Abstract
Background
Diffusion-tensor imaging can be applied to describe the microstructural integrity of the whole brain. As findings about microstructural alterations in migraine are inconsistent, we aimed to replicate the most frequent results and assess a relationship between migraine parameters and changes in microstructure.
Methods
Diffusion-weighted MRI data of 37 migraine patients and 40 controls were collected. Two indices of diffusion of water molecules, fractional anisotropy and mean diffusivity were used in a voxel-wise analysis. Group comparisons were carried out in SPM12 using age and sex as covariates. Statistically significant results survived family-wise error correction (pFWE < 0.05). Migraine intensity, frequency, and duration were self-reported and correlated with mean fractional anisotropy and mean diffusivity values across clusters.
Results
Migraine patients showed significantly lower fractional anisotropy in occipital regions, and significantly higher fractional anisotropy in thirteen clusters across the brain. Mean diffusivity of migraine patients was significantly decreased in the cerebellum and pons, but it was not increased in any area. Correlation between migraine duration and fractional anisotropy was significantly positive in the frontal cortex and significantly negative in the superior parietal lobule.
Conclusion
We suggest that microstructural integrity of the migraine brain is impaired in visual areas and shows duration-related alterations in regions of the default mode network.
Introduction
Magnetic resonance imaging (MRI) offers the possibility to identify structural and functional alterations in the migraine brain. With diffusion-tensor imaging (DTI) technique we can reveal changes in microstructural level to estimate white matter integrity (WMI) by investigating the diffusion of water molecules in the brain. There are two commonly used parameters that quantify white matter integrity: fractional anisotropy (FA) indexing the directionality of water diffusion and mean diffusivity (MD) reflecting the amplitude of water diffusion (1).
In 2022, Rahimi et al. (2) reviewed migraine-related DTI studies and came to the conclusion that the results of DTI studies targeting WMI in migraine compared to controls are inconsistent and inconclusive. The most frequently reported structures showing altered integrity are the corpus callosum, cingulate fibers, brainstem areas, thalamic radiations, and the superior and inferior longitudinal fasciculus (2). However, structural connectivity is able to change dynamically during the migraine cycle. For instance, thalamic fibres (3) and several pain-processing regions of the brainstem (4) demonstrated varying microstructural characteristics depending on the time elapsed since the last attack.
Besides investigating white matter tracts integrity, DTI technology can be used to understand any changes in the diffusion across the brain. For instance, significant differences emerged in FA and MD within the thalamus when investigated between attacks (3). Using a different type of analysis, MD was found to be lower in bilateral thalamus in migraine without aura compared to control subjects, or more precisely, in the left and right anterior thalamic radiations (5), which connects prefrontal lobe to thalamus. Using an extension of DTI (diffusion kurtosis imaging), MD was found to be increased in the periaqueductal gray (PAG) matter among patients with episodic migraine compared to controls (6).
High heterogeneity of results may stem from the method of analysis applied. In addition, inclusion criteria regarding chronicity of migraine, preventive medication, comorbidities also differ across studies. There are also quite a few differences in demographic characteristics (e.g., age and sex) of samples in different studies that might affect the results (7). Finally, a meta-analysis on DTI results obtained with mainly tract-based spatial statistics (TBSS) in migraine pointed out that number of studies on WMI is still quite low (7), thus, more studies are needed. Regarding the association between WMI and clinical variables, the results are also mixed. There are studies in which disease duration was related to tract integrity (5), while other studies reported null findings (8).
Our aim was, using a whole-brain voxel-wise analysis, to test whether WMI in episodic migraine without aura (MwoA) patients differ from headache-free control subjects (HC), and possibly replicate the most frequent findings of migraine DTI studies. In addition, for exploratory purposes, we also aimed to examine in the MwoA group whether clinical characteristics, such as migraine intensity, frequency and duration were correlated with FA and MD values.
