Abstract
Background
Emerging evidence highlights the role of microRNAs (miRNAs) in epigenetic mechanisms related to migraine pain. The expression of miR-382-5p and miR-34a is higher in serum and peripheral blood mononuclear cells of people with migraine, but limited data is available regarding their possible alteration in other cell subtypes. Several lines of evidence support a monocyte dysfunction in migraine pathophysiology. To gain deeper insights into cell-specific miRNAs expression in migraine individuals with different disease severity, this study aims to determine the expression levels of miR-34a and miR-382-5p in monocytes.
Methods
This cross-sectional, controlled study included 47 participants with episodic migraine (EM, 72.3% females, 41.4 ± 10.7 years), 32 with chronic migraine with medication overuse (CM-MO, 81.3% females, 46.1 ± 10.9 years) and 30 healthy controls (HCs, 66.7% females, 42.9 ± 14.8 years). We assessed interictal monocyte-specific miR-382-5p and miR-34a expression by qRT-PCR, normalizing the expression with U6 RNA (relative quantification - RQ).
Results
miR-382-5p monocytic expression was higher in EM (4.21 ± 1.41 RQ) and CM-MO (6.80 ± 4.37 RQ) when compared to HCs (2.02 ± 0.64 RQ) (p = 0.005 for all comparisons). miR-34a monocytic expression was higher in EM (4.50 ± 1.62 RQ) and CM-MO (6.47 ± 1.87 RQ) when compared to HCs (1.94 ± 0.81 RQ, p = 0.005 for all comparisons). Expression of miR-382-5p and miR-34a were higher in CM-MO when compared to EM (p = 0.005 for both comparisons). After adjusting for age, sex, ongoing preventive medications, presence of anxiety or depressive symptoms, and smoking habit, a logistic regression model confirmed the differences in the monocytic expression of miR-34a and miR-382-5p between EM and CM-MO participants.
Conclusions
Our findings underscore the relevance of miR-34a and miR-382-5p in migraine pathophysiology, as evidenced by their altered expression in monocytes from migraine participants compared to HCs. These miRNAs were also associated with disease severity, being higher in CM-MO when compared to EM individuals.
Trial Registration
The study protocol was registered at ClinicalTrials.gov (NCT05891808)
This is a visual representation of the abstract.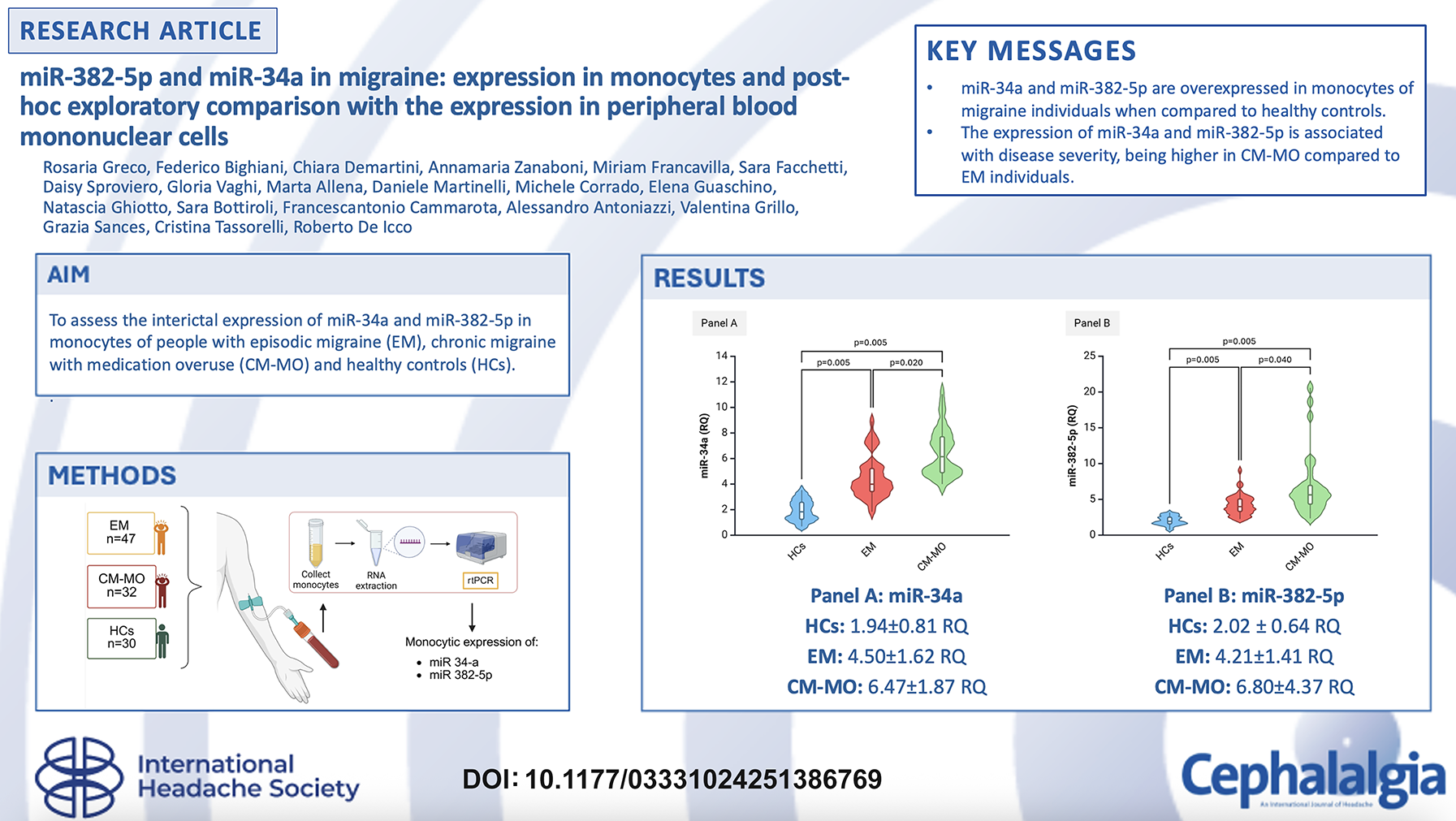
Background
Growing evidence highlights the prominent role of epigenetics in migraine pathophysiology (1). Epigenetic mechanisms include microRNAs expression, histone modifications and DNA methylation, and are responsible for the interaction between endogenous gene expression and environmental factors (1). Among different epigenetic mechanisms, microRNAs (miRNAs) have emerged as particularly intriguing players, due to their potential involvement in both the genesis and perpetuation of chronic pain, including migraine (2–5).
