Abstract
Background
Greater occipital nerve block (GONB) has become an established treatment for migraine. Though numerous systematic reviews and randomised control trials (RCTs) are cited as supporting evidence, the quality and consistency of this data remains unclear.
Methods
An umbrella review of systematic reviews investigating GONB for migraine was conducted. Additionally, an independent systematic review and meta-analysis of relevant RCTs was performed in accordance with PRISMA guidelines. Both evaluated MEDLINE (‘PubMed’), Embase, and CENTRAL databases.
Results
Nine relevant systematic reviews were identified; all had significant limitations and/or contained methodological errors. The reviews had been cited 256 times. None were eligible for statistical analysis.
Sixteen RCTs (930 patients) and seven RCTs (401 patients) were included for qualitative and quantitative analyses respectively. Studies were heterogeneous in their methodologies. No serious adverse effects were identified. With moderate certainty, local anaesthetic (LA) GONB reduces headache severity in acute migraine attacks at 30 min (−2.08; p < 0.001). With low certainty, weekly bilateral LA GONB injections reduce headache severity (−1.33; p < 0.001) and monthly headache days (−4.46; p < 0.001) at one month for chronic migraine. Sustained benefits of GONB remain unclear. Data was insufficient to analyse the efficacy of steroid GONB, LA-steroid GONB, nor unilateral GONB for chronic migraine, and GONB — of any type — for episodic migraine.
Conclusions
There is limited RCT evidence supporting GONB for the treatment of migraine. Existing systematic reviews should be interpreted with caution. RCTs with homogeneous methodologies are required to evaluate GONB in the management of disability in migraine.
Trial Registration
PROSPERO registration ID: CRD42024595492.
This is a visual representation of the abstract.
Background
Migraine affects 14–15% of the global population (1) and is the fourth leading individual cause of patient years lived with disability (4.73%) (2). In the United Kingdom, £150 million/year is spent on migraine treatment and £4.4 billion is lost to the economy due to migraine-related work absence (3).
The majority of this societal economic burden is from individuals with chronic migraine (CM) (4); defined as headache (migraine-like or tension-type-like [sic]) on ≥15 days per month (≥8 with migrainous features) for >3 months (5). Diagnoses with a frequency of <15 days per month are termed ‘episodic migraine’ (EM) (5). The mechanisms underpinning evolution to CM remain unclear (6); however, risk factors include female sex, low socio-economic status, obesity, poor sleep quality, acute-relief medication and caffeine use, psychiatric co-morbidities, and antecedent life events (7). CM raises multiple issues which translate generically to chronic pain, these include: agreed scales of associated disability; the contextual contribution of procedures and thus the need to include a placebo; and the realistic consideration of the value of a treatment in terms of longevity of response, disease modification, and cost effectiveness.
Where oral treatments fail, procedural interventions arise. Greater occipital nerve blockade (GONB) is premised on the neuromodulation of functionally connected trigeminocervical afferents inherent to head pain. In a 2019 survey of BASH members, 80% of respondents (48% response rate; mostly consultant neurologists, general practitioners, and nurses) performed peripheral nerve blocks for headache disorders; GONB was the most common (98%) (8). A dedicated GONB session was included at the 2024 European Headache Congress in Rotterdam, Netherlands. However, as with many historical interventions, evidence promoting its use remains controversial. This paper reviews the evidence for GONB in the management of EM and CM; primary outcome measures were a reduction in headache severity and a reduction in monthly headache days.
Methods
Umbrella review
MEDLINE (‘PubMed’) and Embase databases were evaluated from their inception to 2 February 2025 for systematic reviews with meta-analyses investigating GONB for the treatment of migraine. Search terms (Online Supplemental Material 1) included: ‘greater occipital nerve block’; ‘GONB’; ‘episodic migraine’; ‘chronic migraine’; ‘nerve block’; ‘migraine’; ‘headache’; ‘review’; ‘systematic review’; and ‘meta-analysis’. Reverse citation and online searches (using Google and Microsoft Bing) were performed to identify grey literature. Duplicate records were removed and abstracts screened to exclude: non-relevant records, narrative reviews, primary studies, correspondence articles, and non-English texts. Full-text review was performed with the following inclusion criteria: English texts; systematic reviews which performed individual meta-analyses of GONB for the treatment of migraine; and adult patient cohorts. No limitations were made to publication status. Data was insufficient to allow for planned statistical analysis.
Systematic review
MEDLINE (‘PubMed’), Embase, and Cochrane's Central Register of Controlled Trials (‘CENTRAL’) databases were evaluated in accordance with PRISMA guidelines (9) from their inception to 2 February 2025 for randomised control trials (RCTs) investigating the efficacy of GONB for the treatment of migraine. Search terms (Supplemental 1) included: ‘greater occipital nerve block’; ‘GONB’; ‘migraine’; ‘episodic migraine’; ‘chronic migraine’; ‘headache’; and ‘nerve block’. To identify any grey literature, reverse citation and online searches (using Google and Microsoft Bing) were performed. This review has PROSPERO registration; ID: CRD42024595492.
Screening was performed independently by two reviewers with any conflicts resolved through a consensus meeting with the full research team. Duplicate records were removed and abstracts screened to exclude: non-English texts, conference abstracts, review articles, correspondence articles, non-RCTs, and incomplete studies. Inclusion criteria for full-text review were: RCTs investigating the efficacy of GONB; migraine patient cohorts (or reporting of migraine sub-cohort data if mixed headache diagnoses were enrolled); at least single blinding; a control intervention; successfully completed studies; English full-texts; and adult patient cohorts. Exclusion criteria were interventions performed that are not relevant to this review's focus (e.g., nerve stimulation). No limitations were made to publication status. Included studies underwent rigorous qualitative analysis. Cochrane Risk of Bias 2 (RoB2) assessments were performed.
