Abstract
Abstract
Objective
Since the data regarding the efficacy of greater occipital in episodic migraines are rare, we aimed to examine the efficacy of greater occipital block in the prophylaxis of episodic migraines without aura and compare different injectable drug regimens.
Methods
In a randomized, double-blind placebo-controlled trial, adult patients suffering from episodic migraines without aura were randomized to one of the following: triamcinolone, lidocaine, triamcinolone plus lidocaine, and saline. Patients were assessed at baseline, one week, two weeks, and four weeks after the injection for severity and duration of headaches and side effects.
Results
Fifty-five patients completed the study. Repeated measures ANOVA indicated that the severity and duration decreased significantly after the greater occipital block (P < 0.001, P = 0.001 respectively) in all four groups. However, there was no difference between groups at any study time points (P > 0.05). In paired sample T-test, only groups 2 and 3 with lidocaine as a part of the injection showed a significant decrease in frequency compared to the baseline (P = 0.002, P = 0.019). Three patients reported side effects with a possible association with triamcinolone.
Conclusion
Greater occipital block with a local anesthetic significantly decreases the number of attacks in episodic migraine, whereas no injection was superior to the placebo in regards to the duration and severity of the headaches.
Introduction
Migraine headaches, affecting more than one billion people globally, are responsible for a tremendous amount of decreased productivity and lost working days, roughly five times more than that of tension-type headaches (1). Therefore, a great deal of emphasis is put on developing and evaluating novel treatments that would address the disability caused by migraine. Prophylactic treatment with antidepressants, antiepileptics, calcium channel blockers, beta-blockers, and most recently with monoclonal antibodies against calcitonin gene related peptide (CGRP) have shown different efficacies in reducing the attack frequency, severity, and duration (2,3). However, alternative treatments are also required, as there are patients who do not achieve adequate pain relief or who develop side effects of migraine prophylactic agents. Greater occipital nerve (GON) block with local anesthetics and steroids has been used to treat several types of headaches (4–7). Peripheral nerve blocks, including GON block, have been used for many years (8) as an accessory treatment option in patients with migraine – especially those with chronic migraine. There have been several proposed mechanisms in the literature that could partly explain the possible underlying pathophysiology of the effect of GON block on migraine headaches. The main focus in these studies has been on the central nervous system (CNS) connections of GON and trigeminal afferent fibers, which are believed to be the prime origin of migraine headaches (9–11). On the other hand, neither the results from available clinical trials nor the experts are in full agreement regarding this method's efficacy in reducing the frequency or intensity of the headaches in patients with migraine (4–6,12–15). Furthermore, the medications used for the GON block (16) and the putative duration (7) of effect have been topics of interest in this field. Given the research gaps and paucity of evidence for the use of GON block as a prophylactic treatment in episodic migraine, we aimed to evaluate the efficacy of GON blocks in the prophylaxis of headaches in patients suffering from episodic migraine without aura (MwoA). Our main goal was to determine whether GON block would be effective in reducing the severity and duration of the headache episodes that patients experienced and whether this method would be superior to the placebo in decreasing the number of attacks in patients with episodic MwoA in a month. Additionally, to determine the most effective regimen for the GON block, we decided to use three different regimens and one placebo injection.
Method and materials
We conducted a randomized, double-blind placebo-controlled trial on adult patients suffering from episodic MwoA. These subjects were sequentially chosen from patients referred to the Neurology Clinic at Tehran's Shariati Hospital between 2018–2019. The episodic migraine headache without aura diagnosis was initially made according to the International Headache Society's ICHD-3 beta version published in 2013 (17). Since the final version of ICHD-3 was published in 2018, the diagnosis was made in accordance with the most recent guideline (18). The study protocol was approved by the Ethics Committee of Tehran University of Medical Sciences (Approval ID: IR.TUMS.MEDICINE.REC.1397.130), and written informed consents were acquired from all the participants before entering the study. This study was registered in the Iranian registry of clinical trials (Registration number: IRCT2017070334879N1).
Inclusion and exclusion criteria
The study's inclusion criteria were as follows: 18 to 65 years of age and a history of MwoA with a frequency of at least four attacks per month. The exclusion criteria included: 1) less than fifteen headache-free days per month, 2) start or change of prophylactic migraine treatments within the last month, 3) hypersensitivity to lidocaine or triamcinolone, 4) history of seizure, 5) local infection on the injection site, 6) history of craniotomy, 7) medication overuse headache based on ICHD-3, and 8) a migraine disability assessment score (MIDAS) greater than twenty-one (17–19).
