Abstract
Aim
To evaluate the overlap between commonly reported trigger factors and corresponding premonitory symptoms in individuals with migraine.
Methods
This cross-sectional study analyzed data based on participant recall obtained through semi-structured interviews. Individuals diagnosed with migraine were enrolled from September 2020 to June 2022. Participants underwent semi-structured interviews to record their usual trigger factors, premonitory symptoms, non-headache symptoms during the headache and postdromal symptoms. The primary outcome was to assess whether reporting specific trigger factors increased the likelihood of experiencing corresponding premonitory symptoms. Furthermore, the presence of non-headache symptoms across migraine phases was examined.
Results
Among the 632 participants (mean age 44.6 ± 12.0 years; 89% female), the most frequent triggers were sleep disturbances (70.1%), stress (67.7%) and alcohol consumption (59.0%). Common premonitory symptoms included tiredness (39.9%), concentration difficulties (35.0%) and neck pain (33.2%). Significant associations were found between specific triggers and premonitory symptoms: bright light with premonitory photophobia (odd ratio (OR) = 2.79; 95% confidence interval (CI) = 1.90–4.12; p < 0.001), loud noise with premonitory phonophobia (OR = 4.26; 95% CI = 2.77–6.59; p < 0.001) and sleep disturbances with premonitory tiredness (OR = 1.74; 95% CI = 1.15–2.64; p = 0.009).
Conclusions
Our results reveal a notable overlap between specific migraine triggers and corresponding premonitory symptoms, implicating that some perceived triggers could be early signs of an impending attack. Moreover, the continuation of most premonitory symptoms into the postdromal phase suggest that migraine-related symptoms extend across a broader temporal continuum than previously recognized.
Trial Registration
NCT04603976
This is a visual representation of the abstract.
Introduction
Migraine is a prevalent and disabling neurological disorder, characterized by recurrent attacks of moderate to severe headache and accompanying photophobia, phonophobia, nausea and vomiting (1). The progression of a migraine attack is often divided into distinct phases: premonitory phase, aura phase, headache phase and postdromal phase (2). The premonitory phase occurs up to 48 hours before headache onset and encompasses a wide range of symptoms, including tiredness, neck stiffness, mood changes and concentration difficulties (1,3). The postdromal phase follows the resolution of headache and can involve symptoms similar to those reported during the premonitory phase (1,4).
Individuals with migraine often identify trigger factors that they perceive as precipitating migraine attacks (5). Commonly reported triggers include stress, sleep disturbances, hormonal fluctuations, sensory stimuli (e.g. bright lights, loud sounds), certain foods and alcohol consumption (6). However, emerging evidence suggests that some triggers overlap with premonitory symptoms, potentially representing early manifestations of a migraine attack rather than true precipitants (7–9). This overlap can complicate patient education and self-management strategies because individuals with migraine might misattribute premonitory symptoms as external triggers, leading to unnecessary lifestyle restrictions and ineffective disease management.
Understanding the relationship between perceived trigger factors and premonitory symptoms is essential for improving migraine management. If certain triggers are actually premonitory symptoms, this recognition can facilitate timely interventions and reduce unwarranted lifestyle modifications. Moreover, non-headache symptoms such as photophobia, phonophobia, nausea and neck pain might persist across different migraine phases, but the extent of their persistence is not well characterized.
In the present study, we evaluated whether reporting specific trigger factors increases the likelihood of experiencing corresponding premonitory symptoms among individuals diagnosed with migraine. In addition, we examined the persistence of non-headache symptoms across the premonitory, headache and postdromal phases. We hypothesized that participants who identify specific trigger factors (e.g. bright light, loud noise, sleep disturbances) would be more likely to experience the corresponding premonitory symptoms (e.g. photophobia, phonophobia, tiredness). Furthermore, we hypothesized that these non-headache symptoms would frequently persist across migraine phases, reflecting a continuous symptom trajectory rather than distinct, isolated phases. By investigating whether early symptoms (often perceived as triggers) persist throughout the migraine attack, we aimed to contextualize them not as isolated events, but as part of a continuous pathophysiologic process. This approach should help reframe traditional distinctions between phases, reinforce the unified nature of migraine symptomatology and improve clinical recognition of early attack features.