miRNAs are a particular class of endogenous small noncoding RNAs, typically comprising about 22 nucleotides, that orchestrate post-transcriptional regulation of gene expression (6). They achieve this by either promoting messenger RNA (mRNA) degradation or by inhibiting mRNA translation, thereby serving as indicators of environmental influences on gene expression dynamics (6–8).
The identification of miRNAs as potential peripheral migraine markers has been explored in previous studies (1), yielding heterogeneous results. These inconsistencies are mainly due to variations in laboratory procedures, biological matrices, and a high degree of heterogeneity among study groups. Peripheral blood mononuclear cells (PBMCs), considered to be the primary source of miRNAs, comprise different cell types such as monocytes, lymphocytes, and dendritic cells, and little is known about the interaction between different blood cell types and the release of circulating miRNAs (7,9).
Among the explored miRNAs, miR-382-5p and miR-34a appeared to be consistently altered in the serum, peripheral cells, and saliva (10,11) of migraine subjects, suggesting that they may reflect common aspects of the disease. Indeed, serum alterations of miR-382-5p and miR-34a were found in migraine individuals when compared to healthy controls (11). These miRNAs were found elevated during a migraine attack, with miR-382-5p also showing increased levels during the interictal phase (5,11). Interestingly, miR-382-5p and miR-34a expression was associated with the migraine phenotype, being higher in PBMCs of individuals with chronic migraine (CM) compared to episodic migraine (EM). Additionally, the peripheral expression of these two miRNAs varied in response to drug treatments (5,10,12), with changes observed after a short course of calcitonin gene-related peptide (CGRP)-targeting treatments (13).
We recently showed that miR-155 expression in monocytes is associated with migraine and disease severity (14). miR-155 is considered a key regulator of inflammation due to its ability to modulate many inflammatory mediators (15). Monocytic dysfunction has been suggested in migraine. When compared to a control group, migraine individuals exhibited a distinct monocytic inflammatory phenotype accompanied by a reduced peripheral circulating count (16,17). When evaluated in the interictal period, migraine subjects showed a decreased TNF-α expression in monocytes along with increased activity of their nitric oxide synthase (NOS) cyclooxygenase enzymes and prostaglandin E2 release, suggesting an ongoing subclinical inflammatory state (18,19). During the ictal phase, monocytes from migraine individuals showed increased nuclear factor-kappa B (NF-κB) and NOS expression, further emphasizing their contribution to the underlying migraine symptoms (20).
While miR-34a and miR-382-5p have consistently been found altered in serum and PBMCs of migraine subjects, their expression has not yet been evaluated within specific PBMCs subpopulations, such as monocytes.
The comparative analysis of miRNAs expression in different PBMC sub-populations (monocytes, lymphocytes, natural-killers, and dendritic cells) seems interesting as it may help identifying a cellular subtype that is more sensitive to changes and may therefore be more suitable for investigating migraine-related mechanisms.
In the present study, we aimed to provide further insights into the role of miR-34a and miR-382-5p in migraine pathophysiology focusing our attention on their expression in monocytes. We measured the levels of these two microRNAs in monocytes obtained from subjects with EM, CM and medication overuse (CM-MO), and healthy controls (HCs). Additionally, we conducted a secondary exploratory analysis where we compared the levels of microRNAs derived from monocytes with those obtained from PBMCs of a historical cohort in order to assess the informative contribution to migraine mechanisms of the different cell subtypes.
Methods
Study design
This study comprises two separate analyses:
Analysis of miRNAs expression in monocytes: this analysis includes novel, prospectively collected, data for the assessment of the expression of miR-34a and miR-382-5p in monocytes; Comparison between PBMCs vs. monocytes: this section includes a post-hoc and purely exploratory analysis aimed at comparing the expression of miR-34a and miR-382-5p in PBMCs and monocytes.
Analysis of miRNAs expression in monocytes
The monocytes study is a cross-sectional, controlled, study that involves three experimental groups: CM-MO, EM and HCs. The study protocol was registered at ClinicalTrials.gov (NCT05891808) and was approved by the local Ethics Committee (p-20200048922). All participants provided written informed consent.
Study population
Participants with migraine, aged between 18 and 65 years, were consecutively enrolled from those attending the Headache Science & Neurorehabilitation Unit at the IRCCS Mondino Foundation in Pavia, Italy. The inclusion criteria for EM were: i) diagnosis of migraine with or without aura according to ICHD-III criteria (21); ii) migraine frequency between 2 and 14 migraine days per month; iii) negative lifetime history of CM, with a stable episodic pattern for at least 10 years (to reduce the likelihood of transitioning to CM).
