Abstract
Aim:
To investigate the effect of different cannabinoid compounds on the periorbital mechanical allodynia and photosensitivity in acute and chronic migraine models.
Methods:
Female Wistar rats were treated systemically with different cannabinoid compounds (cannabidiol, CBD, 30 mg/kg; CBD and cannabigerol, CBD/CBG – 2:1; CBD and 0.3% tetrahydrocannabinol (CBD/THC); or CBD/CBG/THC) followed by injection of calcitonin-gene-related peptide (CGRP) or pituitary adenylate cyclase-activating polypeptide (PACAP) into the trigeminal ganglion to induced immediate periorbital mechanical allodynia and late photosensitivity. The effect of CBD and CBD/THC was also assessed on periorbital mechanical allodynia and photosensitivity in the chronic migraine model induced by repeated nitroglycerin (NTG) injections.
Results:
Periorbital mechanical allodynia induced by CGRP was significantly reduced by CBD alone and combined with THC or CBG. CBD/THC was the most effective treatment in this condition since it presented the longer effect (up to three hours) and was the only treatment capable of reducing late photosensitivity associated with CGRP. All four compounds presented antinociceptive effect on acute migraine-like responses induced by PACAP, with CBD alone presenting the longer effect (from 30 minutes up to two hours). Except for CBD/CBG, all compounds also reduced (up to two hours) late photosensitivity associated with PACAP. In the chronic migraine model induced by NTG, CBD reduced periorbital mechanical allodynia on days 5, 7 and 11, while CBD/THC suppressed the development of periorbital allodynia up to day 13 and significantly reduced photosensitivity up to three hours.
Conclusion:
Altogether, these results suggest that cannabinoid compounds may represent effective alternatives for the treatment of episodic and chronic migraine.
This is a visual representation of the abstract.
Introduction
Migraine is a neurological disorder characterized by recurring and unpredictable episodic attacks of intense head pain that affects around 15% of the general population, being considered a leading cause of disability, especially in women between the ages of 15 and 50 years (1).
Migraine has a complex physiopathology that involves vascular, neuroimmune and neurological mechanisms, which are still not fully elucidated. From the neurobiological perspective, migraine initiation is associated with cortical spreading depression, neurotransmitter imbalance, and trigeminal nerve activation. This last process leads to the release of vasoactive peptides, which, in turn, induce neurogenic inflammation (2). In recent years, research efforts have primarily focused on the calcitonin-gene-related peptide (CGRP). However, due to the lack of response in certain patients to anti-CGRP treatments, attention has recently shifted towards the pituitary adenylate cyclase-activating polypeptide (PACAP) (3,4). CGRP and PACAP are vasodilatory peptides that can cause migraine-like attacks when infused into patients and migraine-like responses when injected into rodents, but mechanistic studies suggest that these peptides largely act independently from one another (5–8). Hopefully, a multi-receptor antagonist, targeting CGRP and PACAP receptors will be the next significant advance in migraine therapeutics (9). Still, due to the complex and multifactorial nature of migraine, the search for new treatment options is warranted.
There is growing pre-clinical and clinical evidence that cannabis and cannabis-derived products may provide benefit in the control of headache pain and other migraine-related symptoms. Studies have shown that modulation of the endocannabinoid system can interfere with various migraine-related mechanisms. In animal models of migraine, it has been demonstrated that endocannabinoids can inhibit vasodilation and CGRP release through the activation of the cannabinoid 1 (CB1) receptor, as well as mast cell degranulation through the activation of CB2 receptors (10–12). Through the application of strategies that enhance endocannabinoid levels, reports have been made of a decrease in nociceptive responses and the levels of pro-inflammatory mediators in different animal models of migraine (13–15). Studies using phytocannabinoids are still in early stages, with most research to date focusing on the psychoactive compound Δ9-tetrahydrocannabinol (THC) and on cannabidiol (CBD), which is a non-psychotropic cannabinoid. THC has been shown to produce antinociceptive effects and suppress cortical spreading depression in animal models by activating CB1 receptors (16). Likewise, CBD showed antinociceptive and anti-inflammatory effects in migraine induced by nitroglycerin (NTG) in rats, and reduced allodynia, spontaneous pain, and anxiety-like behavior in migraine induced by CGRP in mice (17,18). Recently, it was also demonstrated that a combination of CBD:THC (100:1) mitigated light aversion induced by CGRP and a nitric oxide donor in male and female mice, and reduced squint in male mice, without causing any emotional, cognitive, or locomotor dysfunction (19). To our knowledge, the effect of the different cannabinoid compounds has never been investigated in migraine-like responses induced by PACAP, and only the effect of CBD alone has been demonstrated in chronic migraine induced by NTG. In the clinical setting, medicinal cannabis has been used in the acute and preventive treatment of migraine. Although preliminary data are encouraging, the evidence is weak due to a lack of high-quality controlled trials (20,21).