Methods
Participants
Participants, aged between 18-39 years, with MwoA (N = 40) and HC (N = 47) were recruited via advertisements and headache clinics. ES, NK and AG were first screened by a trained researcher using the Mini International Neuropsychiatric Interviews (9), GJ then participated in a medical examination carried out by experienced neurologist and psychiatrist researchers. Exclusion criteria were any current or past serious medical, neurological (except migraine for participants with migraine) or Axis I. psychiatric disorder and psychotropic medication use. Inclusion criteria for episodic MwoA were based on the International Classification of Headache Disorders-III criteria (10). All participants were given a headache diary, and only those who reported no headache attack during the study period could remain in the group of healthy controls. Participants with MwoA did not take any prophylactic medication during the preceding three months, they had at least one migraine attack per month, and were included only if they did not report overusing headache medication. All subjects had normal or corrected-to-normal vision and were right-handed. Participants with MwoA underwent MRI scan during the interictal period, i.e., they had no headaches 48 h before MRI scans.
The study was approved by the Scientific and Research Ethics Committee of the Medical Research Council of Hungary (23609-1/2011-EKU; 23421-1/2015/EKU), and written informed consents were received from all subjects in accordance with the Declaration of Helsinki. The study followed the STROBE statement and guidelines.
Self-report measures
The following migraine-related data were collected from all MwoA patients: 1) pain intensity of migraine attacks in the last three months (measured by a 0–10 numerical rating scale); 2) migraine frequency (average number of migraines per month and in the last three months); 3) duration (number of years lived with migraine).
All participants completed the 20-item trait anxiety version of State-Trait Anxiety Inventory (STAI; 11). Items are answered on a 4-point Likert type scale (1 = almost never to 4 = almost always). Current depressive symptoms – covering the past several days – were assessed by the Zung Self-Rating Depression Scale (12). Twenty items are answered on a 4-point Likert-type scale (from 1 = a little of the time to 4 = most of the time). Both questionnaires were filled out on the scan day.
Internal consistency (Cronbach-α) of all self-report measures was excellent (ZSDS = 0.82, STAI-T = 0.90).
Imaging
Diffusion-weighted MRI (DWI) data were acquired at 3T (Philips Achieva scanner, Philips Medical Systems, Best, The Netherlands) and was collected with the following acquisition parameters: single shot SE-EPI sequence with b = 800 s/mm2 diffusion weighting in 32 directions, one
Preprocessing and statistics overview
Preprocessing of the DWI data was performed using ExploreDTI, a Matlab-based (MATLAB 8.3, The MathWorks Inc., Natick, MA, 2000) software package with advanced features for correction steps, robust tensor fitting, deterministic tractography, etc. (13). Subject motion, susceptibility-related and EPI-related (eddy currents) distortions are all corrected in a single interpolation step, combining rigid body transformations for motion, and nonlinear transformations for the latter two. To avoid angular inaccuracies, the b-matrix (the diffusion weighting directions) is also rotated in this step (14). Each subject’s T1-weighted image was used as registration target to correct the distortions related to EPI-acquisition (15); thereby DW-images were spatially aligned to the T1W images.
Tensor fitting was performed using the RESTORE (Robust Estimation of Tensors by Outlier Rejection) (16) algorithm. Two voxel-wise DTI-measures, FA and MD (17–19) were calculated from the tensor eigenvalues, following their well-established definitions, to be used in voxel-level and ROI-based analyses.
Voxel-wise analysis
Images containing these DTI scalar values were ‘co-registered’, i.e., transformed to a study specific, common coordinate system utilizing the DARTEL tools (20) of the SPM12 toolbox (21, 22), Online Supplement B, for a detailed description). Spatial smoothing was performed using a Gaussian kernel with 8 voxel FWHM.
As we aimed at examining microstructure across the whole brain, we preferred the above-described analyses to TBSS. Though TBSS is considered to have high statistical power, it constrains the analysis to the FA skeleton (i.e., the supposed center of WM tracts), thus causing spatial information loss. In addition, TBSS has been reported causing poor spatial alignment performance in regions with complex white matter structures (23).
