Abstract
Background
Calcitonin gene-related peptide plays an important role in migraine pathophysiology. We evaluated eptinezumab, an intravenous (IV) anti-calcitonin gene-related peptide monoclonal antibody, for the prevention of chronic migraine.
Objective
To determine the safety, tolerability, and effectiveness of four dose levels of eptinezumab and to inform the phase 3 development program.
Methods
This was a phase 2b, parallel-group, double-blind, randomized, placebo-controlled, dose-ranging clinical trial. Men and women (N = 616) aged 18–55 years were included if they had a diagnosis of chronic migraine, with onset at age ≤35 years and history of chronic migraine ≥1 year. During the 28-day screening period, patients must have had ≥15 headache days, including ≥8 migraine days, with ≥5 migraine attacks as recorded in the electronic diary. Patients were assigned in a 1:1:1:1:1 ratio to eptinezumab 300, 100, 30, 10 mg or placebo, administered as a single IV infusion. The primary endpoint was the percentage of patients with a ≥75% decrease in monthly migraine days over weeks 1–12 compared with the 28-day screening period.
Results
The ≥75% migraine responder rates over weeks 1–12 for eptinezumab 300, 100, 30, and 10 mg were 33.3%, 31.4%, 28.2%, and 26.8%, respectively, versus 20.7% for placebo (p = 0.033, 0.072, 0.201, 0.294 vs. placebo). Secondary efficacy endpoints (e.g. ≥50% responder rate, change from baseline in frequency of migraine/headache days, and percentage of severe migraines) had results favoring the three higher eptinezumab doses versus placebo. Eptinezumab was well tolerated and adverse event rates were similar to placebo.
Conclusions
The results of this trial demonstrate that eptinezumab appears effective and well-tolerated for the preventive treatment of chronic migraine and justifies the conduct of pivotal phase 3 trials for migraine prevention.
Trial Registration
ClinicalTrials.gov identifier: NCT02275117.
Keywords
Introduction
Migraine, a neurologic disorder with a stable 3-month prevalence of approximately 15% according to the US National Center for Health Statistics, exacts an enormous toll on society and on families (1–3). A recent epidemiologic survey reported that fewer than 50% of people with chronic migraine (CM) receive either acute or preventive pharmacologic treatment (4). For patients who receive oral migraine prevention medication, dose-titration adjustments are often required, and it may take several weeks or months before efficacy is established (5). Side effects are often problematic, efficacy rates are modest, and nonadherence is significant (6–8). There is an unmet need for well-tolerated and rapidly effective migraine prevention medications, with sustained efficacy, to optimize adherence and long-term patient outcomes.
Calcitonin gene-related peptide (CGRP) is a therapeutic target based on evidence that it plays an integral role in migraine pathophysiology (9,10). Recent efforts in migraine prevention are focused on inhibiting CGRP or its receptor using monoclonal antibodies (mAbs), based on the high target specificity, long half-life, and low hepato-renal interference in metabolism of humanized antibodies (11). Of the four mAbs targeting the CGRP pathway, three are administered subcutaneously and one (eptinezumab [previously referred to as ALD403]) utilizes IV administration. Eptinezumab is an immunoglobulin G1 antibody that potently and selectively binds to human α- and β-CGRP (12–14). This ligand-directed mAb was designed to reduce recognition by immune receptors (FcγR) and complement by antibody scaffold glycoengineering (15,16). Intravenous (IV) administration offers 100% bioavailability with maximal plasma concentration, Cmax, occurring at approximately the end of infusion and facilitating the potential for a rapid onset of preventive effect (17). Early-phase studies have demonstrated that eptinezumab has a half-life (t1/2) of 27 days, which is linear and dose-dependent (17). A single IV dose of eptinezumab significantly reduced monthly migraine days (MMDs) versus placebo in a prior phase 2 study and in a recent phase 3 study of patients with episodic migraine (12,13). The proportion of patients achieving greater than 50% reduction in migraine days (≥50% migraine responder rate [RR]) has been used as a historical standard of efficacy in migraine prevention trials (18). This standard emerged from the observed efficacy of available drugs and does not necessarily reflect the level of efficacy desired by patients with migraine. Based on the molecular attributes of eptinezumab, including target selectivity (dissociation constant ∼4 pM), potency, and 100% bioavailability by IV infusion (15), the ≥75% migraine RR was prespecified as the primary efficacy endpoint in this phase 2b, dose-ranging study (Clinical Trials.gov identifier: NCT02275117).
The primary objective of the current study was to evaluate the dose response of a single IV dose of eptinezumab in patients with CM, with secondary objectives evaluating the safety profile and duration of therapeutic effect, as well as to characterize the pharmacokinetics and immunogenicity profile.
Methods
Patients
Men and women aged 18–55 years were eligible if they had a diagnosis of CM according to the International Classification of Headache Disorders-3β criteria (ICHD-3β) (19), including a diagnosis of migraine established at age ≤35 years and history of CM of ≥1 year. During the 28-day screening period, patients were required to have ≥15 headache days, of which ≥8 were assessed as migraine days. Use of hormonal therapy and preventive medications for headache (such as topiramate, beta-blockers, valproate, tricyclic anti-depressants), except botulinum toxin, was allowed if the dosing has been stable for ≥3 months before screening, and was maintained at the same dosing level throughout the trial (unless deemed medically necessary by the investigator). The use of barbiturates or opioids for the acute treatment of CM was allowed if the dosing had been stable for 3 months before screening, and dosing did not exceed 4 days/month. Patients with CM who were diagnosed with medication overuse headache were also included (details in the Supplemental material). Written informed consent was obtained from all participants prior to the conduct of the study; consent forms used at each site were approved by a local or a central Institutional Review Board or Ethics Committee.