Current evidence indicates that a combination of cannabinoids has the potential to offer distinctive analgesic benefits while simultaneously mitigating the adverse effects of specific cannabinoids. For example, CBD and THC operate through distinct mechanisms and target different pathways, and their combination has a broad potential for therapeutic effects. Notably, CBD can counteract the psychotropic effects associated with THC (22). Furthermore, the analgesic potential and the synergistic effect of other cannabinoid compounds remain to be determined. In this regard, cannabigerol (CBG) has garnered attention in the field of pain management due to its anti-inflammatory and neuroprotective effects (23). Recently, it has been shown that it produces acute antinociceptive effect selectively in female mice (24). The antinociceptive effects of CGB may involve multiple targets since it activates several members of the potential transitory receptor family (i.e. TRPV1, TRPV2, TRPV4 and TRPA1), in addition to CB2 and PPAR receptors (23). The effect of CBG in combination with other cannabinoid compounds has remained largely unexplored, but a recent study demonstrated that, in association with CBD, the behavioral profile of the mice is altered compared to its isolated effect (25). This observation highlights the importance of studying the effects of less known phytocannabinoids, as well as their combination with well-studied phytocannabinoids.
In the present study, the acute effects of different cannabinoid-based compounds, alone (CBD) or in combination (CBD/THC, CBD/CBG and CBD/CBG/THC), were assessed in the periorbital mechanical allodynia and photosensitivity induced by two potent migraine triggers, CGRP and PACAP, injected into the trigeminal ganglion (TG) of female rats. The efficacy of the different compounds was also evaluated in a chronic migraine model induced by repeated injections of NTG. In addition, the antinociceptive potential of the compounds was evaluated in the orofacial formalin test, allowing for a comparison of their efficacy in two trigeminal pain models with distinct underlying mechanisms. Finally, animals were tested in the open field to assess the influence of treatments on locomotor activity and exploratory behavior.
Methods
Animals
In this study, a total of 247 adult female Wistar rats (Rattus norvegicus), approximately 60 days old and weighing between 180 and 250 g, were used. The estrous cycle of the rats was not characterized. The animals were supplied by the animal facility of the Federal University of Paraná and housed in groups of four in plastic cages (41 cm x 32 cm x 16.5 cm) lined with wood shavings, with food and water provided ad libitum. They were kept in a separate room with controlled temperature (22 ± 2 °C), humidity (40–60%), and a 12-hour light/dark cycle, with lights on at 07:00 am and off at 07:00 pm. All experimental protocols were approved by the Ethics Committee on the Use of Animals (CEUA/BIO/UFPR #1589) and conducted following the ARRIVE guidelines. All experiments were carried out after a minimum 48-hour acclimatization period in the laboratory. Experimenters were blinded to the treatment condition, with a different experimenter assigning each animal to a group by simple sortition.
Drugs and solutions
The cannabinoid-based compounds used were: decarboxylated cannabidiol (CBD) oil; decarboxylated CBD and cannabigerol (CBD/CBG-2:1) oil; CBD oil with 0.3% tetrahydrocannabinol (CBD/THC); and decarboxylated CBD/CBG oil with 0.3% THC (CBD/CBG/THC). Isospec Laboratories (Fort Lauderdale, FL, USA) produced all formulations under Good Manufacturing Practice (cGMP) certification in scientific partnership with Health Meds Laboratories (USBR-Pharmaceutical, Coral Springs, FL, USA). They were diluted in medium-chain triglyceride oil (100% MTC oil) and administered at a dose of 30 mg/kg relative to CBD. The dose selected for CBD was based in several previous studies that showed antinociceptive effect of CBD, alone or in combination, in different pain models, including migraine models (17,19,26,27).
Formaldehyde (Alphatec, São José dos Campos, SP, Brazil) was diluted to 2.5% in 0.9% sterile saline (Samtec Biotecnologia, Ribeirão Preto, SP, Brazil). Rat calcitonin gene-related peptide (α-CGRP; Sigma-Aldrich, St Louis, MO, USA) was diluted in sterile saline and administered at a concentration of 0.1 nmol/10 µl. Sumatriptan succinate (Libbs, Embu das Artes, SP, Brazil) was diluted in water and administered at a dose of 1 mg/kg. Pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38; Sigma-Aldrich, St Louis, MO, USA) was diluted in 0.9% sterile saline to 1 ng/10 µl. Nitroglycerin (NTG, Cristália, Brazil) was diluted in 0.9% sterile saline and administered at a dose of 5 mg/kg. The doses were based on previous studies (28–30).
Intraganglionic injection
Intraganglionic injections of CGRP, PACAP or vehicle were performed as previously described by our group (28–31). A 50-µL Hamilton syringe was attached to a sterile 27-G needle. Animals were deeply anesthetized with halothane (4%) and oxygen, placed in the prone position and the needle was inserted into the zygomatic process via the infraorbital foramen. The needle was angled at approximately 10 ° from the midline and advanced through the infraorbital canal until it passed the foramen rotundum, reaching the correct TG. A volume of 10 µl was injected.
Assessment of periorbital mechanical allodynia
Animals were individually placed in acrylic boxes for two hours to acclimate them to their surroundings. Von Frey filaments (Semmes-Weinstein Monofilaments; Stoelting, Wood Dale, IL, USA: 0.16, 0.4, 1.0, 2.0, 4.0 and 8.0 g) were applied to the periorbital region in ascending order to determine the baseline periorbital threshold. Each filament was applied three times at three-second intervals. The filament that evoked an aversive response (e.g., withdrawal, escape, aggression, vocalization, or attack attempt) in two or more trials was considered the response threshold (28,31). Only rats showing baseline responses equal to 8 g were included in the experiments. Then, animals were treated systemically (i.p.) with the cannabinoid compounds (CBD, CBD/THC, CBD/CBG or CBD/CBD/THC), sumatriptan or vehicle (100% MTC oil, 3 ml/kg), and 15 minutes later, CGRP, PACAP or vehicle (10 µl) were injected into the TG. The periorbital mechanical threshold was assessed from 30 minutes to four hours after intraganglionic injections.
