Abstract
Background
Pituitary adenylate cyclase-activating polypeptide (PACAP) occurs as either a 27- or 38-amino acid neuropeptide and belongs to the vasoactive intestinal polypeptide/glucagon/secretin family of peptides. PACAP and vasoactive intestinal polypeptide have a 68% homology of their amino acid sequences and share three B-type G-protein coupled receptors: VPAC1, VPAC2 and PAC1 receptors.
Methods/results
The distribution of PACAP-38 and its receptors in the brain is only partly described in the literature. Here, we have performed a study to provide the more general picture of this system in rat brain in order to understand a putative role in primary headaches and partly in relation to the calcitonin gene-related peptide system. We observed a rich expression of PACAP-38 and PAC1 receptor immunoreactivity in many regions throughout the cerebrum, cerebellum and brainstem. The expression pattern points to multiple functions, not least associated with pain and reactions to pain. The expression of VPAC1 and VPAC2 receptor immunoreactivity was very sparse. In several regions such as the cerebral cortex, trigeminal nucleus caudalis, hypothalamus and pons there was a close relation to calcitonin gene-related peptide expression.
Conclusion
The findings suggest that the rich supply of PACAP-38 and PAC1 receptors is associated with basic functional responses in the central nervous system (CNS), and there are important close anatomical relations with calcitonin gene-related peptide in CNS regions associated with migraine pathophysiology.
Abbreviations
Anterior commissure Bed nucleus of the anterior commissure Field CA1 of the hippocampus Field CA3 of the hippocampus Corpus callosum Dentate gyrus Fornix Cerebellar granular layer Habenular commissure Inferior olive Locus coereleus Later leminiscus Lateral septal nucleus Cerebellar molecular layer Motor cortex and somatosensory cortex Medullary reticular nucleus Mesencephalic trigeminal nucleus Paraventricular hypothalamic nucleus Posterior commissure Purkinje cells Pons Posterior thalamic nuclear group Paraventricular thalamic nucleus Reticular thalamic nuclei Septohyopthalamic nucleus Stria medullaris of the thalamus Supraoptic nucleus Spinal trigeminal nucleus Bed nucleus of stria terminalis Tringular septal nucleus Trapezoid body Ventral hippocampal commissure
Introduction
The family of peptides containing pituitary adenylate cyclase-activating polypeptide (PACAP), occurring as either as a 27- or 38-amino acid neuropeptide, and vasoactive intestinal polypeptide (VIP) share an amino acid homology of about 68%. They belong to the VIP/glucagon/secretin family of neuropeptides (1). Originally, VIP was isolated from ovine intestine, and PACAP -27 and -38 from ovine hypothalamus (2). The details of the receptors have emerged over 2 decades and they are described as having many sites of action (3).
The PAC1 receptors are selective for PACAP, whereas VPAC1 and VPAC2 receptors respond to VIP and PACAP with similar high affinity (4). In the periphery, VIP and PACAP play important roles in vasodilation, adrenaline secretion, insulin secretion, and in the control of immunity and inflammation (5) as well as in primary headaches (6).
In the central nervous system (CNS), VIP and PACAP have diverse and important roles, with functions related to the control of sleep and circadian rhythms, learning and memory, anxiety and responses to stress and brain injury (7). PACAP has, in addition, been suggested to have a neuroprotective effect in stroke, in Parkinson’s disease, traumatic brain injury and spinal cord injury (8). In addition, studies have implicated the VPAC2 receptors in susceptibility to schizophrenia and the PAC1 receptors in post-traumatic stress disorder.
In 2002, Hannibal described using immunohistochemistry and in situ hybridization PACAP distribution in the rat brain (9). The development of new specific antibodies towards in particular the receptor subtypes provides new tools to revisit the location of PACAP and its receptors in the CNS, in particular since PACAP and the PAC1 receptors have been implicated in the pathophysiology of migraine and cluster headache (10–12). We focused on PACAP-38 because this peptide has greater expression in intracranial structures, induced migraine effect when infused intravenously while VIP did not (13), and the response to PACAP-27 was less pronounced (14). Here we provide a comprehensive overview of PACAP-38 expression and its three receptors in rat brain. Overall there is an extensive network of nerve fibers that express PACAP-38 and PAC1 receptors. The localization of the VPAC1 and VPAC2 receptors were more focused to a few regions in central structures (Figure 1). In migraine-related regions, we observed co-localizations with the CGRP system in the brain (15,16).
