Abstract
Background
Diffusion tensor imaging (DTI) provides valuable insights into the white matter integrity of people with migraine. The present study compares the microstructural white matter integrity between a large sample of people with migraine and healthy controls, as well as across different migraine subtypes.
Methods
This cross-sectional case-control study included adults with migraine and age- and sex-matched healthy controls. Each participant and control underwent a single brain magnetic resonance imaging session, including DTI, to assess microstructural white matter integrity using tract-based spatial statistics by voxel-wise comparison using a general linear model (GLM). The DTI outcome measures included fractional anisotropy, mean diffusivity, axial diffusivity and radial diffusivity. Comparisons were made between participants with migraine and controls, as well as among different migraine subtypes (migraine with aura, migraine without aura, episodic migraine and chronic migraine). Additional comparisons were conducted between participants during and outside of migraine attacks, as well as between hemispheres ipsilateral and contralateral to the pain side in those scanned during unilateral migraine attacks.
Results
In total, 293 participants with migraine and 154 healthy controls provided DTI data eligible for analysis. Among the participants, 181 (61.7%) had chronic migraine, 103 (35.1%) had migraine with aura and 79 (26.9%) were scanned during a migraine attack. No significant differences in white matter integrity were identified between participants with migraine and controls, nor among the different migraine subtypes.
Conclusions
Our results suggest that migraine does not result in microstructural alterations within the cerebral white matter. Further research is necessary to explore other potential radiologic biomarkers and pathophysiologic mechanisms underlying migraine.
This is a visual representation of the abstract.
Introduction
Migraine is a prevalent and disabling neurologic disorder, characterized by recurrent headache attacks and associated symptoms such as nausea, vomiting, photophobia and phonophobia (1). Epidemiologic data estimate that migraine affects about 14% of the global population, with a higher prevalence in females than males (2). Despite its widespread prevalence and disabling nature, the neurobiologic underpinnings of migraine remain incompletely understood.
Advances in magnetic resonance imaging (MRI) techniques offer new opportunities to explore the microstructural integrity of the migraine brain. For this purpose, diffusion tensor imaging (DTI) is particularly useful and can provide insights into the organization and integrity of white matter fiber tracts (3). Key outcome measures derived from DTI include fractional anisotropy (FA), mean diffusivity (MD), axial diffusivity (AD) and radial diffusivity (RD). These measures can reveal subtle alterations in white matter that might be attributed to migraine (4,5).
Previous MRI studies using DTI have reported conflicting results regarding white matter integrity in people with migraine. Some studies have suggested the presence of white matter abnormalities, whereas others have found no discernible differences compared to healthy controls (HCs) (5,6). These inconsistencies might be attributed to small sample sizes and heterogeneity within the migraine population (5). Indeed, the potential differences in white matter integrity between migraine subtypes, such as migraine with and without aura, remain unclear (7–9). Because the presence of white matter pathology raises concern about increased risks of neurodegenerative disorders, it becomes critical to ascertain whether the white matter is intact or not among people with migraine (10).
To address the gaps of previous studies, we compared the microstructural white matter integrity using DTI with voxel-wise comparison using GLM between a large sample of people with migraine and age- and sex-matched HCs, as well as across different migraine subtypes.
Methods
The data presented herein were acquired as part of the MRI Core within the parental Registry for Migraine (REFORM) study (ClinicalTrials.gov identifier: NCT04674020) (11). Some of the data from patients included in this study have been published previously (12–16). The study protocol was approved by the relevant ethics committee. Regular oversight meetings were held to monitor the study's progress, address any ethical concerns and ensure compliance with the approved protocol. All participants provided written informed consent prior to the commencement of any study-related assessments or procedures. The study complied with the principles of the Declaration of Helsinki, with later revisions.
Design and participants
A cross-sectional, case–control design was applied to compare the microstructural white matter integrity between participants with migraine and HCs. A complete account of inclusion and exclusion criteria for both groups are provided in the supplemental material (Doc. S1 and S2).
Eligible participants were adults diagnosed with either migraine without aura, migraine with aura or chronic migraine according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) (17). Furthermore, eligible participants were required to report experiencing at least four monthly migraine days within the past three months. The main exclusion criteria were any history of cluster headache, hemiplegic migraine or post-traumatic headache.