Meta-analysis
Studies were screened with inclusion criteria: double blind RCTs; successful blinding; pain score data; monthly headache days data; diagnoses according to any edition of the International Classification of Headache Disorders (ICHD); control arms receiving placebo GONB injections; and dosages supplied for all interventions. Exclusion criteria were: ‘high’ risk of bias scores; pain pressure thresholds (PPTs) as headache severity measures; and confounding interventions performed during or immediately before trials.
To ensure heterogeneous studies were robustly analysed, data was segregated five times. First, studies investigating episodic relief of migraine over an acute follow-up (≤48 h) were separated from those with a longer-term follow-up (≥1 week). Second, studies were grouped by their GONB injectate: local anaesthetic (LA); steroid; and LA-steroid. The longer follow-up data was then subdivided three more times, by: injection regimens (single-, weekly-, and monthly-injections); laterality of GONB (unilateral; bilateral); and migraine diagnosis (EM; CM). Acute follow-up studies were inherently single-injection in their design. Additionally, for episodic management, assorted laterality of injections and migraine diagnoses were accepted to be pooled. Primary outcome measures were: a reduction in headache severity and, for longer follow-up studies, monthly headache days. The secondary outcome measure was the frequency of adverse effects.
Mean differences (MD) between control and intervention arms were calculated and DerSimonian and Laird random effects analyses performed. Heterogeneity was indicated by an I2 value above 50% or a significant chi-square test. Calculated probability (p-values) below 0.001 were considered significant. Studies were weighted inversely to the variance of their effect estimate and forest plots generated. Uncertainty in calculated values was expressed by confidence intervals. Certainty of outcomes was additionally assessed using a Grades of Recommendation, Assessment, Development, and Evaluation (GRADE) approach (10).
Results
Umbrella review
Figure 1 shows the results of the systematic search. No umbrella reviews were identified. Nine systematic reviews are included; all have significant limitations (Table 1).

Diagram showing the ‘hierarchy of evidence’ of clinical studies.
Qualitative analysis of systematic reviews investigating GONB for the treatment of migraine. Data arranged chronologically.

CM: Chronic Migraine; EM: Episodic Migraine; GONB: Greater Occipital Nerve Block; ICHD: International Classification for Headache Disorders; IV: Intravenous; LA: Local Anaesthetic; N: Number; SONB: Supraorbital Nerve Block; VAS: Visual Analogue Scale.
*Number of studies included in GONB meta-analyses only.
As per Google Scholar (correct as of 10/02/2025).
Data shown for GONB sub-analyses only.
Two reviews from the same Chinese province, by Tang et al. (11) and Zhang et al. (12), have identical methodological flaws. LA GONB arms (13–16) are pooled with LA-steroid GONB arms (17–19). Similarly, saline GONB control arms (13–16) are pooled with LA-saline GONB control arms (17–19). EM (18) and CM (14) patient cohorts are pooled within the authors’ analyses, as are studies with single-injection (13,15,17–19) and weekly-injection (14,16) GONB regimens. Unilateral GONB (15,18) data is also pooled with bilateral GONB (13,16,19) and trigger-point injection (17) data. A study where participants additionally received 20 mg oral propranolol twice daily (19) is included by both reviews. The study additionally does not use ICHD criteria for patient diagnoses, EM/CM populations are not defined, and doses of triamcinolone injections are not specified (only ‘0.5 ml’ volumes are stated). Another included study (18) merged EM and CM patient data as it was insufficiently powered to detect a difference between the subgroups. Both reviews erroneously include ‘one-week’ headache day data (13) in analyses of ‘monthly headache days’. Data collected at heterogeneous time-intervals, — from twenty minutes (17) up to two months (19) — are pooled in pain severity analyses. Tang et al. (11) additionally pool pain scores measured at three months (16). Both reviews erroneously pool ‘average monthly’ pain scores from one study (15) with endpoint pain score data. Similarly, both reviews misreport ‘duration of reduction in headache severity’ data (17) as ‘number of headache days’ data. Zhang additionally erroneously extracted one study's one-month headache duration data (14) in the intervention arm as ‘14.3 days’, rather than the correct value of ‘13.2 days’. The above limitations are not acknowledged by the reviewers nor are risk of bias assessments provided.
Shauly et al. (20) repeat an almost identical list of methodological errors, thereby bringing into question whether the three reviews were performed rigorously and independently. Shauly et al. additionally inappropriately pool a cervicogenic headache study which performed facial nerve blocks (21) and a triptan-overuse headache study (22) into their migraine focused analyses. The authors also make numerous data extraction errors. Figure 3 of Shauly et al.'s review lists ‘monthly headache days’ values for one study (13) as ‘13.6’ and ‘18.0’ for the intervention and placebo arms, respectively. Not only do these values not appear in the original paper, the study only had a one-week follow-up period. Although no such data transformation is acknowledged in their method, it appears Shauly et al. have multiplied ‘one week headache day’ values of ‘3.4’ and ‘4.5’ (13) by four to generate monthly values. This erroneously assumes that each week of the month had uniform headache frequency. Furthermore, in figure 4 of the review, the data extracted as endpoint ‘placebo visual analogue scale (VAS) pain scores’ for one study (17) are instead the placebo arm's baseline pain scores. Similarly, the reported endpoint ‘intervention VAS pain scores’ are actually the endpoint values for the placebo arm (17). Errors in data extraction, such as the misdating of Ashkenazi et al.'s 2008 paper (17) as ‘Ashkenazi et al. 2018’, further calls into question the rigour of analyses performed.
Contrastingly, Barad et al.'s review (23) has laudably robust methodology. However, the authors pool within analyses two studies (18,19) with numerous limitations, as outlined earlier. Additionally, as two GONB analyses each contain only one study, very limited conclusions can be made.