Between-group statistical analysis
Co-registered maps of the diffusion-tensor scalar parameters (i.e., FA and MD) were compared between MwoA patients and HC, using voxel-wise two-sample T-tests from the second level GLM-tools of SPM12. Statistical significance was assessed on p = 0.001 primary threshold (T = 3.21) for defining clusters, and no extent threshold, followed by cluster-level inference (pFWE < 0.05). Subject age and sex were used as covariates in the calculations.
Correlation analysis
For exploratory purposes, we correlated the mean FA and MD values across clusters with the migraine intensity (reported on a 0-10 scale), frequency (average number of migraine attacks in the last three months) and duration (years lived with migraine) across all migraine subjects, including age and sex as covariates. Only results controlled for false discovery rate (FDR < 0.05) were considered as statistically significant.
Visualization
Between-group comparisons and correlation analyses were carried out in DARTEL Template space, then t- and r-score maps corrected for multiple comparisons were transformed to MNI152 space (24). Significant clusters were identified in WFU_PickAtlas v. 3.0.5 (The Functional MRI Laboratory, Wake Forest University School of Medicine, NC, United States) using the Automated Anatomical Labeling atlas (aal; 25) and visualized in MRIcroGL3 (26). Further identification of significant clusters were carried out using the following atlases: 1) CONN (27), 2) JHU DTI-based white-matter atlas (28), and UManitoba – JHU Functionally-Defined Human White Matter Atlas (29,30).
Results
