Abstract
Objective
To compare the efficacy and tolerability of combination treatment of topiramate and greater occipital nerve block to topiramate monotherapy in adult chronic migraine patients.
Background
Options for the preventive treatment of chronic migraine are limited and costly. Combination treatments do not have an evidence base yet.
Methods
This was a parallel group, 3 arms with 1:1:1 allocation ratio randomized controlled study in consecutive adult chronic migraine patients attending Headache Clinic in a tertiary care hospital. Patients received either topiramate monotherapy 100 mg/day (group A), or topiramate plus greater occipital nerve block with 40 mg lidocaine (2%) and 80mg (2 ml) methylprednisolone as the first injection followed by monthly injections of lidocaine for the next 2 months (group B) or topiramate plus greater occipital nerve block with 40 mg lidocaine (2%) injections monthly for 3 months (group C). The primary endpoint was the mean change in monthly migraine days at Month 3. Multiple secondary endpoints were assessed that included among others, achievement of ≥50% reduction in mean monthly headache days compared to baseline at Month 3 and assessment for any adverse events.
Results
One hundred and twenty-five patients were randomized; 41 to group A, 44 to group B, and 40 to group C. Efficacy assessments were done for 121 patients. Patients receiving combination treatment of topiramate and greater occipital nerve block with steroids and lidocaine and greater occipital nerve block with only lidocaine compared to topiramate monotherapy showed greater reductions in monthly migraine days at Month 3 (−9.6 vs −7.3 days; p = 0.003) and (−10.1 vs −7.3 days; p < 0.001) respectively. Greater proportion of patients in both the combination treatment groups (added greater occipital nerve block with and without steroid) achieved ≥50% reduction in mean monthly headache days [71.4% vs 39%; OR (95% CI) 3.9(1.6–9.8); p = 0.004] and [62.4% vs 39%; OR (95% CI) 2.7(1.1–6.7); p = 0.034] respectively, compared to those receiving topiramate monotherapy. Adverse effects between the groups were comparable although patients receiving combination treatment with added greater occipital nerve block reported transient adverse effects like post-injection dizziness, local site swelling, and pain. No serious adverse event was reported.
Conclusion
Combination treatments of topiramate with monthly injections of greater occipital nerve block were more effective in reducing monthly migraine days in chronic migraine than topiramate monotherapy at Month 3. Combination treatments were well tolerated.
Introduction
Chronic migraine (CM) is a severe, highly disabling chronic neurological disorder with a 1-year prevalence of 1.4% to 2% in the general population (1). International Classification of Headache Disorders-3rd edition (ICHD-3) defines CM as headache occurring on ≥15 days/month for more than 3 months, having features of migraine headache on at least 8 days/month (2). Many patients with CM also have a secondary headache in the form of medication overuse headache (MOH) because of frequent use of analgesics, ergots, triptans, and opioids (3). ICHD-3 allows concurrent diagnosis of MOH with CM (2).
Treatment of CM is difficult and becomes even more challenging in presence of co-existent MOH (4). A mixed approach using withdrawal of the offending medication for MOH, careful use of acute medications, optimization of preventive medications, and non-pharmacological interventions have been advocated (3–5). Currently only a few drugs, namely oral topiramate, onabotulinum toxin A (OBT-A) injections, and monoclonal antibodies (mAbs) against calcitonin gene-related peptides (CGRP) receptor or ligands have evidence bases for the preventive therapy of CM (4,5). However, topiramate needs slow titration and has multiple adverse effects which often lead to poor compliance. In the two pivotal trials of topiramate for CM, adverse effects led to treatment withdrawals in 10.9% and 18.8% of patients respectively (6,7). Many CM patients do not respond to topiramate alone and need additional preventive drugs (8). However, an evidence-based combination treatment is not yet available. OBT-A and mAbs against CGRP although efficacious, are costly and beyond the reach of most of the patients in middle to low-income countries. Thus, treatment options for CM are limited.
Greater occipital nerve block (GONB) using a local anesthetic (LA) such as lidocaine or bupivacaine has been investigated as a treatment option for CM and few RCTs have reported good short-term efficacy (9,10). Its long-term efficacy, however, lacks robust evidence (11). It is also uncertain whether the addition of steroids augments the efficacy of GONB for CM patients (11). GONB has been well-tolerated and only minor adverse events have been reported in the RCTs (12). Additionally, GONB is cheap, requires little training for administration, and has minimal potential for the drug-to-drug interactions (12). Therefore, combining GONB to topiramate treatment for CM seems to be a promising option. In particular, the efficacy of GONB in rapidly decreasing headache severity (13) may be very useful for providing an interval time for topiramate to act and facilitating easy withdrawal of the offending medication in MOH.
We therefore, aimed to assess the efficacy and tolerability of monthly injections of GONB using two regimens (lidocaine with and without methylprednisolone) added to topiramate for the prevention of CM and compared them to topiramate monotherapy. Our hypothesis was that the combination treatments will provide greater reduction in migraine and headache days compared to monotherapy.