Methods
The data presented herein derive from the prospective, single-center
Design and participants
We applied a cross-sectional design and analyzed clinical data based on participant recall obtained using semi-structured interviews. The participants with migraine were primarily recruited from the outpatient clinic of the Danish Headache Center, Copenhagen University Hospital – Rigshospitalet. Participants were enrolled from September 2020 to June 2022.
Eligible participants were individuals (age ≥18 years) diagnosed with migraine without aura, migraine with aura or chronic migraine, in accordance with the International Classification of Headache Disorders, 3rd edition (ICHD-3) (1). Furthermore, participants were required to report at least four monthly migraine days during the three months prior to enrollment. Exclusion criteria included the onset of migraine after 50 years of age, in order to reduce the likelihood of including secondary headache disorders that can mimic migraine in older adults, such as those related to cerebrovascular disease or systemic illness. An inability to distinguish migraine from other headache types also lead to exclusion. Additional exclusions applied to persons with continuous and unremitting daily headache. A complete list of inclusion and exclusion criteria is provided in the Supplementary material (Table S1).
Procedures
All participants with migraine underwent a semi-structured interview to obtain data on clinical features and medical history. This interview included inquiries about typical factors that consistently trigger migraine attacks. We first introduced an eight-item list of potential triggers, which included physical activity, bright light, loud sounds, stress, menstruation, alcohol, strong smells and sleep disturbances. Participants were then asked if they experienced any additional triggers not captured by this predefined list. The site investigators explained the phases of a migraine attack without citing specific symptoms, using illustrations when needed to ensure comprehension. Next, participants were asked about premonitory and postdromal symptoms. They first answered open-ended questions, followed by a systematic evaluation of 16 specific symptoms. Detailed findings related to premonitory and postdromal symptoms can be found in previous publications (12,13).
Outcomes and measures
Premonitory symptoms were defined according to ICHD-2 and ICHD-3β (i.e. symptoms taking place 2–48 hours before the onset of headache in a migraine without aura or before the aura in migraine with aura) (14,15). Postdromal symptoms were defined in adherence with ICHD-3 (i.e. symptomslasting up to 48 hours, following the resolution of pain in a migraine attack) (1). The primary outcome was whether reporting specific trigger factors, identified through prompted inquiry, increased the likelihood of experiencing corresponding premonitory symptoms. Because this analysis was not pre-specified in the REFORM protocol, only a subset of trigger factors was included. Specifically, three trigger-symptom pairs were assessed: bright light and premonitory photophobia, loud noise and premonitory phonophobia, and sleep disturbances and premonitory tiredness. The secondary outcome was whether reporting specific premonitory symptoms, identified through prompted inquiry, increased the likelihood of experiencing corresponding postdromal symptoms, also assessed through prompted inquiry.
Statistical analysis
Descriptive statistics were used to summarize demographics, clinical features and frequencies of triggers, premonitory symptoms and postdromal symptoms. Continuous data are presented as the mean ± SD or median with interquartile range (IQR), as appropriate. The Kolmogorov–Smirnov test was used to assess whether continuous variables were normally distributed. Categorical data were reported as frequency counts (n) with percentages (%). For dichotomous or categorical data, Fisher's exact test or the chi-squared test were performed, as appropriate. Simple logistic regression was performed to calculate odds ratios (ORs) with corresponding 95% confidence intervals (CIs). p < 0.05 was considered statistically significant. To control for multiple comparisons, p-values were adjusted for false discovery rate (FDR) using the Benjamini–Hochberg procedure. All analyses were conducted using R statistical software, version 4.2.0 (R Foundation, Vienna, Austria).
Results
In total, 632 participants were included in the analysis after excluding one individual with missing data. The participants’ mean age was 44.6 ± 12.0 years, and 563 (89%) were female. The mean ± SD number of monthly headache days was 17.9 ± 7.1 and 381 (60%) participants met the criteria for chronic migraine. The most common self-perceived trigger factors were sleep disturbances in 443 (70.1%) participants, stress in 428 (67.7%) participants and alcohol consumption in 373 (59.0%). Other frequently reported triggers included menstruation in 342 (60.1%; among female participants), bright light in 266 (42.1%) participants, strong odors in 260 (41.1%), physical activity in 258 (40.8%) and loud noise in 181 (28.6%). The most frequent premonitory symptoms were tiredness in 252 (39.9%) participants, concentration difficulties in 221 (35.0%) and neck pain in 210 (33.2%) participants. Detailed demographic and clinical characteristics are presented in Table 1, whereas Table 2 provides the relative frequencies of trigger factors.