Assessment of photosensitivity
Twenty-four hours after TG injection with CGRP, PACAP or vehicle, the periorbital mechanical threshold was re-evaluated, followed by the animals’ exposure to a bright light (5.000 lux) for one hour (28,31,32). Subsequently, the periorbital mechanical threshold was assessed, as described in item 2.4, from 30 minutes to four hours after light exposure.
NTG model of migraine
Animals were treated with CBD, CBD/THC or vehicle, at the same doses of acute treatments, but once a day, on alternate days (1, 3, 5, 7 and 9). Subjects received a second i.p. injection of NTG (5 mg/kg) or vehicle (sterile saline solution, 1 ml/kg) 15 minutes later and on alternate days for 9 days (days 1, 3, 5, 7 and 9) (29). The periorbital mechanical threshold (item 2.4) was assessed before the treatments (baseline) and on alternate days, two hours after NTG or vehicle injection. The periorbital mechanical threshold was also evaluated after the termination of the treatments, up to day 20, when the animals were exposed to the bright light for one hour to determine photosensitivity (item 2.5).
Orofacial formalin test
The orofacial formalin test was performed as previously described by our group (33), with the purpose of assessing the efficacy of the different cannabinoid compounds in an acute trigeminal pain model not related to migraine. Animals were individually placed in transparent acrylic boxes for 15 minutes to acclimate them and reduce stress-related behaviors. After this period, animals were treated i.p. with the cannabinoid compounds (CBD, CBD/THC, CBD/CBG or CBD/CBD/THC) or vehicle (100% MCT oil, ml/kg), and 30 minutes later, they received a subcutaneous (s.c.) injection of formalin solution (2.5%, 50 µl) or vehicle (0.9% sterile saline, 50 µl) into the right upper lip and were immediately returned to the observation box. The grooming behavior (rubbing the injected area with both forepaws) was recorded for 30 minutes. The phase I of the response was defined as 0–3 minutes, and the phase II was the interval between 12 and 30 minutes post-injection.
Open field test
To assess whether the treatments per se could affect locomotion or potentially induce effects suggestive of sedation or anxiety-like behavior, which might bias the behavioral analysis, the animals were tested in the open field. One hour after the treatment with the vehicle or the different cannabinoid compounds, the animals were placed in the center of an arena (50 cm long, 50 cm wide and 40 cm high) with closed sidewalls and the floor divided into nine quadrants. The room's light was dimmed to provide comfortable conditions for the rats (40 lux). Rats’ behavior was recorded for 5 minutes for posterior analyses using EthoVision XT, version 10 (Noldus, Wageningen, The Netherlands), from which the total distance moved, velocity, average speed, number of entries into the center, and time spent in the center were extracted (34).
Statistical analysis
The sample size was determined using G*Power, version 3.1 (http://www.gpower.hhu.de), which defined an extensive, standardized effect size of F = 0.5, a power of 0.85 and an α of 0.05, estimating nine rats per group across the different experimental designs. The primary outcome was the assessment of the periorbital mechanical threshold. Based on this observation, in previous studies that have used the same animal model (28,31) and considering replacement, reduction and refinement in optimizing animal use, it was determined a sample size of 6 to 10 animals per group.
Data from the orofacial formalin and open field tests were analyzed using one-way analysis of variance (ANOVA), followed by a Bonferroni post-hoc test. Data from periorbital mechanical allodynia and photosensitivity were analyzed by two-way repeated measures ANOVA, followed by a Bonferroni post-hoc test. Outliers in the formalin tests were identified and excluded using Grubbs’ test (α = 0.05). Analyses and graphs were generated using Prism, version 9.0 (GraphPad Software Inc., San Diego, CA, USA). p ˂ 0.05 was considered statistically significant.
Results
Effect of cannabinoid compounds on migraine-like responses induced by CGRP
Figure 1A shows the experimental design. Injection of CGRP in the TG caused a significant reduction of periorbital mechanical threshold in the first 30 minutes up to the third hour after injection (Figure 1B–E). Previous treatment with CBD, CBD/THC and CBD/CBG prevented the development of periorbital mechanical allodynia; however, the effect of CBD/THC (F15,120 = 7.783; p < 0.0001) was longer-lasting, persisting up to three hours (Figure 1B), followed by CBD (F15,120 = 10.85; p < 0.0001) (for two hours) (Figure 1C) and CBD/CBG (F15,120 = 6.120; p < 0.0001) (for one hour) (Figure 1D). CBD/CBG/THC had no antinociceptive effect throughout the evaluated period (Figure 1E).