HCs were selected to match the participants with migraine in terms of age, age distribution and sex, ensuring a balanced comparison group. Controls were screened to confirm the absence of any personal or first-degree family history of headache disorders, except for infrequent episodic tension-type headaches. In addition, persons with any clinically significant disorder, disease or regular medication intake were excluded.
Procedures
Before the MRI scan, all participants underwent a physical and neurological examination, accompanied by a semi-structured interview to record data on sociodemographics, medical and medicines history. To facilitate more accurate and reliable results, participants abstained from using acute, anti-inflammatory or antihistaminergic medications for ≥48 hours before the scan and avoided caffeinated beverages or food for ≥12 hours prior.
Each participant and HC underwent a single non-contrast enhanced brain MRI session. The MRI scans were performed using a 3.0 Tesla Siemens MAGNETOM Prisma MR imaging unit (Siemens Healthineers, Erlangen, Germany) equipped with a 32-channel head coil. To restrict head motion, foam pads were carefully placed at both temple regions, and participants were instructed to remain still during the scan.
The imaging protocol included magnetization-prepared rapid acquisition gradient echo (MPRAGE), fluid-attenuated inversion recovery (FLAIR) and DTI sequences. Detailed specifications for sequence parameters and the full protocol have been published elsewhere (11,12). The total duration of scan session was about 55 min.
The MPRAGE and FLAIR images were transferred to a centralized server and reviewed by a certified neuroradiologist (DT) who was blinded to the group status of the participants and controls. Any incidental findings of clinical relevance were recorded, and the resulting report was sent to the senior author (FMA), who initiated appropriate follow-up.
The DTI acquisition parameters included: b = 1000 s/mm3 and 2b = 0 s/mm3, repetition time = 11,000 microseconds, echo time = 60 microseconds, flip angle = 90°, field of view 256 × 256 mm, 70 slices, slice thickness = 2 mm, voxel size = 2 × 2 × 2 mm.
Preprocessing
The DTI analyses were performed using tract-based spatial statistics (TBSS) with the FMRIB Software Library (FSL), version 6.0 (Analysis Group, FMRIB, Oxford, UK) (18). First, image artifacts were corrected for eddy current distortions and motion by registering the images to the b = 0 image using linear affine registration. The brain was then extracted using the FSL Brain Extraction Tool (BET). Different tensors at each voxel were fitted to obtain voxel-wise maps of FA, MD, AD and RD for all participants and controls.