Demographics and baseline characteristics of the study population.

BMI = body mass index, SD = standard deviation.
The relative frequencies of trigger factors among participants with migraine.

All values are reported as n (%), unless otherwise specified. aThese symptoms were reported by participants when given the option to add other trigger factors following a prompted enquiry where a structured checklist was used. bThe percentage is relative to the number of female participants.
Association between trigger factors and premonitory symptoms
Table 3 provides an overview of the associations between specific trigger factors and their corresponding premonitory symptoms. Among 192 participants reporting photophobia as a premonitory symptom, 114 (59.4%) identified bright lights as a migraine trigger. Our analysis revealed a significant association between bright light as a trigger factor and premonitory photophobia (OR = 2.79; 95% CI = 1.90–4.12; p < 0.001). Likewise, among 155 participants noting phonophobia as premonitory symptom, 76 (49.0%) reported loud noise as a trigger. There was a significant association between loud noise and premonitory phonophobia (OR = 4.26; 95% CI = 2.77–6.59; p < 0.001). In addition, among 252 participants experiencing tiredness as a premonitory symptom, 194 (77.0%) identified sleep disturbances as a trigger. A significant relationship was found between sleep disturbances and premonitory tiredness (OR = 1.74; 95% CI = 1.15–2.64; p = 0.009).
The overlap between perceived triggers and corresponding premonitory symptoms.

Relative frequencies of trigger factors and their corresponding premonitory symptoms, followed by the number of participants who reported the same symptom as both a trigger and a premonitory symptom. Data were collected from 632 participants. These symptoms were reported upon direct enquiry (i.e. symptoms endorsed in response to a structured checklist following open-ended questioning during the semi-structured interview).
Agreement with premonitory symptoms is defined as the number of participants who reported a symptom as both a trigger and a premonitory symptom, divided by the number who reported it as a premonitory symptom.
Non-Headache symptoms across a migraine attack
Table 4 presents a comprehensive summary of non-headache symptoms reported during the premonitory, headache and postdromal phases of a migraine attack. Data were provided by 632 participants. Notably, all participants who experienced photophobia (n = 192), phonophobia (n = 155) and nausea/vomiting (n = 131) as premonitory symptoms also reported these symptoms during the headache phase. However, fewer participants experienced these symptoms consistently throughout all migraine phases: photophobia (n = 75), phonophobia (n = 61) and nausea/vomiting (n = 39).
Relative frequencies of non-headache symptoms reported during the premonitory, headache, and postdromal phase.
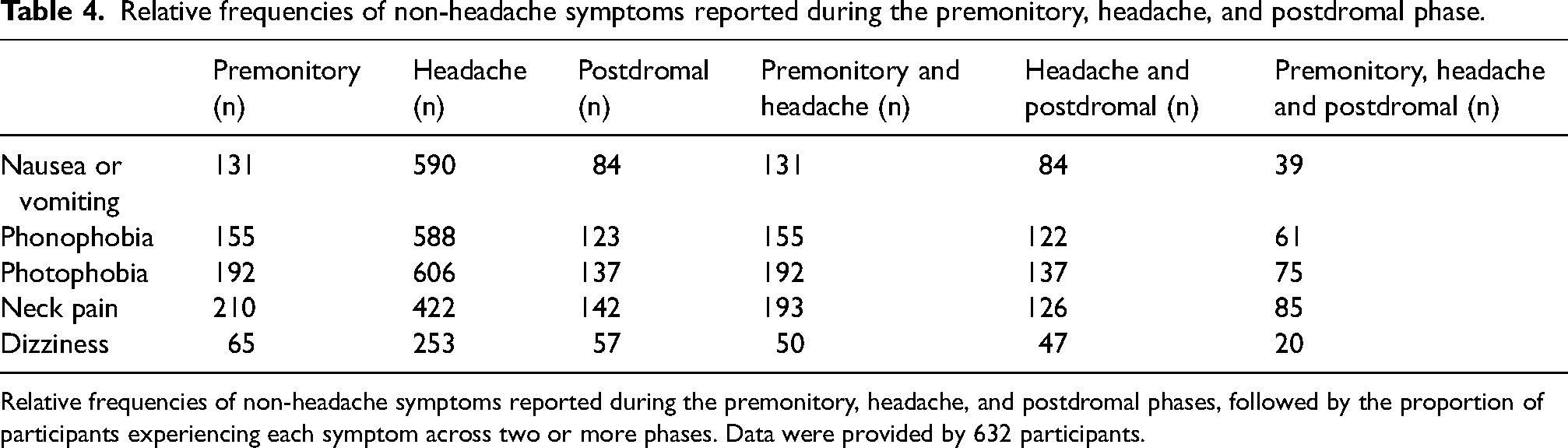
Relative frequencies of non-headache symptoms reported during the premonitory, headache, and postdromal phases, followed by the proportion of participants experiencing each symptom across two or more phases. Data were provided by 632 participants.