The remaining reviews (24–28) repeat several of the aforementioned methodological errors — for brevity, these are summarised in Table 1. Four reviews (24–26,28) additionally include a study (29) where participants received three quarters of their allocated injectate (LA or saline) as a GONB and the remaining quarter as a supraorbital nerve block (SONB). Moreover, within Mustafa et al.’s (26) analysis of ‘headache frequency during the second month’ data collected two month after patients received three (29) and four (16) doses of weekly GONB injections is pooled with data collected between ‘weeks 9–12’ of a four-weekly GONB injection regime (30).
Chang et al.'s review (25) contains several additional errors. The authors’ analysis of ‘changes in headache episodes per month’, contains a mixture of ‘headache days’ (14,16), ‘headache frequency’ (29), and ‘≥moderate migraine frequency’ (18) data. This data was also measured at various timepoints, from one to three months. The analysis is also incorrect; mean values labelled as ‘Inan et al.’ (14) are in fact data from Dilli et al.'s study (18), and vice versa. Likewise, the data labelled as ‘Özer et al.’ (29) is from Gul et al.'s RCT (16), and vice versa. As the N values for all four studies are accurate to their respective study labels (i.e., the rows are not just mislabelled), the analysis is uninterpretable. Similarly, in the review's ‘changes in headache severity’ analysis, heterogeneous studies are pooled and data extraction for one study (14) is incorrectly reported as ‘0.93’ and ‘0.47’ for the intervention and control arms respectively. The correct values are ‘3.1’ and ‘1.4’, respectively (14). This error has significant influence as the study is weighted twelve times more than remaining GONB studies. Additionally, ‘changes in headache duration’ data was attributed to a study (29) where this was not an outcome measure. The source of this data is unclear, and the study is nonsensically weighted ‘0.0%’ in the analysis. This, with other errors — table 1 of their review erroneously reports that a study (31) had a duration of ‘270 weeks’ rather than ‘270 days’ — brings into question the reliability of analyses performed.
Li and Tang's review of GONB for ‘acute migraine’ (27) has numerous further faults. ‘Placebo’ arms within analyses contain data from patients who received 10 mg intravenous (IV) metoclopramide infusions (32). This is an effective dose for the treatment of acute migraine attacks (33) and is misleading to be labelled as ‘placebo’ data. In fact, the cited study concluded that IV metoclopramide outperformed bupivacaine GONB in reducing headache severity scores (32). Similarly, patients from the ‘treatment’ arm of the study also received IV saline infusions. This is methodologically problematic as it raises the possibility that a part of the overall effect observed in this arm might be due to non-specific (i.e., placebo) effects attributable to IV saline, not present in the placebo arm, which have been reported to be equivalent to those of a 4–6 mg IV morphine infusion (34). It is therefore misleading to label this data solely as ‘GONB’. Neither IV intervention is mentioned in the review. The review also includes a study which enrolled patients who failed to respond to parenteral metoclopramide, administered only hours before the GONB investigations (35). This confounds interpretation of the efficacy of GONB. Furthermore, the RCT was stopped before an effective treatment size was achieved, blinding was reported to have likely failed, and 60% of the placebo group and 77% of the GONB group had taken analgesia prior to their arrival (35). Another included study reported that 75% of the metoclopramide group and 63% of the GONB group had taken other medications (32). The remaining two studies excluded any patients with recent medication use. Further unreported heterogeneity between the studies is summarised within Table 1; despite this extensive list, Li and Tang did not perform a sensitivity analysis. Begg and Egger tests were, however, performed by the authors despite analyses containing, at most, only three studies.
Li and Tang's analyses (27) also contain numerous errors. Figure 6 of the review shows ‘the meta-analysis of sustained headache relief’. However, the figure erroneously contains ‘sustained headache freedom’ data from one study (32); the correct values were in fact ‘11 (22%)’ and ‘18 (38%)’ for the LA-GONB (with IV saline) and IV metoclopramide (with saline GONB) arms respectively. Li and Tang also incorrectly define ‘sustained headache relief’ as ‘achieving a pain level of none within 2 h of medication administration and maintaining a level of none without the use of additional analgesic medication for 48 h’ — this definition is more appropriate for ‘sustained headache freedom’. Median value data from one study (36) is erroneously labelled as ‘mean’ value data in figures 3-5 of Li and Tang's review. The unequal distances between the median and interquartile ranges (IQRs) suggest an asymmetrical data distribution; therefore, mean and median values were not equitable. Due to the low sample size of the data, this equivalence may be acceptable but this should nonetheless have been acknowledged as a limitation by the authors. Moreover, in Figure 6 of the review, x-axis values below one are erroneously labelled as ‘favours [experimental]’. An odd's ratio below one suggests that the exposure (GONB) reduces the likelihood of the outcome measure (sustained headache relief). As a positive effect is being analysed, this would therefore favour the control group.
Hidayati et al.'s review (28) contains a study that was discontinued before an a priori sample size was achieved (37). Another included study (14) investigated the effects of LA GONB against a placebo intervention for one month; afterwards, blinding was removed and both arms received the treatment intervention with patents followed up for a further two months. Hidayati et al. erroneously extract data from months 2 and 3 of the study (i.e., post patient crossover). Therefore, the control arms of analyses also include LA GONB data. Hence, in figures 4 and 7 of the review, the control arm for the study (14) is erroneously reported as outperforming the treatment arm. Furthermore, the review's analyses are categorised either by measurement timepoints (but with merged injectate contents) or by injectate contents (with merged data timepoints, from 20 min up to 3 months). The review also includes a study in GONB analyses where treatment and control arms additionally received sham SONB injections (38); this may skew data. There is also erroneous grouping of data. Figure 8 of the review shows the analysis of GONB containing ‘8–20 mg triamcinolone’; however, the figure also includes data for LA-steroid GONB (17,19). Additional sources of heterogeneity (Table 1) unfortunately render the authors’ analyses uninterpretable.