Study design
This randomized, double-blind, placebo-controlled, parallel-group, dose-ranging study was conducted from December 2014 to December 2016 at 92 sites (82 in the United States, four in Australia, and three each in New Zealand and the Republic of Georgia). Patients completed a headache electronic diary (eDiary) daily for 28 days during the screening period to confirm eligibility and establish baseline migraine results. Eligible patients were randomized (1:1:1:1:1) to receive a single IV infusion of eptinezumab 300 mg, 100 mg, 30 mg, 10 mg, or placebo (total volume of 100 mL administered over 1 hour [ ± 15 minutes]) on day 0. A stratified, permuted block randomization was used, with the treatment assignment obtained from an interactive web response system. Randomization was stratified by baseline migraine days (< 20 vs. ≥20) and medication overuse headache status (yes vs. no; defined in the Supplemental material). Patients visited study centers at weeks 4, 8, 12, 24, 36, and 49 after dosing, and completed the eDiary daily until the week 49 visit. Study staff also contacted patients by telephone at weeks 2, 16, 20, 28, 32, 40, and 44. The full trial protocol is available on the Cephalalgia website.
Efficacy assessment
Baseline characteristics and demographics.
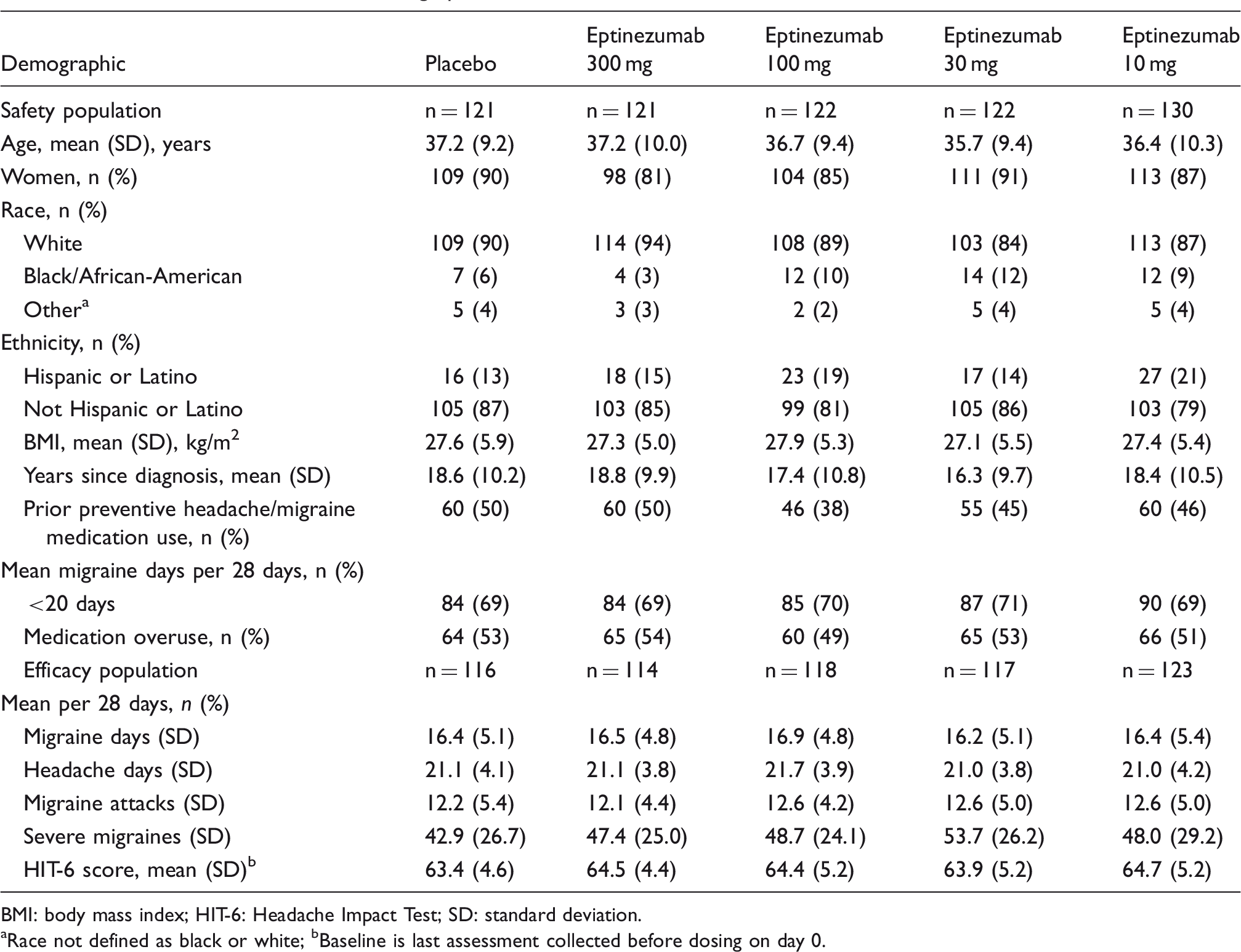
BMI: body mass index; HIT-6: Headache Impact Test; SD: standard deviation.
aRace not defined as black or white; bBaseline is last assessment collected before dosing on day 0.
The primary efficacy endpoint was ≥75% migraine RR, defined as the percentage of patients with an average monthly ≥75% decrease in migraine days over weeks 1–12 compared with the 28-day baseline period. Prespecified secondary efficacy endpoints were: ≥50% and 100% reduction in migraine days and headache days; change from baseline in frequency of migraine and headache days; percent change from baseline in migraine and headache days; migraine and headache hours; migraines and headaches with severe intensity; and percentage of migraines and headaches with acute medication usage.
Other secondary endpoints included change in the Headache Impact Test (HIT-6, version 1.0) score (20). The HIT-6 total score ranges from 36 to 78, with a ≥5-point decrease in score for an individual patient considered clinically meaningful (21,22). In this study, HIT-6 scores were measured on day 0 and at weeks 4, 12, 24, and 49. A prespecified analysis of life impact of migraine based on HIT-6 scores was performed. A post hoc analysis explored the percentage of patients with a migraine on day 1 post-infusion compared with baseline.