Sagittal cryo-sections stained with Hematoxylin and Eosin. The sixth edition of The rat brain in stereotaxic coordinates by Paxinos and Watson (17) was used to identify the different areas. Blue color denotes PACAP-38, green PAC1, pink VPAC1 and yellow VPAC2 imunoreactivity. PACAP-38 expression was mainly found in frontal and motor cortex, septum/thalamus/hypothalamus and the brainstem. PAC1 immunoreactivity was expressed in more areas than VPAC1 and VPAC2. Areas with both PACAP-38 and either of the receptors were motorcortex, reticular nucleus, pons, cerebellar white matter and mesencephalic trigeminal nucleus.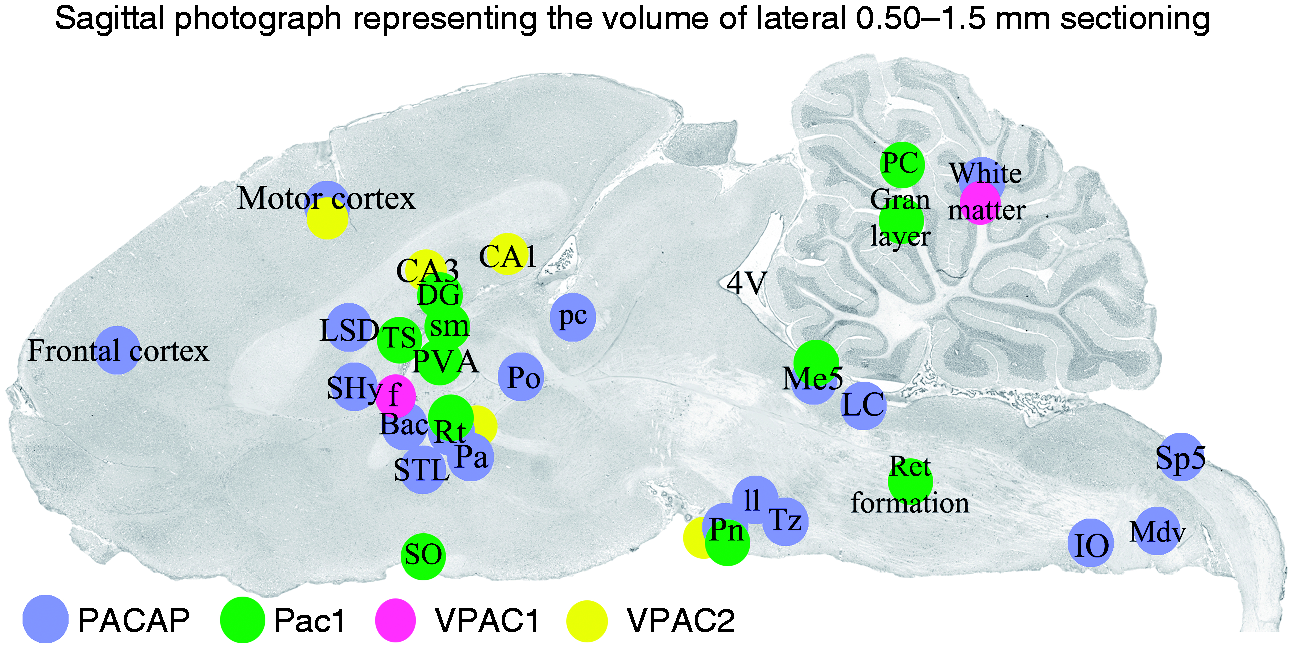
Materials and methods
Ten Wistar male rats (250–320 g) were euthanized by CO2 inhalation followed by decapitation. The brains were carefully dissected, cut sagittal in the midline and placed in 4% paraformaldehyde (PF) for 4 hours, followed by incubation overnight in Sörensen’s phosphate buffer (pH 7.2) containing 10% and 25% sucrose in turn. Thereafter, the tissue was embedded in Yazulla embedding medium (30% egg albumin, 3% gelatin) and cryo-sectioned at 12 µm. The sections were stored at −20℃ until use.
The methods used in the present study are the same as published earlier (15) and hence are suitable in comparison with the expression pattern of the CGRP system. However, the same animals were not used as in the studies previously published, though the methodology was the same.
The study followed the guidelines of the European Communities Council (86/609/ECC) and was approved by the Regional Ethical Committee on Animal Research, Malmö/Lund, Sweden (M43-07).
Hematoxylin-Eosin (HE)
Every 10th slide, with two sagittal cryo-sections of the whole brain including cerebellum, brainstem and C1 spinal cord, were stained using Hematoxylin and Eosin (Htx 4 min, Eosin 1 min). The staining was done in order to examine the morphology and condition of the tissue, and to identify the distance of the section from the midline. The sixth edition of The rat brain in stereotaxic coordinates by Paxinos and Watson (17) and HE staining of sagittal sections spanning over 0.5 mm to 1.5 mm lateral to the midline were used to identify the different areas subjected to the detailed study of the distribution of PACAP-38 and of the receptors (Figure 1).
Immunohistochemistry
Primary and secondary antibodies.

Three different PACAP-38 antibodies were used, one from AMGEN and two from Peninsula Laboratories (see Table 1). The different antibodies all showed consistent and comparable results; however, Peninsulas T-3042 and AMGENs P70670.26 antibodies displayed a clearer and more distinct immunoreactivity and therefore these antibodies were used in the present study. Regarding the PAC1 receptor, two different antibodies were used, one from AMGEN and one from Abcam (see Table 1) (18). The results of the two antibodies were similar; however, the AMGEN antibody showed a more intense immunoreactivity and also a more distinct staining. Therefore, the AMGEN antibody was used in the current presentation. VPAC1 receptor and VPAC2 receptor antibodies were purchased from Santa Cruz and have been used in several publications (19,20).
Omission of primary antibodies served as negative controls. The sections were examined in a light- and epifluorescence microscope (Nikon 80i, Tokyo, Japan) equipped with a motor table, enabling us to get images of a whole section, and with a Nikon DS-2MV camera. Finally, images were processed using Adobe Photoshop CS3 (v0.0 Adobe Systems, Mountain View, CA).
Comparison migraine-related areas, CGRP, and PACAP and its receptors.

Results
HTX Eosin general histology
The brain halves were subjected to immersion fixation with 4% formalin. Since the examination only contained a volume of about 1.5 mm, the tissue fixation was adequate. Every 10th slide of the sagittal sections was subjected to HE staining. The staining showed, in general, excellent morphology. Figure 1 shows a sagittal section, which may well represent the tissues used in the present study.
PACAP-38 positive neurons and fibres
In general, there was an extensive distribution in most parts of the CNS of PACAP-38 expression. This rich distribution of PACAP-38 was found in particular in the cerebrum, cerebellum, septum, thalamus/hypothalamus and brainstem as nerve fibres (Figure 1).
Cerebrum
The cerebral cortex consists of six layers (Layer I–VI). Each layer comprises neuronal cell types that have different genetic, functional and/or structural properties. We did not detect any PACAP-38 immunoreactive cell bodies. However, there was a rich distribution of positive fibres. Cortical fibers can be categorized on the basis of their course and connection into association fibers, transverse fibers and projection fibers. Association fibers connect within the cortex and transverse fibers between the two hemispheres. The largest source of input to the cortex is from the thalamus, which projects to wide areas of cortex. Most regions of the cortex thus receive converging inputs from thalamic nuclei. We observed thin, slender PACAP-38 immunoreactive fibres in layers II–V, both in frontal and in motor and somatosensory cortex (Figure 2(a)). Other cortex regions of the cerebrum displayed no PACAP-38 immunoreactivity.