For CM-MO, the inclusion criteria were: i) diagnosis of “1.3 chronic migraine” and “8.2 medication overuse headache” as per ICHD-III (21); ii) a documented history of CM for at least one year before enrollment. Inclusion criteria for HCs included: i) absence of any primary or secondary headache diagnosis according to the ICHD-III classification, with the sole exception of sporadic tension-type headache; ii) absence of any other chronic pain conditions.
General exclusion criteria were: i) concomitant diagnosis of neurological disease, major psychiatric disorders according to DSM-V, or other clinically relevant pathologies; ii) pregnant or lactating women; iii) intake of acute drugs within 24 h before blood sampling. Presence of mild anxiety and/or depressive symptoms were not considered as exclusion criteria.
All participants underwent a screening visit, where clinical and demographic data were collected, headache diaries were reviewed, and inclusion/exclusion criteria were confirmed. Eligible subjects were then scheduled for a second visit to complete study procedures, which included: i) fasting peripheral venipuncture for assessing miRNAs expression in monocytes; ii) evaluation of anxiety and/or depressive symptoms by a psychologist from the Headache Science & Neurorehabilitation Unit. Migraine subjects were tested in the interictal phase defined as follows: i) for the EM group: no headache or acute migraine medications intake in the previous 24 hours; ii) for the CM-MO group: no headache, or only a mild headache (less than 3 on a 0 to 10 visual analogue scale), not qualifying as a “migraine day” and not requiring acute migraine medications in the previous 24 h (22).
Blood sample vials from migraine participants and HCs were coded before being sent to the laboratory, ensuring that the biologists conducting the biochemical analyses were blinded to the participant's diagnoses and clinical information.
Laboratory procedures
Peripheral blood mononuclear cells (PBMCs) isolation
Blood samples (18 mL) were collected between 08:00 and 12:00 from the cubital vein of fasting subjects into sterile tubes. The isolation of PBMCs commenced immediately after blood collection. The samples were placed in tubes containing ethylenediamine tetra-acetic acid (EDTA) and diluted at a 1:1 ratio with phosphate-buffered saline (PBS) (Sigma Aldrich, Milan, Italy). This mixture was then carefully layered onto 15 mL of Ficoll separating solution (Sigma Aldrich, Milan, Italy) and centrifuged at 800 g for 30 min at room temperature. The PBMCs formed a distinct white layer in the middle, which was collected and washed twice in sterile PBS at 300 g for 15 min each.
Monocyte isolation
Monocytes were isolated from the previously collected PBMCs using the Pan Monocyte Isolation Kit and a MidiMACS Separator (Miltenyi Biotec). Hence, PBMCs were resuspended in PBS containing 0.5% bovine serum albumin and 2 mM EDTA. Non-monocytes, including lymphocytes, granulocytes, dendritic cells and NK cells were indirectly magnetically labeled with a cocktail of biotin-conjugated antibodies, an FcR blocking reagent and Anti-Biotin MicroBeads. The magnetically labeled cells were then depleted, allowing for the isolation of high-purity unlabeled monocytes. Thereafter, monocytes were washed with buffer, pelleted by centrifugation and resuspended in trizol for further analysis via RT-PCR.
RNA extraction and miRNAs analysis
Total RNA including all small non-coding RNAs, was extracted from cell pellets within two weeks using the Direct-zol RNA Mini Prep Plus kit (Zymo Research, Aurogene, Rome, Italy). The quality of the extracted RNA was assessed with a NanoDrop Spectrophotometer (Nanodro Thermo Fisher Scientific, Euroclone, Milan, Italy), ensuring an optical density (OD) 260/280 ratio of ≥ 1.9 and an OD 260/230 ratio of ≥ 1.5. To reduce the impact of any differences in the miRNA expression analysis, the same amount of total RNA (1μg) was used for reverse transcription and an equal volume of cDNA (2μl) for qPCR across all samples.
cDNA synthesis and miRNAs quantification
Synthesis of cDNA was performed using the MirX miRNA First Strand Synthesis Kit (Takara-Diatech Labline, Jesi-Ancona, Italy). Quantitative real-time PCR was performed using TB Green qRT-PCR kit (Takara Bio USA, Inc., Diatech Labline, Jesi-Ancona, Italy) was then employed to measure the expression levels of miR-34a and miR-382-5p. The expression of these miRNAs was normalized using U6, a small nuclear RNA, as the housekeeping gene. All miRNA primers were selected using the Prime3 software and synthesized by Sigma Aldrich (Milan, Italy). The thermocycling conditions were as follows: initial denaturation at 95°C for 10 min, followed by 40 cycles of 15 s at 95°C, and 1 min at 60°C. These reactions were performed using a Light Cycler 480 Instrument RT-PCR Detection System (Roche, Milan, Italy). The Real-time PCR results were expressed as 2−ΔCt values, where ΔCt was calculated by subtracting the Ct of the reference gene from the Ct of the target miRNA (ΔCt = Ct_target−Ct_reference) (relative quantification - RQ). To correct for batch effects arising from different PCR plate runs, we applied the ComBat algorithm using the sva (surrogate variable analysis) package in R within the same cellular population (in PBMC or in monocytes). To improve readability, all real-time PCR-derived values were uniformly multiplied by 1000, which did not affect relative quantification or statistical comparisons.