Voxel-wise statistical comparisons were performed using the TBSS method, which included registering all FA images to a more common space using the non-linear registration tool FNIRT. Mean FA images were created, and a mean FA skeleton representing the center of all common white matter tracts was extracted (Figure 1). The FA skeleton threshold was set to FA ≥ 0.2. Each participants’ and controls’ FA images were projected onto the mean FA skeleton, and voxel-wise cross-subject statistics were performed among voxels fitting with the skeleton. The same analysis was performed for MD, AD and RD based on the FA skeleton (18).
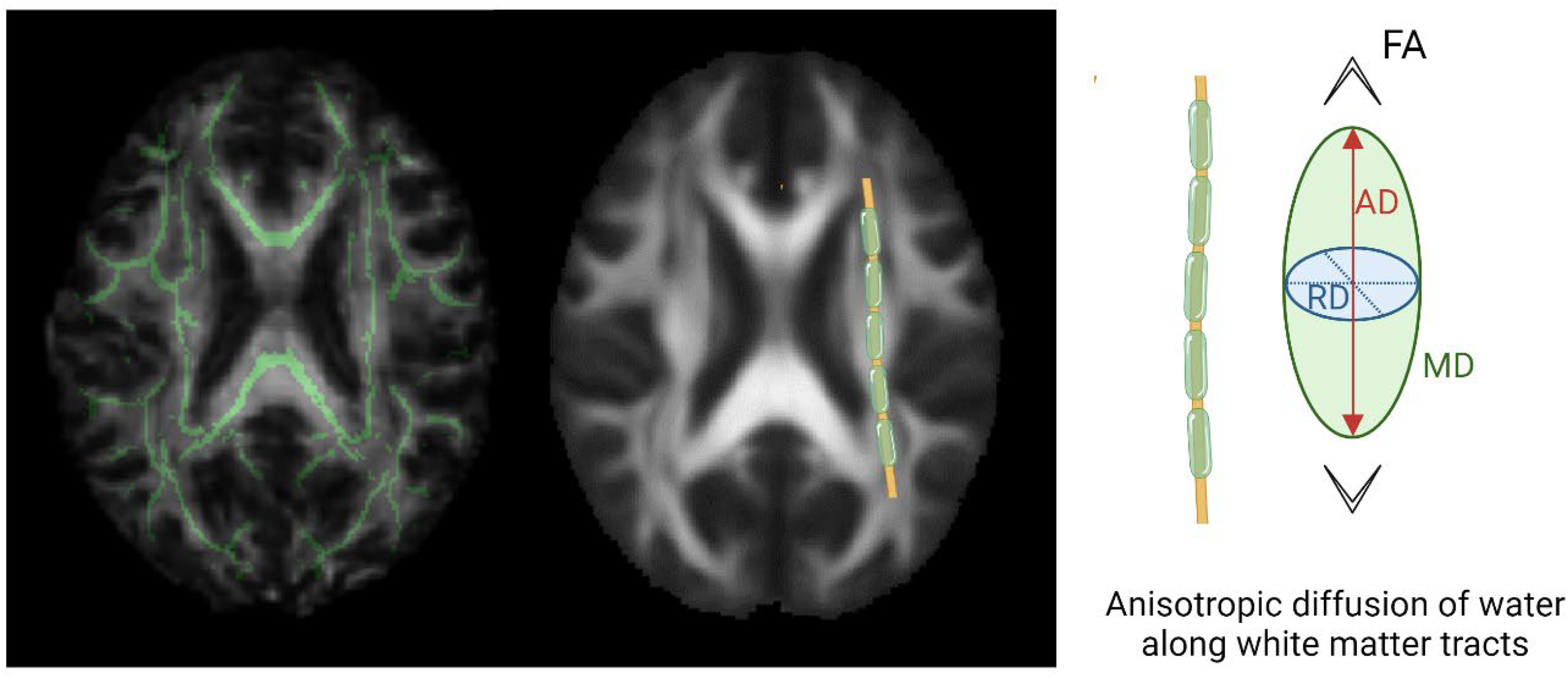
White matter integrity analyzed using diffusion tensor imaging and tract based spatial statistics. Diffusion tensor imaging (DTI) properties were compared between patients with migraine and controls within a FA skeleton of white matter tracts in green on the left. The DTI properties compared were fraction of anisotropy (FA), mean diffusivity (MD), radial diffusivity (RD) and axial diffusivity (AD) represented on the right
To compare hemispheres ipsilateral and contralateral to the pain side during the ictal phase of a unilateral migraine attack, the following steps were performed: (i) the skeleton was symmetrized using the tbss_sym tool for left > right comparison; (ii) brain volumes were flipped, skeletonized, and symmetrized for right > left comparison; and (iii) brain volumes were organized to compare the painful side > pain-free side and vice versa (19).
Participant classification: Categories and subgroups
We applied the ICHD-3 criteria for all headache diagnoses, including migraine with and without aura, episodic and chronic migraine (17). Migraine with (MA) or without aura (MO) pertaining to the presence or absence of aura. Participants with both migraine with and without aura were classified as migraine with aura within this system. Episodic and chronic migraine were defined according to the frequency of migraine. Based on the headache characteristics prior to the scan, we classified participants with migraine as either: (i) ictal participants, who had a headache fulfilling ICHD-3 for a migraine attack without aura (17); the duration criterion of 4–72 hours was exempted to capture the early onset of a migraine attack; (ii) participants with a non-migraine headache, defined as any headache not fulfilling ICHD-3 criteria for a migraine attack; and (3) headache-free participants, who had no headache at all, rated as 0 on a Numerical Rating Scale (NRS) of 0 to 10.
Outcomes
DTI assesses the movement of water molecules in three spatial directions, providing insights into microstructural properties of the cerebral white matter. In this context, the following four measures are used: FA, MD, AD and RD represented in Figure 1 (3,4).
