patients endorsed photophobia, phonophobia and worsening headache with routine physical activity, while unilaterality was reported in just under half of patients. Most experienced daily or near-daily headache, as opposed to infrequent attacks often seen in migraine. This highlights the importance of more specific and granular phenotyping of post-traumatic headache, rather than relying solely on the existing framework for primary headache disorders (4). Advanced neuroimaging studies have identified functional and structural differences in the brain between those with PTH and migraine, although small sample sizes limit their interpretation (19–23). Similarly, electroencephalography (EEG) studies described some differences between these groups (10). In contrast, biochemically, similar to primary migraine disorder, calcitonin gene receptor peptide (CGRP) or pituitary adenylate cyclase activating peptide (PACAP)-38 infusion has been found to exacerbate or trigger migraine-like headache in patients with PTH who predominantly had a migraine-like phenotype and no pre-injury history of headache disorder besides infrequent tension-type headache (24,25). Pre-existing migraine disorder is a risk factor for post-traumatic migraine, and migraine phenotype headache after concussion is a risk factor for the development of persistent PTH (10,26). Collectively, the evidence suggests that while PTH and primary migraine share some overlapping mechanisms and features, PTH is also a distinct entity warranting independent study beyond the migraine paradigm.
In contrast to previous studies, most patients in our cohort were seen within the first few months post-injury. Still, as mentioned above, the frequency of headache was high: daily or nearly daily in most patients. Duration of headache was long, typically either being continuous or lasting hours to days. Similarly, an earlier Danish cohort of 100 patients with PTH with an average disease duration of four years showed a high frequency of headache (mean of 25 ± 7 days per month) (8). Our findings may indicate early sensitization in PTH. Previous research in rodents has demonstrated selective hypernociceptive processing from deep cranial but not extracranial tissues, as well as increased periosteal mast cells, 48 h after mild closed head injury (27). An animal model of allodynia has also suggested orofacial sensitization in rodents after traumatic brain injury (28). Early sensitization may call for a more liberal and early preventive treatment strategy in patients at risk for the development of persistent PTH (2). Agents with demonstrated efficacy in the treatment of chronic migraine or sensitization may be the best candidates for RCTs in PTH, and future studies should consider initiating treatment early (2). As noted, to date there are limited high quality studies to inform preventive pharmacological treatment of PTH (2,7,29). There is a small randomized study (N = 40) investigating botulinum toxin injections for PTH in veterans (30), and another pilot study evaluating prazosin for PTH, also in veterans (31). Otherwise, available PTH-specific data is largely observational (7,29). The convention of treating PTH according to phenotype, while reasonable as an interim approach, remains insufficient: There is a critical need for PTH-specific, evidence-based treatments (2,7).
The daily or near daily headache in our cohort was paralleled by frequent acute medication use, with fewer than half of patients using them less than three days per week, potentially exposing many to a risk of medication overuse headache, and further supporting a need for early and effective preventive treatment. These findings also underscore the importance of counselling both patients and clinicians on strategies to mitigate medication overuse in this population. Future RCTs investigating the preventive treatment of PTH may consider prioritizing pharmacotherapies that also have evidence in the treatment of medication overuse headache. For acute treatment of post-traumatic headache in the emergency department, one RCT suggested benefit from intravenous metoclopramide 20 mg plus diphenhydramine 25 mg compared with placebo (32). Otherwise, as with prevention, there is a lack of high quality RCTs to guide acute treatment. Gepants do not appear to be associated with medication overuse headache and may be worth exploring in this population (33,34). Of note, the infrequent use of gepants in the present cohort must be interpreted in the context of this drug class being new to the market in Canada in the latter part of this study period (35).
In our cohort, just over half of patients preferred lying down or being in a reclined position. With daily or near daily headache after mTBI, and a preference for lying down, cerebrospinal fluid (CSF) leak may be entertained by the clinician as a potential cause. Indeed, there are many overlapping features with concussion, ranging from vestibulocochlear symptoms and posterior neck pain to cognitive symptoms or dysautonomia (36). CSF leak is important to recognize due to available targeted treatment, and orthostatic headache is a hallmark (36–39). However, a study of nearly 100 patients with persistent PTH did not find any cases in which a modified Bern score suggested a high risk for CSF leak (40). A positional preference is nonspecific and may instead reflect kinesiophobia (as seen after concussion or migraine), dysautonomia, or relief of cervical musculoskeletal tension.