Due to the significant errors, methodological incongruence, and poor rigour of these systematic reviews — which have in total been cited 256 times as evidence for GONB in the treatment of migraine — no meaningful statistical analysis could be performed in our umbrella review. An independent systematic review and meta-analysis of RCT evidence was therefore performed.
Systematic review
Sixteen RCTs (930 patients) were included for qualitative analysis (Figure 2). Active GONB arms had diverse local anaesthetic formulations, including: 1 ml 2% prilocaine (39); 1 ml–6 ml 0.5% bupivacaine with (14,16,36) and without (13,15,32,35) 1 ml saline; 2 ml 2% lidocaine with (40,41) and without (30) 0.5–2 ml saline; 1 ml 1% lidocaine with 1 ml saline (38); 1 ml 2% lidocaine with 0.5–1 ml saline (19,29); and 4.5 ml 2% lidocaine with 4.5 ml 0.5% bupivacaine and 1 ml saline (17). LA-steroid GONB regimens included: 2.5 ml bupivacaine with 20 mg methylprednisolone (18); 2 ml 2% lidocaine with 20 mg triamcinolone (40); 2 ml 2% lidocaine with 80 mg methylprednisolone (41); and 4.5 ml 2% lidocaine with 4.5 ml 0.5% bupivacaine and 40 mg triamcinolone (17). One study had a non-specified steroid dosage of ‘1 ml 2% lidocaine with 0.5 ml triamcinolone’ (19). Steroid GONB injections were performed by one study; these contained 20 mg triamcinolone and 2 ml saline (40).

Flowchart showing results of search for systematic reviews with meta-analyses of GONB for the treatment of migraine.
Control arms mostly received saline GONB procedures, except two studies which performed: 2.75 ml saline shams with 0.25 ml 1% lidocaine (18); and 1 ml 0.5% Bupivacaine intradermal injections (35). In three studies, control arms received additional pharmacologic interventions, namely: 20 mg oral propranolol twice daily (19); 10 mg IV metoclopramide (32); and 100 mg oral topiramate once daily (41). Two RCTs were essentially add-on studies with no true control arm (17,19).
Eleven studies (13,16,17,19,29,30,32,35,38,40,41) performed GONB injections bilaterally; while two studies (15,39) performed unilateral injections on the patient's most affected side. Two studies (18,36) performed either bilateral or unilateral injections dependent on the patient's symptoms. One study (14) did not clarify the injection laterality. Primary outcome measures for six studies (17,32,35,36,38,39) were assessed over a period of ≤48 h. The remaining ten studies had follow-up periods of ≥1 week. One study was multi-centre (14). Forty four percent of studies were conducted in Türkiye. Studies included patients both with and without aura. Mean patient age was 37.4 years. Patient cohorts were predominantly female (Mean 86.3%; 803/930).
Studies had numerous limitations, as summarised in Table 2. Inan et al. (14) did not specify whether GONB injections were performed bilaterally or unilaterally. However, as the study shared not only authorship but verbatim sections of text with another RCT (16), it was assumed that the studies had identical methodology (i.e., bilateral GONB). Five studies had ‘high’ overall risk of bias scores (Online Supplemental Materials 2, 3).
Qualitative analysis of randomised controlled trials investigating GONB for the treatment of migraine.
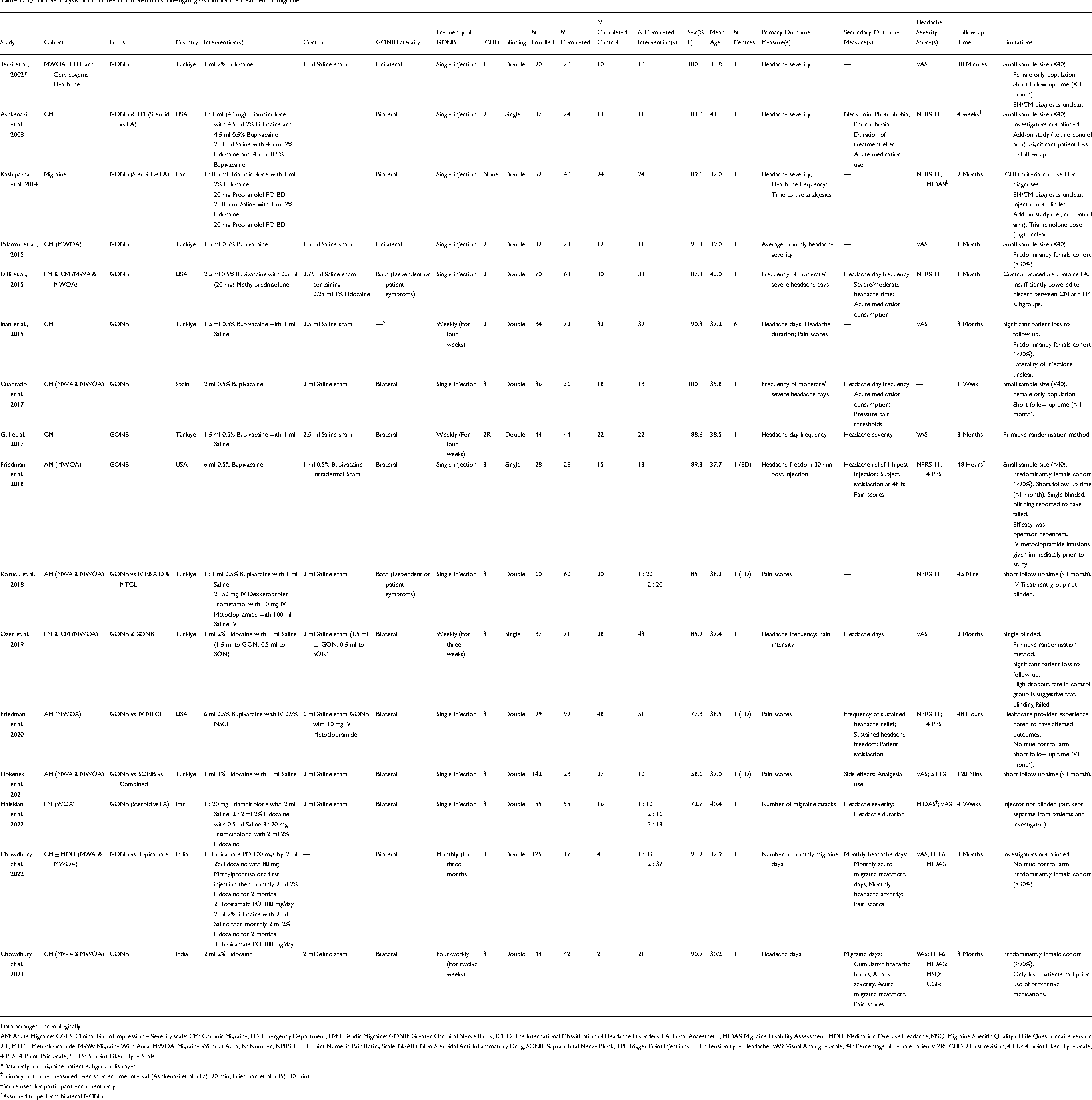
Data arranged chronologically.