Premonitory and postdromal symptoms
Table 5 details the presence and overlap of premonitory and postdromal symptoms in participants with migraine. Positive associations were identified between all examined premonitory symptoms and their corresponding postdromal symptoms (p < 0.05), except for tiredness (OR = 1.51; 95% CI = 0.89–2.57; p = 0.12). All positive associations remained significant following correction for multiple comparisons.
The relative frequencies and overlap of premonitory and postdromal symptoms.
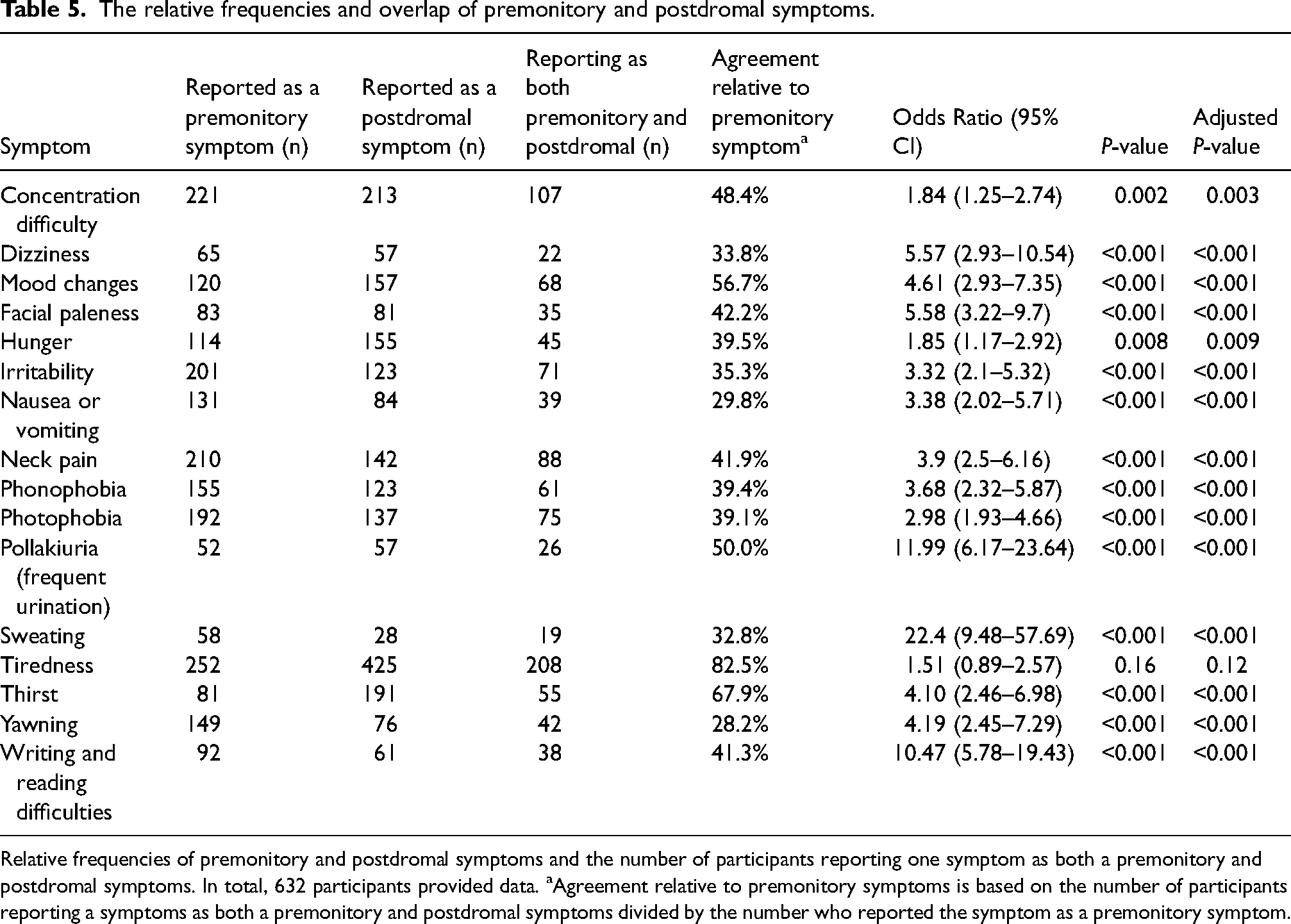
Relative frequencies of premonitory and postdromal symptoms and the number of participants reporting one symptom as both a premonitory and postdromal symptoms. In total, 632 participants provided data. aAgreement relative to premonitory symptoms is based on the number of participants reporting a symptoms as both a premonitory and postdromal symptoms divided by the number who reported the symptom as a premonitory symptom.