PACAP-38 immunohistochemistry of frontal and motor cortex, and the cerebellum. (a) In the frontal and motor cortex, thin and slender fibres expressed PACAP-38 in layers II–V. Lower panel shows higher magnification of the immunoreactive fibers. (b) In the cerebellar white matter, a tight mass of PACAP-38 immunoreactive fibers was found. In addition, the granular layer showed immunoreactive fibers. No or very little expression was found in the molecular layer or the Purkinje cells (Pc).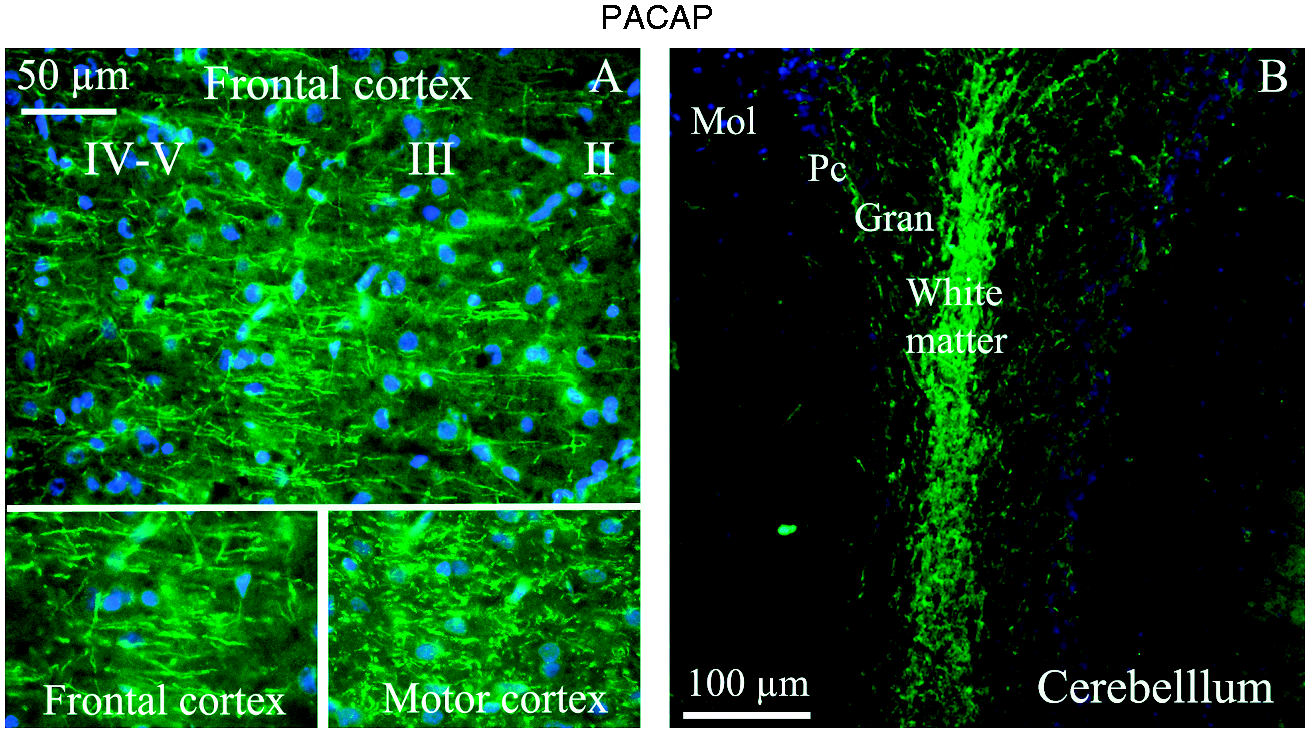
Cerebellum
The cerebellar cortex consists of a highly regular arrangement, the most important cells being the Purkinje cells and the granular cells. Almost all of the output from the cerebellar cortex passes through the deep cerebellar nuclei lying in the cerebellar white matter. Moreover, the cerebellum receives information from the sensory systems, the spinal cord, and other parts of the brain and then regulates, for example, motor movements.
We found that there was a dense tract of PACAP-38 immunoreactive fibres in the white matter in the central parts of cerebellum; however, the cerebellar deep nuclei were not found to be expressing PACAP-38. Furthermore, some positive fibres in the granular layer were also observed. No or very little expression was found in the molecular layer or in the Purkinje cells (Figure 2(b)).
Central CNS structures
Septum
PACAP-38 immunoreactive fibers were observed in the lateral septal nucleus (LSD) with a role in reward and reinforcement function (Figure 3(a)). BAC (Bed nucleus of the anterior commissure, septohypothalamic area) is implicated in the control of anxiety and fear responses (Figure 3(b)), and the septohypothalamic nucleus (SHy) has a key role in enabling human emotions, such as warmth and tenderness (Figure 3(c)). In these nuclei, many PACAP-38 immunoreactive fibers were seen; some of these expressed PACAP-38 in a delicate, pearl-like manner. In addition, we found a few neurons that expressed PACAP-38 in the cytoplasm (Figure 3).
PACAP-38 immunohistochemistry of the lateral septal nucleus (LSD), Bed nu of the anterior commissure (BAC) and the septohypothalamic nucleus (SHy). (a) In LSD, intense immunoreactive fibers were seen, often as a pearl-like structure. Also, occasional neurons expressed PACAP-38 (arrow). (b) As for LSD staining, the pearl-like structure of the immunoreactive fibers was seen in BAC; however, the density of fibers was augmented. It was not obvious if any cell soma immunoreactivity was seen. Both the fornix and anterior commissure are shown in the image. No immunoreactivity was found in these areas. (c) In SHy, thin, slender fibers expressed PACAP-38. These fibers were often observed to have a pearl-like pattern. No neuronal cell soma expression was seen (insert).
Thalamus/hypothalamus
In the thalamus, more specifically the habenular commissure (hbc) is closely linked to reward processing such as encoding negative feedback or negative rewards. PACAP-38 immunoreactivity was found in large PACAP-38 expressing neurons at the border between the commissureand the habenular nuclei (Figure 4(a)). The paraventricular hypothalamic nucleus (Pa or PVN) is well known to be one of two nuclei within the brain that also produce oxytocin. The Pa area displayed both PACAP-38 immunoreactivity fibers and neuronal cell soma (Figure 4(b)). The posterior thalamic nucleus (Po) is involved in visual attention, suppression of irrelevant stimuli and utilization of information to initiate eye movements. PACAP-38 immunoreactivity showed intense expression in slender fibers in the Po (Figure 4(c)).
PACAP-38 immunohistochemistry of the habenular commissure (hbc), paraventricular hypothalamic nucleus (Pa) and posterior thalamic nucleus (Po). (a) hbc PACAP-38 immunoreactivity showed large PACAP-38 expressing neurons at the border between the habenular nuclei and commissure (arrow). (b) Pa displayed both PACAP-38 immunoreactive fibers and neuronal cell soma. The intense immunoreactivity in the Pa area seemed to be limited to approximately 200 µm. (c) The PACAP-38 immunoreactivity in Po was present in very thin and slender fibers.