FA measures the degree of anisotropy of water diffusion in tissue. Lower FA suggest a loss of directional coherence in water diffusion, often associated with axonal loss or damage. MD quantifies the average rate of water diffusion within a voxel, regardless of direction. It provides information about the overall magnitude of diffusion. Higher MD values reflect increased overall water mobility, which can be associated with tissue damage, incl. demyelination. AD refers to the rate of water diffusion along the principal axis of the diffusion tensor, which typically aligns with the direction of the whiter matter fibers. Lower AD values indicate potential axonal damage or loss. RD measures the rate of water diffusion perpendicular to the principal axis of the diffusion tensor. Higher RD values suggest demyelination because it is sensitive to alterations in the myelin sheath surrounding the axons.
The TBSS method uses non-linear registration for voxel-wise analysis of DTI data within a group-wise white matter skeleton. This approach was used to compare FA, MD, AD and RD values between the following groups: (i) participants with migraine vs. HCs; (ii) chronic migraine vs. HCs; (iii) migraine with aura vs. HCs; (iv) migraine without aura vs. HCs; and (v) ictal participants vs. HCs. Furthermore, the following subtypes of migraine were also compared with each other: (vi) chronic migraine vs. episodic migraine; (vii) migraine with aura vs. migraine without aura; and (viii) ictal participants vs. interictal participants.
Exploratory analyses were performed to assess the correlations between DTI metrics and clinical variables in the migraine population. The clinical variables examined included the mean number of monthly migraine days (MMD) and monthly headache days (MHD) over the preceding three months, as well as the peak headache intensity during a usual migraine attack, as measured on a 11-point numeric rating scale. Moreover, hemispheric differences between the pain side and pain-free side were compared in participants who reported experiencing unilateral migraine attacks during the MRI scan.
Statistical analysis
Continuous data are presented as the mean ± SD if normally distributed or as medians with interquartile ranges if not. Normality was assessed using the Shapiro–Wilk test for sample sizes <30, whereas normality was presumed for sample sizes ≥30, in line with the central limit theorem. For normally distributed data, group differences were evaluated using Student's two-sample, unpaired t-tests. Conversely, non-normally distributed data were analyzed using Mann–Whitney U-tests. Age distributions between groups were compared using the Kolmogorov–Smirnov test. Categorical data were presented as absolute numbers and percentages, and group differences were analyzed using chi-squared tests. All statistical analyses were performed using R statistical software, version 4.2.0 (R Foundation, Vienna, Austria). p ≤ 0.05 was considered statistically significant.
The analysis of spatial DTI data was conducted using non-parametric permutation-based methods, including 5000 permutations with FMRIB Software Library (FSL), version 6.0 (Analysis Group, FMRIB). Group comparisons were performed with a threshold-free cluster enhancement, providing better sensitivity than voxel-wise methods (20). Group comparisons were corrected for multiple comparisons using the family-wise error rate with a significance threshold of p ≤ 0.05. The statistical model was delineated with a GLM design. All comparisons were adjusted for age and sex. Further adjustments were applied when comparing migraine with aura to migraine without aura, with adjustments made for chronic migraine, and when comparing episodic migraine to chronic migraine, with adjustments made for migraine with aura.
Results
In total, 306 participants with migraine and 160 age- and sex-matched HCs were enrolled. Of these, DTI data were excluded from 13 participants with migraine and six HCs due to incomplete scan data, imaging artifacts, incidental findings and pre-MRI caffeine intake. The final analysis comprised 293 participants with migraine and 154 HCs, matched for age (mean ± SD = 41.6 ± 12.3 years vs. 41.5 ± 11.6 years, p = 0.99) and sex (female (%) = 259/293 (88%) vs. 133/154 (86%), p = 0.41). Among the participants with migraine, 181 (62%) had chronic migraine, 159 (54%) used preventive medication(s) and 103 (35%) experienced migraine with aura. Furthermore, 79 (27%) participants were scanned during the ictal phase of a migraine attack. Detailed demographic and clinical characteristics of the study population are presented in Table 1.
Demographics and baseline characteristics of the study population.

MHD = monthly headache days; MMD = monthly migraine days.
*Fulfilling International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria for migraine without aura (ICHD-3 code 1.1) during scan.
**Fulfilling ICHD-3 criteria for migraine without aura (ICHD-3 code 1.1) and has headache of at least moderate to severe intensity (Numerical Rating Scale = 4–10) during scan.