Discussion
Our findings provide insights into the relationship between commonly reported triggers factors of migraine and their corresponding premonitory symptoms, revealing an overlap that challenges conventional distinctions between external precipitants and early manifestations of attacks. Among 632 participants with migraine, we identified significant associations between specific triggers (i.e. bright light, loud noise and sleep disturbances) and their corresponding premonitory symptoms: photophobia, phonophobia and tiredness. These findings suggest that what individuals with migraine often perceive as triggers might, in some cases, represent the initial phases of a migraine attack rather than independent causal factors, or vice versa. This has important implications for patient education, self-management strategies and the broader understanding of migraine pathophysiology.
Overlap between triggers and premonitory symptoms
The observed associations, namely bright light with photophobia, loud noise with phonophobia and sleep disturbances with tiredness, underscore a notable convergence between self-reported triggers and premonitory symptoms. This overlap aligns with emerging evidence that some triggers might not initiate migraine attack but instead reflect altered sensory thresholds during the premonitory phase (7–9,16–19). For example, premonitory photophobia might be misinterpreted as light being a trigger when it actually signals an underlying sensory process already underway (16). Likewise, the relationship between loud noise and phonophobia suggests that sensitivity to sound could represent the early stages of a migraine attack rather than act as a true external trigger. The identified association between sleep disturbances and tiredness, although less pronounced, further supports the assertion that fatigue-related triggers might be conflated with prodromal tiredness.
These findings resonate with previous studies suggesting that the premonitory phase might begin hours to days before headache onset (3,20,21). Notably, some reports have identified positive associations between patient-reported triggers, such as light, sound, strong odors, certain foods and missed meals, and their corresponding premonitory symptoms (8,9,22). Our data build on this evidence by quantifying the likelihood of specific trigger-symptom pairings, providing a framework to further explore this overlap. Nevertheless, it is important to emphasize that the retrospective nature of our data precludes us from establishing a definite temporal relationship. We therefore cannot rule out the opposite scenario, where actual migraine triggers are mistaken for premonitory symptoms. In this context, prospective diary studies incorporating real-time symptom tracking might better determine whether perceived migraine triggers precede or coincide with their corresponding premonitory symptoms, but lack the ability definitively attribute causality, or lack thereof, to a putative migraine trigger.
Important causal insights might instead derive from controlled experimental studies, which indicate that self-reported triggers often fail to reproducibly induce migraine attacks (23–27). For example, in one controlled experiment (23), none of 11 participants with migraine developed an attack after intense photic stimulation. When this stimulation was combined with strenuous physical exercise, only two of 11 participants experienced a migraine attack. These findings suggest that isolated trigger exposure might be insufficient to reliably induce an attack. However, they also underscore the potential importance of parameters such as stimulus intensity, duration and timing, comprising factors that were not captured in our study but could significantly influence migraine induction (16). Alternatively, it could be the lack of a lower threshold during the premonitory phase that did not facilitate the onset of a migraine attack to the exposure of the pre-defined trigger. Ultimately, the seen overlap aligns with emerging evidence that some triggers might not initiate migraine attack but instead reflect altered sensory thresholds during the premonitory phase. A recent review further emphasized these diagnostic and conceptual ambiguities, advocating for prospective designs and clearer phenotyping to disentangle true precipitants from early manifestations of a migraine attack (16).