The activity of the STL (Bed nucleus of stria terminalis) is known to be involved in anxiety responses (Figure 5(a)). The reticular thalamic nucleus (Rt) is a key area putatively involved as a gatekeeper in information flow between the thalamus and cortex, and implicated in attention behavior (Figure 5(b)). Both these nuclear areas (STL and Rt) showed intense networks of PACAP-38 immunopositive fibers. In this network of fibers, large cell soma were embedded. Some of these cell soma were PACAP-38 immunoreactive; however, it was difficult to judge whether immunoreactivity was present in the cell body or in the surrounding network of fibers (Figure 5).
PACAP-38 immunohistochemistry of Bed nu of the stria terminalis (STL) and reticular thalamic nucleus (Rt). (a) and (b) Both nuclear areas (STL and Rt) showed intense fiber PACAP-38 immunoreactivity. In addition, neuronal cell soma displayed PACAP-38 expression.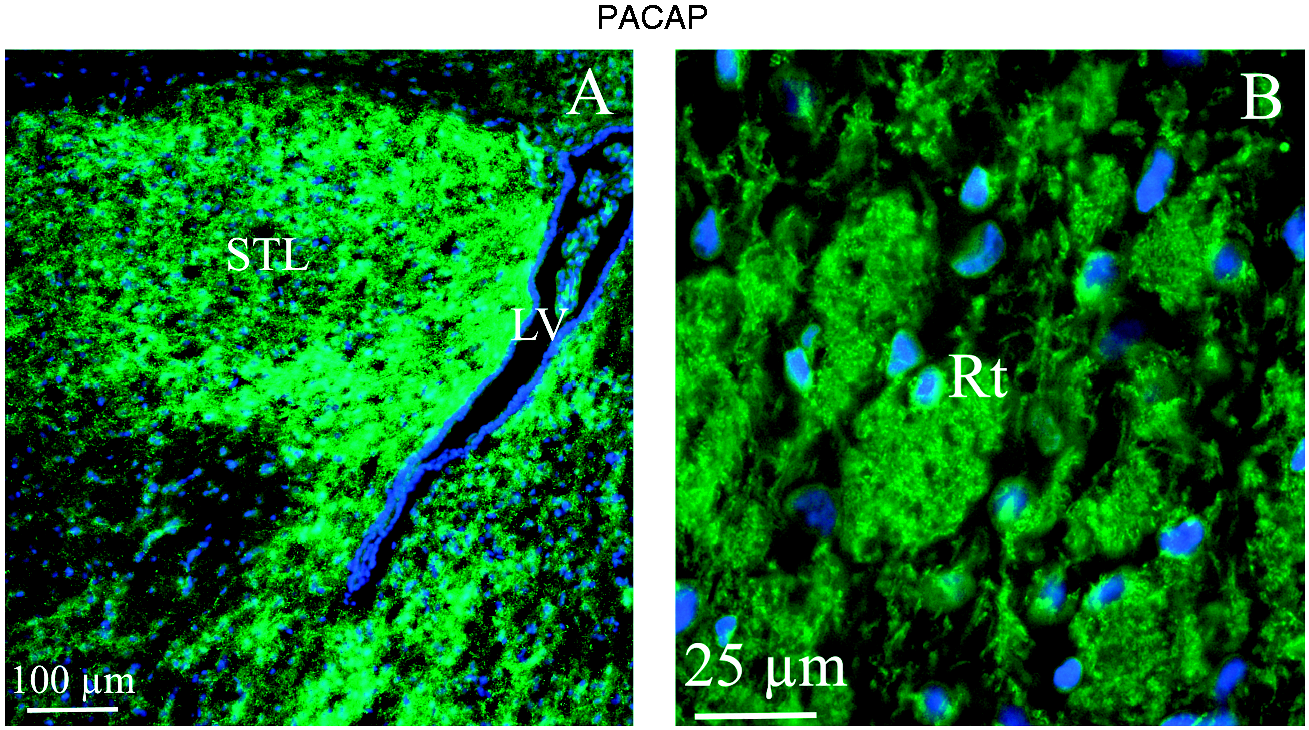
Brainstem
The lateral lemniscus (ll) is a tract of axons in the brainstem that carries information about sound from the cochlear nucleus to various brainstem nuclei. We observed that the ll contained a network of slender PACAP-38 positive fibers (Figure 6(a)) but no cell bodies.
PACAP-38 immunohistochemistry of the lateral lemniscus (ll), Pons (Pn) and mesencephalic trigeminal nucleus (Me5). (a) The lateral lemniscus contained a network of slender PACAP-38 positive fibers resembling the network found in the cortex. (b) In the pons nuclei, PACAP-38 immunoreactive neurons were found. It was among the few nuclei found where the PACAP-38 immunoreactivity was distinctly present in the cell soma (insert). (c) In the mesencephalic trigeminal nucleus, some PACAP-38 immunoreactive pearl-like fibers were observed.
The pons (Pn) provides the cerebellar hemispheres with the majority of their mossy fiber afferents and receives their main input from the cerebral cortex. In different parts of the Pn we found PACAP-38 immunoreactive neurons (Figure 6(b)).
The mesencephalic trigeminal nucleus (Me5) consists of unipolar neurons and the nucleus is the single best-known instance of primary sensory neurons enclosed in the central nervous system instead of in a peripheral sensory ganglion. In the present study, we observed numerous intense PACAP-38 fibers, but no immunoreactive cell soma (Figure 6(c)).
The locus coeruleus (LC) is a reticular nucleus and part of the reticular formation. Signals from the locus coeruleus are involved in mechanisms related to the overall attentiveness level of the brain. The inferior olive (IO) coordinates signals from the spinal cord to the cerebellum to aim to regulate motor coordination and learning. In the LC and IO, intense PACAP-38 immunoreactive fibers and some neuronal cell soma were found (Figure 7(a),(b)).
