Abstract
Background
Whether migraine with aura (MA) is a biologically independent entity from migraine without aura (MO) is still debated. Similarities and differences between MO and MA have been extensively investigated in recent years through several neuroimaging studies, providing valuable insights into their underlying pathophysiology. To provide a better understanding of functional and structural differences between MO and MA, we conducted a narrative review of neuroimaging studies in these two conditions.
Methods
A comprehensive PubMed search for neuroimaging studies in MO and MA was conducted in May 2025. We included studies that directly compared the two conditions using diffusion tensor imaging, voxel-based morphometry, surface-based morphometry, functional MRI and arterial spin labeling studies. No publication date restrictions were applied.
Results
Overall, patients with MA exhibited heightened engagement of the visual regions, cerebellum and thalamus. However, both MO and MA shared common activation of parts of the salience network and involvement of similar visual areas, including the striate and extrastriate cortices. However, contrasting results and several inconsistencies emerged from the analysis of different imaging studies. These included the lack of specification regarding the phase of the migraine cycle during which the scans were conducted, the inclusion of patients under migraine prevention, small sample sizes, and different approaches to data and statistical analysis (including a more liberal approach to interpreting results).
Conclusions
Although several biases influence the reliability of most findings, patients with MA exhibited higher involvement of visual processing regions, decreased cerebellar antinociceptive activity and impaired thalamic information filtering. Whether this pattern represents the consequence of the recurrence of cortical spreading depression or a primary predisposition to it remains to be determined. Future studies with a rigorous and standardized approach are needed to understand the differences between MO and MA.
This is a visual representation of the abstract.
Introduction
Migraine is a widespread and debilitating neurological disorder and, despite its global reach, its pathophysiology remains elusive. This is mainly due to its complex interplay of genetic predispositions, environmental triggers and neurobiological cascades (1). In addition, the pathophysiology of migraine is further complicated by the presence of several subtypes, including migraine with aura, for which the relationship with migraine without aura is still debated.
Migraine aura can be defined as the experience of a series of transient neurological symptoms preceding or accompanying the pain-phase of the migraine cycle (2). Common manifestations include flashing lights, jagged zigzag lines (fortification spectra) or shadowy blind spots (scotomas). Less frequently, an aura may manifest as sensory disturbances, aphasia, motor or brainstem-related symptoms (3–5). A key mechanism implicated in aura is cortical spreading depression (CSD), a wave of cortical hyperactivity followed by prolonged neuronal suppression. Supported by animal studies and limited human evidence, such as intraoperative electrophysiological recordings, CSD is widely accepted as a driver of the symptoms characterizing the aura phenomenon (6,7). However, also understanding the bonding between CSD, aura and the eventually succeeding or accompanying headache, whether migraine with aura (MA) is a biologically distinct entity from migraine without aura (MO), and whether aura is a cause, consequence, or both of migraine in MA patients, remains unresolved. Given the symptomatic experience of aura and its repeatability across multiple attacks in MA patients, it is reasonable to hypothesize that peculiar structural and/or functional changes may exist in the brains of those with MA. Electroencephalography and magnetoencephalography excel at capturing high-resolution temporal data on neural activity, while imaging techniques such as positron emission tomography (PET), structural magnetic resonance imaging (MRI), and fMRI offer critical insights into brain structure, function, and connectivity. Due to their accessibility and extensive supporting literature, imaging methods are valuable for studying the pathophysiology of migraine. These approaches have revealed critical insights, including activation patterns associated with aura and pain, as well as structural and connectivity differences in the cortex (8–14). This narrative review synthesizes neuroimaging studies comparing MA and MO, aiming to highlight differences and similarities between these subtypes. Given the symptomatic experience of patients during aura, which reflects consistent patterns of brain activity, exploring the presence or absence of imaging peculiarities may enhance our understanding of migraine and the aura phenomenon itself.
Methods
A comprehensive literature search was performed on the PubMed database in May 2025. No restrictions were placed on publication dates.
