Abstract
Background
Neurovascular coupling (NVC) reflects the interaction between cerebral blood flow (CBF) and functional activity. However, the relationship between NVC and migraine chronification remains unclear. This study investigated the state of NVC in migraine patients and evaluated its potential as an imaging feature for migraine chronification using arterial spin labeling (ASL) combined with resting-state functional magnetic resonance imaging (rs-fMRI).
Methods
This was a cross-sectional study. Thirty-nine episodic migraine (EM), 61 chronic migraine (CM) patients (25 with medication overuse headache, MOH) and 42 healthy controls (HCs) were recruited in the same period. Imaging data were acquired using a 3.0 T MRI. Regional homogeneity (ReHo) represented functional activity, whereas CBF was quantified via ASL. Correlation coefficient between CBF and ReHo values of each participant in voxel level represented the whole-brain NVC status, whereas the CBF/ReHo ratio represented regional NVC status. Correlations between NVC metrics and clinical characteristics were analyzed in CM patients. Exploratory mediation analysis was conducted to identify mediators between NVC alterations and the clinical characteristics of CM patients. Finally, receiver operating characteristic (ROC) curve was generated to evaluate the diagnostic performance of NVC metrics for migraine chronification.
Results
Compared to HCs, both EM and CM patients presented significantly reduced whole-brain CBF-ReHo coupling. Compared to EM patients, CM patients presented a decreased CBF/ReHo ratio in the right precuneus. Correlation analysis revealed that z value of the CBF/ReHo ratio in the right precuneus was negatively correlated with both HIT-6 score and PHQ-9 score; HIT-6 score was positively correlated with PHQ-9 score in CM group. Exploratory mediation analysis indicated that depression mediated the relationship between abnormal NVC and clinical characteristics in CM patients. Finally, ROC curve indicated that the CBF/ReHo ratio in the right precuneus (AUC = 0.75) exhibited high sensitivity and specificity in distinguishing CM from EM patients.
Conclusion
Abnormal NVC in the precuneus was involved in migraine chronification, with depression potentially serving as a mediator in this process. NVC metric may serve as an imaging feature for migraine chronification in the future.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine affects approximately 10–14% of the population worldwide (1,2). Approximately 3% of episodic migraine (EM) progresses to chronic migraine (CM) (3). Previous studies have shown that the chronification of migraine is associated with various factors, including overuse of acute analgesics, ineffective treatment, stress, obesity, gender, and age (4–6). Additionally, CM patients often have comorbidities such as anxiety and depression (7). It is currently believed that frequent headache attacks and risk factors lower the threshold for migraine onset, thereby increasing the risk of chronic migraine (6). While many studies have attempted to address the progress of migraine chronification, the pathophysiological mechanisms underlying migraine chronification remain obscure.
Magnetic resonance imaging (MRI) has widely been used to explore the pathophysiology of migraine chronification. Some studies revealed that CM patients presented many abnormal brain regions at the brain function and networks level, such as the somatosensory cortex, visual cortex, thalamus, caudate nucleus, default mode network (DMN), and the executive control network (ECN), through resting-state functional magnetic resonance imaging (rs-fMRI) (8–11). Moreover, some studies have shown that CM patients present abnormal cerebral blood flow (CBF) in several brain regions such as the primary somatosensory cortex, nucleus accumbens, ventral lateral nuclei of the thalamus, and amygdala, using the arterial spin labeling (ASL) technique (12–15). To some extent, these studies have contributed to our understanding of migraine pathogenesis. The trigeminovascular theory posits that migraine results from activation of trigeminal nerve terminals releasing neuropeptides such as calcitonin gene-related peptide (CGRP) and substance P, which induce dural vasodilation and transmit pain signals to the somatosensory cortex (1,16). Recurrent attacks lead to sensitization of the trigeminovascular system, lowering the pain threshold and contributing to migraine chronification (17,18). Thus, neurovascular dysfunction and interactions play a central role in the pathophysiology of migraine and its chronification.
The normal function of the human brain depends on a sufficient supply of CBF (19). Previous studies have revealed a linear relationship between CBF and brain functional activity, whereby increased CBF was associated with greater functional activities (20,21). This physiological process, known as neurovascular coupling (NVC), plays a crucial role in maintaining normal brain functional activities (22,23). ASL combined with rs-fMRI can reflect the CBF and functional activities of the human brain, as confirmed in previous studies (20,21). This method has been widely applied in various diseases, such as schizophrenia, neuromyelitis optica, primary open angle glaucoma, and cervical spondylotic myelopathy, and has been used to explore the status of NVC under pathological conditions (24,25). Moreover, previous studies have shown that the ratios of CBF to brain functional activity in the parietal and occipital gyrus of CM patients differ from those in healthy controls (HCs), suggesting potential abnormalities in NVC in CM patients (26). However, it has remained unclear whether there is abnormal NVC during migraine chronification.