Persistence of symptoms across migraine phases
Our analysis of premonitory and postdromal symptoms reveals a striking continuity of non-headache symptoms across migraine phases. Positive associations were observed between all examined premonitory and postdromal symptom, except for tiredness. These findings align well with previous reports, suggesting that certain non-headache symptoms persist across multiple migraine phases (21,28,29). In an observational study, almost all symptoms reported as warning symptoms (i.e. premonitory) were present during the migraine headache (21). A retrospective study reported similar, but slightly stronger agreements, for most of the symptoms that were also investigated here (28). This study further highlighted that non-headache symptoms presented at different stages during an attack, suggesting that the migraine attack is not linear but rather sequential with overlapping phases. Our findings are in line with these findings and, taken together, it appears evident that certain non-headache symptoms, such as photophobia and phonophobia, persist beyond headache resolution, framing migraine as prolonged, multi-phase disorder.
Clinical and practical implications
Our findings provide insights into the relationship between commonly reported trigger factors of migraine and their corresponding premonitory symptoms, revealing an overlap that challenges conventional distinctions between external precipitants and early manifestations of attacks. Moreover, by demonstrating that many non-headache symptoms persist into the headache and postdromal phases, our results suggest that migraine-related symptoms unfold across a broader temporal continuum than previously appreciated. Together, these analyses highlight the need to reconsider phase-based definitions of migraine, recognizing that symptoms often attributed to the prodrome or misclassified as triggers might, in fact, represent the early and evolving features of a single pathophysiological process.
Clinically, our results suggest a potential shift in how patient education addresses trigger factors and symptom recognition, but caution is warranted given the limited body of evidence. Misinterpreting premonitory symptoms as triggers (e.g. avoiding bright light due to photophobia) might impose unnecessary restrictions without reliably preventing attacks. Conversely, failure to recognize a trigger might inadvertently increase the propensity towards migraine attacks. Therefore, it becomes critical to guide patients to distinguish early warnings from reliable triggers. Moreover, the persistence of many symptoms in the postdromal phase implies that management might need to extend beyond headache resolution.
However, implementing such changes in clinical practice requires a stronger evidence base, as current data, including our findings, remain preliminary and lack much-needed prospective validation. While this dual focus on anticipation and extended care could empower patients to mitigate the impact of migraine, the scarcity of longitudinal studies limits confidence in distinguishing true triggers (e.g. bright light) from prodromal cues (e.g. photophobia) or in predicting symptom trajectories across phases. Tools such as symptom diaries or mobile apps hold promise for tracking these patterns and informing individualized care, but their utility hinges on further research to confirm the trigger-symptom overlap and its consistency. Until prospective studies substantiate these relationships, clinicians should approach treatment adjustments, such as prolonging acute therapies or tailoring plans to postdromal persistence, with restraint, prioritizing established protocols over speculative shifts.
Limitations and future perspectives
The present study relies on participants’ ability to recall triggers and non-headache symptoms which introduces recall bias because participants might conflate triggers, premonitory symptoms and postdromal effects. Likewise, the use of a predefined list of symptoms introduces acquiescence bias. The cross-sectional design limits causal inference, particularly regarding whether triggers precede symptoms or symptoms persist due to attack severity (18). In line with this, we did not assess the intensity or duration of exposure to a potential trigger, which may have a crucial impact. The predominantly female (89%) and middle-aged (mean 44.6 years) sample, primarily from a tertiary clinic, may not generalize to all individuals with migraine. In particular, excluding participants with migraine onset after age 50 years could limit our ability to understand how aging, comorbidity or medication burden influences symptom recognition and attribution. Even though, the exclusion of participants with migraine onset after age 50 years was intended to reduce diagnostic ambiguity associated with late-onset secondary headache disorders, this criterion limits the generalizability of our findings to older populations. The clinical course of migraine may change with age, with some individuals experiencing reduced attack frequency but more frequent comorbidities that could alter symptom expression and perception. In older adults, the ability to distinguish true triggers from early symptoms could also be attenuated by cognitive changes or diminished sensory reactivity. These age-related factors could warrant further investigation in cohorts that include a broader age spectrum. At last, the frequent occurrence of migraine and headache days introduces some risk of misclassifying premonitory and postdromal symptoms.