We included original neuroimaging studies using MRI that directly compared MO and MA employing the following techniques: diffusion tensor imaging (DTI), voxel-based morphometry (VBM), surface-based morphometry (SBM), fMRI and arterial spin labeling (ASL). We excluded studies that investigated patients with chronic migraine as well as pediatric patients. The search utilized the following string: (“migraine with aura” AND “migraine without aura”) AND (“neuroimaging” OR “neuro-imaging” OR “structural” OR “functional” OR “MRI” OR “fMRI” OR “PET” OR “DTI” OR “VBM” OR “SBM” OR “ALS”) AND (“compar*” OR “versus”). To ensure thoroughness, the reference lists of included studies and relevant review articles were also examined to identify any additional research that may have been overlooked. The literature search did not find any PET studies that directly compared patients with MA and MO.
Results
Neuroimaging studies on brain structure: DTI, VBM and SBM
Among the different methodologies employed to obtain MRI data, DTI is the most responsive to microstructural alterations because it assesses the diffusivity of water molecules along the fibre tracts of white matter in the three principal orthogonal diffusion directions (15).
Few studies have examined changes in white matter comparing MO to MA patients. Mandl et al (16). using a low-field MRI scanner, identified a decrease in radial diffusivity, a surrogate marker of axonal damage, in the parieto-occipital, corpus callosum and cingulate white matter (17). By contrast, other studies with a higher magnetic field and larger sample sizes did not replicate these findings, revealing no substantial differences among the groups (18–21). Additionally, one study showed a decrease in the magnetization transfer ratio (MTR), a method for identifying myelin-related anomalies, in the crus I of the cerebellum in patients with MO compared to patients with MA and healthy controls (22). In another study, the same study group (19) revealed microstructural variations in T1 relaxation times and MTR in the thalamus, with MA exhibiting lower values than MO.
Using both DTI and resting-state fMRI, Fu et al.(23) investigated the structural and functional brain connectivity between MA, MO and healthy volunteers. In particular, they explored the connectivity strength between two arbitrary nodes computed as the average fractional anisotropy value of all the tracts through the corresponding two nodes after the diffusion tensor tractography (24). In comparison to MO and healthy volunteers, they found that MA had reduced structural and functional connectivity in the frontal, temporal, visual, amygdala and thalamic regions (23).
VBM and SBM are two primary approaches used to obtain structural information on white and grey matter volumes, as well as evaluate brain morphology, from MRI scans. VBM is an accessible and automated technique that evaluates alterations in both grey and white matter volumes. By contrast, SBM can be used to obtain information on the characteristics of cortical areas, such as cortical thickness (25).
Using region of interest (ROI) VBM analysis, a single study identified a decrease in grey matter volume in the visual regions V3 and V5 in both MO and MA compared to controls (26). Nevertheless, SBM analyses in a small cohort of patients have revealed an increase in cortical thickness in several brain regions, particularly within the visual and somatosensory areas, among patients with MA and MO compared to healthy controls (27). This study failed to show any changes between the two groups, nor any associations between thickness and clinical aspects of migraine. In a different study with a larger cohort of patients from four academic headache centres (28), a thinner cortex was identified among patients with MO relative to healthy individuals in different areas involved in the multisensory elaboration of information, including the somatosensory cortex, middle frontal gyrus, primary and secondary visual areas, left supramarginal gyrus, and in the right cuneus. In contrast, compared with healthy subjects, patients with MA showed alterations in the thickness of cortices of several areas actively involved in higher cognitive functions, such as memory and learning, arousal and awareness. These included a thinner cortex in the subparietal sulcus, intraparietal sulcus and anterior cingulate cortex. Furthermore, a thickened cortex was observed in the middle temporal gyrus (28).
Neuroimaging studies on brain function: fMRI and ASL
fMRI is a technique that investigates brain connectivity across different and distant cerebral nodes. It can be carried out during specific tasks (i.e. task-related) or in the absence of any specific activities (i.e. resting-state), facilitating the detection of spontaneous interactions among diverse brain regions (29). The conceptual basis of fMRI relies on the evaluation of blood oxygenation level dependent (BOLD) signals, whose elevation correlates with increased cerebral metabolic activity. However, whether an increase in the BOLD signal reflects inhibitory or excitatory activity is yet to be determined (Table 1).