PACAP-38 immunohistochemistry of the locus coeruleus (LC), inferior olive (IO) and medullary reticular nucleus (MdV). (a) Immunoreactive pearl-like fibers were observed. In addition, some neuronal cell soma expressing PACAP-38 were found. (b) In the inferior olive PACAP-38 immunoreactive fibers and some neuronal cell soma were found. However, many cells nor expressing PACAP-38 were also observed (insert). (c) Neurons expressing PACAP-38 were a frequent finding (arrows). Fibers expressing PACAP-38 were also observed.
The medullary reticular nucleus (MdV) is another region implicated in the modulation of pain. PACAP-38 immunohistochemistry revealed large immunoreactive neurons and, in addition, immunoreactive fibers in this nucleus (Figure 7(c)). The immunoreaction was not as intense as for the IO and LC.
The trapezoid body (tz) is part of the auditory pathway. After nerves from the cochlear nucleus cross over in the trapezoid body and go on to the superior olivary nucleus, they continue from there to the lateral lemniscus. PACAP-38 immunoreactive neuronal fibers were richly displayed in tz (Figure 8(a)).
PACAP-38 immunohistochemistry of trapezoid body (tz) and spinal trigeminal nucleus (Sp5). (a) PACAP-38 immunoreactive neuronal fibers were displayed in the trapezoid body. Insert show a larger magnification of the immunoreactive fibers. (b) In the spinal trigeminal nucleus, there was intense PACAP-38 immunoreactivity in thin fibers; these often showed a pearl-like pattern. The insert shows a higher magnification of the pearl-like pattern.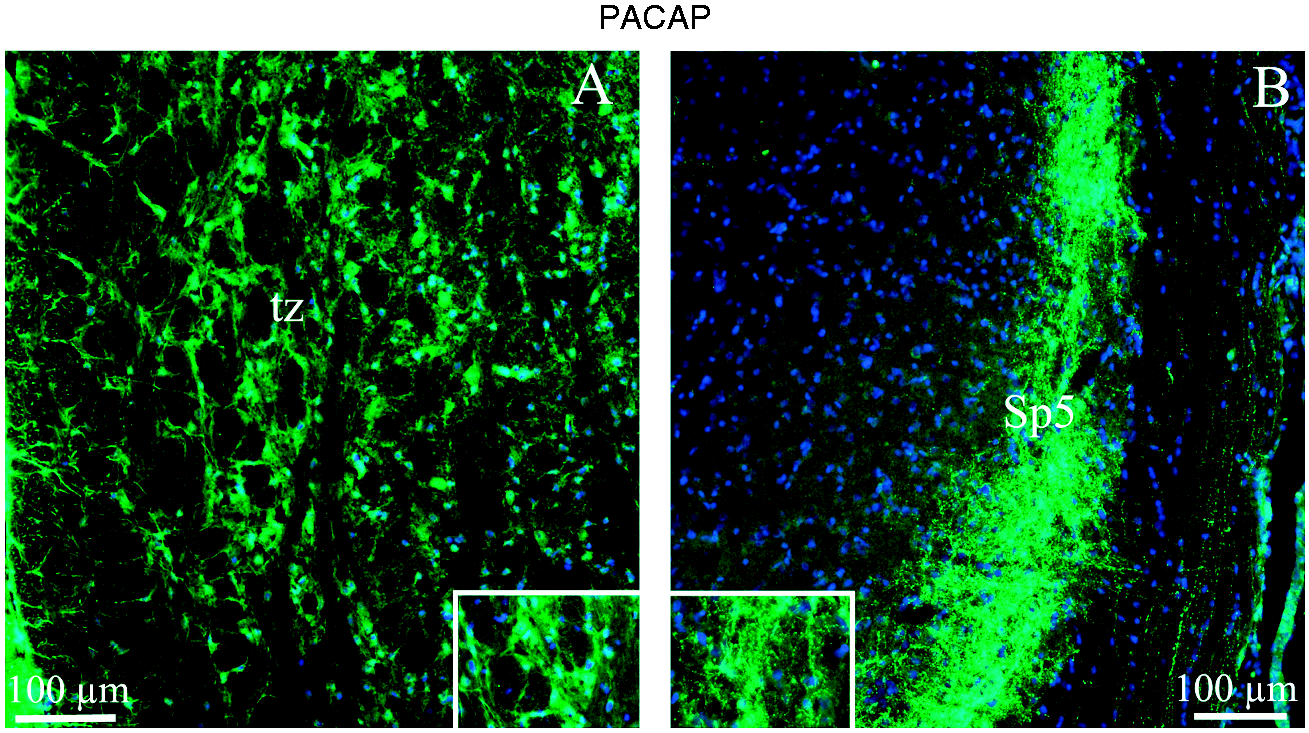
The spinal trigeminal nucleus (Sp5) is a nucleus in the medulla oblongata that receives information about touch, pain and temperature from the face and the head in general. In Sp5, we observed intense PACAP-38 immunoreactivity in thin fibers; these fibers often showed a pearl-like pattern and were located in lamina I–II (Figure 8(a)).
PAC1 receptor positive neurons and fibers
A schematic drawing of the PAC1 expression distribution is shown in Figure 1. In general, there is a rich expression in many regions of the CNS. There was no PAC1 expression either in fibers or in cell bodies of the cerebral cortex.
In the dentate gyrus (DG), PAC1 receptor immunoreactivity was present in the nuclear layers (Figure 9(a)). In addition, in the stria medullaris of the thalamus (sm) intense expression was found in the neuronal cell soma (Figure 9(a)).
PAC1 immunohistochemistry of the dentate gyrus of the hippocampus (DG), the stria medullaris of the thalamus (sm), the triangular septal nucleus (TS), ventral hippocampal commissure (vhc), subfornical organ (SFO) and the paraventricular thalamic nucleus (PVA). (a) In the dentate gyrus, PAC1 immunoreactivity was present in the nuclear layers (arrows). The insert shows a higher magnification of the the dentate gyrus, which reveals that all cells showed PAC1 expression. In addition, in sm (the stria medullaris of the thalamus) intense expression was found in the neuronal cell soma. (b) Intense PAC1 immunoreactivity was found in the fibers and cell soma of the triangular septal nucleus. The insert shows a higher magnification of the triangular septal nucleus. In addition, few fibers in the ventral hippocampal commissure expressed PAC1. On the other hand, in the subfornical organ, intense PAC1 expression was found in the fibers. Expression of PAC1 was very intense in the paraventricular thalamic nucleus, both in fibers and neuronal cell soma.