Safety and tolerability
Safety endpoints included treatment-emergent adverse events (TEAEs) and changes in laboratory parameters, vital signs, 12-lead electrocardiograms, and physical examination findings. Blood samples were collected at each study visit for analysis of immunogenicity, including monitoring development of anti-eptinezumab antibodies and assaying for neutralizing potential.
Pharmacokinetics
Blood samples were collected pre-dose, immediately post-dose, and 4 hours post-dose on day 0 and at all subsequent study visits. Plasma samples were analyzed for concentrations of free, pharmacologically active eptinezumab. Pharmacokinetic parameters, including maximum observed plasma concentration (Cmax), time of Cmax, area under the plasma concentration-time curve from time 0 to the time of last quantifiable concentration (AUC0-last), and terminal half-life (t½), were derived from the free eptinezumab plasma concentrations using standard noncompartmental methods (23).
Statistical analysis
Sample size calculation is described in the Supplemental material. Efficacy endpoints were analyzed using data from all randomized patients who received study treatment, excluding patients at one study site where anomalous data were deemed unreliable for the efficacy analyses. The decision to remove this site was made prior to unblinding and study completion. Had these data been included, p-values would have been more favorable to eptinezumab than the current analysis. Safety endpoints were evaluated using data from all patients who received study treatment. For the primary efficacy endpoint, pairwise comparisons between each eptinezumab dose and placebo were made using a Cochran-Mantel-Haenszel (CMH) test controlling for stratification factors. Step-down testing was used to control for multiplicity, starting with the 300 mg dose versus placebo and then proceeding down the dose range, with a 10% two-sided alpha level. Normalization was used to address missing migraine data in the primary efficacy analysis. If the headache diary was completed for ≥21 days of a 4-week interval, the observed frequency was normalized to 28 days by multiplying by the inverse of the completion rate. If the diary was completed for < 21 days of a 4-week interval, the results were a weighted function of the observed data for the current interval and the results from the previous interval, with the weight proportional to how many days the diary had been completed. The ≥50% migraine RRs (weeks 1–12) were evaluated using a CMH test and changes in migraine days (weeks 1–12) were evaluated using an extended CMH test (24). Multiplicity control was not used for the secondary endpoints. Following the 2016 American Statistical Association recommendation, p-values were not added to each summary measure at each timepoint (25). Adverse events were coded using the Medical Dictionary for Regulatory Activities (MedDRA), version 15.0.
Results
Patients