Comparative table illustrating the similarities and differences in brain morphometry, functional connectivity and cerebral blood flow in migraine with aura versus migraine without aura.
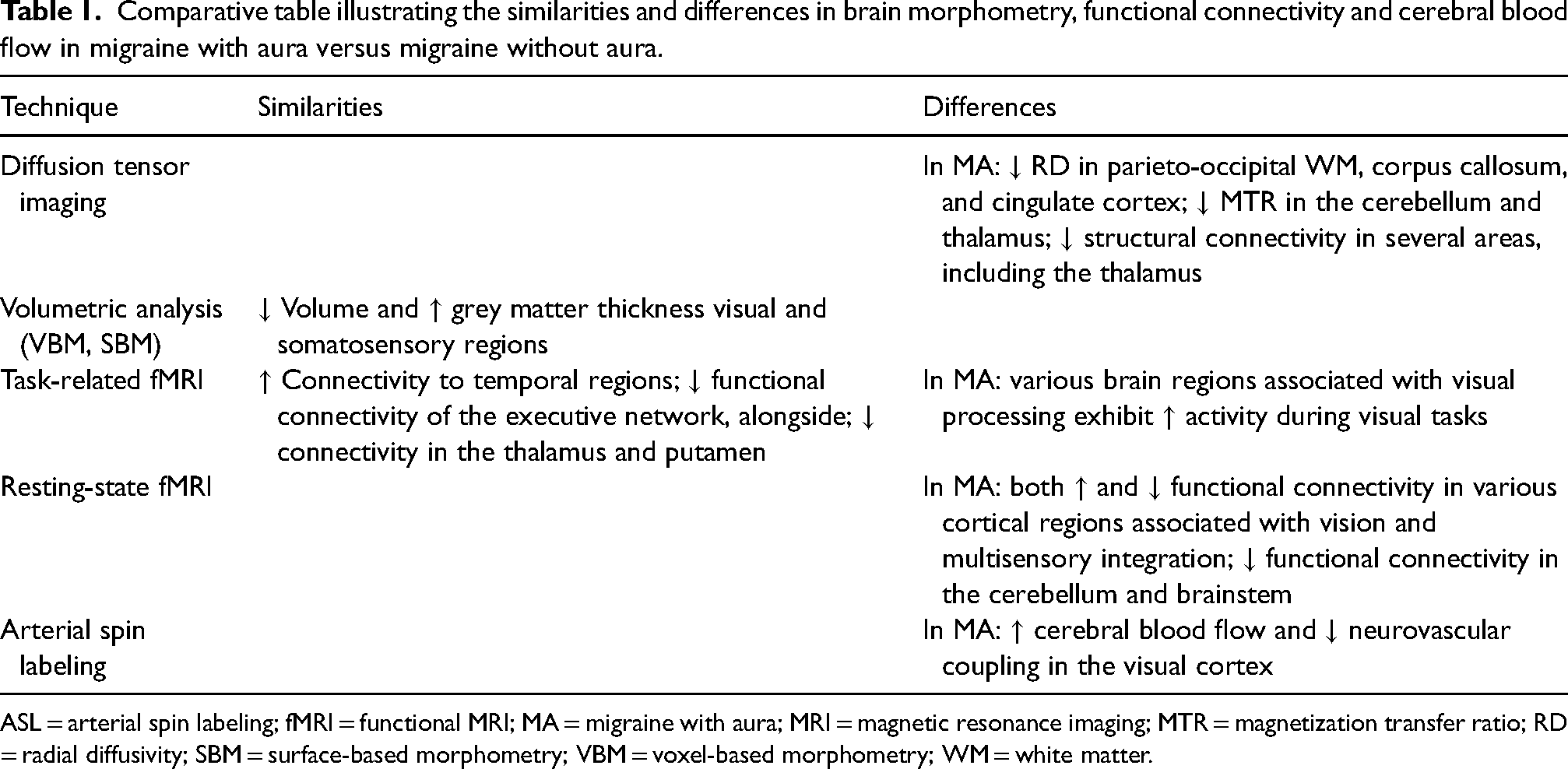
ASL = arterial spin labeling; fMRI = functional MRI; MA = migraine with aura; MRI = magnetic resonance imaging; MTR = magnetization transfer ratio; RD = radial diffusivity; SBM = surface-based morphometry; VBM = voxel-based morphometry; WM = white matter.