Prospective, longitudinal studies with real-time data collection, via wearables or apps, could clarify the sequence of triggers, symptoms, and phases, whereas controlled exposure experiments (e.g. to light or noise) could disentangle causation from correlation. Biomarker studies might reveal whether premonitory and postdromal symptoms share a common substrate, enhancing pathophysiological models. For individual-level prediction, tracking 10–20 attacks per patient could uncover personalized patterns, leveraging machine learning to forecast attack onset and duration with precision.
Conclusions
Our findings demonstrate a considerable overlap between certain migraine triggers and their corresponding premonitory symptoms, suggesting that some perceived precipitants might be early attack indicators. Furthermore, we identified notable symptom persistence from premonitory to postdromal phases. Collectively, these insights support patient education to distinguish premonitory symptoms from reliable triggers and highlight the potential need to extend care across all migraine phases. Although limitations underscore the need for prospective and experimental validation, our results offer a foundation for rethinking migraine as a continuous, multi-phase neurologic disorder, paving the way for more individualized strategies to alleviate its burden.
Article highlights
Bright light, loud noise and sleep disturbances were significantly associated with corresponding premonitory symptoms, suggesting that they may be early signs of migraine rather than true triggers.
Non-headache symptoms frequently persisted throughout all migraine phases, supporting a more continuous progression of symptoms.
These findings suggest that some commonly reported migraine triggers could instead reflect early symptoms of an attack, highlighting the need for further research into how migraine triggers are identified and understood.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251364234 - Supplemental material for Overlap between perceived triggers, premonitory symptoms and symptom persistence across migraine phases: A REFORM study
Supplemental material, sj-docx-1-cep-10.1177_03331024251364234 for Overlap between perceived triggers, premonitory symptoms and symptom persistence across migraine phases: A REFORM study by Janu Thuraiaiyah, Rune H. Christensen, Haidar M. Al-Khazali, Astrid Wiggers, Messoud Ashina and Håkan Ashina in Cephalalgia
Footnotes
Acknowledgements
We sincerely thank the investigators and staff of the Danish Headache Center for their invaluable contributions to this study. Sub-investigators: Andreas Vinther Thomsen, Betel Tesfay, Christopher Kjær Cullum, Lili Kokoti, Nadja Bredo Rasmussen, Navid Noory, Rogelio Domínguez-Moreno, Thien Phu Do, William Kristian Karlsson and Zixuan Alice Zhuang; clinical research nurses: Anne Mette Autzen, Susanne Leed and Marianne Hestad; Medical secretaries: Ane Lundgaard Dahl, Dianna Bartolin Christiansen and Pia Frydendall; research administrator: Kateryna Kolkova; and medical students: Amanda Poulsen, Amenah Ayyoub, Amir Al-Saoudi, Emil Gazolov, Johanne Gry Larsen, Kathrine Rose, Mikkel Johannes Henningsen, Mohammed Bakir Ahmad Lafta, Sarra Al-Khazali, Sarah Hugger and Shan Elahi Goandal. Their dedication and expertise were instrumental in the success of this study. We are also deeply grateful to the study participants for their invaluable contribution.
Author contributions
HA and MA conceived and initiated the study. JT, RHC, HMA and AW participated in data acquisition. JT and RHC performed analyses of the data. JT drafted the first version of the manuscript with contributions from RHC, HMA and AW. All authors revised the manuscript for intellectual content, approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Data availability
Upon reasonable request, the corresponding author will provide the necessary data and materials to interested researchers for the purpose of academic scrutiny, reproducibility and further scientific investigation.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: JT reports receiving personal fees from Lundbeck and Pfizer, outside of the submitted work. RHC reports receiving personal fees from Pfizer, outside of the submitted work. HMA reports receiving personal fees from Lundbeck, Pfizer and Teva, outside of the submitted work. AW reports no conflicts of interest. MA reports receiving personal fees from AbbVie, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals outside of the submitted work. MA received institutional grants from Lundbeck Foundation, Novo Nordisk Foundation, Lundbeck A/S and Novartis. MA reports serving as associate editor of The Journal of Headache and Pain and associate editor of Brain. HA has received personal fees from AbbVie, Lundbeck, Pfizer and Teva, outside of the submitted work. HA is also an Editorial Board Member of The Journal of Headache and Pain.
Ethical statement
The protocol was approved by the relevant ethics committee (NCT04603976), and all participants provided written informed consent prior to the commencement of any study-related assessments.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MA was supported by the Lundbeck Foundation professor grant (R310-2018-3711).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