Comparison between PBMCs vs. monocytes
In the post-hoc PBMCs vs. monocytes, data from the monocytic population were compared with miRNAs expression in PBMCs. miR-34a and miR-382-5p expression in PBMCs were sourced from a historical cohort of our group, which was previously published (see Greco et al. (23) for details). The previous study did not include a control group formed by HCs, thus the post-hoc indirect comparison was performed between EM and CM-MO. With the limit of an indirect comparison, the inclusion/exclusion criteria, the study design, and the laboratory procedures were comparable between the present and previously published studies. A comparison of the methodologies adopted in the two studies is presented in supplementary materials (Online Supplementary Table 1).
Statistical analysis
For the monocytes analysis, sample size calculation was performed considering our previous findings regarding microRNAs expression in monocytes (14). The sample size was computed with www.openepi.com and pwr2 package. The primary objective was to compare miR-382-5p expression in monocytes between EM and CM-MO groups. Thus, the following parameters were used: power of 80%; significance level: 95%; microRNAs expression of 2.7 ± 2.4 RQin CM-MO and 1.4 ± 0.8 RQ in EM group. The minimum suggested sample size was 60 participants (30 for CM-MO group and 30 for EM group). Considering possible variability, we enrolled a final population of 85 migraine subjects.
Statistical analysis was conducted with the SPSS software, ver. 21 (IBM Corp., USA) and with “R: A language and environment for statistical computing” (R Foundation for Statistical Computing, Vienna, Austria), Version 1.2.5033, for Windows. Shapiro-Wilk test showed a non-normal distribution of the biochemical variables; therefore, we used non-parametric tests for all the analyses.
Categorical variables were compared among groups with Chi-square test or Fisher exact test. For continuous variables, differences among groups were tested with non-parametric tests (the Mann–Whitney U test or the Kruskal–Wallis test). Spearman test was used to measure the correlations between biochemical and clinical variables. To assess monocytic expression of miRNAs, we performed three separate ROC curve analyses (HCs vs. EM, HCs vs. EM, EM vs. CM-MO) evaluating the area under the curve with 95% confidence interval, the level of significance against the reference line and the optimal cut-off point for differentiating between the two groups. To assess the best cut-off value, we considered the RQ with the maximized Youden index (“sensitivity + specificity−1”). We completed the analysis with a binomial logistic regression with “EM vs. CM-MO (reference category)” as dependent variable, age, miR-382-5p, and miR-34a as continuous covariates, and sex, presence of anxiety symptoms, presence of depressive symptoms, ongoing prevention, and smoking habit as categorical factors. The HCs group could not be included in the logistic regression analysis because of the pronounced difference in miRNAs expression between HCs and migraine groups, which would have resulted in an overfitting of the model. The goodness of fit of the final model was assessed with a Hosmer and Lemeshow test.
For the post-hoc analysis, differences in continuous variables were assessed with a general linear model with two between-subjects factors: factor CELL (2 levels: monocyte vs. PBMCs) and factor GROUP (2 levels: EM vs. CM-MO).
For all of the previous analyses, the level of statistical significance was further corrected using the Bonferroni method to account for multiple comparisons.
Statistical significance was set at α < 0.050.
Results
Analysis of miRNAs expression in monocytes
Clinical and demographic features of the study population
We enrolled a total population of 50 EM subjects, 35 CM-MO subjects and 30 HCs. Following batch correction, three EM and three CM-MO participants exhibited high variability in miRNAs expression and were identified as outliers and excluded from the final analysis.
The final study population included 47 EM subjects (72.3% female; age 41.4 ± 10.7 years), 32 CM-MO subjects (81.3% female; age 46.1 ± 10.9 years), and 30 HCs (66.7% female; mean age 42.9 ± 14.8 years). Age and sex distributions were comparable among the three groups (H(df = 2): 2.780; p = 0.249 and χ2(df = 2): 1.733; p = 0.420, respectively).
EM participants experienced an average of 6.0 ± 3.7 monthly migraine days (MMDs) and reported using 7.0 ± 5.2 doses of acute migraine medications per month. CM-MO participants experienced an average of 22.4 ± 5.9 MMDs. They reported using 34.6 ± 19.5 doses of acute migraine medication per month. The prevalence of ongoing pharmacological prevention was comparable between EM (27.7%) and CM-MO (31.3%) groups (χ2(df = 1): 0.119; p = 0.803). However, CM-MO participants had a higher rate of other comorbidities (56.3%) compared to EM participants (36.2%) and HCs (6.7%) (χ2(df = 2): 17.160; p = 0.001). The most prevalent (>5%) comorbidities were: asthma (6.3%), hypertension (7.6%), and thyroid disorders (7.6%). Detailed clinical and demographic characteristics are presented in Table 1.
Clinical and demographic characteristics of the study population.

CM-MO, chronic migraine with medication overuse; EM, episodic migraine; HCs, healthy controls; MHDs, monthly headache days; MMDs, monthly migraine days; NSAIDs, nonsteroidal anti-inflammatory drugs; NRS, numeric rating scale; MIDAS, migraine disability assessment score questionnaire; HIT-6, headache impact test; ASC-12, allodynia symptom checklist. A previous smoking habit is defined as a positive lifetime history of smoking, but no active smoking in the previous 12 months. Continuous variables are reported as mean ± standard deviation.
miR-34a and miR-382-5p expression in monocytes
We found a difference in the expression of miR-382-5p and miR-34a in monocytes among the three study groups (H(df = 2): 61.171; p = 0.005 for miR-382-5p and H(df = 2): 68.295; p = 0.005 for miR-34a). Specifically, miR-382-5p and miR-34a expression in monocytes of CM-MO participants (6.80 ± 4.37 RQ and 6.47 ± 1.87 RQ, respectively) was higher when compared to EM (4.50 ± 1.62 RQ, p = 0.020 for miR-34a and 4.21 ± 1.41 RQ, p = 0.040 for miR-382-5p) and HCs (1.94 ± 0.81 RQ, p = 0.005 for miR-34a and 2.02 ± 0.64 RQ, p = 0.005 for miR-382-5p). Furthermore, miRNAs levels differentiated EM participants from HCs, being lower in the latter (p = 0.005 for both miR-34a and miR-382) (Figure 1).