A total of 616 patients received study treatment and comprised the safety population (Figure 1). Patients (n = 28) at one US site were excluded due to multiple protocol violations; thus, efficacy endpoints were evaluated in the remaining 588 patients. Overall, 434 patients (70.5%) completed the study through week 49, with > 95% remaining on study through week 12. Withdrawal rates were low, with only 25 patients withdrawing during weeks 1–12 (300 mg, n = 2; 100 mg, n = 4; 30 mg, n = 9; 10 mg, n = 6; placebo, n = 4). Of those patients withdrawing during weeks 1–12, 15 patients withdrew consent (with n = 9 due to lack of efficacy), nine were lost to follow-up, and one violated the study entry criteria. Early discontinuations before the week 49 visit were due most frequently to withdrawn consent (n = 85) or lost to follow-up (n = 66). Reasons for withdrawal were study burden (across all dose groups) and lack of efficacy (placebo group). Demographics and baseline characteristics were balanced across treatment groups (Table 1). The study cohort had a median age of 37 years (range, 18–55); 87% were women and 89% were white. Migraine started after menarche in most women (76%) and after puberty in most men (69%). Previous preventive medication use was reported by 281 patients (46%), with topiramate (137 [22.2%]) being the most common. In total, 65.1% of patients did not receive any concomitant preventive migraine medication. The median number of preventive medications for weeks 0–12 was 0, with an overall mean of 1.3. The most common concomitant headache or migraine prevention medications were topiramate (95 [15.4%]), amitriptyline (32 [5.2%]), and propranolol (20 [3.2%]).
Patient disposition.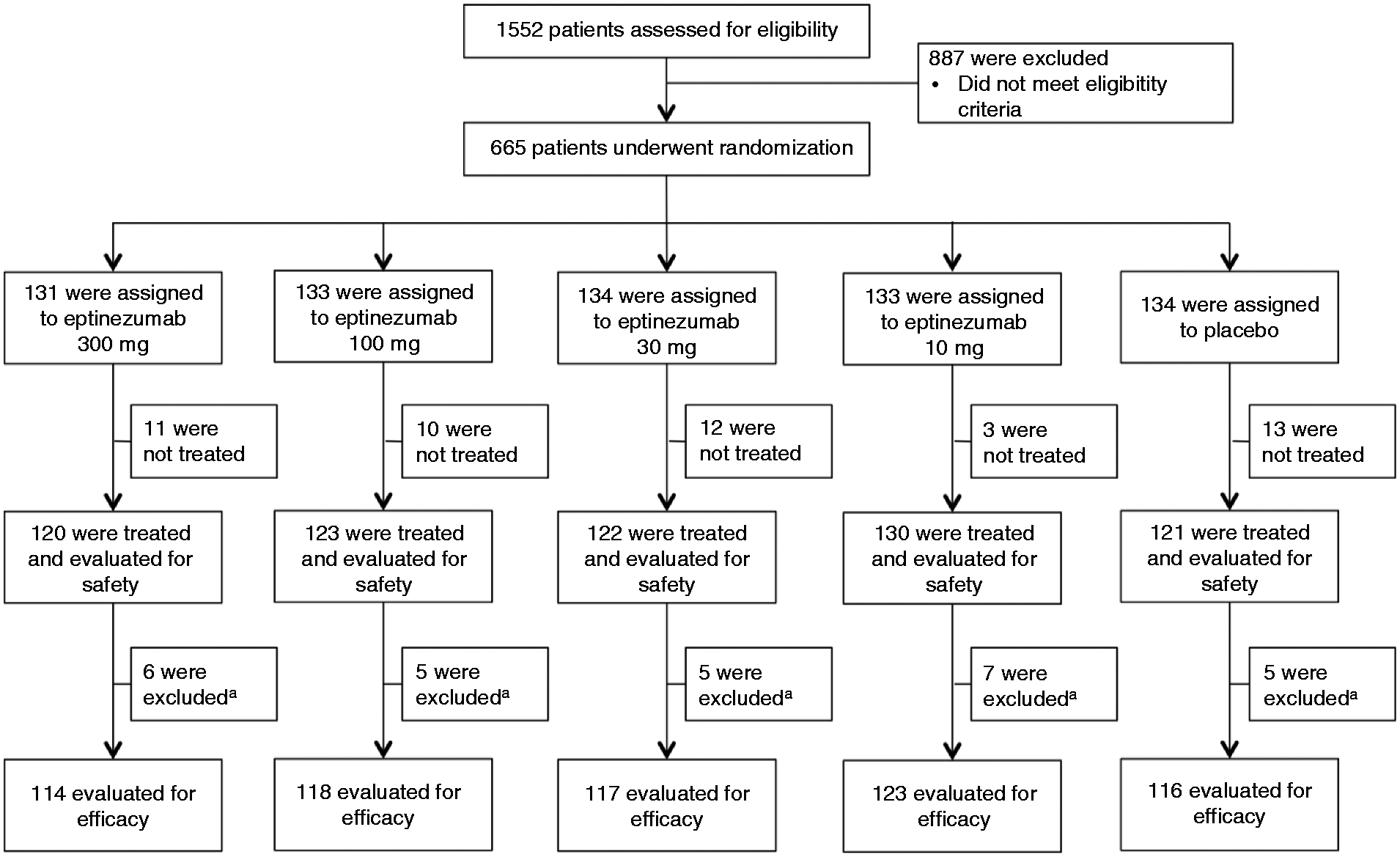
Efficacy
Migraine day frequency was approximately 16 days over the 28-day baseline period in all treatment groups. In the primary efficacy analysis, ≥75% migraine RRs over weeks 1–12 were 33.3%, 31.4%, 28.2%, and 26.8% for eptinezumab 300, 100, 30, and 10 mg, respectively, compared with 20.7% for placebo (p = 0.033, 0.072, 0.201, and 0.294 vs. placebo) (Figure 2(a)). The ≥50% migraine RRs over weeks 1–12 were 57.0%, 55.1%, 55.6%, and 43.9% for eptinezumab 300, 100, 30, and 10 mg groups, respectively, compared with 40.5% for placebo (p = 0.013, 0.029, 0.024, and 0.621 vs. placebo). The ≥75% migraine RRs for eptinezumab groups over weeks 1–4, 5–8, and 9–12 were similar to the rates over weeks 1–12 (Figure 2(b)). The ≥50% migraine RRs across each of these time intervals were also consistent with ≥50% migraine RRs over weeks 1–12. Percentages of patients with freedom from migraine (i.e. no migraine) over weeks 1–4, 5–8, and 9–12 were 7.9%, 14.0%, and 13.2%, respectively, for eptinezumab 300 mg and 7.6%, 13.6%, and 11.9% for eptinezumab 100 mg versus 5.2%, 6.9%, and 9.5% for placebo.
(a) ≥ 75% migraine responder rates over weeks 1–12, and (b) ≥ 75% and ≥ 50% migraine responder rates over weeks 1–12 by 4-week intervals.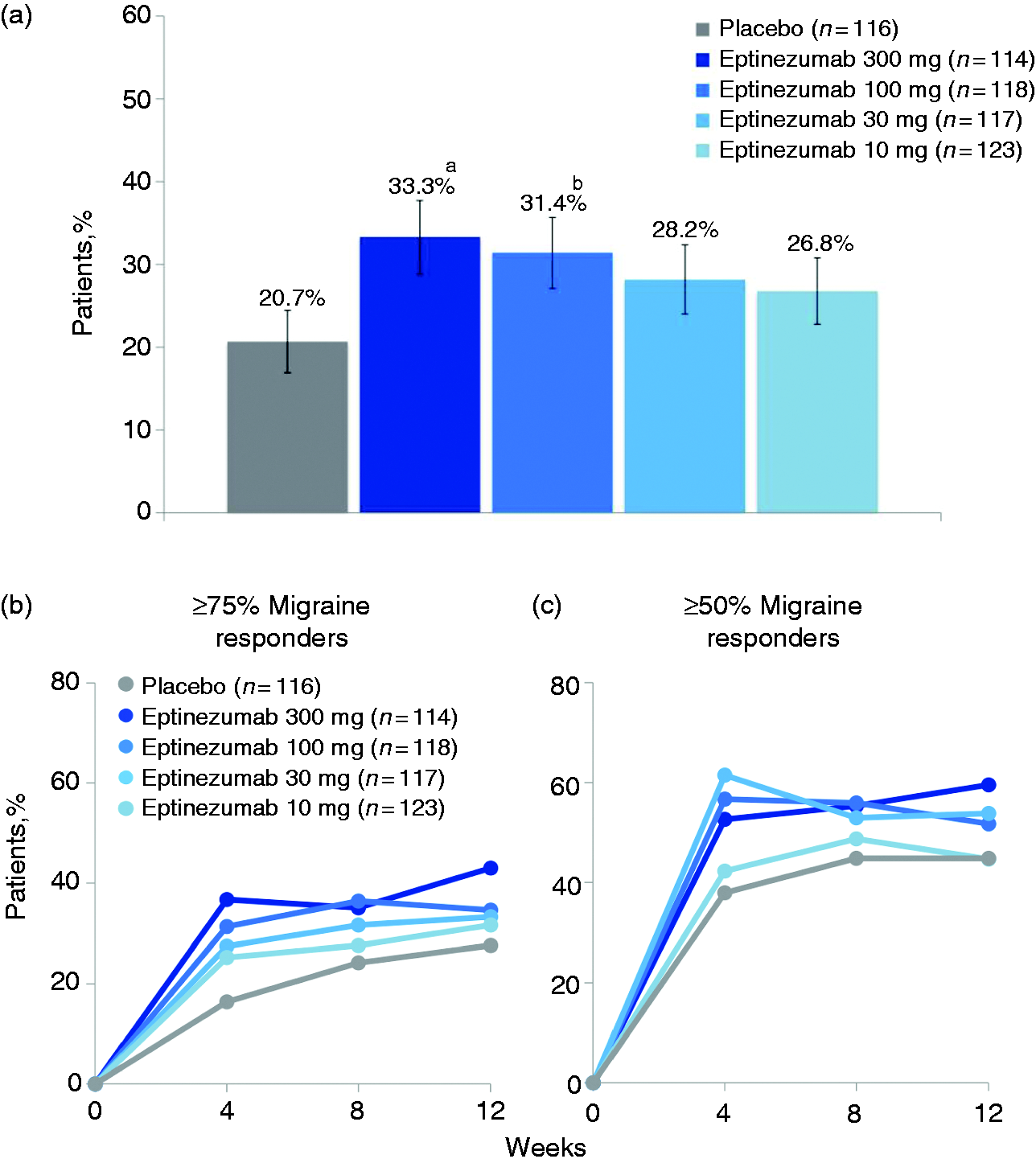
Treatment-emergent adverse events (TEAEs).