Materials and methods
Standard protocol approvals and patient consents
The final study protocol, informed patient consent forms, and accompanying materials were reviewed and approved by the institutional ethics committee of Maulana Azad Medical College and associated G B Pant Institute of Post Graduate Medical Education and Research (GIPMER), New Delhi, India. Informed written consent for participation in the study was taken from all participants. The study was conducted as per the International Council for Harmonisation Good Clinical Practice regulations and guidelines. The study follows the tenants of the Declaration of Helsinki. This was an academic study conducted as a part of a dissertation for grant of post-doctoral degree in neurology under University of Delhi, India.
Patients
This was a parallel group randomized controlled study with 3 arms and 1:1:1 allocation ratio. The patients were recruited from the Headache Clinic GIPMER, New Delhi, India. Consecutive patients aged 18 years or above who fulfilled the diagnostic criteria laid down by ICHD-3 for CM with or without MOH (2) were screened for four weeks by a neurology resident (AM). All other primary or secondary headache patients were excluded. Pregnant women, patients with known allergies against lidocaine or topiramate, history of glaucoma or renal stones, history of cervical or cranial surgery, patients with any bleeding diathesis, any local site infection, and with any cardiac disease were also excluded from the study.
Trial profile
The trial profile is shown in Figure 1. All patients were assessed as per a structured proforma. Demographics (age, sex, education level, and occupation), duration of the attack, frequency, location, laterality, character, premonitory symptoms, aura, and postdrome features were recorded. During the screening period, patients were required to fill a paper headache diary, recording the days with or without headache, the days with or without migraine, visual analog scale (VAS) score for each day of migraine or headache, and the days with or without acute migraine treatment (AMT) (analgesics, triptans) from which baseline averages of these measures were calculated. Mean headache impact test-6 (HIT-6) scores and migraine disability assessment scale (MIDAS) scores were also generated at the end of the baseline period. Only acute treatments were allowed during the screening period.

Flow of the study.
Randomization and intervention
After excluding the screen failures, the patients were randomly allocated to groups A, B, and C based on a computer-generated simple randomization chart by a neurology resident (DD) using sequentially numbered opaque sealed envelopes attached to the patient's proformas. All the three groups received oral topiramate which was up titrated from 25 mg per week slowly over four weeks to the maximum dose of 100 mg (50 mg twice a day). Group A received only oral topiramate. Group B received oral topiramate and GONB with 40 mg (2 ml) 2% lidocaine plus 80 mg (2 ml) methylprednisolone as the first injection followed by monthly injections of 40 mg (2 ml) 2% lidocaine alone for the next 2 months. Group C received oral topiramate and GONB with 40 mg (2 ml) 2% lidocaine plus 2 ml saline as the first injection followed by monthly injections of 40 mg (2 ml) 2% lidocaine alone for the next 2 months. GONB injection was prepared by DD and administered by the neurology consultant (DC) who was blinded to the composition of the injections. The follow-up evaluation was done by another neurology resident (AM) who was blinded to the intervention given to the patients in groups A, B and C.
Efficacy endpoints
The primary efficacy endpoint was the mean change in monthly migraine days (MMD); migraine day was defined as a calendar day when the patient reported ≥4 continuous hours of headache meeting ICHD-3 criteria for migraine) at the end of Month 3 compared to baseline. The key secondary efficacy endpoint was proportions of patients achieving more than 50% reduction in monthly headache days (MHD) compared to baseline at Month 3. Other secondary efficacy endpoints were mean change in MHD (headache day was defined as a calendar day [00:00 to 23:59] when the patient reported ≥4 continuous hours of headache episode in the diary), mean migraine-free and headache-free days at Month 3, mean change in monthly headache severity scores (by VAS), mean change in monthly acute migraine treatment (AMT) days, mean change in monthly headache impact scores (by HIT-6) and mean change in MIDAS scores compared to baseline at Month 3.
Safety evaluations
These included the documentation and reporting of treatment-emergent adverse events (TEAE) including serious adverse events (SAE).
GONB Injection Protocol
GONB was administered using a standardized protocol (14). Patients were asked to sit on the chair in a comfortable room and the entire procedure was explained to the patient. Patients were made comfortable and relaxed before being given the GONB. The site of injection was marked (i.e. two-thirds of the distance on a line drawn from the center of the mastoid to the external occipital protuberance on both sides). These points were tested for conspicuous pain sensitivity to pressure. The injection was given on both left and right sides. The patient was positioned properly by slight neck flexion. The injection site was cleaned with spirit to give GONB under aseptic precaution. Fanning technique was used and one-third of the injection was injected in that area marked, one-third slightly medially and one-third slightly laterally. 26 gauge needles were used for injection. The patients were tested for analgesic effect at the site of GONB injection after 20 minutes following each GONB injection. They were observed for at least 30 minutes for any immediate adverse events. The assessment was done at the end of each month after the first GONB injection. The final assessment of efficacy including primary and secondary endpoints was carried out at the end of Month 3.