Based on previous studies, we hypothesized that abnormal NVC occurred during migraine chronification and may serve as a potential imaging feature. Therefore, this study aimed to provide new insights into migraine chronification from the perspective of NVC by using a combination of rs-fMRI and ASL techniques.
Method
Participants
This study included migraine patients who visited Beijing Tiantan Hospital, Capital Medical University, between 2020 and 2023. The study recruited 142 participants, including 42 patients with EM and 61 patients with CM, of whom 25 CM patients also had MOH and 39 healthy HCs. For migraine patients, the following criteria were used: (i) A confirmed diagnosis of EM or CM based on the International Classification of Headache Disorders, 3rd edition (ICHD-3), with all patients experiencing migraine without aura. (ii) Aged between 18 and 65 years. (iii) Right-handed. (iv) No contraindications to MRI scanning. (v) No use of preventive migraine treatments for at least three months prior to recruitment. (vi) Voluntary participation in the study. For HCs, the inclusion criteria are as follows: (i) Aged between 18 and 65 years. (ii) Right-handed. (iii) No contraindications for MRI. (iv) Voluntary participation in the study. The following exclusion criteria were applied to both migraine patients and HCs: (i) Participants with other primary headache types or pain conditions in regions beyond the head. (ii) Individuals who were pregnant or breastfeeding at the time of the study. (iii) Those who were diagnosed with neurological, cardiovascular, or endocrine disorders were excluded from participation. (iv) A history of substance abuse or alcohol use. (v) Participants with a first-degree relative who had a history of headaches. (vi) MRI scans with poor data quality, such as excessive head motion or missing raw data. (vii) Clear signs of brain injury or high white matter signals (Fazekas score ≥ 2, especially around the lateral ventricles). (viii) Patients who experienced headache attacks during MRI scanning.
Demographic data and neuropsychological tests
General clinical data, including age, gender and body mass index (BMI), were collected from all participants. For migraine patients, additional information including migraine history, headache frequency, and pain intensity visual analog scale (VAS) score was collected. In addition, migraine patients were also required to complete the following standardized scale, the Migraine Disability Assessment Scale (MIDAS), Headache Impact Test-6 (HIT-6), Patient Health Questionnaire-9 (PHQ-9), Generalized Anxiety Disorder-7 (GAD-7), Pittsburgh Sleep Quality Index (PSQI). Higher scores on HIT-6, PHQ-9, GAD-7, and PSQI scales reflected greater severity of headache, depression, anxiety, and sleep disturbances, respectively.
Ethical approval for this study was obtained from the Ethics Committee of Beijing Tiantan Hospital, Capital Medical University (Approval No. KY2022-044), and all research procedures adhered to the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all participants prior to their inclusion in the study. Furthermore, this research was part of a registered clinical trial (NCT05334927), and its protocol and procedures were reviewed and approved by the Institutional Review Board of Beijing Tiantan Hospital.
Image acquisition
Please see Online Supplementary Material for more information in image acquisition.
Cerebral blood flow analysis
In this study, the ASL sequence was used to calculate the CBF images for each participant. Arterial transit time (ATT) maps were derived using a signal-weighted delay approach as outlined by Dai et al. (27) For each pair of post-labeling delay (PLD) and labeling duration (LD), the arrival-time-corrected CBF maps were quantified as follows:
Here, δ denotes the ATT, τ the LD, and ω the PLD. T1a represents the longitudinal relaxation time of arterial blood (1.6 s), while
The CBF images were preprocessed via Statistical Parametric Mapping version 12 (SPM12, http:// www.fil.ion.ucl.ac.uk/spm), which specifically includes the following steps: (i) Image quality check: the CBF images of each participant were inspected to ensure the quality of the images. (ii) Image registration: The CBF images of each subject were registered to the positron emission tomography (PET) brain template. (iii) Normalize the registered CBF images to the Montreal Neurological Institute (MNI) standard space. (iv) Image quality check: Quality checks are conducted on the CBF images after spatial standardization to ensure the quality of image registration. (v) Smoothing: The preprocessed CBF image was smoothed, and the Gaussian smoothing kernel size was selected as 4 × 4 × 4 mm³.
Functional MRI data preprocessing
Please see Online Supplementary Material for more information on functional MRI data preprocessing.
Regional homogeneity calculation
Kendall's coefficient of concordance (KCC) was applied to assess the regional homogeneity (ReHo) of each voxel by evaluating its synchronization with the time series of its 26 nearest neighboring voxels on a voxel wise basis within the gray matter mask. The resulting standardized ReHo maps were subsequently smoothed via a Gaussian kernel with a full width at half maximum (FWHM) of 4 × 4 × 4 mm³.