Data of two participants with MwoA had to be excluded from the analysis as they suffered a migraine attack during the MRI scans, and data of one participant with MwoA had to be excluded due to anti-headache medication 24 h before the MRI scan. Out of 47 HC, seven participants had to be excluded due to high level of motion during the MRI scans, insufficient co-registration performance, or missing data. The final sample consisted of 37 MwoA patients (30 females, mean age = 26.86, SD = 4.73) and 40 HC (25 females, mean age = 25.75, SD = 4.25). See Figure 1 for inclusion procedure.
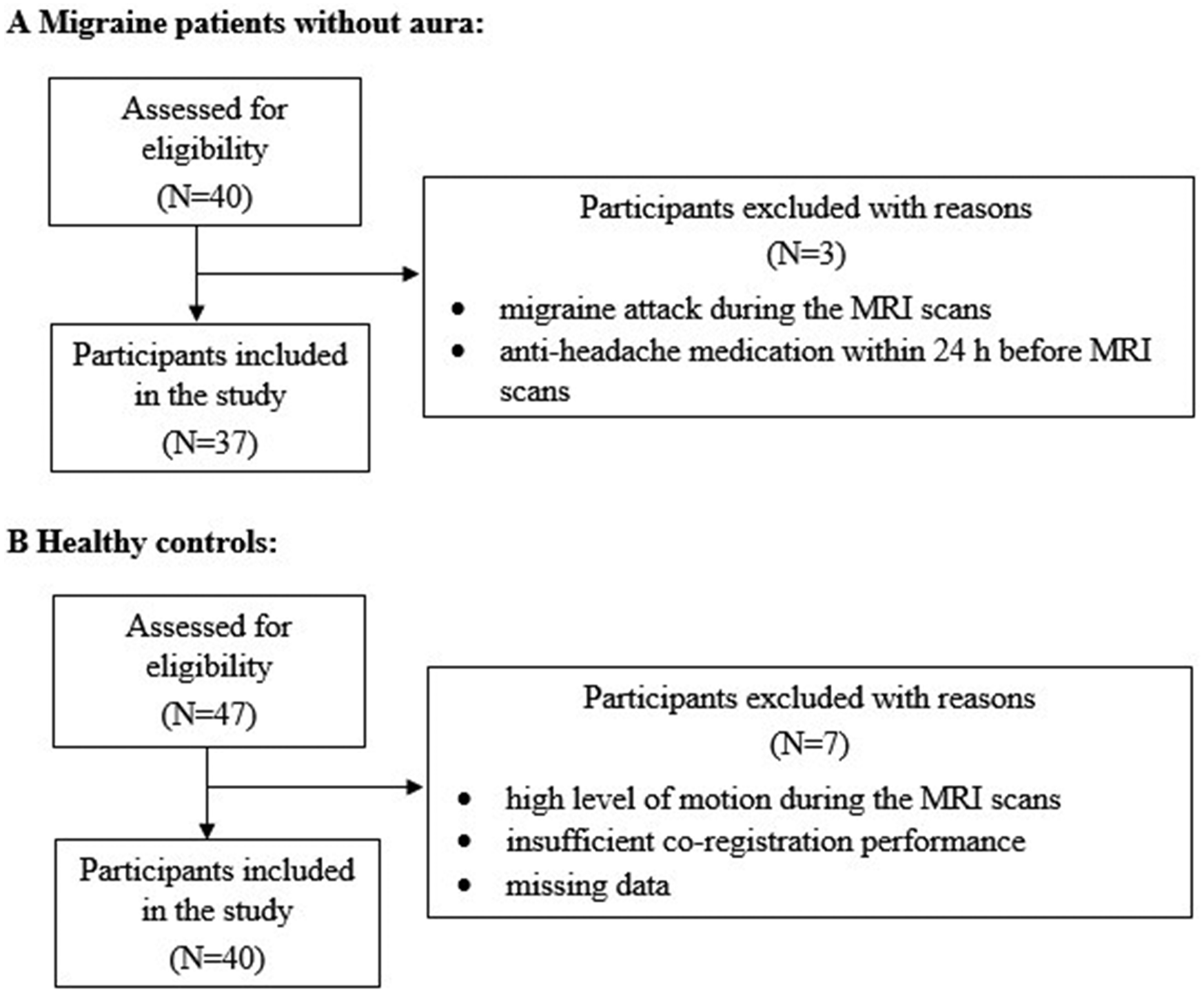
Inclusion of participants in the study.
The descriptive data and clinical characteristics of participants are shown in Table 1. The proportion of female participants in the two groups did not differ from each other (χ2 = 3.25, p > 0.05). When presenting descriptive results and group comparisons of questionnaire data, we preferred means to ranks, as it is easier to interpret differences in means than in ranks. Therefore, we performed these analyses using a series of bootstrapped independent t-tests. Using a series of bootstrapped independent t-tests, HC and MwoA patients did not differ on mean age (t = 0.068, p > 0.05), trait anxiety (t = 0.909, p > 0.05), or current depressive symptoms (t = 0.050, p > 0.05).
Demographic data and clinical characteristics of participants (N=77).

Note. Migraine frequency per month and Pain severity of migraine attacks variables had no normal distributions, their medians (and interquartile range, IQR) are 1.50 (IQR: 3.00) and 6.00 (IQR: 2.25), respectively. HC, healthy controls; MwoA, migraine patients without aura; STAI-T, trait anxiety version of state-trait anxiety inventory; ZSDS, zung self-rating depression scale.
Voxel-based analysis: Group differences
There were significant differences in FA (see Table 2) and MD (see Table 3) between MwoA patients and HC. See Online Supplementary Table 1 for classification of clusters according to the aforementioned atlases.
Fractional anisotropy in episodic migraine without aura patients vs. healthy controls.