Statistical analysis
Sample Size Calculation
The study compared oral topiramate monotherapy for 3 months versus combination treatment of topiramate with three monthly GONB injections. The study made two comparisons of effectiveness. First was oral topiramate monotherapy versus combination treatment of topiramate with GONB using LA and steroid in the first injection. The second was oral topiramate treatment versus combination treatment of topiramate with GONB using LA and saline in the first injection. The key outcome variable was the mean change in MMD at the end of Month 3 compared to baseline. Based on the estimates provided by a previous randomized controlled trial (RCT) comparing topiramate with a placebo (7) (baseline mean MMD of 15.5 ± 4.6 days and reduction in mean MMD with topiramate by 3.5 days) and expected reduction in mean MMD migraine days with additional GONB (without steroids) of 7 days and GONB (with steroids) of 8 days, and 10% dropout, the sample size was estimated to be 40 patients in each of the three groups assuming an alpha error of 5%, and a beta error of 20% (power of 80%).
Demographic characteristics were summarized using descriptive statistics. Categorical data were summarized as frequencies and percentages. Continuous data were summarized as means. The primary and secondary efficacy endpoints (except proportion of patients achieving >50% reduction in MHD) were analyzed using a linear model with baseline value as a covariate and treatment as a factor for all randomized patients who received ≥1 dose of randomized treatment and completed at least one post-baseline monthly headache diary measurement. The efficacy analyses were done on a modified intent-to-treat basis, with patients analyzed according to the assigned treatment group and the missing data were imputed using the last observation carried forward (LOCF) method. Based on observations per each month data during the study period of three months, pair-wise comparisons between group B versus group A and group C versus group A were obtained. A post-hoc Bonferroni adjustment was done to control for Type I errors for multiple efficacy assessments. The Cochran-Mantel-Haenszel pairwise test was used to assess treatment differences in the proportion of patients between the groups who achieved >50% reduction in headache days at Month 3 compared to baseline along with the estimation of odds ratio (OR) and 95% confidence interval (CI). A post-hoc exploratory analysis of the efficacy of GONB in CM patients with and without MOH at Month 3 compared to baseline using the same statistical methods as for the assessment of primary and secondary efficacy endpoints was performed in patients receiving either of the two GONB regimens. Statistical analysis was done with the SPSS version 25.0 software package. The level of significance was set at p < 0.05.
Results
Demography and baseline headache variables. Between January 2018 to May 2019, 180 consecutive chronic migraine patients were assessed for eligibility. Of these, 55 were screen failures and 125 were randomized (41 for group A, 44 for group B, and 40 for group C) (Figure 1). Eight patients were lost to follow up at the end of Month 3 (5 in group B and 3 in group C). Of these, 1 patient in group C refused further continuation in the study immediately following randomization and did not receive any treatment, 2 patients in group B and 1 patient in group C were lost to follow-up within 1 month. These 4 patients were excluded from the efficacy analysis. For 3 patients from group B and 1 patient from group C who were lost to follow up after 1 month, the missing data were imputed using the data available at one month. The majority of the study population consisted of female patients (11 males and 114 females). The mean age at presentation and duration of migraine was 32.9 ± 9.7 years and 67.5 ± 59.2 months respectively. The mean duration of CM was 4.4 ± 1.3 months. Ninety-five patients also had co-existent MOH. Mean MMD and MHD at baseline were 14.1 ± 5.1, and 23.5 ± 5.3 respectively. The average monthly days of AMT use was 12.8 ± 5.4. Mean VAS, HIT-6, and MIDAS scores at baseline were 7.8 ± 0.8, 64.9 ± 3.6, and 64.2 ± 12.6 respectively. The three treatment groups were well balanced in terms of demographics, headache profiles, and baseline characteristics (Table 1).
Baseline demographic and clinical characteristics.

GONB: greater occipital nerve block; SD: standard deviation; VAS: visual analog scale; HIT-6: headache impact test-6; MIDAS: migraine disability assessment scale.