Whole brain gray matter CBF-ReHo coupling analysis
To quantitatively assess the coupling between CBF and ReHo across the whole-brain gray matter, a cross-voxel correlation analysis was performed for all participants in this study (29,30). Therefore, a correlation coefficient between the CBF and ReHo was obtained for each participant.
Analysis of the CBF/ReHo ratio
To quantify the NVC status, the CBF/ReHo ratio was computed for each brain region. To improve data normality, these voxel-wise ratios were standardized into z scores for each participant. Voxel-wise analysis of covariance (ANCOVA) was performed to compare CBF/ReHo ratio across groups. For three group comparisons, age and gender were included as covariates. For the post hoc comparison between EM and CM group, age, gender, PHQ-9, GAD-7 were included as covariates to control for potential emotional confounding.
Correlation analysis between NVC and clinical features in CM group
To explore the relationships between NVC and clinical characteristics in CM patients, the CBF/ReHo ratios from brain regions showing significant abnormalities were extracted and converted into z scores. Pearson's correlation analysis was then performed between these NVC values and the clinical data, with age and gender included as covariates.
Exploratory mediation analysis
Based on the results of the correlation analysis, this study hypothesized that NVC alterations in CM patients may be associated with migraine clinical symptoms. To further explore the potential pathways underlying this association, exploratory mediation analysis was conducted. The bias-corrected bootstrap method was used for estimation, with 5000 resamples used to calculate the 95% confidence interval (CI) for the mediation effect. A mediation effect was considered statistically significant if the CI did not include zero.
Statistical analysis
All statistical tests were two-tailed and performed using SPSS (version 27.0, IBM Corp, Armonk, NY, USA). The normality of continuous variables was assessed via the Shapiro–Wilk test. For normal distributed data, the results were presented as mean ± standard deviation, and statistical comparisons were performed using independent-samples t-tests or one-way analysis of variance (ANOVA). For data that did not meet the normality assumption, values were expressed as medians and interquartile ranges (IQR), and nonparametric tests were used. Specifically, the Mann-Whitney U test was applied for comparisons between two groups, whereas the Kruskal Wallis H test was used for comparisons among three or more groups. Categorical variables were analyzed using the chi-square test.
Multiple comparisons were corrected in imaging analyses using Gaussian random field (GRF) correction with a voxel level threshold of P < 0.001 and a cluster level threshold of P < 0.05. Correlation analyses were corrected using the Bonferroni method.
In addition to significance testing, effect sizes were reported to quantify the magnitude of group differences. Cohen's f² was used for ANOVA and Cohen's d for pairwise comparisons, interpreted according to Cohen: small/medium/large effects at d ≈ 0.2/0.5/0.8 and f² ≈ 0.02/0.15/0.35 (31,32).
Results
Demographics and clinical data
This study initially recruited 163 participants, including 71 patients with CM, 43 with EM, and 49 HCs. Following quality control procedures, 10 CM participants were excluded due to poor fMRI data quality (n = 6) or missing ASL images (n = 4). Similarly, four EM participants were excluded because of poor fMRI data quality. Seven HCs were also excluded, four for poor fMRI data quality and three for missing ASL images. As a result, the final sample included 142 participants: 61 with CM (25 with MOH and 36 without MOH), 39 with EM, and 42 with HCs (Figure 1). No significant differences were observed in age, gender, or BMI among the three groups. Compared with EM patients, CM patients had significantly higher headache frequency, MIDAS scores, PHQ-9 scores, and GAD-7 scores (Table 1). Within the CM subgroups, patients with MOH presented a longer migraine history and higher VAS scores than did those without MOH (Table 2).

Flowchart of the process for participant inclusion.
Demographic characteristics and general clinical data among HCs, EM and CM groups.

Note: a one-way ANOVA; b, chi-square test; c, Mann–Whitney U; d, two sample t test;
Demographic characteristics and general clinical data of CM patients with MOH and non MOH.

Note: a, chi-square test; b, Mann–Whitney U; c, two sample t test; *, significant difference (p < 0.05).
Spatial distributions of the CBF, ReHo and CBF/ReHo ratios
The CBF, ReHo, and CBF/ReHo ratio maps for HCs, EM, and CM patients are presented in Figure 2(a). All three groups presented higher values in the bilateral middle frontal gyrus, superior temporal gyrus, angular gyrus, and occipital cortex. A similar spatial distribution was observed in CM patients with and without MOH (Figure 2(b)).

(a) spatial distribution of the CBF, ReHo and CBF/ReHo ratios values in HCs, EM and CM groups. Warm colors indicate higher values, and cool colors indicate lower values. (b) spatial distribution of the CBF, ReHo and CBF/ReHo ratios values in CM patients with MOH and non MOH. Warm colors indicate higher values, and cool colors indicate lower values.