The triangular septal nucleus (TS) has been shown to heavily innervate the medial habenula. We found intense PAC1 receptor immunoreactivity in fibers within the TS (Figure 9(b)). The ventral hippocampal commissure (vhc) is a large bundle of axons from the pyramidal neurons that connects the right and left hippocampal formations. Few fibers in vhc expressed the PAC1 receptor (Figure 9(b)). On the other hand, the subfornical organ (SFO), thought to be a chemoreceptor zone involved in cardiovascular regulation, showed intense PAC1 receptor expression in nerve fibers (Figure 9(b)). The paraventricular thalamic nucleus (PVA) is particularly sensitive to stressors. Expression of the PAC1 receptor was very intense in PVA, both in fibers and neuronal cell soma (Figure 9(b)).
In the cerebellum, the fibers of the granular layer displayed PAC1 receptor expression, in addition to the surface of the Purkinje cells (PC), either in the PC cell membrane or surrounding glial cells (Figure 10(a)). In the mesencephalic trigeminal nucleus (Me5) fibers showed PAC1 receptor immunoreactivity, but not in the neurons (Figure 10(b)).
PAC1 immunohistochemistry of the cerebellum and mesencephalic trigeminal nucleus. (a) In the cerebellum, the fibers of the granular layer displayed PAC1 expression, in addition to the surface of the Purkinje cells (insert). It could not be determined whether the immunoreactivity was present in the Purkinje cell membrane or surrounding Bergmann glial cells. (b) In the mesencephalic trigeminal nucleus, fibers showed PAC1 immunoreactivity, but not the neurons.
The reticular thalamic nucleus (Rt) is made up of neurons that lie in a complex meshwork of intertwining thalamocortical and corticothalamic axons. It was shown that all these intertwining axons expressed intense PAC1 receptor; in contrast, the neuronal soma were negative to the PAC1 receptor staining (Figure 11(a)). The reticular formation is a diffuse network of nerve pathways in the brainstem connecting the spinal cord, cerebrum, and cerebellum, and mediating the overall level of consciousness. In general, the reticular formation displayed PAC1 receptor expression in the fibers (Figure 11(b)).
PAC1 immunohistochemistry of the reticular thalamic nucleus (Rt) and reticular formation. (a) PAC1 immunohistochemistry showed that all the intertwining axons in the reticular thalamic nucleus expressed intense PAC1, in contrast to the neurons, which were negative to the PAC1 staining (arrow). (b) Reticular formation displayed PAC1 expression in the fibers.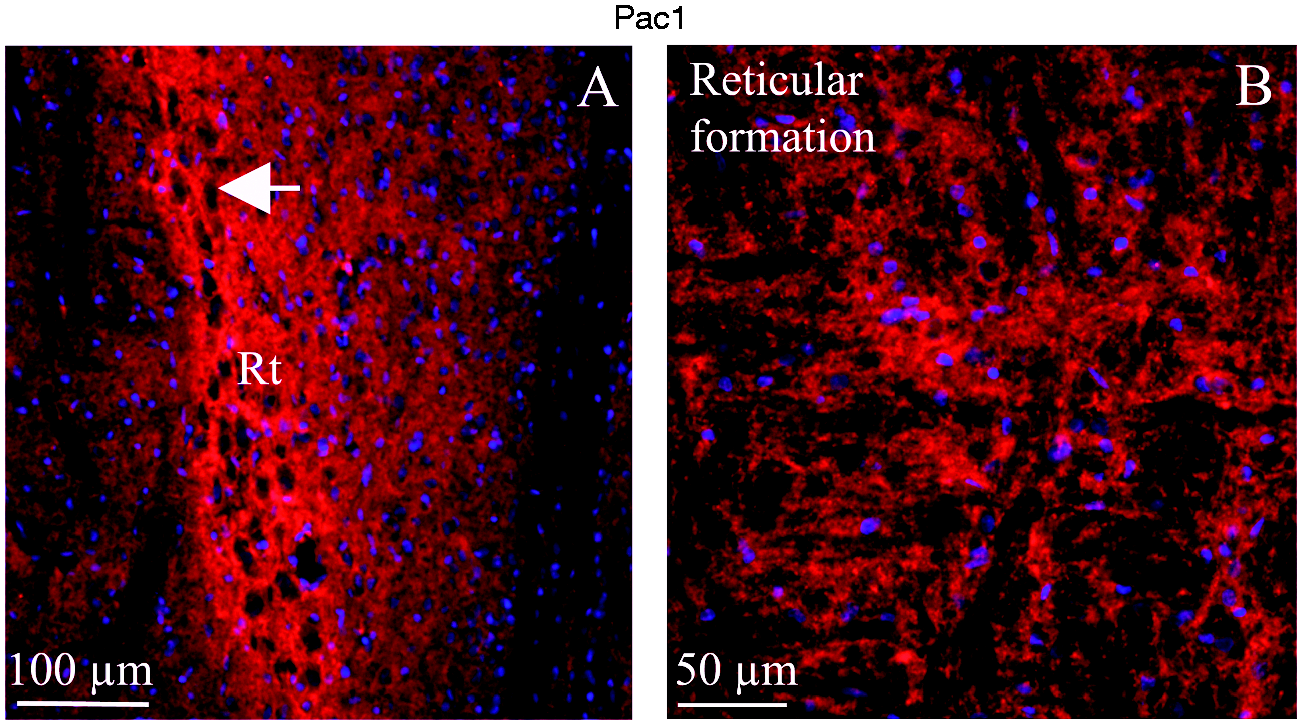
In the supraoptic nucleus (SO or SON), oxytocin is synthesized in magnocellular neurosecretory. PAC1 receptor immunohistochemistry showed expression in the fibers surrounding the neurons in SO (Figure 12(a)). Pn showed a similar pattern, PAC1 receptor expression was found in the nerve fibers surrounding the neurons (Figure 12(b)).
PAC1 immunohistochemistry of the supraoptic nucleus (SO) and Pons (Pn). (a) PAC1 immunohistochemistry showed expression in the fibers surrounding the neurons in the SO (insert). No immunoreactivity was observed in the optic chiasm (opt). (b) In the pons, PAC1 expression in the fibers surrounding the neurons was observed, but also in the cell soma (insert).