Effect of treatment with cannabinoid compounds on periorbital mechanical allodynia and photosensitivity induced by calcitonin-gene-related peptide (CGRP). Experimental protocol (A). Effect of cannabidiol (CBD) (B), CBD/tetrahydrocannabinol (THC) (C), CBD/cannabigerol (CBG) (D) or CBD/CBG/THC (E) on periorbital mechanical allodynia induced by CGRP. Effect of CBD (F), CBD/THC (G), CBD/CBG (H), and CBD/CBG/THC (I) on photosensitivity assessed 24 h after CGRP injection. BL = baseline; VF = von Frey. *p < 0.05 and **p < 0.0001 compared to the Vehicle (Veh) – Veh group; #p < 0.05 and ##p < 0.0001 compared to the Veh – CGRP group. Two-way analysis of variance with repeated measures followed by a Bonferroni post-hoc test. Data are expressed as the mean ± SEM (n = 6-8).
Twenty-four hours after CGRP injection, periorbital mechanical allodynia was reactivated by light exposure to assess the influence of the treatments on the sensitization of trigeminal afferents. The photosensitivity induced by CGRP was observed at the first hour after light exposure (Figure 1F–I), but only the treatment with CBD/THC (F15,120 = 1.671; p = 0.0379) was able to prevent this sensitization (Figure 1G). No significant results were observed with CBD, CBD/CBG and CBD/CBG/THC treatment (Figure 1F and H–I, respectively).
Effect of cannabinoid compounds on migraine-like responses induced by PACAP
Figure 2A shows the experimental design. Injection of PACAP in the TG caused a significant reduction of periorbital mechanical threshold in the first 30 minutes up to the second hour after the injection (p > 0.0001) (Figure 2B–E). Previous treatment with all cannabinoid compounds caused a significant reduction of PACAP-induced periorbital mechanical allodynia (Figure 2B–E). Still, the effect of CBD (F10,126 = 3.431; p = 0.0005) was more evident since it fully prevented the development of periorbital mechanical allodynia (Figure 2B).

Effect of treatment with cannabinoid compounds on periorbital mechanical allodynia and photosensitivity induced by pituitary adenylate cyclase-activating polypeptide (PACAP). Experimental protocol (A). Effect of cannabidiol (CBD) (B), CBD/tetrahydrocannabinol (THC) (C), CBD/cannabigerol (CBG) (D) or CBD/CBG/THC (E) on periorbital mechanical allodynia induced by PACAP. Effect of CBD (F), CBD/THC (G), CBD/CBG (H), and CBD/CBG/THC (I) on photosensitivity assessed 24 hours after PACAP injection. BL = baseline; VF = von Frey. *p < 0.05 and **p < 0.0001 compared to the Veh (Vehicle) – Veh group; #p < 0.05 and ##p < 0.0001 compared to the Veh – PACAP group. Two-way analysis of variance with repeated measures followed by a Bonferroni post-hoc test. Data are expressed as the mean ± SEM (n = 8).
Twenty-four hours after PACAP injection, animals were exposed to bright light. The photosensitivity induced by PACAP was observed at the first 30 minutes up to the second hour after light exposure (Figure 2F–I). Again, CBD (F10,105 = 4.829; p > 0.001) treatment fully prevented the development of photosensitivity (Figure 2F). Treatment with CBD/THC (F10,126 = 3.387; p = 0.0006) significantly reduced photosensitivity at a one-hour time point (Figure 2G). Still, CBD/CBG (F10,126 = 2.699; p = 0.0049) did not affect the development of photosensitivity (Figure 2H). A complete prevention of photosensitivity induced by PACAP was also observed with CBD/CBG/THC (F10,105 = 4.773; p < 0.0001) treatment (Figure 2I), contrasting the effect of this compound in CGRP-induced sensitization.
Comparative effect of cannabinoid compounds on migraine-like responses induced by CGRP and PACAP
To compare the efficacy of the different treatments, the analysis of periorbital mechanical threshold at the first-hour time point was performed, with the effect of sumatriptan used as a positive control. Figure 3A shows that CGRP promotes a reduction in the mechanical threshold, which is fully reversed by CBD, CBD/THC, CBD/CBG, and sumatriptan treatment (F6,43 = 23.98; p < 0.0001). In contrast, CBD/CBG/THC did not promote any change at the mechanical threshold. Likewise, PACAP caused a significant reduction in the mechanical threshold, which was entirely prevented by CBD, CBD/THC, CBD/CBG, and sumatriptan treatments (F6,47 = 4.575; p = 0.0010). At the same time, CBD/CBG/THC slightly increased the mechanical threshold compared to vehicle treated animals (Figure 3B). In the photosensitivity test, CGRP (Figure 3C) promoted a reduction in the periorbital mechanical threshold at the first hour after light exposure that was fully reversed by CBD/THC treatments, but the other treatments did not have a significant effect (F5,37 = 4.510; p = 0.0026). The periorbital mechanical allodynia was also reactivated by light exposure in female rats previously injected with PACAP (Figure 3D). This effect was fully prevented by treatment with CBD, CBD/THC and CBD/CBG/THC (F5,41 = 19.19; p < 0.0001). However, CBD/CBG did not promote any change in the periorbital mechanical threshold of PACAP-injected animals.