Primary endpoint. Absolute mean MMD changed from 14.1 days at baseline to 6.7 days at Month 3 in the topiramate monotherapy group as compared to 12.9 days to 4 days in patients who received combination treatment of topiramate and GONB with steroids and lidocaine and 15 days to 4.2 days in patients who received combination treatment of topiramate and GONB with lidocaine only (Figure 2). The adjusted mean changes in MMD at Month 3 were greater in patients who received combination treatment of topiramate and GONB with steroids and lidocaine [−9.6 vs −7.3 days; mean difference (95% CI) −2.3 days (0.7–4.1); p = 0.003] and GONB with only lidocaine [−10.1 vs −7.3 days; mean difference (95% CI) −2.8 days (1.1–4.5); p < 0.001] compared to patients who received topiramate monotherapy (Figure 3). These differences were also statistically significant at Month 1 [−7.0 vs −4.2 days; mean difference (95% CI) −2.8 days (0.4–5.2); p = 0.014] and Month 2 [−9.1 vs −6.4 days; mean difference (95% CI) −2.7 days (0.8–4.7); p = 0.003] for the group receiving combination treatment of topiramate and GONB with steroids and lidocaine. For the group receiving combination treatment of topiramate and GONB with only lidocaine, the differences were significant at Month 1 [−7.6 vs −4.2 days; mean difference (95% CI) −3.4 days (0.9–5.8); p = 0.003] but was non-significant (though numerically superior) at Month 2 [−8.3 vs −6.4 days; mean difference (95% CI) −1.9 days (−0.1–3.9); p = 0.068].

Absolute (nonadjusted) mean monthly migraine days at baseline and at months 1 to 3 by treatment and visit (modified intention-to-treat analysis set). Error bars represent the standard error.

Change from baseline in mean monthly migraine days (primary efficacy endpoint) by treatment and visit (modified intention-to-treat analysis set) analyzed using a linear model with baseline value as a covariate and treatment as a factor. Error bars represent the standard error. p < 0.05 was considered as statistically significant. CI, confidence interval; MMD, monthly migraine days.
Key secondary endpoint. At the end of the Month 3, 30/42 (71.4%) patients who received combination treatment of topiramate and GONB with steroids and lidocaine (OR:3.9; 95% CI:1.6–9.8; p = 0.004) and 24/38(62.4%) patients who received combination treatment of topiramate and GONB with only lidocaine (OR: 2.7; 95% CI: 1.1–6.7; p = 0.034) achieved ≥50% reduction from baseline in MHD at Month 3 compared to 16/41 (39%) patients receiving topiramate monotherapy (Figure 4).
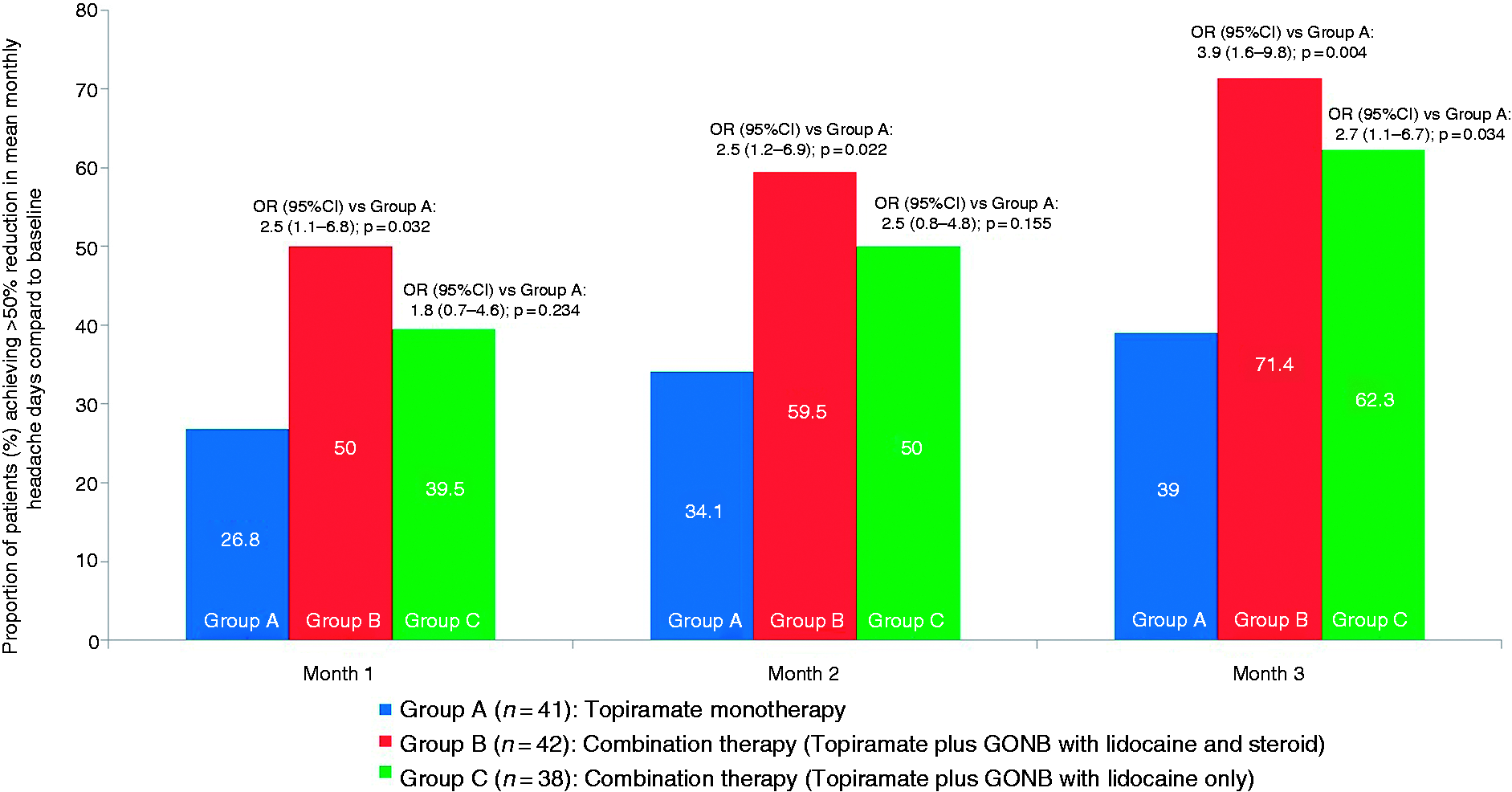
Proportion of patients with: ≥50% reduction from baseline in monthly as visit headache days by treatment and visit (modified intention-to-treat analysis set). Cochran-Mantel-Haenszel pairwise test was used to assess treatment differences. p < 0.05 was considered as statistically significant. CI, confidence interval; OR, odds ratio.