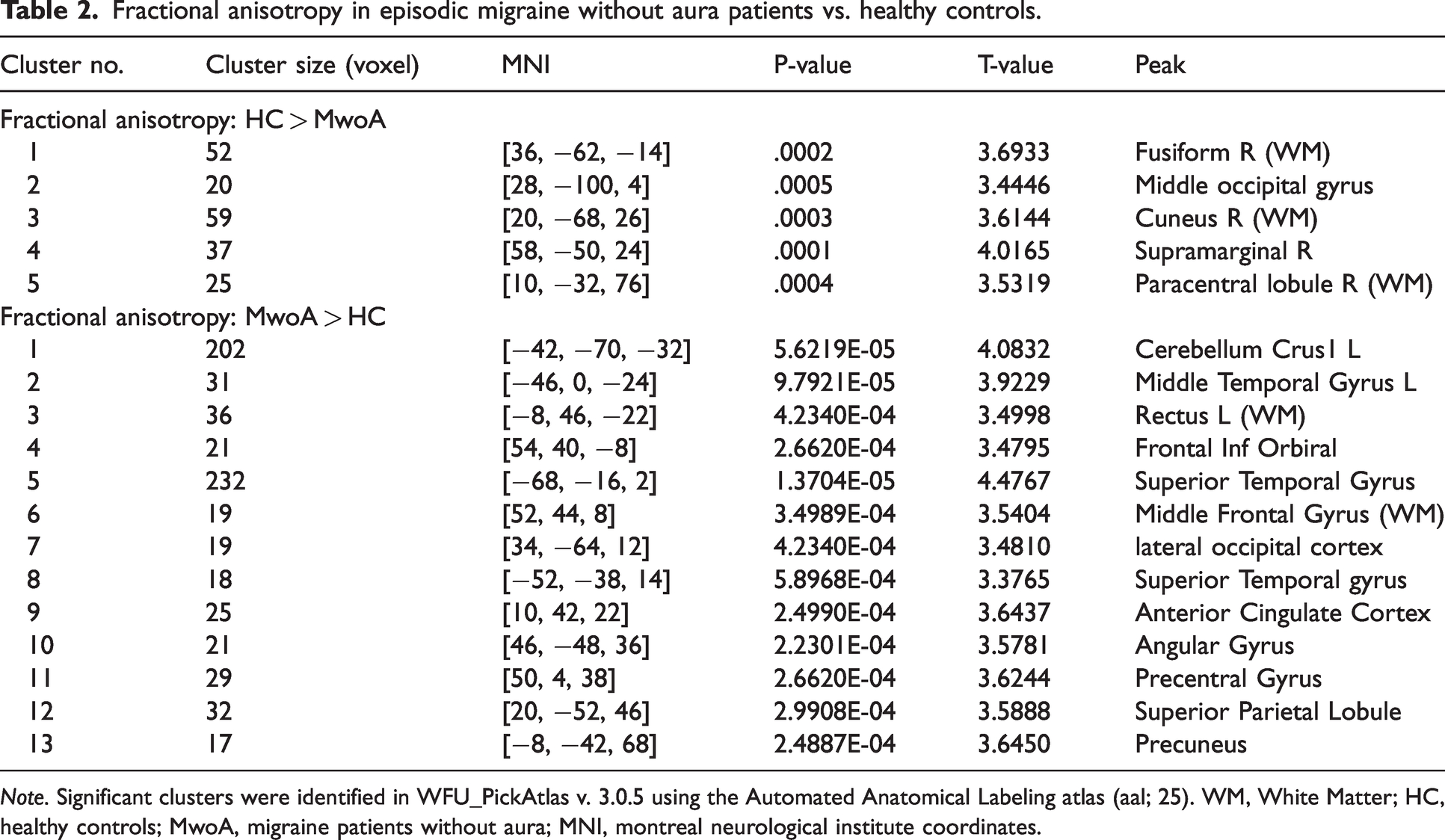
Note. Significant clusters were identified in WFU_PickAtlas v. 3.0.5 using the Automated Anatomical Labeling atlas (aal; 25). WM, White Matter; HC, healthy controls; MwoA, migraine patients without aura; MNI, montreal neurological institute coordinates.
Mean diffusivity in episodic migraine without aura patients vs. healthy controls.

Note. Significant clusters were identified in WFU_PickAtlas v. 3.0.5 using the Automated Anatomical Labeling atlas (aal; 25). HC, healthy controls; MwoA, migraine patients without aura.
Higher FA was found in five (right sided) clusters in HC compared to MwoA patients, including clusters in the paracentral lobule (precentral and postcentral gyri), occipital lobe (occipital pole, lingual gyrus), fusiform gyrus (occipital fusiform gyrus and temporal occipital fusiform cortex) and parietal lobe (angular gyrus, precuneus). Higher FA in the MwoA group was found in thirteen clusters, including clusters in the cerebellum, temporal (middle and superior temporal gyri, temporal pole, planum polare, planum temporale) and frontal lobes (frontal pole, frontal medial cortex) bilaterally. FA in the left postcentral gyrus and in the right precentral gyrus, angular gyrus, and paracingulate cortex was also significantly increased in the MwoA group compared to HC (see Table 2 and Figure 2).