Sample size and randomization
The calculated sample size for the study was 80 patients (20 patients in each group). However, due to the disruption of routine clinic visits during the COVID-19 pandemic and to avoid unnecessary exposure of patients, we stopped recruiting patients after 55 patients were registered. For randomization, we used computer-generated blocks of four in random order. Based on this method, patients were assigned to one of the following groups: 1) Triam (triamcinolone): 20 mg (0.5 ml) of triamcinolone and 2 ml of saline (n = 10), 2) Lido (lidocaine): 2.0 ml of lidocaine 2% and 0.5 ml of saline (n = 16), 3) Lido + Triam (lidocaine plus triamcinolone): 20 mg (0.5 ml) of triamcinolone and 2.0 ml of lidocaine 2% (n = 13), and 4) NS (normal saline): 2.5 ml of 0.9% saline solution (n = 16).
Method of intervention
Each patient underwent a single injection session after registration. The injections were given bilaterally at a point 2 cm lateral on the line that connects the occipital protuberance to the mastoid process and medial to the occipital artery pulse with 22-gauge needles (20). The person responsible for preparing and administrating injections was not blinded to the intervention but had no contact with the patients or the research team during registration or follow-up. The patients were kept under observation for 30 minutes after receiving injections to note possible immediate side effects.
Outcomes and patient evaluation
A neurologist blinded to the type of treatment was assigned to evaluate the patients at the first and follow-up visits. The patients were assessed at baseline, one week, two weeks, and four weeks after the injection for pain severity and duration. The frequency of headaches was assessed as the number of attacks in four weeks; the baseline was the number of attacks patients experienced in the four weeks preceding the study, and the outcome was assessed as the number of attacks patients experienced in the four-week-period following the intervention. Patients were asked to record every headache episode and rate the severity by the Visual Analogue Scale (VAS) of pain. The duration of headaches was recorded in hours. We used the number of attacks and the average of headache severity and duration in our final analysis.
Adverse events
Three of the patients reported side effects associated with the injection site. Upon examination, cutaneous atrophy and alopecia were observed and recorded in these patients. In order to review the incidence, after halting the study, an independent party was asked to review the type of injection that these patients received. After reviewing the groups, it was noted that two patients were in the group Triam (triamcinolone), and one patient was in the group Lido + Triam (lidocaine plus triamcinolone). As a result, considering the possible association of local corticosteroids with mentioned side effects, we stopped recruiting patients in these two groups. The blinding and recruitment were maintained for the other two groups. We included the patients who already received either of the mentioned injections in our final analysis.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics 26.0. The results were presented as means ± standard deviations (SD) for quantitative variables and were summarized by frequency (percentage) for categorical variables. T-tests and repeated measures analysis of variance (ANOVA) were used to compare the groups' means. Chi-square was used to compare variable frequencies in different groups, and where the cell size requirements for chi-square were not met, Fisher's exact test was used instead. Paired sample T-tests were used for variables for comparisons within the groups. Profile plots were produced using the repeated measures ANOVA model for each of the variables. P-value <0.05 was considered as significant.
Results
In total ninety-five patients were considered for the study; 8 patients were excluded due to the previous history of seizure, and 32 patients declined to consent for the study. Fifty-five patients completed the study and were included in the analysis (Figure 1). The mean age of the participants was 40.42 ± 12.23, and 72.7% of the patients were female. The baseline characteristics of the patients in all four groups, as summarized in Table 1, show no significant difference between the groups at baseline. Sixteen patients were already on preventive migraine medications with a stable dose for at least two months. There was no significant difference between groups in regards to the number of patients receiving prophylactic medication (P = 0.442). The data regarding headache severity and duration were collected in four different time points, and the frequency was assessed once in baseline and once four weeks after intervention. Supplemental Table 1 outlines mean measures and standard deviations for severity, duration, and frequency in each time point.

Flow chart of the study.
Baseline characteristics.

*Triamcinolone: 20 mg (0.5 ml) of triamcinolone and 2 ml of 0.9% saline.
†Lidocaine: 2.0 ml of lidocaine 2% and 0.5 ml of 0.9% saline.
‡Lidocaine Plus triamcinolone: 20 mg (0.5 ml) of triamcinolone and 2.0 ml of lidocaine 2%.
§Saline solution: 2.5 ml of 0.9% saline solution.
**Calculated by Fisher’s Exact test.
††Calculated by one-way analysis of variances (ANOVA).