Other secondary endpoints. Patients receiving combination treatment of topiramate and GONB with steroids and lidocaine had a superior outcome in all other secondary endpoints except AMT days at Month 3 compared to patients who received topiramate monotherapy (Table 2). However, patients receiving combination treatment of topiramate and GONB with only lidocaine achieved statistically superior outcomes only in mean change in monthly migraine-free days. Both the GONB groups showed a greater mean change in monthly headache impact scores (by HIT-6) and mean change in MIDAS scores compared to baseline at Month 3 (Table 2). In a head-to-head comparison between the two groups of GONB (with and without steroids), no difference was observed in any of the primary or secondary efficacy endpoints (Supplementary Table 1)
Secondary efficacy endpoints at Month 3.

Adjusted means were compared using a linear model with baseline value as a covariate and treatment as factor. The Cochran-Mantel-Haenszel pairwise test was used to compare treatment differences between the proportions of patients who achieved ≥50% reduction in mean monthly headache days compared to baseline. OR: odds ratio; CI: confidence interval; GONB: greater occipital nerve block; SE: standard error; VAS: visual analog scale; AMT: acute migraine treatment; HIT-6: headache impact test-6; MIDAS: migraine disability assessment scale.
Post-hoc analysis of the efficacy of GONB in migraine patients with and without MOH. Among 84 patients who received either of the two regimens of GONB, 80 patients were analyzed. Of these, 58 had MOH, and 22 did not have MOH. The two groups were balanced in terms of age, sex, and baseline headache variables except those with MOH had worse baseline MIDAS scores (Supplementary Table 2). CM patients with MOH compared to those without MOH did not differ significantly in terms of mean change in MMD (−10.1 ± 0.41 vs −9.0 ± 0.67; p = 0.162) but a greater proportion of them achieved ≥50% decrease in MHD [71.4% vs 50%; p = 0.043; OR (95% CI): 2.9(1.0–8.0)] at Month 3. Other secondary efficacy endpoints did not differ between the two groups (Table 3).
Efficacy of GONB in CM patients with and without MOH at Month 3 compared to baseline.
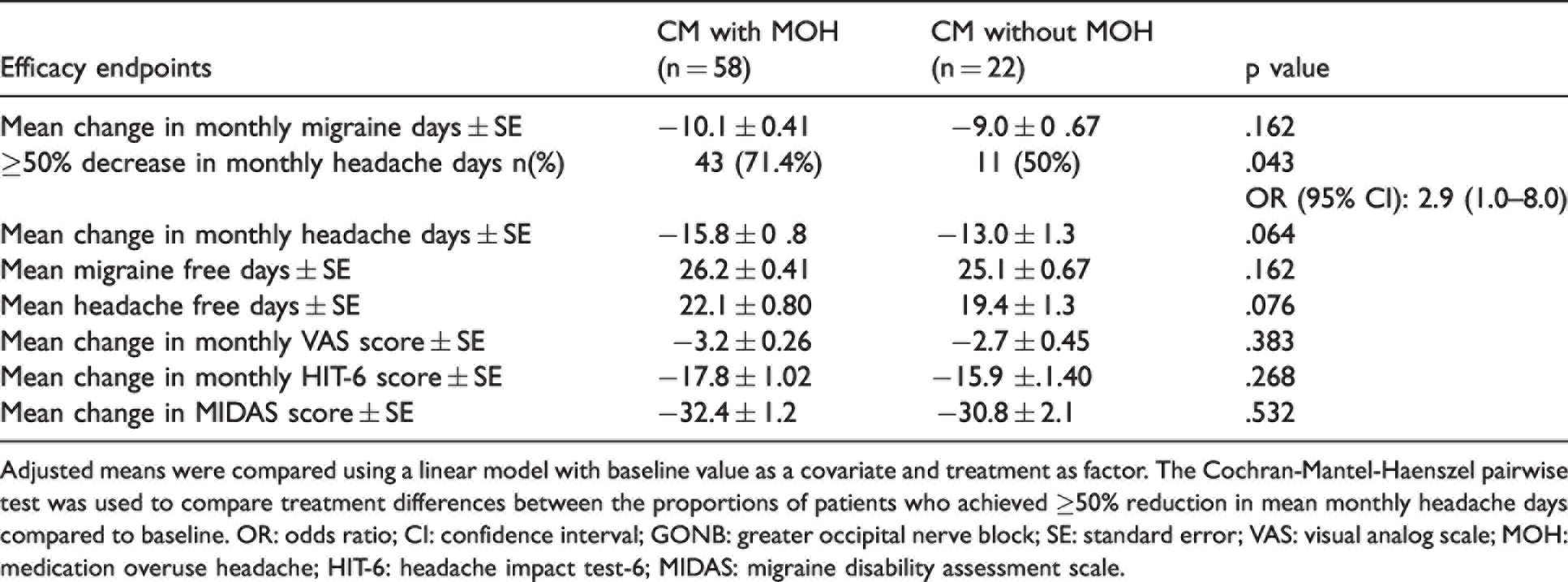
Adjusted means were compared using a linear model with baseline value as a covariate and treatment as factor. The Cochran-Mantel-Haenszel pairwise test was used to compare treatment differences between the proportions of patients who achieved ≥50% reduction in mean monthly headache days compared to baseline. OR: odds ratio; CI: confidence interval; GONB: greater occipital nerve block; SE: standard error; VAS: visual analog scale; MOH: medication overuse headache; HIT-6: headache impact test-6; MIDAS: migraine disability assessment scale.
Tolerability. All the treatment arms were well tolerated and except for mild TEAE reported by patients in all 3 groups, no SAE was noted during the study. In all, 15 types of TEAE were noted during the study. Paresthesias in limbs were reported by 29.3%, 22.7%, and 17.5% of patients in groups A, B, and C respectively. However, 5 patients (11.4%) in group B and 2 patients (5.3%) in group C were lost to follow-up as compared to none in group A.