VPAC1 receptor positive neurons and fibres
A schematic drawing of the VPAC1 receptor distribution is shown in Figure 1.
VPAC1 receptor expression was only found in the fibers of the fornix (f) (Figure 13(a)), in the fibers of the cerebellar white matter and in the granular layer (Figure 13(b)).
VPAC1 immunohistochemistry of the fornix and cerebellum. (a) In the fornix, VPAC1 expression was found in the fibers (arrow). (b) In the cerebellum, VPAC1 immunohistochemistry was observed in fibers in the cerebellar white matter and in the granular layer (arrow).
VPAC2 receptor positive neurons and fibres
Schematic drawing of the VPAC2 receptor distribution is shown in Figure 1.
The most intense VPAC2 receptor immunoreactivity was found in the pyramidal cells of the hippocampus CA1. Some positive neurons were also found in the pyramidal cells of the cortex, of the hippocampal neurons in CA3, as well as in the Pn and in the reticular nucleus (Rt) (Figure 14).
VPAC2 immunohistochemistry of the hippocampus, cortex, pons and reticular thalamic nucleus. In the hippocampus area CA1, the most extensive and intense expression of VPAC2 was found. The large pyramidal neurons and their extensive elaborated axons and dendrites were VPAC2 immunoreactive. The panel to the right shows areas (the cortex, hippocampal area CA3, pons and reticular thalamic nucleus) where occasional cells showed VPAC2 expression in their cell soma.
Match and mismatch
In some regions of the brain (Figure 1), both PACAP-38 and PACAP receptors were expressed (match). The most complete overlaps were found in the cerebral cortex, Rt, Pn, and Me5. In addition, the hypothalamus was a region where both PACAP-38 and PACAP receptors’ expression was observed. The mismatch was primarily confined to the hippocampus (PACAP receptor expression) and Sp5, Mdv and IO (PACAP-38 expression).
Migraine-related regions, CGRP and PACAP
Several regions in the brain are thought to be involved in migraine (Table 2). In the present study, we compared these regions with CGRP, and PACAP-38 and PACAP receptor expression.
We found that PACAP-38 immunoreactivity coincided with the migraine regions in the cerebral cortex and cerebellum, in the pons, amygdala, thalamus, and hypothalamus, and showed a rich expression of fibers in Sp5 of PACAP-38.
We compared the PACAP-38 and receptors expression with that of our earlier CGRP distribution study (15,16). As expected, CGRP expression coincided with migraine-related regions (Table 2). Consequently, the cerebral cortex, cerebellum, thalamus, hypothalamus, the pons and Sp5 were regions where CGRP and PACAP-38 expression matched well. In addition, in the thalamus, hypothalamus, pons and cerebellum area, PACAP receptor and CGRP expression overlapped to some extent.
Discussion
Pituitary adenylyl cyclase-activating polypeptide (PACAP) and its receptors (PAC1, VPAC1, and VPAC2) have a widespread distribution in brain regions implicated in stress response, reward seeking and aversive responses. PACAP-38 and PAC1 receptors are concentrated at sites within the hypothalamus and the extra-hypothalamic brain, which indicates direct neurotransmitter actions. In these locations, current studies implicate activation in migraine attacks (21). These studies, using fMRI, did not reveal cellular localizations; however, in the present study we found in two important parts, the oxytocin-producing SO (SON) and Pa (PVN) regions, rich expression of PACAP-38 in cell somas and in nerve fibers. These regions are linked with many CNS related behaviors as well as with neuroendocrine regulation and stress responses (22). Notably, the Pa region has a direct connection with the key migraine pain region Sp5 in the lower brainstem (23).
PACAP is a multifunctional neuropeptide that is associated with migraine, much on the basis of release during attacks and after infusion in man (24). Current ongoing clinical trials are taking place with mAbs towards PACAP-38 (12), and with PAC1 receptor mAbs (18). The aim of the present paper was to demonstrate localization of PACAP-38 and PACAP-acting receptors in the CNS to understand possible sites of localization-related centres implicated in primary headaches and thereby add data to answer the question, what is the role of PACAP in migraine?
As shown in Figure 1, there is a concentrated appearance of PACAP-38 and its receptors in the central CNS structures. The hypothalamus-thalamus represents a key area in initiation of migraine, observed in fMRI studies by several groups. May and Schulte described upon daily scanning of migraine for one month that the day before the attack there were already changes in the hypothalamus; consequently, they coined their view as “migraine generator revisited” (21). Subsequently, the connectivity data showed that this activation would spread to the thalamus and to the brainstem during the complete attack (25). The imaging data suggest involvement of many regions. We have previously mapped the CGRP system (15) and now the current and second peptide proposed to be involved in migraine pathophysiology – PACAP.
Several regions in the brain are thought to be involved in migraine: The cortex, thalamus, hypothalamus, amygdala, midbrain, pons, cerebellum, vestibular nucleus and spinal trigeminal nucleus (Table 2). Much of the pertinent PACAP-38 localization is compared with our recently-published findings on CGRP expression (15,16) and migraine-related regions. We found that PACAP-38 immunoreactivity coincided with the migraine regions in the cerebral cortex and cerebellum, in the pons, amygdala, thalamus, and hypothalamus and showed a rich expression of fibers in Sp5 of PACAP-38. Furthermore, PACAP-38 and CGRP expression coincided in the cortex, thalamus, hypothalamus, pons, cerebellum and Sp5. One interesting difference between CGRP and PACAP-38 distribution is that CGRP expression was observed only in the neuronal cell soma, while PACAP-38 expression was mainly seen in fibers. Central regions such as the hypothalamus showed rich expression of PACAP-38 in cell soma as well as in fibers. The neuropeptides should perhaps be discussed in terms of more general effects in opposition to the small amino acids or amines, which are more precise neurotransmitters. However, they are both peptides, which means that they could be delivered to the surrounding tissue whether they are expressed in the cell soma or the fibers. Our reasoning regarding the absence of CGRP-positive fibers (15,16) raised the possibility that CGRP release is not localized within the effector area but rather may be released at a distant location of the receptors, acting as a neurohormone (26). This reasoning might be relevant when it comes to the distribution of PACAP-38 within mainly axons and, in addition, the match and mismatch between PACAP-38 and its receptors.