AM: Acute Migraine; CGI-S: Clinical Global Impression – Severity scale; CM: Chronic Migraine; ED: Emergency Department; EM: Episodic Migraine; GONB: Greater Occipital Nerve Block; ICHD: The International Classification of Headache Disorders; LA: Local Anaesthetic; MIDAS: Migraine Disability Assessment; MOH: Medication Overuse Headache; MSQ: Migraine-Specific Quality of Life Questionnaire version 2.1; MTCL: Metoclopramide; MWA: Migraine With Aura; MWOA: Migraine Without Aura; N: Number; NPRS-11: 11-Point Numeric Pain Rating Scale; NSAID: Non-Steroidal Anti-Inflammatory Drug; SONB: Supraorbital Nerve Block; TPI: Trigger Point Injections; TTH: Tension-type Headache; VAS: Visual Analogue Scale; %F: Percentage of Female patients; 2R: ICHD-2 First revision; 4-LTS: 4-point Likert Type Scale; 4-PPS: 4-Point Pain Scale; 5-LTS: 5-point Likert Type Scale.
*Data only for migraine patient subgroup displayed.
Primary outcome measured over shorter time interval (Ashkenazi et al. (17): 20 min; Friedman et al. (35): 30 min).
Score used for participant enrolment only.
Assumed to perform bilateral GONB.
Meta-analysis
Seven studies (401 patients) were eligible for quantitative analysis (Online Supplemental Material 4). Three analyses were viable (i.e., contained at least two studies); these included data for 304 patients.
Figure 3 shows the analysis of headache severity for LA GONB over an acute follow-up period (≤48 h). Three studies were included, each performed different LA regimens, namely: 1 ml 2% prilocaine (39), 1 ml 0.5% bupivacaine with 1 ml saline (36), and 1 ml 1% lidocaine with 1 ml