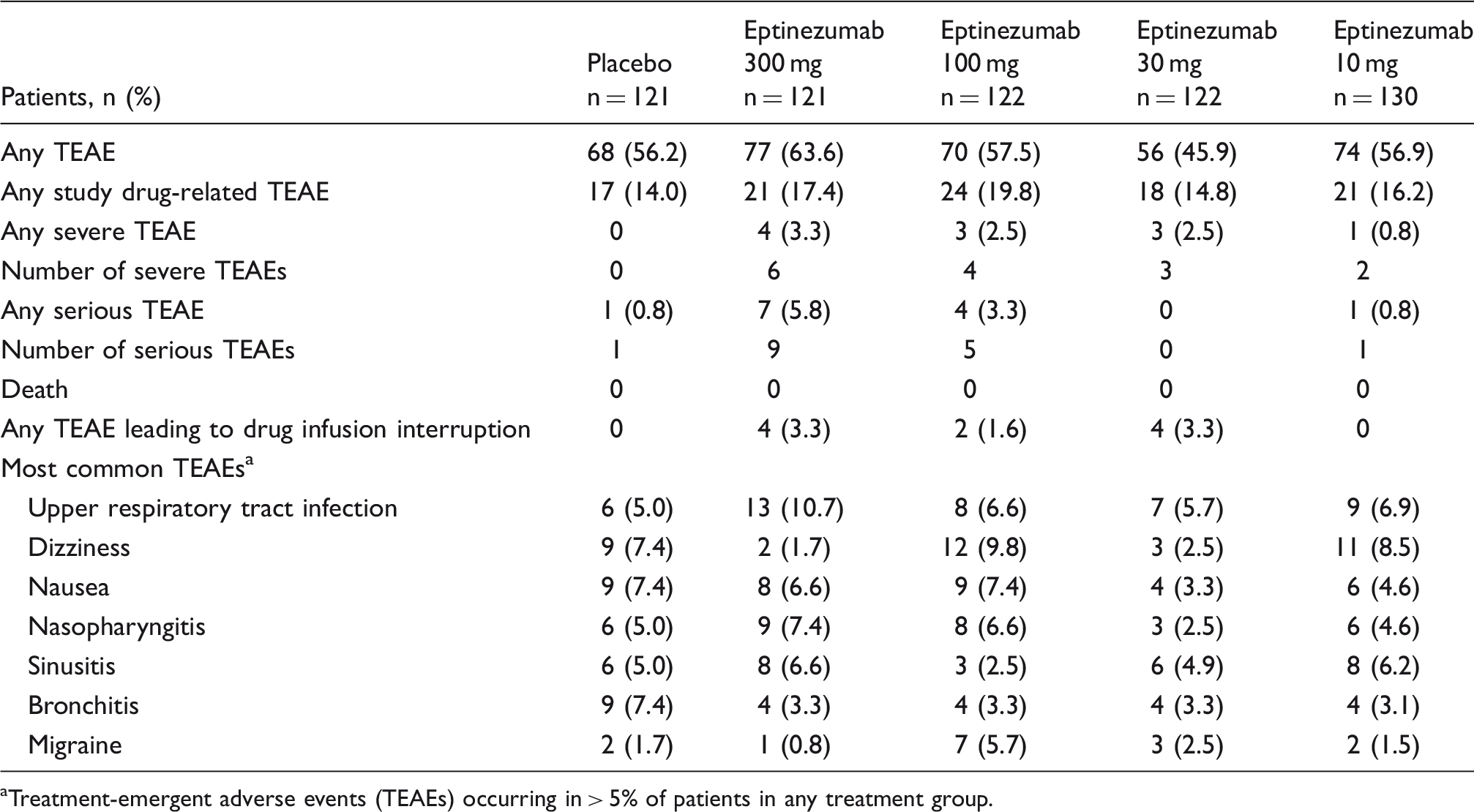
Treatment-emergent adverse events (TEAEs) occurring in > 5% of patients in any treatment group.