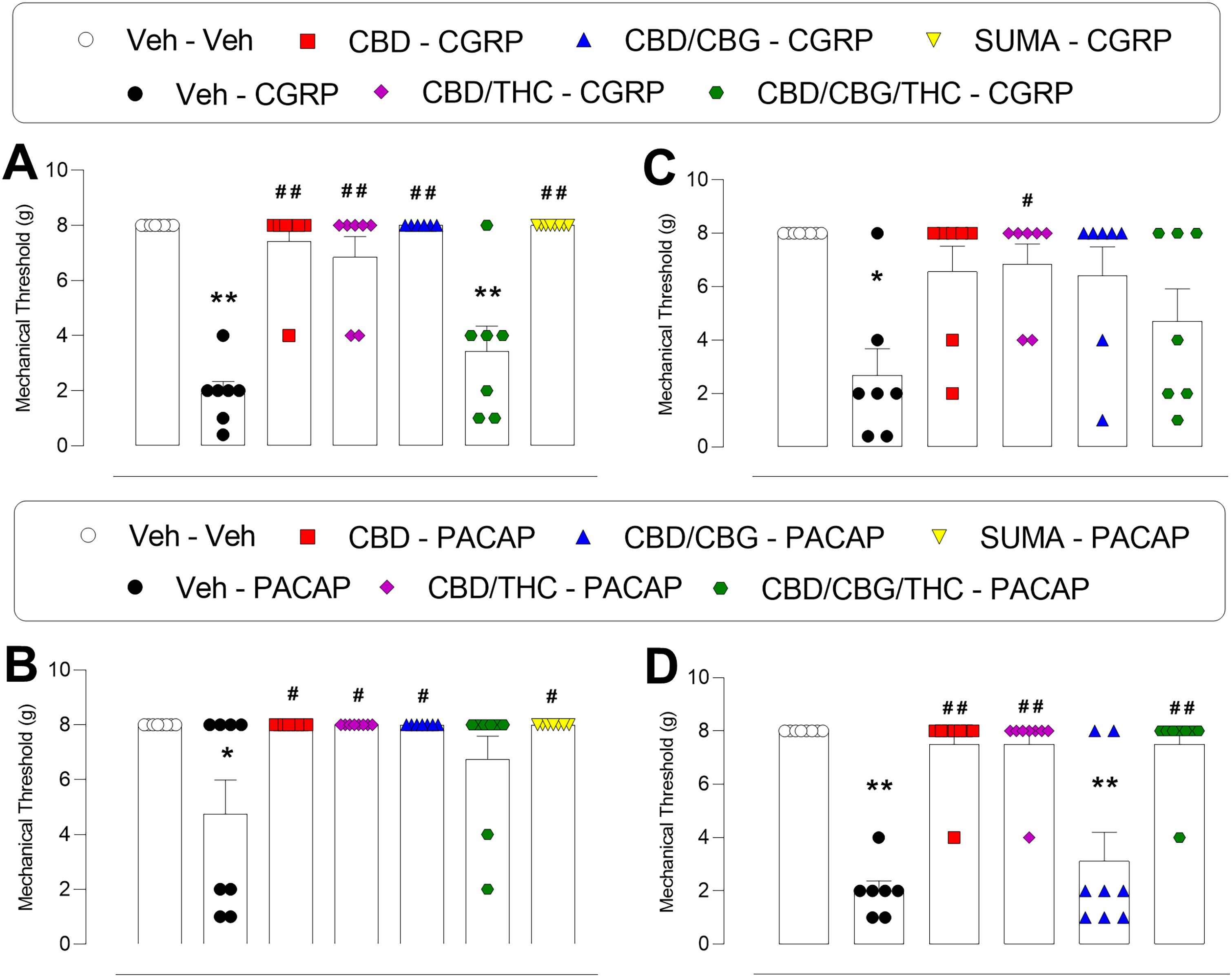
Comparative effect of cannabinoid compounds on periorbital mechanical allodynia and photosensitivity induced by calcitonin-gene-related peptide (CGRP) and pituitary adenylate cyclase-activating polypeptide (PACAP) assessed at the first hour after the intraganglionic injection. Periorbital mechanical allodynia induced by CGRP (A). Periorbital mechanical allodynia induced by PACAP (B). Photosensitivity induced by CGRP (C). Photosensitivity induced by PACAP (D). SUMA = sumatriptan. *p < 0.05 and **p < 0.0001 compared to the Veh (Vehicle) – Veh group; #p < 0.05 and ##p < 0.0001 compared to the Veh – CGRP or Veh – PACAP group. One-way analysis of variance followed by Bonferroni post-hoc test. Data are expressed as the mean ± SEM (n = 6-8).
Effect of cannabinoid compounds on migraine-like responses induced by NTG
Figure 4A shows the experimental design. Repeated NTG injections promoted a significant reduction of the periorbital mechanical threshold on days 5, 7, 9, 11 and 15 compared to vehicle-treated animals (Figure 4B and C). The chronic treatment with CBD (F18,162 = 3.094; p < 0.0001) was able to increase the threshold on days 5, 7 and 11 (Figure 4B), while the CBD/THC (F18,162 = 3.952; p < 0.0001) fully prevented the development of the periorbital mechanical allodynia up to day 13 (Figure 4C). On day 20, the rats were exposed to bright light for the reactivation of allodynia, which was significant from two hours up to four hours after light exposure (Figure 4D and E). CBD (F10,90 = 2.294; p = 0.0190) did not prevent the reactivation of allodynia (Figure 5D), while CBD/THC (F10,90 = 3.724; p = 0.0003) impeded the reduction of the periorbital mechanical threshold in the second and third hours after light exposure (Figure 4E).