saline (38). One study performed unilateral GONB (39), another bilateral GONB (36), and the third performed a mixed lateralisation of injections (38). None of the included studies stated whether patients had EM nor CM diagnoses. This methodological heterogeneity was deemed acceptable for an analysis of GONB for acute migraine episodes. Our analysis showed that LA GONB offered significant benefit at 30 min post-injection (MD = −2.08; p < 0.001). Further inference was limited due to remaining timepoints containing two or fewer studies.
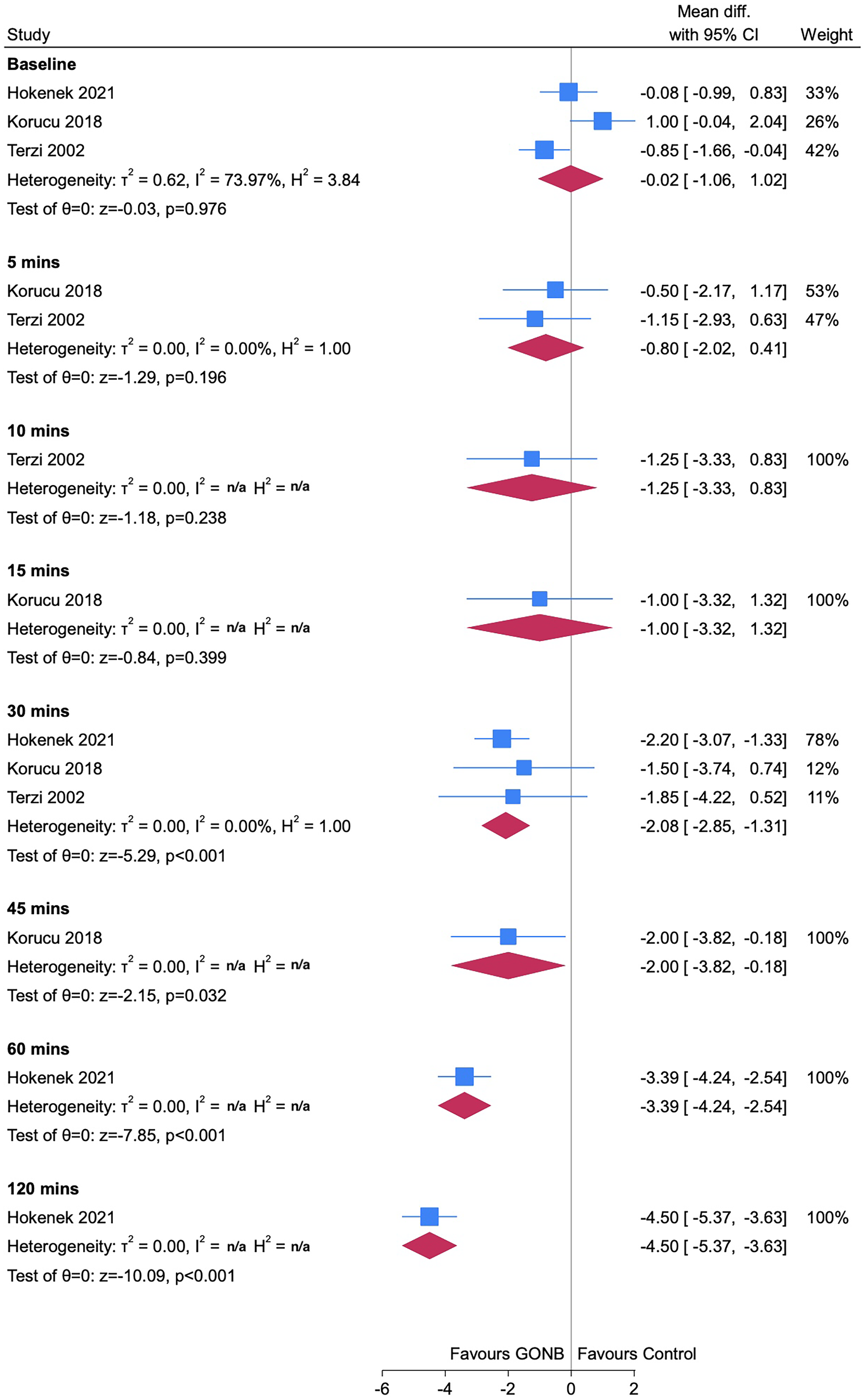
Forest plot showing analysis of headache severity data for studies with an acute follow-up period (i.e., ≤48 h). All studies performed local anaesthetic greater occipital nerve block.
Figure 4 shows the analysis of headache severity for weekly bilateral LA GONB injections in CM patient cohorts over a longer follow-up period (≥1 week). Intervention arms in both studies received GONB injections containing 1.5 ml 0.5% bupivacaine with 1 ml saline (14,16). All timepoints within our analysis contained two or fewer studies; interpretation was therefore limited. However, our analysis suggested that LA GONB may reduce headache severity at one month (MD −1.33; p < 0.001). This effect may be greater at longer timepoints.
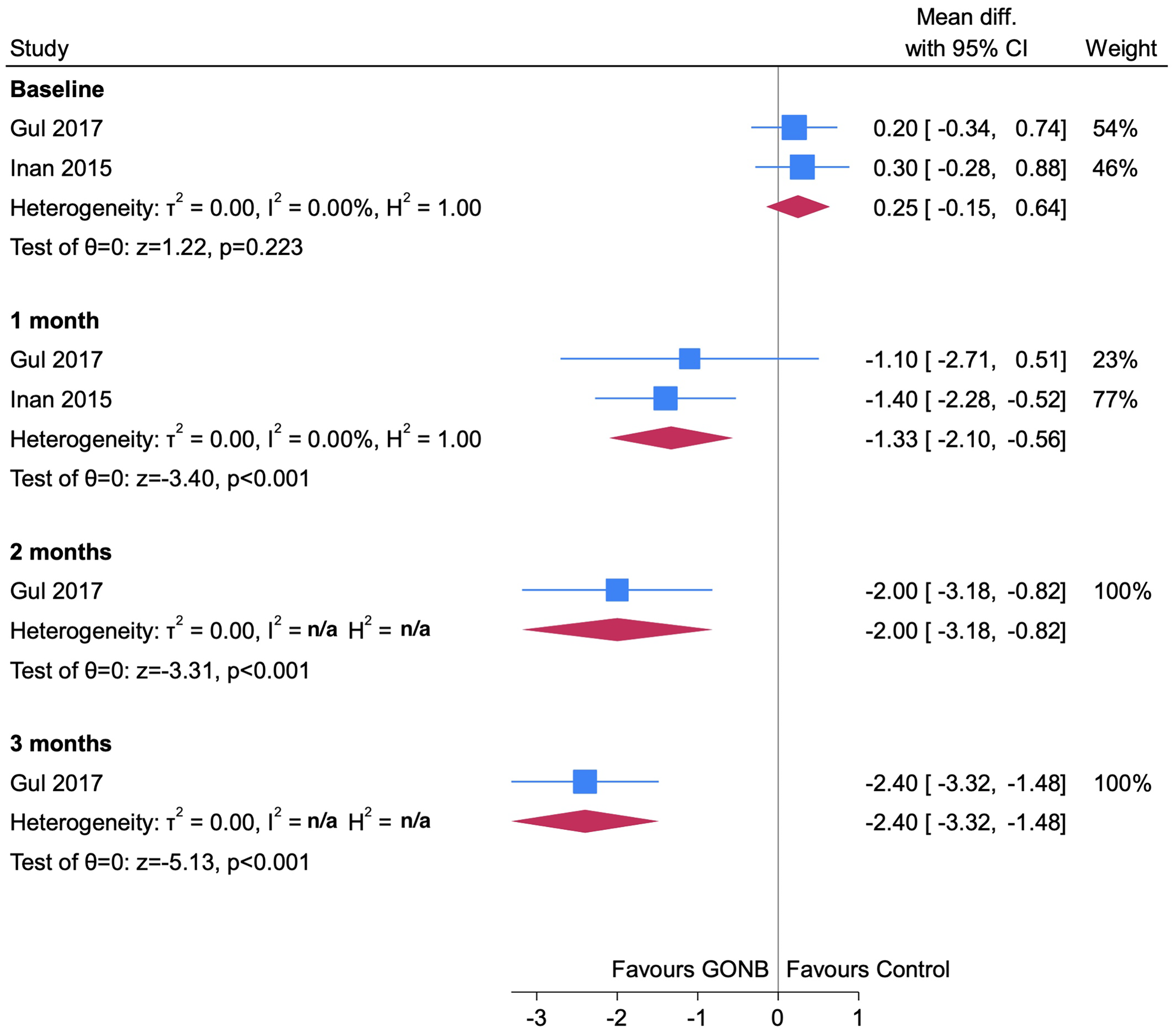
Forest plot showing analysis of headache severity data for studies with a longer follow-up period (≥1 week). Both studies performed weekly bilateral injection regimens of local anaesthetic greater occipital nerve block for solely chronic migraine patient cohorts.