‡‡Mean headache severity in the preceding week assessed by visual analogue scale (VAS).
§§Mean headache duration in the preceding week in hours.
***Number of attacks in the preceding month.
†††Number of patients who were already on preventive medication.
‡‡‡Calculated by Chi Square.
Headache severity and duration
Repeated measure analysis of variance (ANOVA) with time as within-subject factor and group as a between-subject factor was conducted to analyze the effect of different interventions through time on headache severity and duration. The analysis shows that all four groups experience a significant reduction in headache severity and duration with a P-value <0.001 and P-value = 0.001 for severity and duration, respectively. On the other hand, there was no significant interaction between group and time; as a result, no significant difference was observed between groups at any of the time points (P-value = 0.871, P-value = 0.819 for severity and duration, respectively) (Table 2, Figure 2, and Figure 3). Supplemental Table 2 summarizes the pairwise comparison of the estimated means for each time point with Bonferroni adjustment of confidence intervals. This shows that compared to baseline, headache severity was significantly lower at all three time points (P < 0.001), and while at the end of the fourth week, the mean severity was still lower compared to baseline, it had significantly increased compared to the end of the second week (mean difference = 1.537 ± 0.421, P-value = 0.004). In the case of headache duration, in pairwise comparison with Bonferroni adjustment, only the mean duration of headache at the end of the first and second week were significantly lower than the baseline (P-value = 0.003, P-value = 0.007 respectively), and compared to the baseline, the mean duration of headache was not significantly different at the end of the fourth week (P-value = 0.196).
Results obtained from repeated measure ANOVA for the effect of time and interaction of Group and Time on headache severity and duration.
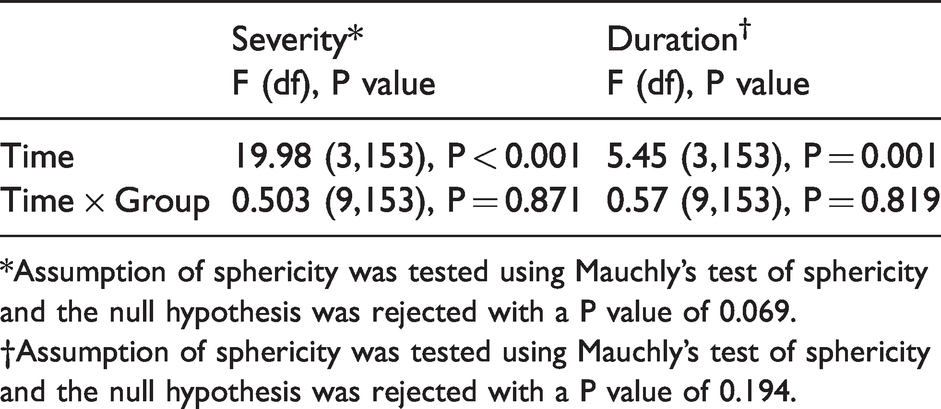
*Assumption of sphericity was tested using Mauchly’s test of sphericity and the null hypothesis was rejected with a P value of 0.069.
†Assumption of sphericity was tested using Mauchly’s test of sphericity and the null hypothesis was rejected with a P value of 0.194.

Profile plot of headache severity by time where each line represents the changes of a group within time. Error bars represent 95% confidence interval.

Profile plot of headache duration by time where each line represents the changes of a group within time. Error bars represent 95% confidence interval.
Headache frequency
We analyzed the before and after data for headache frequency with paired sample T-test and used one-way ANOVA to compare the data between groups at baseline and at the end of week 4. Although there was no significant difference between groups at the end of the 4th week (P-value = 0.306), groups 2 and 3 had a significantly lower frequency of headaches compared to the baseline. Patients in group 2 who received lidocaine injection experienced a significant average decrease of 5.81 attacks per month compared with the baseline (95% CI of the difference = −2.52–−9.09) (Figure 4). In group 3, where patients received a combination of lidocaine and triamcinolone, compared to the baseline, the number of attacks dropped significantly by an average of 5.69 attacks per month (95% CI of the difference = −1.11–−10.27) (Figure 4). Despite a downward trend in the number of attacks in group 1 (P-value = 0.266) and group 4 (P-value = 0.085), the reduction was deemed statistically insignificant (Figure 4).