HIT-6 results
Baseline HIT-6 scores were 65, 64, 64, and 65 for eptinezumab 300, 100, 30, and 10 mg, respectively, versus 63 for placebo. The greatest effect of eptinezumab, as measured by the HIT-6, was observed at week 12, with changes in baseline scores of −10.0, −6.9, −6.5, and −6.5 for the 300, 100, 30, and 10 mg groups, respectively, compared with −5.8 for the placebo group (eTable 3). A prespecified analysis of the percentage of patients for whom migraine had a severe impact on life demonstrated a reduction from 90.3% at baseline to 29.9% at week 12 with eptinezumab 300 mg, 86.4% to 43.0% with eptinezumab 100 mg, compared with 79.3% to 50.9% with placebo (eTable 3).
Onset of action
A post hoc analysis evaluated migraine-preventive effects on the day following IV infusion. On average, 59.1%, 60.4%, and 58.7% of patients in the eptinezumab 300 mg, 100 mg, and placebo groups, respectively, experienced a migraine on any given day during the 28-day baseline period. Percentages of patients who experienced a migraine on day 1 post-infusion of the 300 and 100 mg doses were 26.3% and 29.3%, versus 48.7% for placebo (Figure 3). This represents, for the eptinezumab groups, a > 50% reduction in the likelihood of a migraine day in the 24 hours post-infusion compared with baseline.
Percentages of patients experiencing a migraine on any given day during baseline (BL) period and on day 1 following IV infusion of eptinezumab 300 and 100 mg and placebo.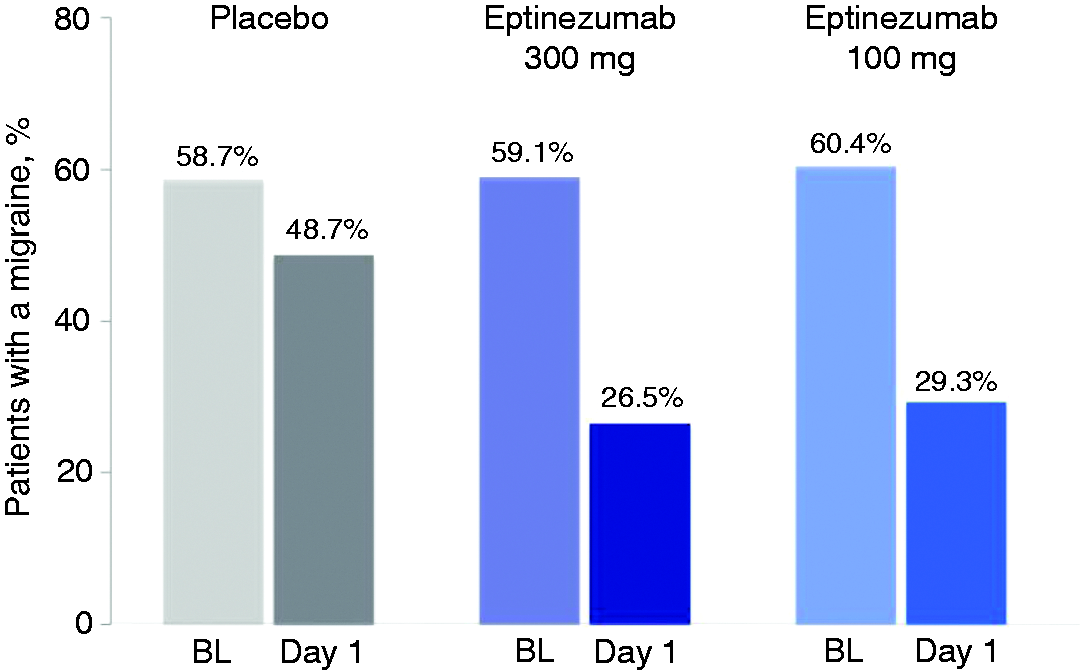
Tolerability and safety
TEAEs were reported in 345 patients (56.0%) (Table 2), and the most frequent were upper respiratory tract infection (eptinezumab 300 mg, 10.7%; eptinezumab 100 mg, 6.6%; eptinezumab 30 mg, 5.7%; eptinezumab 10 mg, 6.9%; placebo, 5.0%) and dizziness (eptinezumab 300 mg, 1.7%; eptinezumab 100 mg, 9.8%; eptinezumab 30 mg, 2.5%; eptinezumab 10 mg, 8.5%; placebo, 7.4%). Drug-related TEAEs were reported in 101 patients (16.4%), with similar incidences across active and placebo groups (eptinezumab 300 mg, 17.4%; eptinezumab 100 mg, 19.8%, eptinezumab 30 mg, 14.8%; eptinezumab 10 mg, 16.2%; placebo, 14.0%). Most TEAEs were mild or moderate; 11 eptinezumab-treated patients had severe TEAEs (grade 3). Thirteen patients reported 16 serious TEAEs; none were considered related to the study drug. Ten patients had TEAEs leading to interruption of eptinezumab infusion, including six patients with TEAEs that were coded as the MedDRA preferred term of hypersensitivity. Patient symptoms varied and included sneezing, cough, nasal congestion, scratchy throat, burning and watery eyes, edema in the face and eyelids, shortness of breath, hives, and itching. Each hypersensitivity TEAE was mild to moderate in severity and was effectively treated with antihistamines or corticosteroids; all symptoms resolved within 24 hours. No cases of delayed hypersensitivity were identified. Four of the six patients received the complete infusion per protocol (100 mL), and two received only partial infusion (21 mL, 33 mL). Laboratory tests, including liver function, vital signs, electrocardiogram, and physical examination, were unremarkable and did not exhibit any dose-related signals.
Anti-drug antibodies
Incidence of anti-eptinezumab antibody formation, at any time during the study, was dose dependent from 10 to 100 mg and was similar at 100 and 300 mg, ranging from 18.2% to 18.9%. The time to maximal anti-drug antibody (ADA) positive response for the 100 and 300 mg dose groups was 24 weeks, with the incidence markedly declining through the week 49 End of Study visit where the incidence decreased to ≤2.5%. The incidence of ADAs exhibiting potential neutralizing activity in the 100 and 300 mg groups was approximately 5.8%. At the end of trial, ADAs exhibiting neutralizing potential had resolved in six of seven, five of seven, and one of two patients receiving eptinezumab 300, 100, and 10 mg, respectively. Importantly, formation of ADAs with or without neutralizing potential did not impact efficacy (change in MMDs over weeks 1–12 for ADA-positive versus ADA-negative patients: Eptinezumab 300 mg, −8.5 vs. −8.1; eptinezumab 100 mg, −9.0 vs. −7.3, respectively). Similarly, safety was not influenced by the formation of ADAs.