Clusters with significantly different fractional anisotropy (FA) in migraine without aura patients vs. healthy controls. Blue clusters: lower FA in migraine without aura patients. Red clusters: lower FA in healthy controls.
Mean diffusivity was increased in the cerebellum and brainstem in HC compared to MwoA patients, while the other contrast (i.e., MwoA > HC) did not produce any significant voxels (see Table 3).
Voxel-based correlation analysis
For exploratory purposes, we examined in the MwoA group whether migraine characteristics including migraine intensity (reported on a 0-10 visual analogue scale), frequency (average number of migraine attacks in the last three months), and duration (number of years lived with migraine) were correlated with FA and MD values. A positive significant correlation (Spearman rho = 0.48, p = 0.0032) between duration and FA values in the cluster including frontal pole and medial frontal cortex [−8, 46, −22] was found. Duration had a negative relationship (Spearman rho = −0.35, p = 0.0391) with FA in the superior parietal lobule [20, −52, 46]. Other characteristics did not correlate with FA, and MD did not correlate with any of the migraine characteristics.
Discussion
In this study, a whole-brain diffusion-MRI analysis was used to investigate the microstructural integrity of both white and grey matter in episodic migraine patients without aura during interictal phase. MwoA group showed significantly higher FA in several regions throughout the brain compared to HC, while significantly lower FA was found mainly in occipital areas. MD was significantly decreased in the MwoA group in brainstem and cerebellum relative to HC. Those suffering from migraine for a longer period of time demonstrated higher FA in frontal pole and medial frontal cortex, but lower FA in the superior parietal lobule.
While interpreting our results, it is important to bear in mind that we carried out voxel-wise analyses. In this way, our findings are not limited to white matter. Also, it is worth noting that DTI indices are unsuitable for revealing underlying causes of brain tissue changes. Thus, care must be taken when interpreting DTI metrics. It has to be pointed out that changes in FA values refer to different alterations depending on the type of brain tissue. In white matter, lower FA indicates an impaired axonal integrity, which may arise from demyelination, axonal loss, inflammation, etc. (2). In the more isotropic grey matter, however, lower FA is desirable, and increases as a result of dendrite pruning, for instance (31). The direction-independent degree of diffusion (i.e., MD) is greater when barriers of the diffusion break down, or the volume of the extracellular space increases (32). Migraine-associated microstructural alterations are typically decreased FA and increased MD values in the white matter (2). We found both lower and higher FA in the MwoA group compared to HC. It is striking that MwoA patients showed lower FA values in occipital regions, because changes in visual cortex functioning of migraine patients have been revealed by numerous studies (33,34). Therefore, FA alterations in these areas might be either the reason or the consequence of the functional alterations.
To date, only a few studies have reported a relationship between migraine duration and FA value. A negative correlation in corpus callosum was reported by Yu et al. (35) and Yuan et al. (36) in episodic and by Li et al. (37) in chronic migraine patients. The main finding of our study is a similar result in the superior parietal lobule and an opposite one (i.e., positive correlation) in the frontal pole and medial frontal cortex. Both clusters are located in the default mode network (DMN; see Figure 3), which is one of the most affected networks in migraine in terms of altered functional connectivity (38). The region in the superior parietal lobule includes fibers linking precuneus and thalamus and was more isotropic the longer a participant has suffered from migraine. In healthy people, structural and functional connectivity between precuneus and thalamus is strong, but they change with age (39). As we corrected for age, the lower FA value may be indicative of a disrupted connection owing to the higher number of years lived with headache. DMN has an important role in the coping with stressful situation (40), and its alterations have been shown by studies investigating chronic pain including migraine (41,42). Therefore, a disturbed structural connectivity within this network may result in impaired pain processing (43) and stress response (44) ascribed to migraine.