Effect of treatment with cannabinoid compounds on periorbital mechanical allodynia and photosensitivity induced by nitroglycerin (NTG). Experimental protocol (A). Effect of cannabidiol (CBD) (B), CBD/tetrahydrocannabinol (THC) (C) on periorbital mechanical allodynia induced by NTG. Effect of CBD (D) and CBD/THC (E) on photosensitivity assessed 20 days after the first treatment. BL = baseline; VF = von Frey. *p < 0.05 and **p < 0.0001 compared to the Veh (Vehicle) – Veh group #p < 0.05 and ##p < 0.0001 compared to the Veh – NTG group. Two-way analysis of variance with repeated measures followed by a Bonferroni post-hoc test. Data are expressed as the mean ± SEM (n = 7).
Effect of cannabinoid compounds on the orofacial formalin test
The time course of the facial grooming behavior after formalin injection into the upper lip, 30 minutes after treatment with cannabinoid compounds, is shown in the supplementary material (Figure S1A). Formalin significantly increased grooming behavior in the first (F9,86 = 18.22; p < 0.0001) and second phases (F9,86 = 11.66; p < 0.0001) of the response compared to vehicle-treated animals. None of the treatments altered the facial grooming behavior in phases I (see supplementary material, Figure S1B) and II (see supplementary material, Figure S1C) of the test.
Effect of cannabinoid compounds on the open field test
As demonstrated in the supplementary material (Figure S2A), CBD increased the distance moved in the open field in female rats, but the other cannabinoid compounds did not change this parameter (F9,50 = 5.748; p < 0.0001). Additionally, the speed (see supplementary material, Figure S2B), the number of entries into the center (see supplementary material, Figure S2C) and time spent in the center (see supplementary material, Figure S2D) were analyzed; however, none of the treatments caused significant changes compared to vehicle-treated animals. It is noteworthy that this test was performed on animals that do not receive intraganglionic injections of CGRP or PACAP since this procedure does not alter any of the parameters analyzed in the open field test (data not shown).
Discussion
The neuropeptides CGRP and PACAP have been identified as key mediators of migraine; however, the extent of their overlapping mechanisms remains unclear. Similar to CGRP, PACAP infusion can trigger migraine in individuals, as both peptides exhibit comparable vasodilatory and pronociceptive properties (5). The injection of CGRP, systemic or into the TG, in mice and rats is a well-established model of episodic migraine (28,31,35,36). It has been reported that this procedure induces immediate periorbital cutaneous allodynia, which has been measured in animal models as a surrogate of headache-like pain. Moreover, it induces latent sensitization, a persistent state of pain hypersensitivity that can be reactivated by specific triggers, such as NTG, stress and exposure to bright light (19,28,31,35–38). The results of the present study showed that CBD and CBD/THC prevented the development of periorbital cutaneous allodynia induced by intraganglionic injection of CGRP. CBD/CBG also showed antinociceptive effect, but it was short-lasting, while CBD/CBG/THC did not cause a significant change in the periorbital mechanical threshold. Moreover, CBD/THC prevented the sensitization of the trigeminal system since there was no reactivation of the periorbital allodynia triggered by the bright light exposure 24 hours later. Altogether, these results suggest that CBD/THC present a beneficial effect on acute head pain and photophobia associated with CGRP. These findings corroborate previous observations that CBD (at 15 and 30 mg/kg) reduces the nociceptive behavior induced by NTG plus formalin and that the CBD/THC combination mitigates light aversion induced by CGRP in male and female mice (17,19). However, in contrast to the previous study, this combination also attenuated periorbital cutaneous allodynia. One possibility for this discrepancy is the difference in the ratio of CBD: THC, which was 30 mg/kg:0.3% in the present study and 100:1 in the previous study (19).
On the other hand, all four compounds caused a significant reduction of periorbital cutaneous allodynia induced by intraganglionic injection of PACAP. CBD and CBD/CBG/THC also fully prevented latent sensitization, as animals that received these treatments did not develop periorbital allodynia after light exposure. CBD/THC caused a transitory effect in photophobia, but CBD/CBG showed no significant effect. There are a few reports that PACAP systemic injection at low doses in mice induces periorbital mechanical allodynia and light aversion (6,39,40). Herein, it was demonstrated for the first time that PACAP injection into the TG induces periorbital mechanical allodynia and latent sensitization similar to CGRP (28). Similar to CGRP, PACAP and its receptors are found in TG neurons, but PACAP is found in considerably fewer neurons than CGRP (41). However, the TG has been suggested as the main source of PACAP in the trigeminovascular system, as there is no PACAP release from the dura mater, in contrast to CGRP (42).
According to our results, migraine-like responses induced by CGRP and PACAP exhibited different sensitivities to the various cannabinoid compounds tested, suggesting the involvement of distinct mechanisms in their pronociceptive effects. However, the periorbital mechanical allodynia induced by both CGRP and PACAP was susceptible to inhibition by sumatriptan, which is supported by previous clinical and preclinical studies (4,28,39,43).