Patients in our cohort were considerably impaired, with almost half completely off their baseline occupational activities and few continuing with full attendance. With regards to baseline vulnerabilities, there were high rates of psychiatric comorbidities in this cohort, with half reporting one or more diagnoses. Nearly half of patients also had ongoing litigation/insurance claims. More work is needed to elucidate the potentially multidirectional relationships among baseline psychosocial vulnerabilities, legal or insurance claim status, and headache related as well as functional outcome.
Strengths
Strengths of this study include that it is a large cohort evaluated relatively early after injury, with detailed phenotyping. It focuses on an adult population with a broad range of injuries, improving generalizability. Additionally, assessments were conducted by a neurologist, likely increasing diagnostic precision.
Limitations
Our study carries some limitations. First, the data are from a single center, which may limit generalizability – particularly to patients who are not referred to neurology, as our cohort likely reflects individuals with more severe disease. Second, there is a necessary reliance on self-reported, subjective symptoms, which are susceptible to recall bias and other reporting inaccuracies. Third, the present analysis is cross-sectional; although patients were seen at different time points and typically early post-injury, a longitudinal analysis demonstrating the evolution of these clinical characteristics over time would add value to these data. Fourth, causal inferences cannot be drawn due to the observational design. Fifth, as a retrospective cohort study, the findings are subject to inherent limitations such as incomplete or missing data; for example, the temporal pattern of light and sound sensitivity (continuous or attack-related) was not specified. Additional potentially relevant factors, including socio-economic status, other associated symptoms and medication adherence and impact, were outside the scope of this analysis but will be considered in the prospective TRANSCENDENT cohort.
Conclusions
While select migraine features such as photophobia, phonophobia and worsening with routine physical activity are common in PTH, there are also distinct features, including daily or near daily headache of long duration. The latter may suggest early sensitization in PTH. There is an associated high risk of medication overuse headache, given frequent administration of acute medications, as well as high rates of psychiatric comorbidities and of functional impairment. Future studies should aim to further delineate the longitudinal clinical, pathophysiological, and treatment response differences between post-traumatic headache and primary migraine.
Clinical implications
While select migraine features are common in PTH, there are distinct features, including a tendency for daily or near daily headache of long duration.
There is a high risk of medication overuse headache in PTH. Early and effective preventive strategies are required, including patient and clinician education.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251404912 - Supplemental material for Post-traumatic headache phenotypes and clinical characteristics
Supplemental material, sj-docx-1-cep-10.1177_03331024251404912 for Post-traumatic headache phenotypes and clinical characteristics by Achelle Cortel-LeBlanc, Miguel Cortel-LeBlanc, Richard J. Webster, Kitty Chen, Henrik Winther Schytz, Kaelan Jolliffe, Andrew B. Dodd, Ivan Terekhov, Farzaneh Dashti, Roger Zemek and in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251404912 - Supplemental material for Post-traumatic headache phenotypes and clinical characteristics
Supplemental material, sj-docx-2-cep-10.1177_03331024251404912 for Post-traumatic headache phenotypes and clinical characteristics by Achelle Cortel-LeBlanc, Miguel Cortel-LeBlanc, Richard J. Webster, Kitty Chen, Henrik Winther Schytz, Kaelan Jolliffe, Andrew B. Dodd, Ivan Terekhov, Farzaneh Dashti, Roger Zemek and in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024251404912 - Supplemental material for Post-traumatic headache phenotypes and clinical characteristics
Supplemental material, sj-docx-3-cep-10.1177_03331024251404912 for Post-traumatic headache phenotypes and clinical characteristics by Achelle Cortel-LeBlanc, Miguel Cortel-LeBlanc, Richard J. Webster, Kitty Chen, Henrik Winther Schytz, Kaelan Jolliffe, Andrew B. Dodd, Ivan Terekhov, Farzaneh Dashti, Roger Zemek and in Cephalalgia