Pharmacokinetics
Maximum free eptinezumab plasma concentrations were achieved at the end of IV infusion (eFigure 2). Exposure to eptinezumab was dose-proportional based on Cmax and AUC0-last. For the 300, 100, 30, and 10 mg groups, mean (standard deviation) Cmax values of free eptinezumab were 115.2 (31.1), 39.8 (17.5), 11.7 (5.7), and 4.7 (2.5) µg/mL, respectively; corresponding mean AUC0-last values were 69630 (21010), 21990 (7728), 6441 (2799), 2600 (1480) h·µg/mL. Mean elimination t1/2 was independent of dose, ranging from 27 to 30 days across groups.
Discussion
In this phase 2b study, eptinezumab 300 mg demonstrated superior efficacy for the primary endpoint of ≥75% reduction in monthly migraine days (weeks 1–12) compared with placebo. After eptinezumab infusion, one-third of patients had a ≥75% reduction in MMDs over 3 months. Eptinezumab 300, 100, and 30 mg showed superiority over placebo for the conventional ≥50% migraine RR, as well as reductions in mean MMDs over weeks 1–12.
Results from a phase 2 clinical trial in patients with episodic migraine suggested that eptinezumab has the potential to significantly reduce migraine frequency. To explore further and confirm these findings, the current study used – for the first time in a migraine prevention trial – a rigorous primary endpoint of ≥75% migraine RR. In meeting this endpoint, this study established a precedent and rationale for pursuing the ≥75% migraine RR benchmark of efficacy as an endpoint in future migraine prevention trials. A separate analysis of patient-reported outcomes obtained in this study, presented elsewhere, provided evidence that patients who achieved a ≥75% reduction in migraine days showed significantly greater improvements from baseline in all SF-36 domain scores (26), including normalization of Bodily Pain and Role-Physical domains, compared with patients not reaching this magnitude of migraine response (27). These results, combined with the lack of dose titration, quarterly administration, and favorable side effect profile, have the potential to improve adherence and long-term outcomes compared with currently available preventive treatments (12).
Several oral CGRP-receptor antagonists have demonstrated acute migraine treatment efficacy, indicating the potential for an IV-administered CGRP mAb to provide acute treatment efficacy and possibly a rapid onset of preventive effect (28–33). Post hoc analysis results suggested a clinically meaningful achievement of migraine prevention on day 1 post-infusion as well as persistence of benefit in the days following infusion. This observation informed the design of the phase 3 PROMISE-1 and PROMISE-2 trials (13,14). Both studies included the percentage of patients with a migraine on day 1 post-infusion as a prespecified endpoint.
The overall incidence of ADAs and NAbs were comparable across the eptinezumab 100 mg and 300 mg dose groups. The immunogenicity analyses revealed no influence of ADA or NAb positive/negative status on efficacy or safety in any of the treatment groups.
A limitation of this study is the single administration design; therefore, the efficacy and safety of repeated doses of eptinezumab was not evaluated. Additionally, the primary efficacy evaluation period was 12 weeks rather than 24 weeks, which would have provided an opportunity to determine sustained responses over a longer period of time. However, efficacy evaluation periods of 12 weeks have historically been the standard in migraine prevention trials; dropout rates are lower with shorter duration trials, and this study measured the effect over the entire 12-week period, rather than over just the last month (weeks 9–12). A second infusion was included in the PROMISE-2 phase 3 study design to investigate multiple quarterly infusions (14). The placebo response rates in the trial were higher than expected. While placebo response rates are generally higher in pain-related clinical studies (34,35), there are a number of previously documented factors that could have attributed to the higher than expected placebo response, including the number of active treatment arms, the potential novelty of treatment, and the observation that 54% of patients in the current study were naïve to preventive therapy (36–38). Other potential contributing factors include the intensive patient contact with migraine care experts, including professionally supervised IV administration and frequent follow-up contact via telephone and in-person visits.
Conclusions
In this exploratory phase 2b study, both the eptinezumab 100 and 300 mg dose groups met the prespecified level of significance, as defined in the protocol. A post hoc data analysis suggests that a clinically meaningful reduction of migraine activity can be achieved as early as day 1 post-infusion. These results provide preliminary, yet robust and promising evidence that eptinezumab administered by IV infusion to patients with CM is associated with a rapid migraine preventive effect that is sustained for 3 months after a single dose and is associated with acceptable safety and favorable tolerability profiles. These results supported the advancement of eptinezumab into phase 3 clinical development to generate additional evidence and confirm its effectiveness for the preventive treatment of episodic and chronic migraine.
Supplemental Material
Supplemental material for Eptinezumab for prevention of chronic migraine: A randomized phase 2b clinical trial
Supplemental Material for Eptinezumab for prevention of chronic migraine: A randomized phase 2b clinical trial by David W Dodick, Richard B Lipton, Stephen Silberstein, Peter J Goadsby, David Biondi, Joe Hirman, Roger Cady and Jeff Smith in Cephalalgia
Footnotes
Article highlights
This randomized, double-blind, placebo-controlled, parallel-group, dose-ranging study evaluated the efficacy and safety of eptinezumab, an anti-CGRP monoclonal antibody, administered as a single IV infusion for the preventive treatment of chronic migraine.
In the primary efficacy analysis, treatment with eptinezumab 300 mg and 100 mg resulted in more patients achieving a ≥75% reduction in migraine frequency compared with placebo across weeks 1–12 post-infusion. Eptinezumab 100 mg and 300 mg were also favored over placebo in analyses of the secondary efficacy endpoints, including the ≥50% reduction in migraine frequency, the overall reduction in monthly migraine days, and the reduction in severe migraine frequency.