TEAE specifically reported after GONB injection. Local site swelling and bleeding and dizziness were the three most common complaints in patients who received GONB. Dizziness and vasovagal syncope occurred only during the first injection. 7 patients had dizziness, 3 had vasovagal syncope and 3 had minimal local site bleeding in group B whereas 3 patients had dizziness, 1 had vasovagal syncope and 5 had minimal local site bleeding in group C. Additionally, local site swelling that remained for 3-4 days was complained of by 11 patients in group B and 8 patients in group C. Mild local site alopecia occurred in 6 patients in group B (observed during 2nd/3rd month) and none in group C. The frequency of various TEAE is presented in Table 4.
Treatment emergent adverse events.

GONB: greater occipital nerve block.
Discussion
This study showed that the combination treatment of topiramate and GONB was superior to topiramate monotherapy for the prevention of CM at Month 3. The combination treatments using two GONB regimens with topiramate achieved statistically significantly greater reductions of 2.3 and 2.8 mean MMD (primary efficacy endpoint) as compared to topiramate monotherapy respectively. Further, a greater reduction in MMD in both the combination treatment groups was evident after 1 month and the trend continued until 3 months, thereby suggesting that the beneficial effect of the combination treatment accrued over time and remained consistent. In terms of the key secondary outcome, a greater proportion of patients in both the combination treatment groups (using the two GONB regimens) achieved ≥50% decrease from baseline in MHD compared to topiramate monotherapy at Month 3.
Currently, CM patients have limited evidence-based treatment options for preventive treatment (4). OBT-A and mAbs against CGRP receptors or ligands have a good evidence base (15,16) and have been approved by FDA for CM. However, these are costly treatment options. Both these treatments are recommended for patients who have failed two to three oral migraine preventives drugs (17,18). Amongst the oral preventives, only topiramate has shown to be effective in CM based on two RCTs (6,7). However, many migraine patients on topiramate become non-compliant due to TEAE such as paresthesias, cognitive dysfunctions, and fatigue (19). Contrarily, slow titration of topiramate up to a dose of 100mg/day has been shown to reduce the adverse events and the rate of discontinuation significantly (20). This has been seen in our study also using a similar dose and titration schedule. Further, many CM patients despite receiving an optimal dose of topiramate do not derive adequate benefit and hence require combination treatment with other migraine preventives (8). An added treatment with propranolol to topiramate for CM, however, did not show any additional benefit (8). There are no other RCTs of combination therapy for CM except a small study that found that propranolol alone and its combination with nortriptyline were significantly better than nortriptyline alone in terms of achievement of ≥50% decrease from baseline of headache frequency and reduction in the mean number of headache days per month (21). A recent retrospective study showed additive effects of OBT-A and CGRP mAbs in CM (22). Similar beneficial results have been reported in 10 refractory CM patients with the combination of both erenumab and OBT-A compared to each therapeutic strategy alone (23). Thus, there is emerging evidence that proper combination treatment in CM may provide additional benefits although the cost may become prohibitive. No previous randomized study, to the best of our knowledge, has prospectively assessed the role of the combination of GONB with topiramate in CM. The salutary results of this study may thus fulfil an unmet need for the preventive treatment of CM and provide a new option.