Supplemental Material
sj-png-4-cep-10.1177_03331024251404912 - Supplemental material for Post-traumatic headache phenotypes and clinical characteristics
Supplemental material, sj-png-4-cep-10.1177_03331024251404912 for Post-traumatic headache phenotypes and clinical characteristics by Achelle Cortel-LeBlanc, Miguel Cortel-LeBlanc, Richard J. Webster, Kitty Chen, Henrik Winther Schytz, Kaelan Jolliffe, Andrew B. Dodd, Ivan Terekhov, Farzaneh Dashti, Roger Zemek and in Cephalalgia
Supplemental Material
sj-png-5-cep-10.1177_03331024251404912 - Supplemental material for Post-traumatic headache phenotypes and clinical characteristics
Supplemental material, sj-png-5-cep-10.1177_03331024251404912 for Post-traumatic headache phenotypes and clinical characteristics by Achelle Cortel-LeBlanc, Miguel Cortel-LeBlanc, Richard J. Webster, Kitty Chen, Henrik Winther Schytz, Kaelan Jolliffe, Andrew B. Dodd, Ivan Terekhov, Farzaneh Dashti, Roger Zemek and in Cephalalgia
Footnotes
Acknowledgments
Thank you to the clinical and research staff at 360 Concussion Care where the TRANSCENDENT research program is integrated into the learning health system. A special thank you to the patients who participated in the study; their contribution made this research possible.
Author contributions
ACL: conceptualization, study design, interpretation of results, writing original draft
MCL: conceptualization, interpretation of results, review and editing
RJW: methodology, analysis, review and editing
KC: methodology, analysis, review and editing
HWS: interpretation of results, review and editing
KJ: data acquisition, interpretation and curation
ABD: database engineering and data extraction, technical infrastructure, review and editing
IT: database engineering and data extraction, technical infrastructure, review and editing
FD: data acquisition, interpretation and curation
RZ: acquisition of funding, supervision, interpretation of results, review and editing
Consent to participate
All included participants provided written consent to 360 Concussion Care's secondary use of data policy.
Data availability statement
Anonymized data not published within this article can be made available on request from any qualified investigator.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.C.L. has received personal fees from Abbvie, Lundbeck, Organon, Pfizer, and Teva for presentations and consultations, and is involved in research funded by Abbvie, the University of Ottawa Brain and Mind Research Institute (uOBMRI), and the Ontario Brain Institute (OBI) – research funding goes directly to the institution. She is a minority shareholder and is on the leadership team at 360 Concussion Care. She has received personal fees for the provision of medicolegal opinions. M.C.L. has received personal fees from Abbvie, Lundbeck, and Pfizer, and for the provision of medicolegal opinions. R.J.W. reports no disclosures relevant to the manuscript. K.C. reports no disclosures relevant to the manuscript. H.W.S. reports receiving personal fees from AbbVie, Lundbeck, Pfizer and Teva, and non-restricted research grants from NovoNordisk Foundation. K.J. reports no disclosures relevant to the manuscript. A.B.D. reports no disclosures relevant to the manuscript. I.T. reports no disclosures relevant to the manuscript. F.D. reports no disclosures relevant to the manuscript. R.Z.’s program of research has received financial support through competitively funded research grants from Canadian Institutes of Health Research (CIHR), Ontario Neurotrauma Foundation (ONF), Physician Services Incorporated (PSI) Foundation, CHEO Foundation, uOBMRI, OBI, National Football League (NFL), Ontario Ministry of Health (MOH), Public Health Agency of Canada (PHAC), Health Canada, Parachute Canada and Ontario SPOR Support Unit (OSSU). RZ is supported by a Tier 1 Clinical Research Chair in Pediatric Concussion from University of Ottawa. All grant funding goes directly to the institution. R.Z. sits on the board of directors for North American Brain Injury Society (NABIS), which is a volunteer (unpaid) role. Finally, R.Z. is Scientific Director and a minority shareholder of 360 Concussion Care (a learning health system and network of interdisciplinary concussion clinics in Ontario); no proceeds have been transferred to R.Z.
Ethical considerations
The Children's Hospital of Eastern Ontario Research Board approved the secondary use of data for this study (#24/99X).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ontario Brain Institute,
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