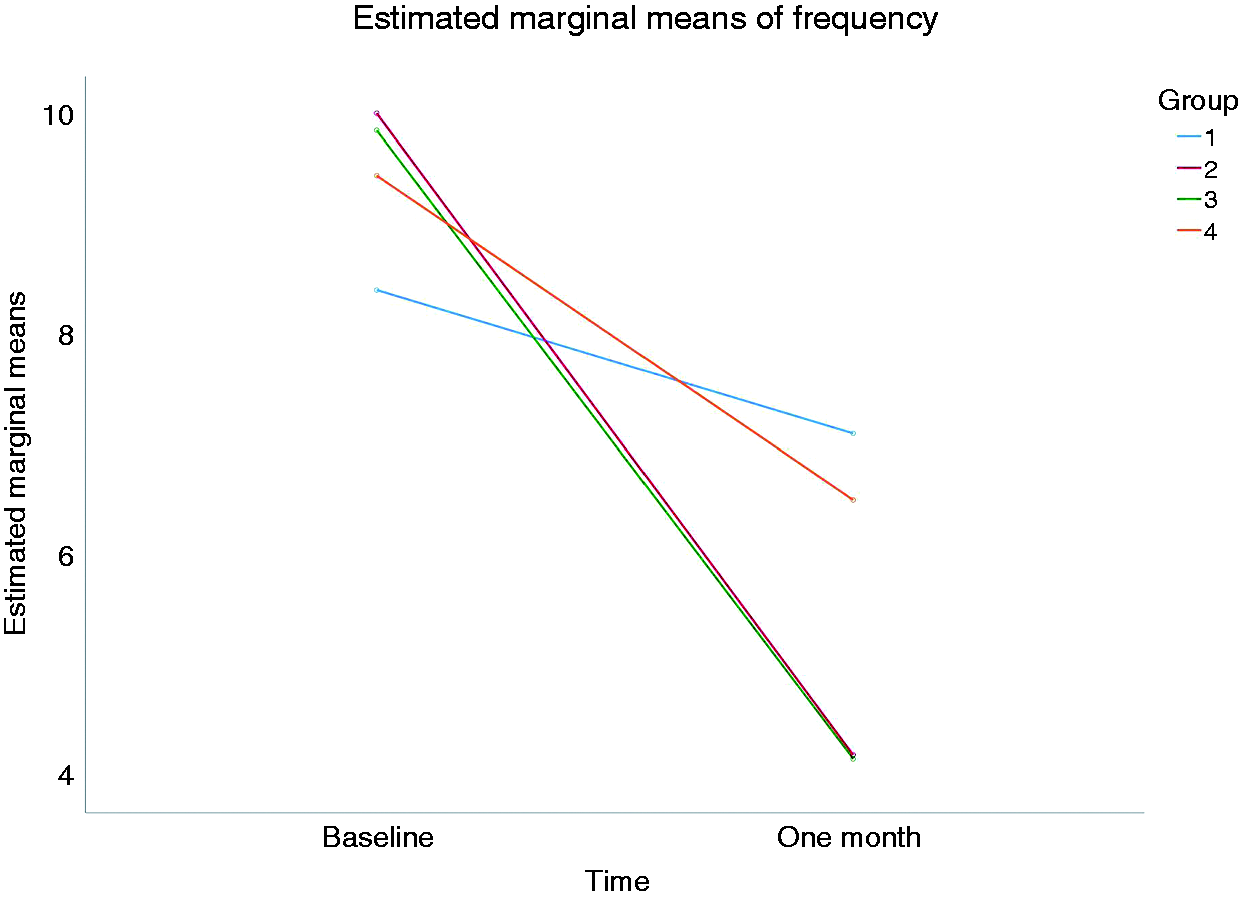
Profile plot of headache frequency by time where each line represents the changes of a group within time.
Discussion
Although there are several randomized placebo-controlled studies on this subject, the main focus of the previous studies has been on patients with chronic migraine, and the data on patients with episodic migraine is scarce (21). Apart from this, to evaluate the efficacy of GON block, patients should avoid the use of new prophylactic medication, which makes it difficult to conduct studies with a longer duration of follow-up. Unlike other available studies, which comprised only two groups, this study had four groups, including a group with just triamcinolone as the intervention. The results of this study demonstrate that there is a significant improvement in the mean severity and duration of headaches in all four groups through time, with no injection being superior to the placebo. Within-subject changes during the time show a sustained effect of GON block on the severity and duration of the headaches beyond the duration of the effect of each of the agents in the solution (7,22). These findings are in keeping with previous studies, which found GON block to have a duration of effect for as long as four weeks (15). Furthermore, the number of the attacks in the four-week period following the intervention was significantly lower compared to the number of headaches in the four-week period preceding the intervention in groups that had lidocaine in their injections.