Global CBF-ReHo coupling changes in HCs, EM and CM groups
Significant voxel wise correlations between CBF and ReHo were observed for each participant by calculating the Pearson correlation coefficient between the voxel-level CBF and ReHo values. At the group level, both CM and EM patients exhibited significantly reduced global CBF-ReHo coupling than HCs did (Figure 3(a)). No significant difference in global CBF-ReHo coupling was found between CM patients with and without MOH (Figure 3(b)).

The whole brain CBF-ReHo coupling changes in HCs, EM and CM groups. (a) The whole brain CBF-ReHo coupling was significantly decreased in EM and CM patients compared to HCs. (b) There were no differences in whole-brain CBF-ReHo coupling between MOH and non MOH patients.
Changes in the CBF/ReHo ratios in HCs, EM and CM groups
There was a significant difference in the CBF/ReHo ratios of the right precuneus among HCs, EM and CM (Table 3, Figure 4(a)). Compared with EM, patients with CM exhibited decreased CBF/ReHo ratios in the right precuneus (Table 3, Figure 4(b)). Compared to HCs, CM patients exhibited decreased CBF/ReHo ratios in the left cerebellar area IX and Crus I (Table 3, Figure 4(c)). Compared to CM patients without MOH, CM patients with MOH exhibited decreased CBF/ReHo ratios in the bilateral fusiform gyrus (Table 3, Figure 4(d)). (GRF correction, voxel level P < 0.001, cluster level P < 0.05).
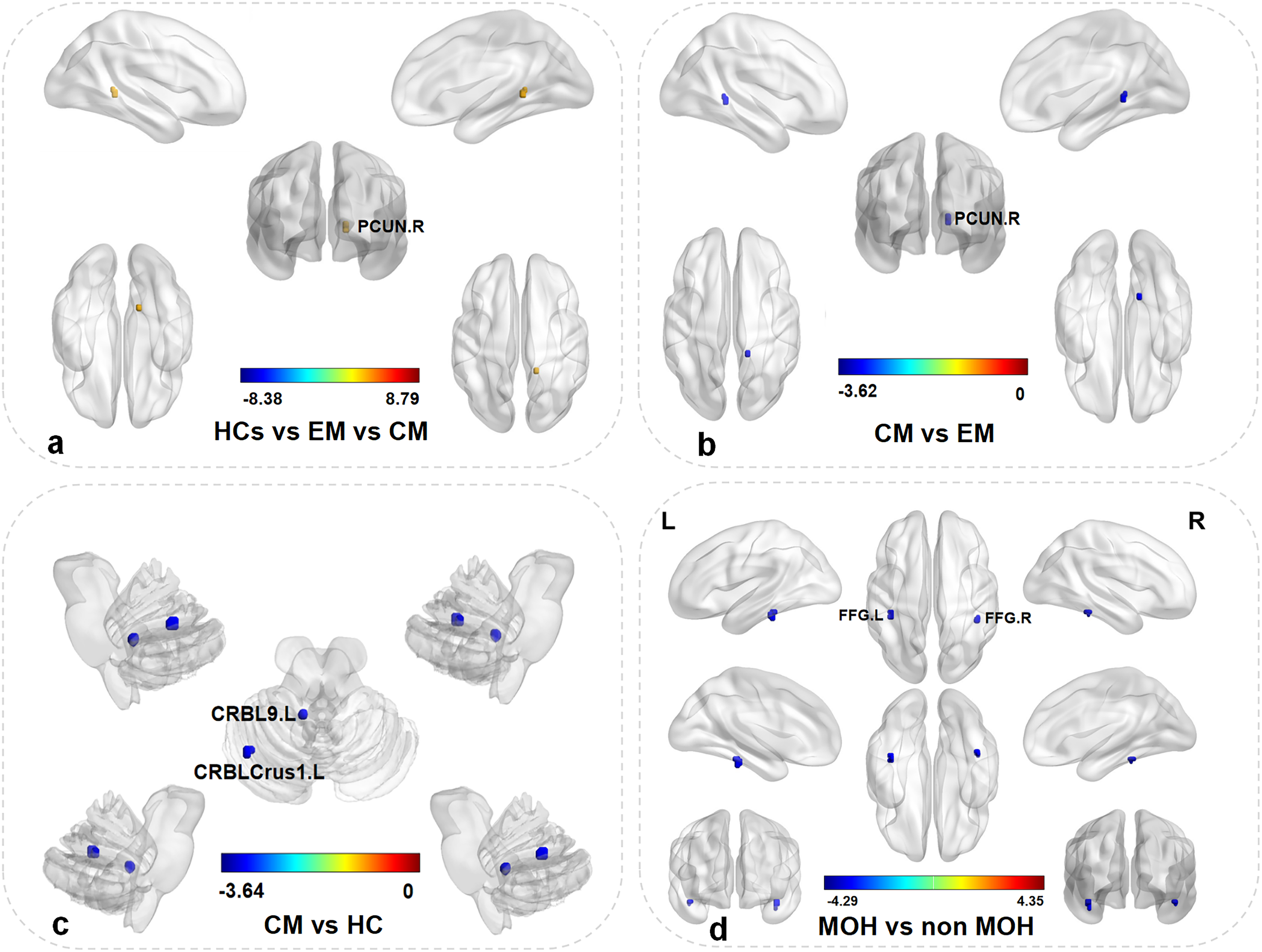
Comparison of CBF/ReHo ratios in HCs, EM and CM groups. (a) There was significant difference in the CBF/ReHo ratios of the right precuneus among HCs, EM, CM. (b) Compared with EM, patients with CM exhibited decreased CBF/ReHo ratios in the right precuneus. (c) Compared to HCs, CM patients exhibited decreased CBF/ReHo ratios in the left cerebellar areas IX and Crus I. (d) Compared to CM patients without MOH, CM patients with MOH exhibited decreased CBF/ReHo ratios in the bilateral fusiform gyrus. (GRF correction, voxel level P < 0.001, cluster level P < 0.05). Abbreviations: PCUN, the right precuneus; FFG, fusiform gyrus; CRBL9, the cerebellar area IX; CRBLCrus1, the cerebellar area Crus I, L, left; R, right. The blue color indicates lower values.
The brain regions where there were differences in CBF/ReHo ratios among the HCs, EM and CM groups.
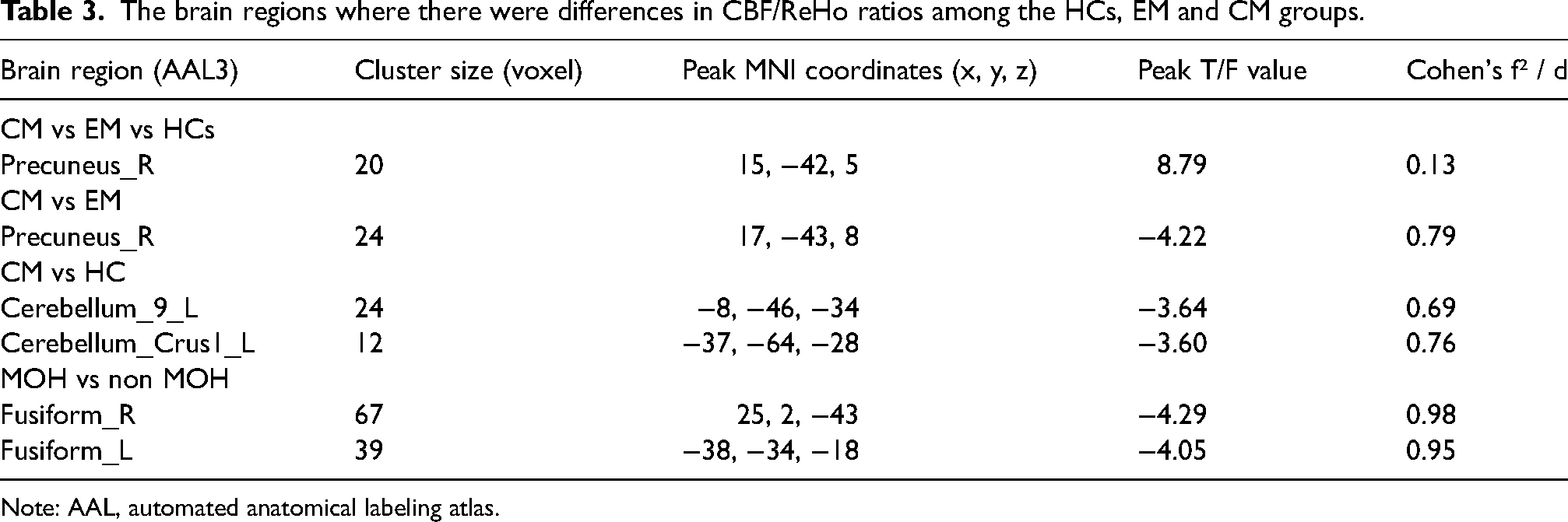
Note: AAL, automated anatomical labeling atlas.