TBSS analysis
The participants with migraine did not exhibit any significant differences in any DTI measures (i.e. FA, MD, RD and AD) compared to HCs (all p > 0.05). Likewise, subgroup analyses revealed no significant differences in DTI measures across all comparisons (all p > 0.05) (Table 2). These comparisons included: migraine with aura vs. HCs, migraine with aura vs. migraine without aura, migraine without aura vs. HC, chronic migraine vs. HCs, chronic migraine vs. episodic migraine, episodic migraine vs. HC, ictal participants vs. HCs and ictal participants vs. interictal participants.
Tract based spatial statistics in migraine and subgroups of migraine with aura, migraine without aura, episodic migraine, chronic migraine and headache phase of migraine. The p-values corresponded to the lower p-value obtained from the voxel-wise analysis.
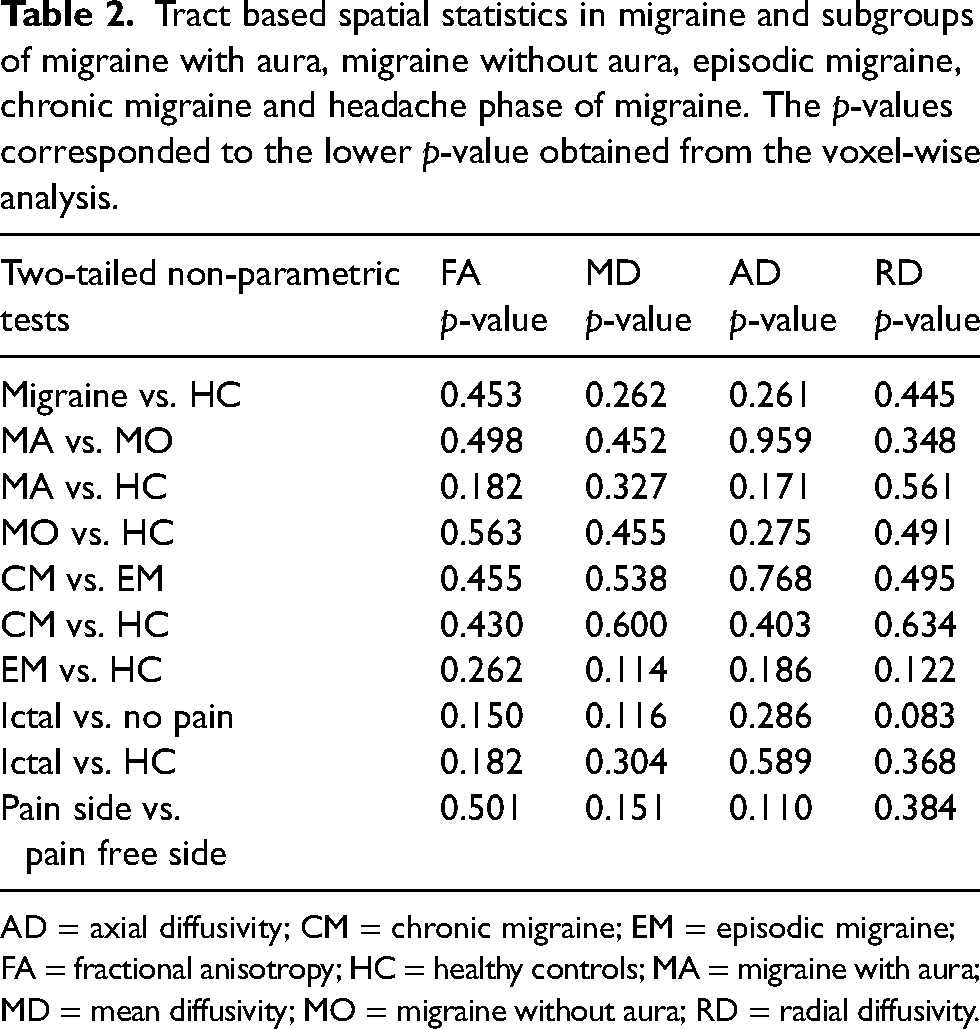
AD = axial diffusivity; CM = chronic migraine; EM = episodic migraine; FA = fractional anisotropy; HC = healthy controls; MA = migraine with aura; MD = mean diffusivity; MO = migraine without aura; RD = radial diffusivity.