In a systematic review published in 2019, Shauly et al. (23) pooled the data of nine placebo-controlled studies on GON block from 2006 to 2018 with a total number of 440 patients. The meta-analysis showed that GON block significantly decreased headache days per month (95% CI = −1.39 to −5.81 days) and VAS (95% CI = −1.56 to −2.84) (23). When considering the randomized clinical trials, there were some differences regarding timing of injection, duration of follow-up, and the volume of injection. Each of these could potentially influence the results. We evaluated the effect of a single injection session, while some studies had weekly (24) or monthly (25) injections with follow-ups as long as six months (13). This difference in the method and timing of intervention is notable in the context of clinical practice, especially when patients are to refrain from prophylactic treatment; thus, we considered a single injection with shorter follow-up. The volume of injection also varies between studies, ranging from 1.5 ml (26) of total injection volume to 3 ml (21) in studies that we reviewed. To bring it into the context of our study, one of the hypotheses that we find plausible for the observed results from this study is that the compression itself plays an important role in the observed effects; hence the greater the volume of injection, the greater the effect of compression and the better the outcomes. This hypothesis is in concordance with clinical and functional connections of upper cervical afferents and trigeminal nuclei known as trigeminal-cervical convergence (9–11) in the medulla and can explain the lack of significant between-group difference in our study while a positive effect is seen among all of the groups. Similar findings have been previously reported. In a study published in 2017, Gul et al. (24) compared the efficacy of weekly GON block using 1.5 ml of Bupivacaine + 1 ml of saline with 2.5 ml of saline as a placebo. There was no significant difference between placebo and GON block at the end of the first month. However, both groups experienced a significant reduction of headache days per month (24). These findings raise the possibility that at least some degree of the improvement experienced after GON block could be attributed to the injection itself rather than the medication that is used. Nevertheless, the role of the placebo effect in the treatment of migraine has been previously raised in the literature, and it is believed that the more invasive the placebo (e.g., injection), the more the placebo effect it might exert (27). Even so, these findings are insufficient to draw such a conclusion with an acceptable degree of certainty, and there is a need for further studies where the placebo group receives no amount of injection and needling is done in order to maintain the blinding. The Trigeminocervical complex is also of importance in explaining the role of local anesthetic in reducing the frequency of migraine attacks as this complex acts as a bridge between peripheral nociceptive stimuli and the intracranial nociceptive receptors (28). Lidocaine's effect in blocking voltage-gated sodium channels has been well established in previous studies; hence, it is a plausible assumption that by suppressing peripheral triggers, lidocaine decreases the number of migraine attacks (29,30).
There is one issue regarding the blinding when some groups do not receive anesthetics as a part of their intervention. One might argue that given the hypoesthesia expected after the injection of the local anesthetic, the patients in the intervention group would not be properly blinded to the treatment. In one of the studies, in order to overcome this issue, a small amount of lidocaine (0.25 ml of 1% solution) was mixed with 2.75 ml of saline solution to mimic the hypoesthesia; this study was unable to find any difference between the intervention (2.5 ml of 0.5% Bupivacaine solution + 0.5 ml of methylprednisolone) and the placebo in terms of headache duration and severity. The authors of this article believed the lack of difference to be partly due to the small amount of anesthetic in the placebo group (21). Given this and the lack of significant documented difference between active and placebo groups in terms of hypoesthesia in other studies (23), we did not believe that the use of local anesthetic in all groups was necessary to maintain the blinding.
The use of corticosteroids as a part of the regiment is also debated. In this study, we had to stop recruiting patients in groups receiving triamcinolone - alone or in combination with local anesthetic - following adverse effects, including cutaneous atrophy and alopecia, that were reported in these patients. Although there has been no report of such adverse events in the previous randomized clinical trials, there have been case reports of alopecia and skin atrophy in the use of local corticosteroid injection in combination with local anesthetic in GON block. These adverse effects have been previously reported both in the use of triamcinolone and methylprednisolone (31,32). Studies on the side effects of the local corticosteroid injection in other sites of the body have found these side effects to be reversible, but there has been no study on the duration and reversibility of these side effects following GON block (33). Corticosteroids have been added to the GON block solution due to the understanding that the added benefit weighed more than the local reversible side effects. However, in our study, there was no difference between triamcinolone combination with lidocaine and lidocaine alone. This has been previously studied in patients with chronic migraine, and no significant difference was observed in those studies with addition of 20 mg of methylprednisolone or 40 mg of triamcinolone as well (16,34). However, this lack of significance should be considered with caution as the sample size of these groups was smaller in our study, and the dose of triamcinolone was relatively low (20 mg). Considering adverse events associated with local triamcinolone injection, until further studies confirm the efficacy of local injection of corticosteroids in GON block and a preferred agent and dose with least complication is established, their routine use is rather discouraged.