On the other hand, the addition of CBG to the cannabinoid compounds does not appear to result in an additional antinociceptive effect and, in fact, it seems to impair the effect of CBD and of CBD/THC on the CGRP model. Otherwise, the CBD/CBG/THC compound expressed a better performance on the PACAP model. This can lead to speculation that cannabinoid compounds modulate vasoactive peptides, such as CGRP and PACAP, differently.
Although there is increasing evidence about the benefits of different cannabinoid compounds in acute migraine models, little is known about their antinociceptive potential in chronic migraine. Repeated injections of NTG have been widely used as a chronic migraine model, as it induces persistent hind paw and periorbital hypersensitivity, particularly in females, that return to baseline levels around day 15, approximately 6–7 days after the last NTG injection (17,29,44). This procedure also results in latent sensitization, as after the animals have fully recovered to baseline mechanical threshold levels, a lower dose of NTG or exposure to other migraine triggers (i.e. light, stress) is sufficient to mediate a reduction in periorbital withdrawal thresholds (29,45). In the present study, the effects of CBD alone and the CBD/THC combination were assessed in chronic migraine induced by NTG because these compounds have been shown to provide better antinociceptive effects compared to CBD/CBG and CBD/CBG/THC in acute models. According to our results, CBD attenuated the periorbital mechanical allodynia induced by NTG, while CBD/THC entirely prevented allodynia development. Moreover, only CBD/THC impeded the reactivation of the periorbital mechanical allodynia induced by exposure to bright light, even several days after the termination of the treatment. In line with these observations, a previous study demonstrated that a peripherally-restricted mixed CB1R/CB2R agonist suppressed the development of acute periorbital and hind paw allodynia in the same migraine model in female mice, in addition to preventing the increase in nNOS, p-PKA and TRPA1 protein levels in the TG induced by NTG (46). Moreover, in male rats, it was shown that CBD at 30 mg/kg caused a reduction in interleukin-6 protein levels in the medulla–pons and TG, as well as in serum levels of CRRP induced by repeated injections of NTG (17). These findings indicate the potential of CBD and CBD/THC in the treatment of frequent migraine, but further studies are necessary to investigate potential risks associated with their application for chronic migraine treatment. A summary of the effects of all four cannabinoid compounds tested in the different migraine models is presented in Table 1.
Summary of the effects of the cannabinoid compounds on migraine models.

CGRP = calcitonin-gene-related peptide; PACAP = pituitary adenylate cyclase-activating polypeptide; CBD = cannabidiol; CBG = cannabigerol; THC = tetrahydrocannabinol.
The effect of CBD, CBD/THC, CBD/CBG and CBD/CBG/THC in the periorbital mechanical allodynia and photosensitivity in the acute (CGRP- and PACAP-induced) and chronic (nitroglycerin-induced) migraine models is summarized. Bold text indicates the treatment associated to the longest duration of inhibition of both allodynia and photosensitivity.
Cannabinoids present multiple mechanisms with anti-migraine potential, and the differential relevance of these mechanisms in each migraine model may account for the differential efficacy of each cannabinoid compound tested. For instance, in the periphery, the activation of CB1 receptors leads to the suppression of CGRP release, while within central processes, endocannabinoids act to inhibit glutamate release, which in turn modulates the transmission of nociceptive signals to second-order neurons in the brainstem (47). In the TG, CB1 receptors are expressed by neurons, while CB2 are expressed by satellite glial cells, and their activation results in inhibition of neuropeptides release and neurons-glia-interaction (47). Cannabinoids have also been shown to reduce neurogenic inflammation and cortical spreading depression (47). Apart from interacting with the conventional inhibitory CB1 and CB2 receptors, cannabinoids can also participate in neuromodulation mediated by non-cannabinoid receptors, which adds an extra layer of complexity to their effects (47). One limitation of the present study is the lack of investigation of the main mechanisms that underly the antinociceptive effects of the compounds in each model, considering that there are differences in the migraine models. Likewise, differences in the biological activity of each compound may contribute to their differential efficacy in each migraine model. It is already known that CBD and THC bind differently to CB1R, which determines the different potency and effects observed. While THC binds orthosterically to this receptor, similar to anandamide, a recognized endocannabinoid with analgesic function, CBD binds in an allosteric site, having, among other effects, the ability to modulate the undesirable effects of THC (48). Furthermore, the effects of phytocannabinoids do not occur exclusively through their binding to the receptor since, in the case of CBD, there is evidence of its inhibitory action on fatty acid amide hydrolase, leading to a decrease in the degradation of anandamide and an increase in its analgesic effect (23). What we observed in the results of this study is in line with the so-called entourage effect, where a better action of cannabinoids is observed when used together, either due to greater efficiency in the effect and in the mitigation of unwanted effects that may occur due to the need for a lower dosage required to achieve the desired effects than when used alone (49).
Another important consideration regarding the cannabinoid compounds tested in the present study is their pharmacokinetic profile. Studies have shown that following systemic administration higher levels of CBD and THC have been reported in the plasma and brain from 30 minutes to two hours post-injection (50). This profile is consistent with the rapid anti-allodynic effect demonstrated in the present study, in which the effect of the compounds was significant between 30 minutes up to three hours after the treatment. However, the late effect of some compounds in blocking photosensitivity is unlikely to be a direct effect of the drug, but a result of the blockade of a signaling pathway that is relevant to the sensitization of the trigeminal system. Interestingly, after systemic administration of CBD/THC, the THC maximum mean peak concentration was five times higher and CBD concentration about half the value than administration of THC or CBD alone in both the serum and brain tissue, indicating a significant inhibition of THC metabolism by CBD (51). Moreover, plasma and brain concentrations of the psychoactive THC metabolite 11-OH-THC were twofold to sevenfold higher in female animals compared with males, indicating sex influence on the metabolism (52).