miR-34a and miR-382-5p expression levels in peripheral blood monocytes among the three study groups. CM-MO, chronic migraine with medication overuse; EM, episodic migraine; HCs, healthy controls; RQ, relative quantification: 2−ΔCt = 2−(ΔCt = Ct_target−Ct_reference); Ct cycle threshold. Panel A: expression of miR-34a in monocytes in HCs, EM and CM-MO groups. Panel B: expression of miR-382-5p in monocytes in HCs, EM and CM-MO groups. Violin-plot: the violine plot includes the median (horizontal line), the interquartile range (light colored boxes), and minimum and maximum values (whiskers). The area of the violin plot depicts the density of data distribution. Kruskal-Wallis Test was used for intergroup comparisons.
In the CM-MO group, 13 out of 32 participants (40.6%) exhibited a mild headache pain during blood samples; There was no difference in the expression of miR-382-5p and miR-34a between CM-MO participants who experienced mild headache pain and those who did not (U(n = 13,19): 87.000; p = 0.170 for miR-382-5p, and U(n = 13,19): 138.000; p = 0.215 for miR-34a).
miR-382-5p expression positively correlated with monthly headache days (MHDs) (Spearman's rho: 0.266, p = 0.020), MMDs (Spearman's rho: 0.286, p = 0.012), days of acute drugs intake (Spearman's rho: 0.264, p = 0.021) (Online Supplementary Table 2). miR-34a expression positively correlated with MHDs (Spearman's rho: 0.536, p = 0.001), MMDs (Spearman's rho: 0.567, p = 0.001), days of acute drug intake (Spearman's rho: 0.546, p = 0.001) (Online Supplementary Table 2).
ROC curve analysis demonstrated that miR-34a and miR-382-5p were able to discriminate between HCs and EM (AUC: 0.950, p = 0.001 for miR-34a, and 0.940, p = 0.001 for miR-382-5p), HCs and CM-MO (AUC: 1.000, p = 0.001 for miR-34a, and 0.979, p = 0.015 for miR-382–5p), as well as EM and CM-MO (AUC: 0.800, p = 0.001 for miR-34a, and 0.725, p = 0.001 for miR-382-5p) (Figure 2).

Receiver operating characteristic (ROC) curves for miR-34a and miR-382-5p expression. CM-MO, chronic migraine with medication overuse; EM, episodic migraine; HCs, healthy controls; RQ, relative quantification. Panel A: ROC curve for HCs vs. EM (reference category) comparison. Panel B: ROC curve for HCs vs. CM-MO (reference category) comparison. Panel C: ROC curve for EM vs. CM-MO (reference category) comparison. For each comparison, we reported the area under the curve (Area) with the 95% confidence interval, and the p-value comparing the single ROC curve with the reference line. For each comparison, we reported the best cut-off that identifies positive cases and minimizes misclassification of negative cases. The best cut-off was defined according to Youden index (“sensitivity + specificity−1”).
In the adjusted logistic regression model (which accounted for confounding factors including age, sex, ongoing preventive medications, anxious symptoms, depressive symptoms and smoking habits) the monocytic miR-34a and miR-382-5p expression remained associated with the migraine phenotype. The adjusted odds ratio of being CM-MO was 1.669 [1.075–2.590] (p = 0.023) for a 1 RQ step increase in miR-34a, and 1.670 [1.098–2.539] (p = 0.017) for a 1 RQ step increase in miR-382-5p (Nagelkerke R Square: 0.619; Hosmer and Lemeshow Test: χ2(df = 8): 14.301; p = 0.074) (Table 2 and Online Supplementary Table 3).
Results of logistic regression model comparing episodic migraine (EM) to chronic migraine with medication overuse (CM-MO).