We observed an extensive network of PACAP-38 positive fibres in frontal and motor/somatosensory cortex, localized to lamina II-V, which resembles the expression of the CGRP receptor component RAMP1 (15,16). Other cortex regions showed some PACAP-38 fibres, but the network was less developed. The largest source of input to the cortex is from the thalamus, which projects to wide areas of cortex. Most regions of the cortex thus receive converging inputs from thalamic nuclei. Interestingly, we observed intense PACAP-38 immunoreactivity in the thalamus-hypothalamus migraine-related region, which agrees with the fiber staining in the cortex. Regarding the receptors, only VPAC2 receptors were seen in the occasional neuronal cell body in the cortex. This is in contrast to findings reported by Joo et al. (27), where both VPAC1 and VPAC2 showed intense staining in layers II–V. Part of these differences might be due to the antibodies used.
Interestingly, the main central part of the cerebellum expressed a massive network of PACAP-38 positive fibres in the central white matter. Other parts had no PACAP-38 expression apart from some occasional fibers in the granular layer. We found no cell bodies with PACAP-38 immunohistochemistry in the cerebellum. We could not confirm the results of Hannibal (9), who reported on PACAP expression in the Purkinje cells. Furthermore, VPAC1 receptor immunoreactivity was seen in the white matter. More interestingly, PAC1 receptors showed a more abundant presence and were in particular located in relation to the Purkinje cells in the folia. The detailed study suggests presence on the outermost part of the Purkinje cell membrane or close to the Bergmann glial cells. A similar distribution pattern was found using antibodies against CGRP receptor RAMP1/CLR (15). However, confocal microscopy revealed that the CGRP receptor was localized not in the Bergmann glial cell, but the Purkinje cell, which could be the case for PAC1 localization as well. However, we know so far little of peptidergic influence on the cerebellar function.
The most extensive and intense expression of VPAC2 was found in the hippocampus area CA1. The large pyramidal neurons and their extensive elaborated axons and dendrites were VPAC2 immunoreactive. In the dentate gyrus, PAC1 immunoreactivity was present in almost all cells in the nuclear layers. The functional role of PACAP-38 and its receptors in the hippocampus is not clear, but this region is often discussed in relation to memory functions.
The brainstem is a centre for much co-ordination and includes many regions of importance in pain modulation. We observed PACAP-38 expression in the pons, which is a large part of the brainstem that includes neural pathways and tracts that conduct signals from the brain down to the cerebellum and medulla, and tracts that carry the sensory signals up though the brainstem into the thalamus.
The lateral lemniscus is a tract of axons in the brainstem that carries information about sound from the cochlear nucleus to various brainstem nuclei. Phonophobia is a symptom sometimes noted in migraine symptomatology. Here we found a delicate network of slender PACAP-38 positive fibers but no cell bodies. The trapezoid body is another part involved in the auditory system pathway. Nerves from the cochlear nucleus cross over in the trapezoid body and pass on into the superior olivary nucleus, from which they continue to the lateral lemniscus. PACAP-38 immunoreactive neuronal fibers were richly displayed in this region. The inferior olive is known to coordinate signals from the spinal cord to the cerebellum to regulate motor coordination and learning. However, recent neurophysiology studies suggest that the IO is an area also involved in pain regulation (28).
The medullary reticular nucleus is possibly implicated in the modulation of pain. PACAP-38 immunohistochemistry revealed large immunoreactive neurons and, in addition, immunoreactive fibers in this nucleus. A more special region in the brainstem is the locus coeruleus. This is a group of cells that are known to be involved in activation, cardiovascular function, and stress reactions, and this is supported by the dense supply of neurons that contain catecholamines and neuropeptide Y. The locus coeruleus is a reticular nucleus and part of the reticular formation. Signals from the locus coeruleus appear to enhance the overall attentiveness level of the brain. We observed a fair supply of PACAP-38 immunoreactive fibers and some neuronal cell soma.
The mesencephalic trigeminal nucleus consists of unipolar neurons and the nucleus is the single known instance of primary sensory neurons enclosed in the central nervous system instead of in a peripheral sensory ganglion. In the present study, we observed numerous intense PACAP-38 fibers. Sp5 is a nucleus in the medulla that receives information about touch, pain and temperature from the face and the head in general. It is the place where the trigeminal nerve ends with its C- and A-fibers to connect to the second order of neurons, which travel upwards in the brainstem. In the Sp5, we observed intense PACAP-38 immunoreactivity in thin fibers; these fibers often showed a pearl-like pattern (29).
The distribution of PACAP-38 receptors in the brainstem contrasts with the peptide localization by itself; only a few were observed. We found some PAC1 receptors in fibres in the pons encircling neurons that were not PACAP-38 positive; this observation did not provide a clear indication of their role. In the pons and in the reticular nucleus we found neurons that were VPAC2 positive. The rest of the brainstem was devoid of positive immunoreaction to VPAC1 and VPAC2 receptor expressions. Immunohistochemical data have shown that PACAP-positive cell bodies and fibers exist in the REM sleep induction zone of the pons, which suggested a role of PACAP in long-term regulation of REMS (30).
As far as is known, PACAP, like other neuropeptides, are found only in large dense-core vesicles. They are released upon high frequency or burst stimulation, unlike classical transmitters that are released in a graded fashion across all stimulation frequencies (31,32). This has important implications for the roles of these neuropeptides and neuropeptides in general, for modulation of synaptic function, especially when co-released with classical neurotransmitters under conditions in which specific neuronal circuits are activated.
We have here described a wide distribution of PACAP-38 and its receptors in the rat brain. The expression suggests multifaceted involvement in many important regions and mechanisms, but these requires separate and detailed study. The involvement of the PACAP system in migraine may be multifaceted and perhaps linked to early activation of the symptomatology due to, for example, stress with behavioral implications. The overall role of neuropeptides in the brain remains to be elucidated, but as a starting point it is important to know the distribution and localization of PACAP and its receptors.
Key findings
PACAP-38 and PAC1 receptor expression was found in many regions throughout the cerebrum, cerebellum and brainstem. VPAC1 and VPAC2 receptor expression was very sparse. Expression pattern points to multiple functions, not least associated with pain and reactions to pain. CGRP and PACAP expression coincided in the cerebral cortex, cerebellum, thalamus, hypothalamus, the pons and Sp5, regions that are said to be migraine related.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Lundbeckfondens Grant of Excellence 2010.