Compared to topiramate monotherapy, patients who received combination treatment of topiramate and GONB with steroid performed significantly better in the four other secondary endpoints namely, mean change in MHD, mean migraine-free and headache-free days, and mean change in headache severity at Month 3 whereas the group with topiramate and GONB with only lidocaine was superior to topiramate monotherapy for mean migraine-free days at Month 3. The addition of steroids to lidocaine for the first injection might have produced these differences in efficacy endpoints between the two groups. A head-to-head comparison between the two regimens of GONB (with and without steroids) did not show any differences. However, the study was not powered to detect the differences between these two groups. Corticosteroids are postulated to reversibly block nociceptive C fibers in substantia gelatinosa, reduce dynamic mechanical allodynia, and prolong the effects of LA (24–26). Two small RCTs of single GONB using steroids with LA for CM patients were negative (27,28) but many open-label studies have shown good results (11). Similarly, GONB with corticosteroids has been found to be useful as a transitional preventive in cluster headaches (29,30). We injected LA with steroids only during the first injection because recurrent GONB with corticosteroids can potentially cause local and systemic adverse effects (31,32). Except for mild reversible alopecia seen in few patients which had been reported in previous studies as well (33), patients in the steroid group did not show any major TEAE in our study.
Interestingly, mean change in monthly AMT days did not differ between monotherapy and combination treatment groups. This apparent discrepancy between reduction in MMD and AMT days between the groups may be due to the use of acute pain medications for even low-intensity headaches by the patients as was previously observed in the Phase III REsearch Evaluating Migraine Prophylaxis Therapy (PREEMPT) study of OBT-A for CM (34). Previous RCTs in CM patients comparing topiramate with placebo (6,7) and GONB with placebo (9,10) also could not find a significant reduction in monthly AMT days for the test drugs.
The impact of migraine treatment on quality of life and disability outcomes is gaining importance (35). This study found that patients receiving either of the combination treatment regimens had significantly greater improvements in headache-related impact and disability compared to those receiving topiramate monotherapy as assessed by HIT-6 and MIDAS scores. For HIT-6 scores, the minimally important difference (MID) between the groups has been estimated to be 2.3 points (36). We found a difference of 5.5 and 2.9 points between the combination treatment groups (with and without steroids) and topiramate monotherapy respectively. The corresponding score for MIDAS is not yet available although a change of 5 days of MIDAS score has been considered clinically meaningful (37). We found a difference of 4.8 and 5.1 days between the combination treatment groups (with and without steroids) and topiramate monotherapy respectively. Thus, combination treatments not only yielded statistically significant superior outcomes for headache-related disability and impact, they also showed clinically meaningful differences compared to topiramate monotherapy.
The combination treatments also proved to be well tolerated, although 11.4% and 5.3% of patients dropped out without giving any reason by 3 months compared to none in the topiramate monotherapy group. The reasons for the overall low rate of discontinuation as compared to previous studies of topiramate versus placebo from USA and Europe may be accounted for by the open nature of the study in which the patients knew that they were receiving active drugs for their headaches, were counseled about the possible adverse effects at the time of randomization and at every subsequent visit and had a good response to intervention in all three arms of randomized treatment. At each follow-up visit, their adverse effects were addressed and the patients were encouraged to continue with the treatment. Frank discussions with patients about potential adverse effects of topiramate and making them aware of the same have been noted to improve compliance (38). This leads to the continuation of topiramate by the patients until they derive maximum benefits (38). Possibly because of this, many open labeled studies of topiramate reported lower dropout rates compared to the RCTs. For example, in the studies by Bartolini et al., (39) Yang et al. (40) and Lai et al. (41) the reported dropout rates for migraine patients treated with topiramate were 3/22, 3/33 and 4/31 respectively. Further, the main adverse effect observed in all three arms was paresthesias and less commonly cognitive effects (difficulty in concentration) and generalized body-ache. It has been shown by a previous study that although paresthesias are the most common adverse effects reported with topiramate treatment in migraine patients, these were a less common cause of discontinuation compared to other adverse effects such as cognitive dysfunctions and fatigue (42). GONB did not produce any SAE and the minor TEAE attributable to GONB were short-lasting and self-limiting. The minor TEAE were comparable to previous studies on GONB in CM (11,12).