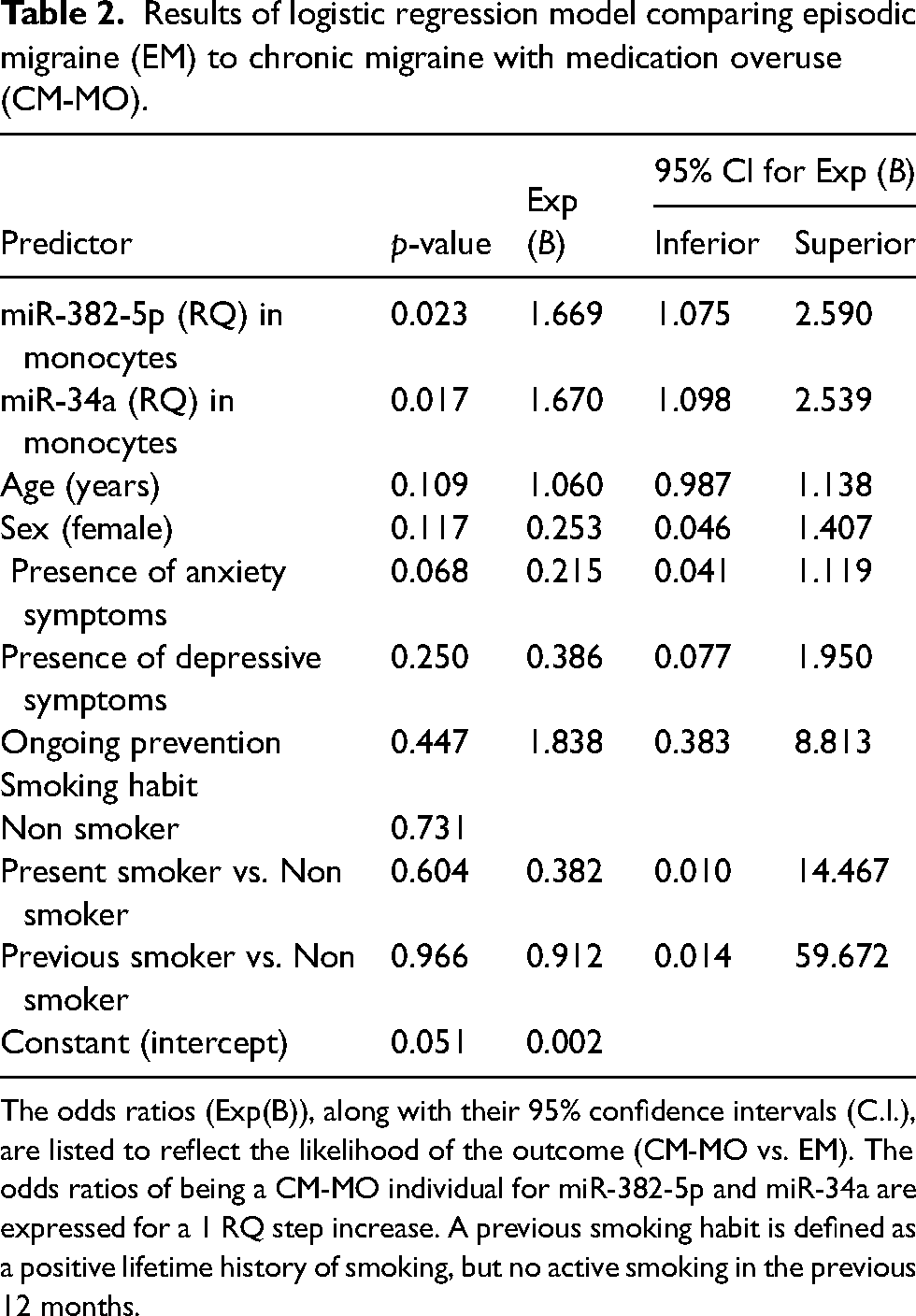
The odds ratios (Exp(B)), along with their 95% confidence intervals (C.I.), are listed to reflect the likelihood of the outcome (CM-MO vs. EM). The odds ratios of being a CM-MO individual for miR-382-5p and miR-34a are expressed for a 1 RQ step increase. A previous smoking habit is defined as a positive lifetime history of smoking, but no active smoking in the previous 12 months.
Comparison between PBMCs vs. monocytes
Clinical and demographic features of the two cohorts
A detailed description of the population analyzed for miRNAs expression in PBMCs has been published elsewhere (23). Following batch correction, two CM-MO participants from the historical cohort exhibited high variability in miRNAs expression, resulting in their classification as outliers and exclusion from the final analysis. A comparison between the historical population in which miRNAs expression was analyzed in PBMCs and the present population in which miRNAs expression level was assessed in monocytes is shown in Table 3. Age, gender, current preventive medication, and migraine characteristics were comparable between the study populations.
Clinical and demographic features of the populations used for the post-hoc analysis.

PBMCs, peripheral blood mononuclear cells; CM-MO, chronic migraine with medication overuse; EM, episodic migraine; HCs, healthy controls; MHDs, monthly headache days; MMDs, monthly migraine days; NSAIDs, nonsteroidal anti-inflammatory drugs; NRS, numeric rating scale; MIDAS, migraine disability assessment score questionnaire; HIT-6, headache impact test; ASC-12, allodynia symptom checklist. Continuous variables are reported as mean ± standard deviation. GLM: two factors generalized linear model. Factor CELL comparison between monocyte and PBMCs, factor GROUP comparison between EM and CM-MO. *: chi-square test.
Post-hoc comparison of miR-34a and miR-382-5p expression between monocytes and PBMCs
The expression of miR-382-5p was significantly higher in PBMCs compared to monocytes (21.20 ± 14.70 RQ vs. 5.29 ± 3.26 RQ; factor CELL: F(1,123): 124.491; p = 0.001). Independently from the PBMCs or monocyte cohorts, miR-382-5p expression was confirmed to be elevated in participants with CM-MO (monocytes: 6.80 ± 4.37; PBMCs: 30.11 ± 14.53) compared to those with EM (monocytes: 4.21 ± 1.41; PBMCs: 13.61 ± 9.92) (factor GROUP: F(1,123): 42.42; p = 0.001). We described a significant CELLxGROUP interaction (F(1,123): 22.506; p = 0.001), suggesting that the increased expression of miR-382 in PBMCs was mainly driven by the higher levels observed in CM-MO subjects (Figure 3).

miR34a and miR-382 expression levels in monocytes and PBMCs in EM and CM-MO groups. CM-MO, chronic migraine with medication overuse; EM, episodic migraine; PBMCs, peripheral blood mononuclear cells; RQ, relative quantification: 2−ΔCt = 2−(ΔCt = Ct_target−Ct_reference); Ct, cycle threshold. Panel A: expression of miR-34a in monocytes and PBMCs in EM and CM-MO groups. Panel B: expression of miR-382-5p in monocytes and PBMCs in EM and CM-MO groups. Circles and whiskers depict mean ± standard deviation.