Using seed-based functional MRI connectivity, it was shown that patients with MA have reduced connectivity between the insula and occipital regions compared to patients with MO, regardless of the clinical characteristics of the aura (30). By contrast, MO and MA patients shared a heightened connection to temporal areas compared to healthy subjects (30). Functional connectivity changes in the insula were also observed in another study with a smaller sample size, which revealed increased functional coupling between the dorsal insula and the cerebellum (vermis VI) in patients with MA but not in those with MO. Similar to the previous study, these alterations of connectivity were not related to the characteristics of aura (31).
During an fMRI study conducted on a painful caloric task in a small, non-replicated study, Russo et al.(32) observed increased spontaneous activity in regions associated with visual processing, including the lingual gyrus, inferior parietal lobule, inferior frontal gyrus and cerebellum in patients with MA compared to those with MO. Nonetheless, when fMRI was conducted in reaction to visual stimuli in a study with a larger sample size, other researchers observed heightened activation of the visual pathway in MA patients compared to those with MO (33,34). Interestingly, this heightened activation strongly correlated with levels of photophobia, whereas no differences in perfusion levels, as assessed by ALS, were noted (33,34). During an fMRI assessment with visual stimulation conducted during a migraine attack in a limited cohort of patients, the an elevation in the BOLD signal was noted within the right brainstem, left cerebellum, and right middle temporal gyrus in patients with MA compared to those with MO. By contrast, they observed a similar activation in the occipital lobe, cuneus, lingual gyrus, and several frontal regions (35).
Numerous studies have examined, frequently with diverse statistical methodologies, the functional brain connectivity at rest in the two cohorts of migraine patients, in contrast to healthy individuals (20,26–40). In a resting-state fMRI study (voxel-level threshold was set to p = 0.05 uncorrected), compared with controls, both patient groups exhibited reduced activity in the right middle frontal gyrus and the dorsal anterior cingulate cortex, regions belonging to the executive control network (20). In another small study, both the MO and MA groups showed reduced functional connectivity in the thalamus and putamen (36), compared to healthy subjects. By contrast, patients with MA exhibited reduced connectivity in the cerebellum and brainstem compared to those with MO (36). Furthermore, enhanced connectivity was observed in the occipital lobe (36), the lingual gyrus (37), the fronto-parietal regions, the anterior cingulate cortex, the cerebellum (38), the right angular gyrus, the right precentral gyrus, the right postcentral gyrus and the right insular cortex (39).
Driven by the objective to ascertain if more complex focal clinical manifestations of aura correlate with distinct functional abnormalities, Silvestro et al.(40) employed a resting-state fMRI methodology (voxel-level threshold was set to p = 0.001 uncorrected) and observed that patients exhibiting complex auras (visual followed by parenthetic and/or dysphasic aura) had enhanced functional connectivity within the visual network (left lingual gyrus) and sensorimotor network (right anterior insula) compared to patients with solely visual auras and those without aura. In a logistic regression analysis, the functional connectivity of the right anterior insula effectively distinguished patients with complex aura from those with pure visual aura (40).
ASL is an MRI technique employed to evaluate cerebral blood perfusion by labeling blood water during its transit in the brain, thereby eliminating the need for gadolinium contrast (41).