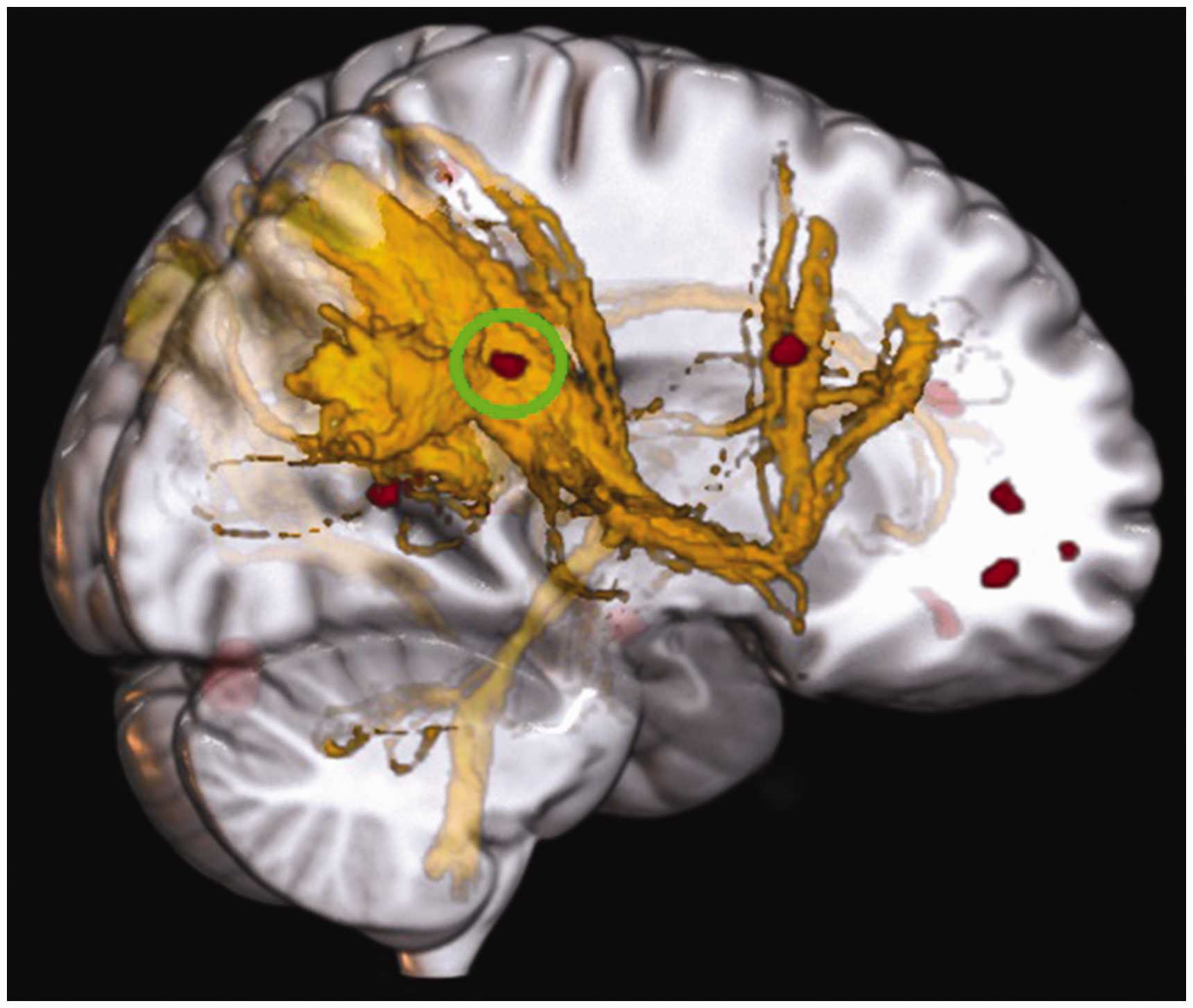
Cluster [MNI: 20,-52,46] in the default mode network (DMN; yellow) with fractional anisotropy showing a negative correlation with migraine duration. Visualization of DMN: (29).