The expression level of miR-34a was higher in PBMCs compared to monocytes (87.82 ± 93.87 RQ vs. 5.28 ± 1.97 RQ; factor CELL: F(1,113): 86.227; p = 0.001). Independently from the PBMCs or monocyte cohorts, miR-34a expression was higher in participants with CM-MO (monocytes: 6.47 ± 1.87; PBMCs: 142.56 ± 104.79) compared to the EM (monocytes: 4.49 ± 1.63; PBMCs: 33.08 ± 28.12) (factor GROUP: F(1,113): 39.498; p = 0.001). We described a significant CELLxGROUP interaction (F(1,113): 36.749; p = 0.001) (Figure 3). This suggests that the increased expression of miR-34a in PBMCs was mainly driven by the higher levels observed in CM-MO participants.
Discussion
The possible roles of miRNAs in migraine diagnosis and treatment have lately been investigated, although the therapeutic implications of the findings need to be explained and proven (24).
The present study aimed to evaluate the expression of miR-34a and miR-382-5p in monocytes of migraine subjects. The key findings may be summarized as follows: i) expression of miR-34a and miR-382-5p in monocytes of migraine subjects was higher compared to HCs; ii) CM-MO participants had higher expression of both miRNAs when compared to EM; iii) miR-34a and miR-382-5p were confirmed as an independent predictor of the CM-MO phenotype in a multivariate regression analysis corrected for several possible confounders. In a secondary purely explorative analysis, we observed that expression of both miR-34a and miR-382-5p was higher in PBMCs when compared to monocytes, in particular in the CM-MO group. This secondary post-hoc analysis was conducted using an historical cohort from our center. It should therefore be interpreted solely as generating hypotheses for future studies (23).
This study further supports the involvement of epigenetic mechanisms in migraine pathophysiology. In particular, our findings reinforce the role of miR-34a and miR-382-5p by demonstrating differential monocytic expression between individuals with migraine and HCs. Additionally, a ROC curve analysis proved that miR-34a and miR-382-5p monocytic expression was able to discriminate between migraine participants and healthy controls with good accuracy. Indeed, the area under the ROC curves was above 0.90 for both EM and CM-MO when compared to healthy controls. This aligns with previous literature, which reported increased serum levels of these miRNAs during spontaneous migraine attacks, with miR-382-5p also elevated during the interictal period when compared to HCs (25). We have previously characterized the interictal expression of miR-34a and miR-382-5p in PBMCs of individuals with EM and CM-MO (23). We found that miR-382-5p and miR-34a expression levels effectively discriminated between EM and CM-MO phenotypes, with higher expression in the chronic group. Noteworthy, this association remained significant after adjustment for age, gender, disease duration, and plasma CGRP levels, further supporting the potential role of these miRNAs in migraine pathophysiology.
miR-34a and miR-382-5p were also studied as possible markers of the therapeutic response. miR-34a was found lower in the saliva of migraine subjects after acute treatment (25). In CM-MO subjects, miR-382-5p and miR-34a expression in PBMCs was reduced in both responders and non-responder to a detoxification protocol, thus lacking association with clinical improvement (23). In a population of CM-MO individuals, miR-382-5p and miR-34a levels were suppressed by a three-month course of treatment with erenumab, a monoclonal antibody targeting CGRP (12). In line with previous results, miRNAs reduction was not associated with clinical outcomes, being comparable between responders and non-responders to erenumab.
From a mechanistic point of view, several genes involved in pain transmission and soluble factor homeostasis are predicted targets of miR-382-5p and miR-34a and are of interest from a migraine pathophysiological perspective. These include various subunits of the GABAA and GABAB receptors, the ion-dependent GABA transporter (SLC6A1), and Interleukin 10 receptor alpha subunit (IL10RA) (11). miR-34a expression is also correlated with pro-inflammatory cytokine IL-1B levels (26). With these premises, the overexpression of miR-382-5p and miR-34a in people with migraine may contribute to a reduction in the GABA inhibitory signals and to a promotion toward a pro-inflammatory state.
The present study was completed by a secondary, post-hoc, analysis in which we compared miR-34a and miR-382-5p expression in monocytes and PBMCs. This was achieved by using a cohort of subjects previously enrolled in Greco et al. (23). We interpreted the results of the post-hoc approach as “hypothesis generating”, without attributing a possible biological role. With these premises, two intriguing observations can be discussed: i) miRNAs expression was confirmed to be higher in CM-MO participants compared to EM, regardless of the cell type, and ii) miRNAs expression was higher in PBMCs when compared to monocytes, with this difference being mainly explained by a pronounced alteration in the CM-MO group.
This latter finding is particularly intriguing, as it suggests that CM-MO individuals may experience upregulation of these miRNAs in immune cell subpopulations besides monocytes. PBMCs include various subtypes, with lymphocytes (T cells, B cells, and natural killer cells) corresponding to the 70–90% of the total, monocytes approximately 10–20%, and dendritic cells (∼1%) (27). It is therefore reasonable to hypothesize that the lymphocyte population may represent the immune cell population that affects the discrepancy of miRNAs expression in PBMCs and monocytes.