Limits of the study
We intended to conduct the study with 80 patients, but due to the COVID-19 pandemic, considering the patient and staff safety, we decided to stop recruiting patients. In addition to that, given the observed adverse events with a possible link to local corticosteroids, we had stopped recruiting patients in the groups receiving triamcinolone as a part of their injection. Since the desired sample size was not met in each group, the study is prone to being underpowered in finding between-group differences. Nevertheless, one must consider that despite the smaller sample size, all of the groups experienced significant improvements over time.
As we mentioned earlier, there are some challenges that limit the follow-up time. Besides, we included patients whose preventive medications have not been changed in the month preceding the registration, but guidelines recommend a three-month period of stable preventive treatment (35). However, given the even distribution of patients on preventive medication between all groups and the fact that none of the patients registered in the study had a change in their preventive medication in the two months leading to the study, the confounding effect of preventive medication would be attenuated. Nevertheless, one must bear in mind this possible overlap.
The issue we raised in regards to the hypoesthesia experienced over the area of GON following the infiltration with a local anesthetic still persists and must be considered as a possible confounder to the blinding.
We mainly evaluated the effect of GON block on the headache, but the amount of the medication used during attacks was not recorded and analyzed. This issue should be addressed in future studies. Furthermore, we did not study the effect of GON block on other symptoms, including nausea and vomiting, photophobia, and phonophobia. It would be logical if future studies take these symptoms into account as well since the data around this issue is less developed.
While we used a 20 mg triamcinolone solution as our corticosteroid agent, equivalent to 20 mg of methylprednisolone, some studies used higher doses (16). Considering the use of a different corticosteroid like methylprednisolone with a higher dose (40-80 mg) in future studies might yield a significant effect and lower complication. Nevertheless, until further studies could provide us with more concrete evidence, caution must be taken in using adjunctive local corticosteroids.
Conclusion
Our study concludes that all of the four types of injections used were effective in reducing the severity and the duration of headaches in episodic migraines, and no block solution was superior to the 0.9% saline solution as a placebo at any of the time points. Further studies are needed to explore whether these results were caused by the compressive effect of injected solution or by the placebo effect. On the other hand, there was a significant decrease in the number of headaches in patients receiving lidocaine alone or in combination with triamcinolone as opposed to 0.9% saline injection or triamcinolone. Overall, the findings of this study support use of lidocaine as an agent for greater occipital nerve block should one be considered.
Clinical implications
GON block with a local anesthetic significantly reduces the number of headaches per month in patients with episodic migraine without aura. GON block, regardless of the type of injection medication, significantly reduces the severity and duration of headaches for at least two weeks. With current data at hand, the use of local corticosteroids for GON is not recommended.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211058182 - Supplemental material for Preventive effect of greater occipital nerve block on patients with episodic migraine: A randomized double‐blind placebo‐controlled clinical trial
Supplemental material, sj-pdf-1-cep-10.1177_03331024211058182 for Preventive effect of greater occipital nerve block on patients with episodic migraine: A randomized double‐blind placebo‐controlled clinical trial by Nazila Malekian, Pouya B Bastani, Shahram Oveisgharan, Ghaemeh Nabaei and Siamak Abdi: on behalf of the RELEASE investigators in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024211058182 - Supplemental material for Preventive effect of greater occipital nerve block on patients with episodic migraine: A randomized double‐blind placebo‐controlled clinical trial
Supplemental material, sj-pdf-2-cep-10.1177_03331024211058182 for Preventive effect of greater occipital nerve block on patients with episodic migraine: A randomized double‐blind placebo‐controlled clinical trial by Nazila Malekian, Pouya B Bastani, Shahram Oveisgharan, Ghaemeh Nabaei and Siamak Abdi: on behalf of the RELEASE investigators in Cephalalgia
Footnotes
Data availability
All the data pertaining to the published work would be made available upon reasonable request from the corresponding author.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study did not receive any funding from sources outside Tehran University of medical sciences (TUMS). Iranian center of neurological research (ICNR) in affiliation with TUMS provided the funding for the equipment and medications used in this research, and the compensation of non-medical staff.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