Sex differences have been reported in the efficacy of cannabinoid compounds and one limitation of the present study is the exclusive use of female rats in the experiments. The reasons for this choice were the higher prevalence of migraine in the female sex and the increased sensitivity of female rats to CGRP and PACAP. Although the compounds tested showed anti-allodynic effect in the migraine models in females, none of the treatments presented an antinociceptive effect in the formalin test, a widely used model of acute pain in rodents. Studies that have assessed the impact of different cannabinoids in the paw formalin test corroborate these observations, showing that higher doses (not tested here) are necessary to produce analgesia in females compared to males (53–55). For example, in male mice, CBD significantly attenuated phases I and II of the paw formalin test at a dose of 30 mg/kg. In female mice, the antinociceptive effect was only detected in both phases at 100 mg/kg (55). Thus, the efficacy of the different cannabinoid-based compounds observed in the present study in the migraine model, but not in the formalin test, in female rats, suggests that antinociceptive mechanisms related to the physiopathology of migraine are involved. It is worth mentioning that previous studies using migraine models have shown the efficacy of some compounds in males (17,19), indicating that both sexes can benefit from cannabinoid-based therapy.
Overall, the cannabinoid compounds tested herein did not change the locomotion of the rats assessed in the open field, except for a slight increase in the distance moved by CBD. This finding aligns with previous reports that CBD treatment at doses of up to 30 mg/kg does not alter locomotor and exploratory behaviors assessed in the open-field test (17,56). There are reports that THC causes changes in locomotor activity, exploratory, and anxiety-like behaviors in the open field test, but at doses higher than the one used in the present study (56,57). The low dose of THC used in the present study represents a clear advantage over other studies since its combination with CBD can improve analgesia without increasing side effects.
In conclusion, all four cannabinoid compounds tested presented antinociceptive effects on acute migraine-like responses induced by two potent migraine triggers, CGRP and PACAP. CBD alone and the combination of CBD/THC presented better comparative results in immediate periorbital mechanical allodynia and late photosensitivity induced by CGRP and PACAP in female rats. Moreover, CBD attenuated periorbital mechanical allodynia, while CBD/THC suppressed the development of periorbital allodynia and photosensitivity in the chronic migraine model induced by NTG. None of the compounds were associated with sedative or anxiety-like effects. Altogether, these results suggest that cannabinoid compounds may represent safe and effective alternatives for the treatment of episodic and chronic migraine.
Clinical perspectives
The present findings highlight the potential of specific cannabinoid formulations, particularly the low-THC and CBD/THC combination, as candidates for migraine management. This compound consistently attenuated periorbital allodynia and photosensitivity across acute (CGRP- and PACAP-induced) and chronic (nitroglycerin-induced) migraine models, without producing locomotor or anxiety-like effects. CBD alone demonstrated moderate efficacy, with shorter duration of action and limited effects on light-induced sensitization, while CBG-containing combinations showed variable results depending on the trigger, suggesting distinct interactions with CGRP- and PACAP-mediated pathways. These data support further controlled clinical studies to evaluate CBD- and CBD/THC-based therapies as potential preventive or adjunctive options for patients with episodic or chronic migraine, particularly those with suboptimal responses to current targeted treatments.
Article highlights
CBD, CBD/THC and CBD/CBG reduced periorbital mechanical allodynia, but only CBD/THC reduced photosensitivity induced by CGRP.
All four compounds reduced periorbital mechanical allodynia induced by PACAP, but CBD/CBG was ineffective against photosensitivity.
None of the compounds were associated with sedative or anxiety-like effects.
CBD and CBD/THC reduced periorbital mechanical allodynia in a chronic migraine model.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251386794 - Supplemental material for Efficacy of different cannabinoid compounds on migraine-like responses in female rats
Supplemental material, sj-docx-1-cep-10.1177_03331024251386794 for Efficacy of different cannabinoid compounds on migraine-like responses in female rats by Fernanda Mariano Ribeiro da Luz, Alexandre Ottoni Kaup, Darciane Favero Baggio, Flavio Henrique de Rezende Costa and Juliana Geremias Chichorro in Cephalalgia
Footnotes
Author contributions
All authors are listed and have contributed substantially to the manuscript.
Consent for publication
All authors have approved the final version of the manuscript submitted for publication.
Data availability
The data is available from the authors with a reasonable request.
Declaration of conflicting interests
The authors declare that there are no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
All experimental protocols were approved by the Ethics Committee on the Use of Animals (CEUA/BIO/UFPR #1589) and conducted by ARRIVE guidelines.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a grant from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES, Finance Code 001). HealthMeds Pharmaceutical Industry provided material support for the study. CAPES also granted scholarship support for DFB. FMRL is the recipient of a scholarship from CNPq and JGC is the recipient of CNPq research productivity fellowship.
ORCID iDs
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