Correlation analysis between NVC and clinical features in the CM group
Correlation analysis between NVC values and clinical features in the CM group revealed that the CBF/ReHo z value in the left cerebellar area IX was negatively correlated with the VAS score (r = -0.50, p = 0.03, Bonferroni corrected) (Figure 5(a)). The CBF/ReHo z value in the right precuneus was negatively correlated with both the HIT-6 score (r = -0.41, p = 0.03, Bonferroni corrected) and the PHQ-9 score (r = -0.49, p = 0.003, Bonferroni corrected) (Figure 5(b) and 5(c)). A positive correlation was also found between the HIT-6 and PHQ-9 scores (r = 0.49, p = 0.003, Bonferroni corrected) within the CM group (Figure 5(d)). Additionally, the CBF/ReHo z value in the left fusiform gyrus was negatively correlated with headache frequency in CM patients with MOH (r = -0.60, p = 0.03, Bonferroni corrected) (Figure 5(e)).

Correlation analysis of CBF/ReHo ratios with clinical characteristics in CM group. (a) The CBF/ReHo z-value in the left cerebellar area IX was negatively correlated with VAS score. (b, c) The CBF/ReHo z-value in the right precuneus showed a negative correlation with both HIT-6 score and PHQ-9 score. (d) A positive correlation was found between HIT-6 and PHQ-9 scores. (e) The CBF/ReHo z-value in the left fusiform gyrus was negatively correlated with headache frequency in CM patients with MOH.
Exploratory mediation analysis
Previous neuroimaging studies have shown that precuneus is part of DMN and plays a key role in emotional regulation (33,34). Moreover, emotions, especially depression, have been identified as important contributors to migraine chronification, which has been confirmed by previous studies (6,35). Based on previous studies and the correlation results in this study, we hypothesized that abnormal NVC may contribute to migraine clinical symptoms and depression may act as a mediating factor in this process.
After adjusting for covariates including age, gender, medication overuse, GAD7, and PSQI, the exploratory mediation analysis indicated that the emotion of depression (PHQ-9 score) significantly mediated the relationship between NVC in the precuneus and migraine-related disability (HIT-6 score). The indirect effect was statistically significant (β = -1.74, 95% CI = [-3.29, −0.20], p = 0.025), whereas the direct effect was marginally significant (β = -2.50, 95% CI = [-5.15, 0.16], p = 0.071). The total effect was significant (β = -4.25, 95% CI = [-6.74, −1.71], p = 0.001), with the emotion of depression accounting for 41.1% of the total effect. The path coefficients were as follows: the CBF/ReHo ratio in the right precuneus was significantly associated with PHQ-9 scores in CM patients (β = –4.75, p < 0.001), and PHQ-9 scores were significantly associated with HIT-6 scores (β = 0.37, p = 0.009, Table 4, Figure 6).

Exploratory mediation analysis with depression as a mediator. In this study, the abnormal CBF/ReHo in the right precuneus of CM patients was considered as the exposure factor, the emotion of depression as the mediator, and migraine-related disability (HIT-6 scale) as the outcome.
The results of the mediation analysis of NVC metrics and migraine characteristics.

Receiver operating characteristic curve analysis
To evaluate the diagnostic performance of NVC metrics in distinguishing different types of migraine and HCs, we extracted CBF/ReHo values from brain regions showing significant abnormalities among the CM, EM, and HC groups and conducted receiver operating characteristic (ROC) analysis. Age and gender were first included as predictors in the ROC models. The results showed that age and gender alone could differentiate CM from EM with an area under the curve (AUC) of 0.60, CM from HCs with an AUC of 0.61, and MOH from no MOH in CM patients with an AUC of 0.59.
The CBF/ReHo ratio from brain regions with significant intergroup differences were subsequently used to construct additional ROC models, with age and gender included as covariates. The results indicated that the CBF/ReHo ratios could distinguish CM from EM with an AUC of 0.75, CM from HC with an AUC of 0.73, and MOH from no MOH in CM patients with an AUC of 0.80 (Figure 7).

Comparison of the diagnostic efficacy of demographic characteristics and NVC metrics.
Discussion
Our study revealed that the whole NVC of EM and CM patients was lower than HCs. CM patients had lower CBF/ReHo ratio in the right precuneus than EM patients did after adjusting for age, gender and mood as covariates. Correlation analysis revealed that the z value in the right precuneus was negatively correlated with both the HIT-6 score and the PHQ-9 score, whereas the HIT-6 score was positively correlated with PHQ-9 scores in CM patients. The exploratory mediation analysis revealed that depression may mediate the relationship between NVC alterations and migraine clinical symptoms in CM patients. Finally, this study revealed that the CBF/ReHo ratio in the right precuneus distinguished CM from EM with an AUC of 0.75.
Previous studies have shown that approximately 3% of EM patients gradually progress to CM, which is influenced by various risk factors, such as age, gender, sleep disturbances, depression, stressful life events, and so on (6,36). Our study also revealed that patients with CM presented higher levels of anxiety and depression than EM patients did.