As our goal was to compare our findings to those of previous studies, we summarized in Table 4 which of the most common findings we were able to replicate (3,35,36,45–48).
Replication of the most common findings of migraine-related DTI studies – fractional anisotropy.

Note. HC, healthy controls; MwoA, migraine patients without aura; MwA, migraine patients with aura.
cluster in fibers connecting cingulum and thalamus
Limitations
Apart from small samples size and imbalanced groups in terms of sex ratio, this study has some further limitations. Only episodic migraine patients without aura were included in the study and scanned during interictal period. Therefore, all findings and conclusions apply exclusively to this subgroup. As a cross-sectional study, we had no opportunity to examine either long-term or dynamic structural changes throughout the migraine cycle. Voxel-wise analysis of brain tissue integrity may be unusual, but it aimed at assessing microstructural characteristics not only in white matter fibers, but also in grey matter (22).
Conclusion
This study compared the microstructural integrity of brain between episodic migraine patients without aura and healthy controls using diffusion metrics. We found several differences, the most remarkable of which is the altered integrity in the occipital regions, because it is a highly affected area in migraine. In addition, we found a relationship between migraine duration and integrity metrics of several areas within the default mode network. Altered integrity between the thalamus and precuneus is in line with the literature in terms of migraine-related changes in thalamic radiations. Altered functioning of this network in migraine might be related to structural alterations. Only longitudinal studies could answer the question as to whether structural changes induce functional ones, or the opposite.
Article highlights
Migraine patients have altered brain microstructure integrity in visual areas. The longer a patient lives with migraine, the more the integrity of the default mode network is affected, mainly between thalamus and precuneus.
Supplemental Material
sj-xlsx-1-cep-10.1177_03331024231216456 - Supplemental material for Microstructural differences in migraine: A diffusion-tensor imaging study
Supplemental material, sj-xlsx-1-cep-10.1177_03331024231216456 for Microstructural differences in migraine: A diffusion-tensor imaging study by Dóra Dobos, Gyöngyi Kökönyei, Gyula Gyebnár, Edina Szabó, Natália Kocsel, Attila Galambos, Kinga Gecse, Dániel Baksa, Lajos R. Kozák and Gabriella Juhász in Cephalalgia
Footnotes
Acknowledgements
The fMRI study was conducted as a groupwork at the Magnetic Resonance Research Center, Semmelweis University. The authors thank Ádám György Békésy-Szabó, István Kóbor, Krisztina Koósné Oláh, and Márk Folyovich for their contribution, and also Zsuzsanna Tóth for her support during data collection. Preliminary results of the study were published as a conference abstract: Dobos D, Kökönyei G, Gyebnár G, et al. Microstructural Differences in Migraine: a Diffusion Tensor Imaging Study. Neuroscience Applied 2023; 2: 101019 101105. DOI: 10.1016/j.nsa.2023.101105.
Data availability statement
The datasets generated for this study are available on request to the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Hungarian Brain Research Program [Grants: 2017-1.2.1-NKP-2017-00002, NAP2022-I-4/2022]; by project no. TKP2021-EGA-25 supported by the Ministry of Innovation and Technology of Hungary from the National Research, Development and Innovation Fund, financed under the TKP2021-EGA funding scheme; by the National Research, Development and Innovation Office, Hungary [2019-2.1.7-ERA-NET-2020-00005], under the frame of ERA PerMed (ERAPERMED2019-108); by the Hungarian National Research, Development, and Innovation Office [grant number: K 143391]; and the ÚNKP-22-4-I-SE-10, ÚNKP-22-3-II-SE-17, and ÚNKP-22-3-II-SE-27 New National Excellence Program of the Ministry for Culture and Innovation from the source of the National Research, Development and Innovation Fund. KG and DD are fully funded PhD students at Semmelweis University, Doctoral School of Mental Health Sciences. The sponsors had no further role in the study design, in the collection, analysis, and interpretation of data, in the writing of the report, and in the decision to submit the article for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