The exploratory analyses revealed no significant correlation between DTI measures and clinical measures (all p > 0.05) (Table 3). The clinical measures included MMD, MHD and the pain intensity score during the scan, as measured on an 11-point numeric rating scale. Furthermore, no differences in DTI measures were identified between the pain side and pain-free side among participants who reported experiencing unilateral migraine attacks during the scan session (all p > 0.05).
Correlations of diffusion tensor imaging values with clinical variables in the overall migraine population. The p-values corresponded to the lower p-value obtained from the voxel-wise analysis.
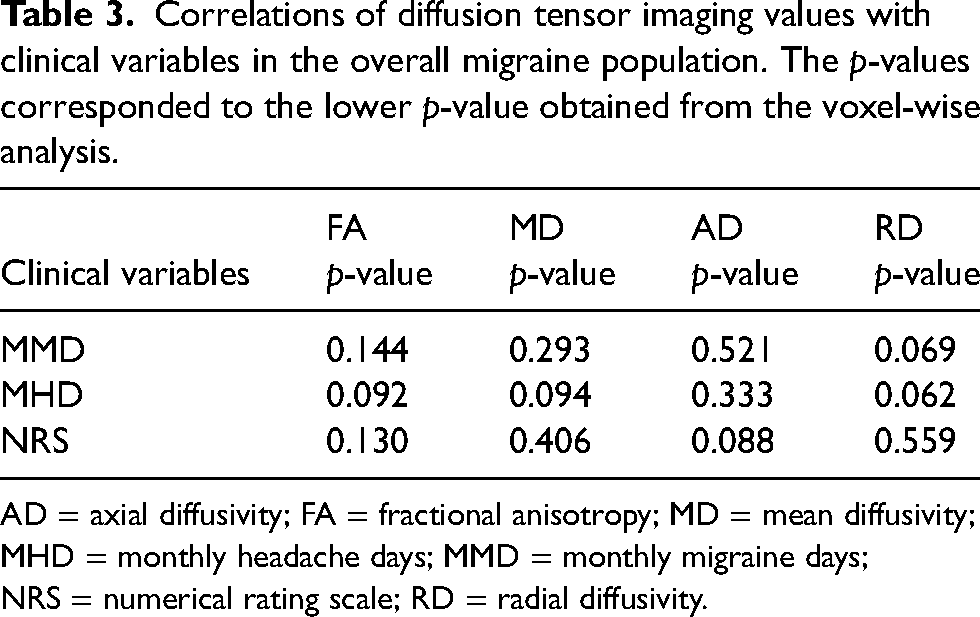
AD = axial diffusivity; FA = fractional anisotropy; MD = mean diffusivity; MHD = monthly headache days; MMD = monthly migraine days; NRS = numerical rating scale; RD = radial diffusivity.
Discussion
The results of this large-scale DTI study provide a compelling perspective on the microstructural integrity of cerebral white matter in people with migraine. Our analyses of all DTI measures (i.e. FA, MD, RD and AD) revealed no significant differences between participants with migraine and HCs. This was consistent across various subgroups within the migraine population, including those with aura, those with chronic migraine, and those scanned during the ictal phase of a migraine attack. This absence of significant differences in DTI measures is an encouraging finding, suggesting that the microstructural integrity of white matter is preserved in people with migraine.
Our results contrast with some previous, smaller DTI studies including generally fewer than 50 participants, which have reported white matter abnormalities associated with migraine, but the smaller sample size could have increased the probability of false positives (5,6,21). Noteworthy, differences in cohort demographics, statistical thresholding and tract-based analyses could also contribute to the differences observed with previous studies. However, it merits emphasis that the current findings are in line with other DTI studies that also failed to identify significant differences between participants with migraine and HCs, such as two studies including 110 and 103 patients with migraine that did not show any difference compared to healthy controls overall but did find significant results in subgroups analyses (22,23). Our study challenged the results previously observed in subgroups analyses of patients with episodic migraine, chronic migraine, migraine with aura and migraine without aura by significantly increasing the sample size (7,8,18). Our exploratory analyses also further reinforced the finding of intact white matter integrity in migraine, revealing no significant correlation between DTI measures and various clinical measures, such as MMD, MHD and pain intensity during the scan. Likewise, our results demonstrated that lateralization of pain during a migraine attack is not associated with lateralized alterations in white matter microstructure.