The brain functional activities require adequate energy, which can be achieved through oxidative phosphorylation and aerobic glycolysis (19,21). Previous studies have demonstrated a linear correlation between CBF and metabolism (20,29). The rs-fMRI method was used to assess the human brain functional activities (37,38). Liang et al. (29) demonstrated that a linear correlation exists between CBF and functional activity in HCs via combination of fMRI and ASL; this tight coupling between CBF and functional activities plays a crucial role in maintaining normal brain function. On the one hand, some previous studies have shown that compared with HCs, CM patients exhibit increased CBF in brain regions such as superior temporal gyrus, somatosensory cortex and so on during the interictal period (39,40). Another study revealed that CM patients exhibited reduced CBF in the nucleus accumbens and increased regional CBF in the bilateral ventral lateral nuclei of the thalamus compared with HCs (13,28). Interestingly, Youssef et al. (41) reported a positive correlation between regional CBF and headache attack frequency. On the other hand, several studies have reported that CM patients exhibit abnormal functional activity and connectivity in multiple brain regions, including thalamus, caudate nucleus, hypothalamus, prefrontal cortex, and limbic structures (9,42,43). Abnormal NVC has been observed in various diseases, including schizophrenia, neuromyelitis optica, primary open-angle glaucoma, and cervical spondylotic myelopathy (24,30,44,45). Our study found that the whole-brain NVC value was significantly lower in migraine patients than in HCs. To the best of our knowledge, this study is the first to report the whole-brain NVC abnormalities in migraine patients. Reduced NVC may contribute to disrupted pain signal transmission, altered brain network function, and impaired sensory regulation, which may be considered key mechanisms in the pathophysiology of migraine attack.
Notably, although the overall CM group exhibited significantly abnormal whole-brain NVC, subgroup analysis revealed no significant difference in the whole-brain NVC between CM patients with and without MOH. This finding suggested that the whole-brain NVC abnormality may not be directly attributable to medication overuse. Instead, these abnormalities may reflect more complex underlying MOH mechanisms.
The CBF/ReHo ratio represents the amount of CBF required to support a unit of brain functional activity, reflecting the degree of coupling between functional activities and CBF in specific brain region (21,30). Our study revealed that CM patients presented a lower CBF/ReHo ratio in the right precuneus than EM patients did. Additionally, CM patients presented decreased ratios in the left cerebellar areas IX and Crus I. CM patients with MOH presented lower CBF/ReHo ratios in the bilateral fusiform gyrus than CM patients without MOH. These reductions in the CBF/ReHo ratios may result from either decreased CBF or increased ReHo in the corresponding brain regions.
However, our study revealed that there were no significant differences in CBF or ReHo values in the brain regions that showed group differences in the CBF/ReHo ratio among the CM, EM, and HC groups (Online Supplementary Table S1, S2; Figure S1, S2). Therefore, we speculated that the CBF/ReHo ratios may serve as an independent indicator, that is distinct from CBF and ReHo metrics alone. The precuneus is in the posteromedial portion of the parietal lobe and is a core region of the DMN (46). It plays a crucial role in visual sensory integration, self-consciousness, and higher-order cognitive functions (47,48). Migraine is considered a disorder of sensory processing, affecting nearly all phases of the migraine attack, particularly in relation to pain, auditory, and visual perception (49–51). Pain transmission involves both ascending pathways, which convey pain signals, and descending pathways, which regulate pain perception (51). These pathways engage multiple brain regions, including the thalamus, locus coeruleus, and parieto-occipital cortex, all of which play crucial roles in the processing and modulation of pain (51). Dysfunction of the right precuneus may impair pain regulation or increase pain sensitivity in CM patients, potentially contributing to migraine attacks. Correlation analysis further revealed that the CBF/ReHo ratio in the right precuneus was negatively correlated with both the HIT-6 and PHQ-9 scores, whereas the HIT-6 and PHQ-9 scores were positively correlated in CM patients. These findings suggested that abnormal NVC in the right precuneus was associated with headache severity and emotional disturbance, further supporting its critical role in pain processing in CM patients. In addition, our study revealed that the CBF/ReHo ratios in the left cerebellar areas IX and Crus I were lower in CM patients than in HCs. Previous research has indicated that the cerebellum plays an important role in pain modulation, with extensive projections to the cerebral cortex that contribute to the regulation of pain signals (52). Li et al. (53) proposed that the cerebellum modulates pain signaling through its functional connectivity with sensorimotor regions, executive control areas, reward circuits, and limbic structures. Mehnert et al. (54) identified functional and structural alterations in the posterior cerebellum, particularly in Crus I and Crus II in migraine patients. This study revealed impaired NVC in the cerebellum of CM patients, which was significantly negatively correlated with the VAS score. These findings suggest that reduced cerebellar NVC is associated with more severe clinical symptoms in CM patients. NVC dysfunction compromises the capacity of the cerebellum to modulate nociceptive processing, consequently facilitating the development of pain symptoms.