Similarly, Figure 5 shows the analysis of monthly headache days for weekly bilateral LA GONB injections in CM patient cohorts over a longer follow-up period. Our analysis suggests that, after one month, LA GONB reduces monthly headache days (MD = −4.46; p < 0.001). Again, these effects may be greater at longer timepoints.

Forest plot showing analysis of monthly headache day data for studies with a longer follow-up period (≥1 week). Both studies performed weekly bilateral injection regimens of local anaesthetic greater occipital nerve block for solely chronic migraine patient cohorts.
Four studies (211 patients) reported figures for adverse effects; the most common (among LA and placebo GONB) were: pain at the injection site; local site bleeding; vertigo/dizziness; and neck pain/spasm (Online Supplemental Material 5). Steroid injections were uniquely and rarely associated with cutaneous atrophy and alopecia. No serious adverse effects (e.g., hypotension, anaphylaxis) were observed.
Due to a moderate risk of bias within included studies, we were able to conclude that, with moderate certainty, GONB reduces headache severity over an acute (i.e., ≤ 48 h) follow-up period (Table 3). Due to a moderate risk of bias, as well as statistical imprecision due to a paucity of studies, we were able to conclude that, with low certainty, GONB reduces headache severity and monthly headache days over a longer (i.e., ≥ 1 week) follow-up period. It is important to note that data was insufficient to generate funnel plots; thus, publication bias could not be evaluated.
Summary of findings from systematic review and meta-analysis.

N: Number; GRADE: Grading of Recommendations Assessment, Development and Evaluation; GONB: Greater Occipital Nerve Blockade; LA: Local Anaesthetic; CM: Chronic Migraine.
*Publication bias could not be assessed.
**As per: Balshem H et al. (10).
Discussion
This paper highlights a significant paucity of robust RCT evidence of GONB for the treatment of migraine. Despite this, multiple relevant systematic reviews have been published, often with erroneous methodologies.
Our quantitative meta-analysis does suggest a potential effect of GONB for migraine, most confidently within the acute setting. However, these findings should be interpreted with caution due to a lack of robust RCTs. The International Headache Society (IHS) guidelines for controlled trials of preventive treatment of chronic migraine in adults recommends primary endpoints of either a change in migraine days, a change in moderate to severe headache days, or a responder rate of at least a 50% reduction in headache days from baseline (42) — although various percentages from 30% to 100% have also been utilized (42,43). A number of Patient Reported Outcome Measures are recommended for assessing functional ability, including the HIT-6. The tool has been shown to outperform Patient Global Impression; a ≥ 6-point improvement in the total score successfully identifies individuals with CM who have experienced meaningful change over time (44).
The majority of RCTs investigating GONB used either the VAS or 11-Point Numeric Pain Rating Scale (NPRS-11) as measures of headache severity. The VAS requires patients to mark their pain intensity along a non-graduated line, usually with verbal anchors such as ‘no pain’ and ‘worst pain imaginable’ on either side. The NPRS-11 is similar to the VAS but replaces the verbal anchors with ‘0’ and ‘10’ and the patient must select a whole number within this range of values. Current IHS guidelines recommend use of a four point pain scale (i.e ‘absent, mild, moderate, or severe’) and/or the NPRS-11 as secondary endpoint measures to assess migraine intensity in controlled trials of prophylactic treatment of CM (42). For acute migraine episodes, the IHS recommends co-primary endpoints of ‘pain freedom at 2 h’ and the ‘absence of the [patient's] most bothersome symptoms (45), all of these scores are self-reported unidimensional scales and arguably do not provide a comprehensive and objective assessment of the impact of migraine on a patient's quality of life, disability status, nor progress towards treatment goals.
The guidelines also recommend the use of patient reported outcomes which measure functional ability in a number of relevant domains. Multiple such tools have been developed. The Migraine Disability Assessment (MIDAS) questionnaire quantifies headache severity and frequency as well as evaluating the effect of migraine on a patient's schoolwork/job, housework, and social life (46). Two RCTs used MIDAS for patient enrolment (19,40) and another two used the score as secondary outcome measures (30,41). Two studies used the six-item Headache Impact Test (HIT-6) as secondary outcome measures (30,41). The HIT-6 assesses the impact that headaches have on a patient's normal activity using 5-point Likert scales (47). One of these RCTs additionally used both the Migraine Specific Quality-of-life (MSQ) and Clinical Global Impression Scale – Severity (CGI-S) questionnaires as secondary outcome measures (30). The MSQ — a 14-item questionnaire that measures patient quality of life over four weeks — has been validated for use in chronic migraine patients (48). It consists of three components: role function-restrictive (RR), role function-preventive (RP), and emotional function (EF). Finally, the CGI-S is a single-item measure that asks clinicians to rate a patient's illness on a scale of ‘normal’ to ‘among the most extremely ill patients’ (49).