The clinical implications of our findings are threefold. First, the lack of microstructural changes in cerebral white matter indicates that migraine affects the brain in a different manner than neurodegenerative disorders, such as Alzheimer's disease and multiple sclerosis (24–26). Second, our results challenge the assertion that migraine is associated with long-term alterations in cerebral white matter (27). This is reassuring for patients and healthcare providers, implying that recurrent migraine attacks do not cause cumulative damage to cerebral white matter. Thus, concerns about migraine attacks being linked to progressive brain damage are unwarranted. Lastly, our findings demonstrate that DTI is not a viable imaging biomarker for migraine. Future research should explore alternative approaches to better understand the neurobiological underpinnings of migraine.
Strengths and limitations
This study benefits from its large and well-characterized migraine population, which also allowed us to compare DTI measures across various migraine subgroups. This enhances the generalizability of our findings. Furthermore, all scans were conducted by the same investigator (RHC) using the same 3T MRI scanner, ensuring consistency in data acquisition. However, the study is not without limitations. Notably, while we adjusted for age, sex, and migraine subtype, not all confounding factors could be taken into account, such as medication overuse. We also did not account for all phases of migraine (i.e. prodromal, headache, postdromal and interictal phases) (28,29), resulting in overall sample heterogeneity. Moreover, the cross-sectional design of our study precludes the assessment of longitudinal changes in white matter microstructure over time. Lastly, our study population was primarily recruited from clinical settings, which might limit the generalizability of our findings to adults with migraine in the broader population. Future research should consider these limitations and aim to include a more diverse sample of people with migraine, account for all migraine phases and use a longitudinal design to track changes in white matter microstructure over time.
Conclusions
Our DTI investigation found no significant differences in the microstructural integrity of cerebral white matter between people with migraine and age- and sex-matched HCs. This finding was consistent across all tested migraine subgroups and was not correlated with clinical measures of migraine severity. These results suggest that the microstructure of cerebral white matter is intact in people with migraine, suggesting that structural alterations in white matter do not play a central role in migraine pathophysiology.
Clinical implications
White matter integrity was assessed using DTI in 293 patients with migraine compared to 154 controls. No significant differences in white matter integrity were identified between participants with migraine and controls, nor among the different migraine subtypes. These results suggest that migraine does not result in microstructural alterations within the cerebral white matter.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251339423 - Supplemental material for Intact white matter in adults with migraine: A REFORM MRI study
Supplemental material, sj-docx-1-cep-10.1177_03331024251339423 for Intact white matter in adults with migraine: A REFORM MRI study by Cédric Gollion, Håkan Ashina, Rune Häckert Christensen, Haidar M Al-Khazali, Daniel Tolnai, Messoud Ashina and Faisal Mohammad Amin in Cephalalgia
Footnotes
Declaration of conflicting interests
C. Gollion reports speaker fees from Teva, Novartis, Lundbeck, Abbvie, Orkyn and Lilly. H. Ashina reports personal fees from AbbVie, Lundbeck, Pfizer and Teva, outside of the submitted work. M. Ashina is a consultant, speaker, or scientific advisor for AbbVie, Allergan, Amgen, Eli Lilly, Lundbeck, Novartis and Teva, and a primary investigator for ongoing AbbVie/Allergan, Amgen, Astra Zeneca, Eli Lilly, Lundbeck, Novartis and Teva trials. M. Ashina has no ownership interest and does not own stocks of any pharmaceutical company. M. Ashina serves as an associate editor of the Journal of Headache and Pain and as an associate editor of Brain. F. M. Amin has received personal fees from Pfizer, Teva, Lundbeck, Novartis and Eli Lilly, outside of the submitted work. R. H. Christensen, H. M. Al-Khazali and D. Tolnai report no relevant conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was funded by the Lundbeck Foundation Professor Grant (R310-2018-3711).
Data availability statement
Anonymized data and materials not published within this article will be made available on reasonable request from any qualified investigator.
Ethical statement
All participants provided written informed consent prior to the commencement of any study-related assessments or procedures. The study complied with the principles of the Declaration of Helsinki, with later revisions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