Further subgroup analysis demonstrated that CM patients with MOH presented significantly lower CBF/ReHo ratios in the bilateral fusiform gyrus than CM patients without MOH did. Previous studies have indicated that the fusiform gyrus plays an important role in facial sensation processing, emotion, facial recognition, and reading (55,56). In this study, correlation analysis revealed that the CBF/ReHo ratio of the left fusiform gyrus was negatively correlated with the frequency of migraine attacks. A significant inverse relationship was observed, whereby greater impairment of NVC in the left fusiform gyrus was correlated with increased headache attack frequency in patients. Prolonged use of acute analgesic medications may disrupt NVC in the left fusiform gyrus, potentially increasing facial sensory sensitivity and predisposing patients to more frequent migraine attacks.
Based on our correlation analysis, we hypothesized that impaired NVC contributes to migraine clinical symptoms, with depression mediating the relationship between NVC dysfunction and clinical symptoms in CM patients. Exploratory mediation analysis revealed that depression played a mediating role in the process by which abnormal NVC led migraine-related disabilities. Several previous studies have shown that depression increases the risk of migraine chronification and plays a crucial role in the process of migraine attack and chronification; at the same time, standardized treatment of depression could improve the clinical symptoms of migraine patients (6,35,57). These findings further confirmed the hypotheses of this study.
Finally, to evaluate the diagnostic value of abnormal NVC metrics for CM patients, this study conducted ROC curve analysis, with the area under the curve representing diagnostic efficacy. This study revealed that abnormal NVC metrics in the right precuneus were better at distinguishing CM and EM patients than age and gender. NVC metrics in the right precuneus may serve as imaging feature for migraine chronification in the future.
Limitations
Although this study provides new insights into migraine chronification from the perspective of NVC, there are still certain limitations. First, while the fMRI and ASL techniques are noninvasive, they represent indirect measurements of brain activity and CBF. Additionally, although correlation analyses revealed associations between abnormal NVC metrics and clinical symptoms in CM patients, they did not allow for causal inference. Therefore, based on prior theoretical frameworks and supporting literature, we propose a hypothesis-driven mediation model to further explore potential mechanisms. However, this analysis remains exploratory in nature, and the results should not be interpreted as evidence of causality. Longitudinal or interventional studies are warranted to validate these findings in the future. Furthermore, as anxiety and depression scores were unavailable for HCs, these factors could not be considered as covariates in the analysis comparing migraine patients with healthy individuals. Finally, in this study, imaging analysis was performed with multiple comparisons correction via the GRF correction method. If other correction methods were chosen, different results would be obtained. This requires further discussion in future larger-scale studies.
Conclusion
Abnormal neurovascular coupling in the precuneus may be associated with migraine chronification, with depression potentially serving as a mediating factor in this process. In addition, neurovascular coupling metrics could serve as new imaging feature for migraine chronification in the future.
Clinical implications
Abnormal neurovascular coupling has been observed in the right precuneus during migraine chronification.
Depression may play a mediating factor in the process by which abnormal neurovascular coupling led to migraine attacks.
Neurovascular coupling metric may serve as objective imaging feature for migraine chronification in the future.
Supplemental Material
sj-doc-1-cep-10.1177_03331024251396031 - Supplemental material for Abnormal neurovascular coupling in the precuneus associated with migraine chronification: A multimodal magnetic resonance imaging study
Supplemental material, sj-doc-1-cep-10.1177_03331024251396031 for Abnormal neurovascular coupling in the precuneus associated with migraine chronification: A multimodal magnetic resonance imaging study by Cunxin Lin, Yuanxiang Li, Zhuang Wang, Chenyang Duan, Geyu Liu, Zhi Guo, Xiaoshuang Li, Zhonghua Xiong, Tong Chen, Mantian Zhang, Tianshuang Gao, Binbin Sui and Yonggang Wang in Cephalalgia
Footnotes
Acknowledgments
We sincerely thank the National Neurological Imaging Centre at Beijing Tiantan Hospital, Capital Medical University, for their essential support in providing technical assistance and imaging equipment for this study. Additionally, we deeply appreciate the contributions of headache specialists, whose expertise is crucial for ensuring accurate clinical diagnoses. Finally, we would also like to thank the other professionals who provided guidance on imaging data analysis for this study
Author contributions
CXL designed the study and wrote the original draft. CXL, YXL, ZW, CYD, and GYL contributed to the collation and acquisition of the data and analyzed the data. ZG, XSL, ZHX, MTZ, TSG, BBS, and YGW verified the results. All the authors contributed to reviewing the final draft and approved the submitted version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Joint Funds of the National Natural Science Foundation of China (U24A20683), National Natural Science Foundation of China (32170752), and Beijing Natural Science Foundation (F252058).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