Using ALS, Fu et al.(42) observed that patients with MA had elevated cerebral blood flow (CBF) levels in the bilateral superior frontal gyrus, postcentral gyrus and cerebellum. By contrast, they found reduced CBF values in the bilateral middle frontal gyrus, thalamus and medioventral occipital cortex. Additionally, a recent study revealed elevated cerebral blood flow levels and diminished neurovascular coupling in the visual cortex of MA patients compared to those with MO, suggesting that this neurovascular decoupling in the visual cortex could be a possible link between exposure to trigger factors and the initiation of aura phenomena (43).
Discussion
Overall, the results obtained using different MRI methods reported varying and sometimes inconsistent results. This is mainly due to various study biases, such as inconsistency in scan timing, the inclusion of patients undergoing migraine prevention, small sample sizes (Table 2), the use of more liberal statistics and the lack of replicability of the studies.
Overview of studies using various MRI techniques to investigate differences between migraine with and without aura (legend below).

ASL = arterial spin labeling; DSC-MRI = dynamic susceptibility contrast magnetic resonance imaging; DTI = diffusion tensor imaging; fMRI = functional MRI; HC = healthy controls; MA = migraine with aura; MO = migraine without aura; MRI = magnetic resonance imaging; MTC = magnetization transfer ratio; NA = not available; ReHo = regional homogeneity; SBM = surface-based morphometry; TBSS = tract-based spatial statistics; VBM = voxel-based morphometry.
Migraine is a recurrent neurological disorder marked by symptoms that may emerge up to two days prior (pre-ictal or prodromal phase) and one day subsequent (post-ictal or postdromal phase) to the pain phase of an attack. Multiple studies highlighted that both brain structure and function may alter according to the phase of the migraine cycle. Furthermore, structural and functional changes may differ based on the specific moment of scanning during the variable duration of the interictal period (46–49). This highlights the importance of a careful classification of patients based on the timing of the MRI scan in relation to the migraine phase. As shown in Table 2, most studies do not specify the phase of the migraine cycle during which the scan was conducted. Although other studies indicate that patients were scanned when pain-free, they did not assess the occurrence of an attack on the day prior or the days after the scanning session. This lack of specification poses the risk of recording patients during the peri-ictal phase of a migraine episode, when metabolic and cerebral functions may change (50–53), maybe even more so in cases of migraine with aura. This heterogeneity may significantly obscure the assessment of structural and functional differences or similarities between MA and MO.
Another prevalent bias identified in numerous studies is the incorporation of patients taking preventive medications for migraine, which may alter the clinical progression of the disease, as well as both the structure and function of migraine brain. There is evidence today that preventive treatments commonly used in the treatment of migraine, such as beta-blockers, antiepileptics and monoclonal antibodies against the calcitonin gene-related peptide (CGRP), may alter the structure and function of the migraine brain (54–58). Furthermore, disease progression in both a favourable and unfavourable direction can result in a different pattern of functional activity (59,60). In essence, migraine prevention therapies, either directly or indirectly, may bias the study findings.
Moreover, inadequate sample numbers, variability in preprocessing pipelines and an expanding array of potential analysis methodologies, collectively termed researcher degrees of freedom, can hinder inter-study comparability. Indeed, a recent Delphi study acknowledged that methodological problems may significantly contribute to the varied findings in MRI research (61).
Greater emphasis ought to be placed on the clinical phenotyping of migraine aura, as underscored by the initial observation of one study (40). There is growing evidence that the anatomical and functional involvement of cortico-subcortical regions varies according to the type and severity of focal neurological symptoms associated with migraine aura (40,62–65).
Notwithstanding these evident limitations, research comparing MA patients with MO patients tends to indicate a heightened engagement of the visual regions, cerebellum and thalamus. Specifically, the visual regions in MA patients relative to MO patients, showed a decrease in radial diffusivity(16) and microstructural integrity of white matter fibre bundles (23), alongside with an augmentation in grey matter thickness (27), enhanced functional connectivity across the visual pathway (33,34,36,37,40), increased connectivity between the occipital cortex and the insula (30), and a decline in regional cerebral blood flow (43). The cerebellum of patients with MA exhibited a lower white matter magnetization transfer ratio (22), reduced resting-state functional connectivity (38), increased regional cerebral blood flow(42) and heightened functional connection with the insula (31). The thalamus of patients with MA had diminished white matter fibre microstructure compared to MO patients (23), characterized by a lower magnetization transfer ratio on DTI (19), decreased resting functional connectivity(36) and reduced regional cerebral blood flow (42).