There are also specific considerations regarding the use of placebo in trials involving injectable treatments. Responses to placebo can vary significantly depending on many factors, including the mode of administration. This is of particular importance in pain syndromes, where there is significant interplay with emotional and cognitive processes. Sham acupuncture and sham surgical procedures cause a greater reduction in migraine frequency than oral placebo interventions; these effects may account for a large component of the overall treatment effects observed by clinicians in their daily practice (50). Injectable treatments for headache episodes have similarly been reported to exhibit higher placebo response rates than oral interventions (51). This is possibly due to their association with contextual factors, the level of physical contact involved and, potentially, the psychological effects associated with the mode of delivery. Additionally, there is growing evidence that placebo and treatment responses interact in non-additive ways, especially when the placebo effect is large, such as in pain studies (52). This raises the possibility that a potentially significant part of the observed effects in the studies included in this meta-analysis are the result of non-specific effects, given the injection nature of the treatment modality and the absence of ‘no-intervention’ arms in the studies included.
Although, often proffered as a quick-fix out of a painful crisis, GONB has not been shown to be consistent in response provided, have disease modifying capability, be cost-effective, nor nurture patient-independence in long-term management of their disability. As such, it would be better for resources to be funnelled towards evidence-based treatments, such as the novel Calcitonin Gene-Related Peptide (CGRP) directed therapies. However, as an acute treatment, GONB may be of interim benefit in regions where alternative options are not available, cost-restricted, and/or not appropriate. Repeated GONB use should be evaluated within a robust RCT that conforms to the IHS’ agreed outcomes and also incorporates measures of functional ability.
It is noteworthy that there are many interventions in chronic pain management that are without established value. Despite many small studies investigating GONB, outcomes suggest benefit is of limited practical value.
Limitations
The major-most limitation this paper faced was a significant paucity in robust studies investigating GONB for the treatment of migraine. Due to this, and significant methodological errors within relevant systematic reviews, we were unable to perform any quantitative analysis within our umbrella review.
Given the heterogeneous methodologies of available RCT evidence, our method of data segregation within our systematic review needed to be robust. Segregating data five times would have, theoretically, optimally produced thirty-nine separate ‘headache severity’ analyses (three with an acute follow-up period and thirty-six with a longer follow-up period) and thirty-six ‘monthly headache day’ analyses (all with a longer follow-up period). A lack of eligible studies limited the accuracy of analyses performed and also prevented the analysis of: steroid GONB; LA-steroid GONB; unilateral GONB; and GONB for EM patient cohorts. Similarly, migraine with and without aura could not be separately analysed. Further inter-subgroup analyses of pertinent value include: ‘landmark vs. ultrasound-guided techniques’; ‘unilateral vs bilateral GONB’; and comparisons of measures of functional ability. This paper highlights the need for multiple robust RCTs with homogeneous and comparable methodologies that evaluate a variety of intervention subgroups. Once this RCT evidence is attained, relevant systematic reviews and umbrella reviews should then be completed.
Several data adjustments were required for statistical analysis. Orbitofrontal and orbitonuchal pain scores were averaged for one study (39). Median values from another study (36) were assumed to be equivalent to mean values and standard deviation values calculated by dividing IQR widths by 1.35. IQRs of the dataset were unequal distances from the median value (suggesting an asymmetrical distribution); however, this approximation was accepted due to the cohort size (n = 60). VAS and NPRS-11 measures were also considered equivalent within analyses. These scores did not give an in-depth impression of patient quality of life nor disability status.
Furthermore, our analysis of headache severity for LA GONB over an acute follow-up period (≤48 h) contained studies that performed GONB with different LA injectates. One study that was included within both of our longer follow-up (i.e., ≥ 1 week) analyses had unclear methodology and was assumed to perform bilateral GONB (14). Finally, adverse effects data was only provided by four of the seven studies included for quantitative analysis.
Conclusions
This paper highlights a significant paucity of robust RCT evidence of GONB for the treatment of migraine. Despite this, multiple relevant systematic reviews have been published, often with erroneous methodologies. These reviews are often cited as supporting evidence for GONB and should instead be interpreted with caution. With moderate certainty, LA GONB improves headache severity in acute migraine episodes (≤48 h). With low certainty, weekly bilateral LA GONB may show benefit at one month for CM. Any sustained benefits offered by GONB remain unclear. Despite the favourability that procedural interventions proffer to both patients and clinicians, there is a severely limited evidence-base to advocate for GONB in the routine management of migraine. There remains a pertinent need for rigorous RCTs with homogeneous and comparable methodologies; systematic reviews and umbrella reviews should then be subsequently performed to definitively establish the role of GONB in the treatment of migraine.
Key findings
Systematic reviews evaluating GONB for migraine have significant limitations and methodological errors.
RCT evidence supporting GONB use is limited; local anaesthetic GONB may improve headache severity in acute migraine episodes and may have a role in regions where alternative therapies are unavailable.
Longer-term benefits from repeated GONB use remain unclear; resources should be diverted towards alternative evidence-based treatments.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251398390 - Supplemental material for Greater occipital nerve block for the treatment of migraine: An umbrella review, systematic review, and meta-analysis
Supplemental material, sj-docx-1-cep-10.1177_03331024251398390 for Greater occipital nerve block for the treatment of migraine: An umbrella review, systematic review, and meta-analysis by Dominic Atraszkiewicz, Emre Ünal, Paul Bassett, Fausto Morell-Ducos and Anish Bahra in Cephalalgia
Footnotes
Author contributions
Project conceptualised and designed by AB, DA, and FM-D.
Umbrella review: record screening, data extraction, and data analysis performed by DA.
Systematic review: record screening and data extraction performed by DA and EU, with conflicts resolved by AB and FM-D. Risk of bias assessment performed by DA and EU. Data analysis performed by DA. Statistical tests performed by PB. Tables and figures created by DA and PB.
Manuscript written by DA with final contributions by AB and FM-D.
All authors read and approved the final manuscript.
Data availability statement
All data generated or analysed during this study are included within this paper and supplementary information files. This data is not available elsewhere.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