Previous scientific evidence indicated T cell alterations in migraine, namely an imbalance between CD4 + and CD8+ T cell thought to cause neuroinflammation and sensitization (28). T helper (CD 4+) lymphocytes were higher during the interictal period (28) while cytotoxic T lymphocytes and in regulatory T cells (29) were lower when compared to HCs, suggesting a polarization towards a pro-inflammatory humoral immune response (28,30). In this context, miR-34a may play a key role in regulating T-cell activation, by targeting multiple modulators of NF-κB signaling and affecting CD8+ T-cell function (31). Numerous studies demonstrated elevated circulating levels of proinflammatory cytokines (IL-1β (20,32–38), TNF-α (32,34,35,39–41) and IL-6 (32,35,37–39,42–44)) accompanied by a reduction in mediators which downregulate the immune response (IL-10 (37,41,45)) in migraine individuals compared to HCs. Our group has shown that the gene expression levels of both pro-inflammatory and anti-inflammatory cytokines in monocytes are altered in people with migraine compared to HCs (14). Notably, for cytokines such as TNF-α and IL-10, these alterations followed a gradient across the migraine spectrum, with more pronounced changes observed in people with CM-MO compared to EM subjects (14). As inflammation is thought to play a pivotal role in migraine, upstream epigenetic processes mediated by miRNAs are likely crucial (46–48).
Limitations of study
One limitation to consider in our study is the enrollment of individuals with CM-MO, as the overuse of acute anti-migraine medications could potentially impact the observed results. However, we aligned our approach with previous studies by our group to maintain consistency. Additionally, we acknowledge that confirmatory studies are required to bolster our findings. As mentioned, we performed a post-hoc analysis using a historical cohort to compare miRNAs expression in monocytes versus PBMCs. This approach seemed acceptable because clinical, laboratory, and methodological settings were comparable between the two studies, but caution is advised in the interpretation of results (see online supplementary materials for a detailed comparison). It is important to note that this post-hoc analysis involved comparing a novel population with historical data from our laboratory. This approach has several limitations: the comparison was indirect, and several confounders may account for the observed differences. In addition, although the analyses were methodologically comparable, they were performed at different times and using different batches, which limits the possibility of performing a normalization analysis. Finally, in the PBMC cohort, the RQ values were characterized by pronounced variability.
Given these limitations, these findings should be replicated and confirmed in future by a well-designed prospective study that aims to assess epigenetic differences among different PBMCs subtypes. Moreover, our study focused on monocytes and total PBMCs without directly examining the other subpopulations, nor did we consider other types of fluids, such as plasma, serum, or cerebrospinal fluid. The comparative analysis of microRNAs expression in different PBMCs subpopulations (monocytes, lymphocytes, natural killer cells and dendritic cells) is interesting, as it could help to identify a cellular subtype that is more sensitive to changes and therefore more suitable for investigating migraine-related mechanisms.
Conclusion
To improve diagnosis and personalize treatment strategies, the search for reliable and validated biomarkers for migraine is essential. Our study strengthened the role of epigenetic mechanisms and a dysregulation of miR-34a and miR-382-5p in migraine pathophysiology, by confirming their altered expression in monocytes and their association with disease severity. Although promising, these miRNAs face limitations due to non-specificity and technical challenges in analysis.
miR-34a and miR-382-5p are overexpressed in monocytes of migraine individuals when compared to healthy controls, highlighting their potential role in migraine pathophysiology. The expression of miR-34a and miR-382-5p is associated with disease severity, being higher in CM-MO compared to EM individuals. miR-34a and miR-382-5p show higher expression in PBMCs compared to monocytes, with this difference being largely driven by CM-MO-related alterations, emphasizing the need for cell-specific analyses.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251386769 - Supplemental material for miR-382-5p and miR-34a in migraine: Expression in monocytes and a post-hoc exploratory comparison with expression in peripheral blood mononuclear cells
Supplemental material, sj-docx-1-cep-10.1177_03331024251386769 for miR-382-5p and miR-34a in migraine: Expression in monocytes and a post-hoc exploratory comparison with expression in peripheral blood mononuclear cells by Rosaria Greco, Federico Bighiani, Chiara Demartini, Annamaria Zanaboni, Miriam Francavilla, Sara Facchetti, Daisy Sproviero, Gloria Vaghi, Marta Allena, Daniele Martinelli, Michele Corrado, Elena Guaschino, Natascia Ghiotto, Sara Bottiroli, Francescantonio Cammarota, Alessandro Antoniazzi, Valentina Grillo, Grazia Sances, Cristina Tassorelli, and Roberto De Icco in Cephalalgia
Footnotes
Acknowledgments
The authors thank the Research Nurse Team of the Headache Science & Neurorehabilitation Unit of the IRCCS Mondino Foundation for their precious assistance in all of the activities.
Author contributions
FB, RG and RDI: Study concept and design; Participants enrollment; Data collection; Statistical analysis; Interpretation of data; Writing of the first draft. DS: batch analysis correction, data normalization, statistical correction, data interpretation, revision of the manuscript. RG, CD, AZ, MF, SF: Laboratory procedures; Data Collection; Data interpretation; Revision of the manuscript for content. MA, DM, EG, NG, SB, MC, FC, AA, VG, GS: Participants enrollment; Revision of the manuscript for content. CT: Study concept and design; Participants enrollment; Interpretation of data; Drafting/revision of the manuscript for content. All authors read and approved the final manuscript.
Consent to participate
The research received approval from the local Ethics Committee (p-20200048922), and all participants provided written informed consent upon enrollment. The present study adhered to the principles outlined in the Declaration of Helsinki as well as prevailing national ethics regulations.
Data availability statement
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Italian Ministry of Health (Ricerca Corrente - RC2025-2027).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