A comprehensive pathophysiological understanding of the role of these brain regions in migraine with aura is challenging. The participation of regions associated with visual processing in the pathophysiology of aura is apparent, while the role of the cerebellum is less clear. The deep nuclei of the cerebellum are recognized for their ability to receive nociceptive information and engage in pain perception and modulation via their connections with the brainstem and thalamus (66,67). Some studies indicated increased engagement of crus I and vermis VI, mostly involved in spatial processing functions, somatosensory integration, and visual working memory(68,69); nevertheless, it is unfeasible to assign a somatotopic relevance to their participation, as morphological investigations have identified diverse representations of identical body regions across several cerebellar sites (70). We might hypothesize that cerebellar antinociception may be less effective in individuals with MA. Both the primary and accessory visual areas, along with the cerebellum, are intricately linked to many thalamic nuclei. Thalamic nuclei are widely recognized for their significant role in the pathogenesis of migraine, both anatomically and functionally (48,59,71,72). The thalamus serves as a central hub for processing and filtering multisensory information and is a crucial pacemaker of cerebral rhythms. Given that all investigated MRI metrics of the thalamus are diminished, we can hypothesize that this structure processes sensory information with less efficiency in MA patients compared to MO patients.
Consequently, despite the study's limitations, we can hypothesize that patients with migraine with aura exhibit altered cerebellar antinociceptive capacity, reduced information filtering at the thalamic level and increased engagement of the regions responsible for visual processing compared to those without aura. Whether this pattern represents the consequence of the recurrence of CSD or a primary predisposition to it remains to be determined. Nevertheless, while research has revealed distinctions, it has also revealed shared alterations between the two cohorts. Indeed, both MO and MA cohorts exhibited diminished grey matter volume in the accessory visual areas V3 and V5 (26), augmented thickness of the visual and somatosensory cortex (27), decreased functional connectivity within the frontal executive network (20), and reduced functional connectivity of the thalamus and putamen (36). While the majority of this data remains un-replicated, these findings may lead to several conjectures. The involvement of the primary and extrastriate visual regions in both cohorts underscores their significance in the pathophysiology of migraine. Moreover, it has to be noted that the somatosensory cortex, the frontal regions of the executive network, the thalamus and the putamen (part of the basal ganglia) are components of the salience network (SN) (73). In humans, the SN identifies and filters significant environmental inputs while modulating attention and behaviour in conjunction with the neurolimbic system. The SN may be crucial in the emotional and attentional modulation of pain perception, as well as in antinociception and interoception (74). Again, a malfunction of this system may be either primary or subsequent to the recurrence of attacks.
In conclusion, these findings suggest that, whereas MA patients and MO patients exhibit certain common alterations, distinct functional and structural abnormalities in some areas distinguish the two conditions and may influence the different clinical manifestations of the disease. An improved phenotypic characterization of the aura, the elimination of pharmacological confounding and controlled MRI acquisitions throughout the migraine cycle phase could enhance the understanding of the cerebral mechanisms by which focal neurological symptoms precede migraine attacks in certain patients. Consequently, we are likely to uncover, as the author Oliver Sacks articulates, ‘A variety of different syndromes may be recognized within the migraine-complex, and these may overlap, merge, and metamorphose into one another’(75).
Article highlights
Migraine with and without aura shared activation of common areas belonging to the salience and visual network.
Structural and functional alterations in certain brain areas, including the cerebellum, thalamus, and visual areas, characterize and distinguish MA from MO.
More studies with a rigorous and standardized approach are needed to understand the differences between the two conditions.
Footnotes
Acknowledgments
The contribution of the G. B. Bietti Foundation to this paper was supported by the Italian Ministry of Health and Fondazione Roma.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