Mechanisms of action of topiramate and GONB in CM are not fully known. Topiramate is believed to modify several receptor-gated and voltage-sensitive ion channels, including voltage-activated Na+ and Ca2+ channels and non-NMDA receptors resulting in inhibition of neuronal hyper-excitability (43). Lidocaine acts by reversibly blocking sodium channels within unmyelinated C-fibers and myelinated nerve fibers. This causes blockage of depolarization within the nerve, thereby preventing the pain signal transmission (44). The greater occipital nerve derives most of its fibers from the second cervical dorsal root and is the main sensory nerve of the occipital area. The trigeminocervical complex receives the convergence between the trigeminal and upper cervical sensory afferents and is connected to higher pain modulating structures (45–47). It has been shown by a previous study that the effect of a single GONB can last up to a month, far longer than its anesthetic effect (48). Further, the anesthetic effect may not be relevant in terms of its efficacy (9). Rather the effect of GONB has been proposed to be due to a modulatory effect of GONB through trigeminocervical complex to the higher pain pathway (45). In our study, however, of all patients who received GONB, only a minority did not achieve full analgesia at the site of GONB injection after 20 minutes (2, 3 and 1 patient in group B during first, second and the third injections respectively, and 0, 2 and 1 patient in group C, during first, second and the third injections respectively) and this did not impact the overall outcome. The quick and sustained response of repeated GONB (13) not only provides an interim relief but provides a window of time for the preventive actions of topiramate to set in. Despite the uncertainties regarding the mechanisms of actions of these drugs, our study showed that combination treatment of topiramate to GONB performed better than topiramate monotherapy and thus the combination was likely to have an additive effect.
The primary efficacy endpoint did not differ between the CM patients with or without MOH who received GONB treatment. Interestingly, greater proportions of patients of CM with MOH achieved ≥50% reduction in MHD at Month 3 compared to baseline although the rest of the secondary efficacy endpoints did not differ between the two groups. This is consistent with two previous observational studies that found good efficacy of GONB in CM irrespective of co-existent MOH (48,49). Contrarily, a retrospective study reported that symptomatic medication overuse almost tripled the risk of GONB failure in a mixed group of headache patients including refractory migraine (50). Most of the RCTs of GONB in CM did not include MOH patients (12). One RCT however, involving exclusively “triptan overuse headache” patients found a beneficial effect of repeated GONB (51).
An important limitation of our study is that the investigators were not blinded and there was no placebo arm. Therefore investigators were prone to biases for the novel intervention arms. However, we chose not to include a placebo arm (sham GONB injections) because we were interested to know the effect of combination treatment versus monotherapy and not the efficacy of GONB in CM per se. Secondly, post-hoc analysis of the impact of coexistent MOH on GONB resulted in two unequal groups for comparison and hence the results should be viewed with caution. Finally, CM patients in our study had lesser disease duration than previous studies. Therefore, it is uncertain whether CM patients with a longer duration of illness will have a similar response to the interventions. Despite these limitations, the findings of our study provide reasonable evidence for a new efficacious and well-tolerated option for the preventive treatment of CM.
Clinical implications
The combination treatment of three monthly injections of GONB using lidocaine (2ml, 2%) alone or with methylprednisolone (80mg) in the first injection added to oral topiramate (100mg/day) were more effective in reducing mean monthly migraine days and achieving ≥ 50% reduction of monthly headache days at Month 3 than topiramate monotherapy in patients with chronic migraine. Both the added GONB regimens achieved superior outcomes in terms of reduction in headache impact and disability compared to topiramate monotherapy. All the three treatment arms were well tolerated although patients receiving added GONB reported transient adverse effects like post-injection dizziness, local site swelling, and pain. No serious adverse event was reported in any of the three treatment arms.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221082077 - Supplemental material for Efficacy and tolerability of combination treatment of topiramate and greater occipital nerve block versus topiramate monotherapy for the preventive treatment of chronic migraine: A randomized controlled trial
Supplemental material, sj-pdf-1-cep-10.1177_03331024221082077 for Efficacy and tolerability of combination treatment of topiramate and greater occipital nerve block versus topiramate monotherapy for the preventive treatment of chronic migraine: A randomized controlled trial by Debashish Chowdhury, Ankit Mundra, Debabrata Datta, Ashish Duggal, Anand Krishnan and Arun Koul in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024221082077 - Supplemental material for Efficacy and tolerability of combination treatment of topiramate and greater occipital nerve block versus topiramate monotherapy for the preventive treatment of chronic migraine: A randomized controlled trial
Supplemental material, sj-pdf-2-cep-10.1177_03331024221082077 for Efficacy and tolerability of combination treatment of topiramate and greater occipital nerve block versus topiramate monotherapy for the preventive treatment of chronic migraine: A randomized controlled trial by Debashish Chowdhury, Ankit Mundra, Debabrata Datta, Ashish Duggal, Anand Krishnan and Arun Koul in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