A post hoc analysis suggested that patients could achieve clinically meaningful prevention of migraine as early as day 1 post-infusion, which led to the consideration of this endpoint in the Phase 3 clinical program.
Treatment with eptinezumab demonstrated acceptable safety and tolerability across doses as compared to placebo, with no apparent dose-related trend in the nature, frequency, or severity of treatment-emergent adverse events.
Author contributions
All authors had full access to all study data and take responsibility for all aspects of the work, including the integrity of the data and the accuracy of the data analysis. All authors provided final approval of the version to be published. Substantial contributions to conception or design of the work: JS, DWD, JH. Substantial contributions to the acquisition, analysis, or interpretation of data for the work: DWD, RBL, SS, PJG, DB, JH, RC, JS. Drafting of manuscript and critical review/revision of the manuscript for important intellectual content: DWD, RBL, SS, PJG, DB, JH, RC, JS.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DWD reports the following conflicts: Personal fees: Amgen, Association of Translational Medicine, University Health Network, Daniel Edelman Inc., Autonomic Technologies, Axsome, Aural Analytics, Allergan, Alder, Biohaven, Charleston Laboratories, Dr Reddy’s Laboratories/Promius, Electrocore LLC, Eli Lilly, eNeura, Neurolief, Novartis, Ipsen, Impel, Satsuma, Supernus, Sun Pharma (India), Theranica, Teva, Vedanta, WL Gore, Nocira, PSL Group Services, University of British Columbia, Zosano, ZP Opco, Foresite Capital, Oppenheimer. CME fees or royalty payments: Healthlogix, Medicom Worldwide, Medlogix Communications, Mednet, Miller Medical, PeerView, WebMD Health/Medscape, Chameleon, Academy for Continued Healthcare Learning, Universal meeting management, Haymarket, Global Scientific Communications, Global Life Sciences, Global Access Meetings, UpToDate (Elsevier), Oxford University Press, Cambridge University Press, Wolters Kluwer Health; Stock options: Aural analytics, Healint, Theranica, Second Opinon/Mobile Health, Epien, GBS/Nocira, Matterhorn/Ontologics, King-Devick Technologies. Consulting without fee: Aural Analytics, Healint, Second Opinion/Mobile Health, Epien. Board of directors: Epien, King-Devick Technologies, Matterhorn/Ontologics. Patent: 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis, without fee. Professional society fees or reimbursement for travel: American Academy of Neurology, American Brain Foundation, American Headache Society, American Migraine Foundation, International Headache Society, Canadian Headache Society. Other: Use agreement through employer: Myndshft. RBL holds stock options in Biohaven Holdings and eNeura; serves as consultant or advisory board member and/or has received honoraria from Alder BioPharmaceuticals, Allergan, American Academy of Neurology, American Headache Society, Amgen, Autonomic Technologies, Avanir Pharmaceuticals, Biohaven Pharmaceuticals, Biovision, Boston Scientific, Dr. Reddy’s Laboratories, electroCore, Eli Lilly and Company, eNeura, GlaxoSmithKline, Merck, Pernix, Pfizer, Supernus, Teva Pharmaceuticals, Trigemina, Vector, and Vedanta; and receives royalties from Informa, Oxford University Press (2009), Wolff’s Headache 7th and 8th Edition, and Wiley. SS receives or has received honoraria as a consultant and/or advisory panel member from Alder Biopharmaceuticals, Allergan, Amgen, Avanir Pharmaceuticals, Cefaly, Curelator, Dr. Reddy’s Laboratories, Egalet Corporation, eNeura, electroCore Medical, Eli Lilly and Company, Medscape, National Institute of Neurological Disorders and Stroke, Satsuma Pharmaceuticals, Supernus Pharmaceuticals, Teva Pharmaceuticals, Theranica, and Trigemina; and receives consulting fees from Alder BioPharmaceuticals, Allergan, Amgen, Avanir Pharmaceuticals, Curelator, Dr. Reddy’s Laboratories, eNeura, electroCore, Eli Lilly and Company, Medscape, National Institute of Neurological Disorders and Stroke, Supernus, Teva Pharmaceuticals, Theranica, and Trigemina. PJG reports grants and personal fees from Amgen and Eli-Lilly and Company; and personal fees from Alder Biopharmaceuticals, Allergan, Autonomic Technologies Inc., Biohaven Pharmaceuticals Inc., Dr Reddy’s Laboratories, Electrocore LLC, eNeura, MundiPharma, Novartis, Teva Pharmaceuticals, Trigemina Inc., MedicoLegal work, Massachusetts Medical Society, Up-to-Date, Oxford University Press, and Wolters Kluwer; and a patent Magnetic stimulation for headache, assigned to eNeura without fee.
RC is a full-time employee of Alder Biopharmaceuticals.
DB was a full-time employee of Alder Biopharmaceuticals at the time of study conduct and completion.
JH is a contracted service provider to Alder Biopharmaceuticals.
JS is a full-time employee, founder, and shareholder of Alder Biopharmaceuticals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored and funded, and its publication supported by Alder BioPharmaceuticals, Inc (Bothell, WA, USA). The sponsor participated in the design and conduct of the study; data collection, management, analysis, and interpretation; and preparation, review, and approval of the manuscript. All statistical analyses were performed by a contracted research organization and were directed or designed by Pacific Northwest Statistical Consulting under contractual agreement with Alder BioPharmaceuticals. All authors and Alder BioPharmaceuticals prepared, reviewed, and approved the manuscript and made the decision to submit the manuscript for publication. Editorial support for the development of this manuscript was funded by Alder BioPharmaceuticals and was provided by BioScience Communications (New York, NY, USA) and The Medicine Group (New Hope, PA, USA).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
