Abstract
Trigger factors, signs and symptoms of the preheadache phases of episodic tension-type headache (ETTH), typical aura with non-migraine headache (TANMH), migraine with (MA) and without aura (MwA) may show similar features. Our objective was to investigate the preheadache phases and trigger factors of these headache types. Questionnaires including trigger factors, signs and symptoms of preheadache phases were answered by all headache patients. A total of 96 patients, 31 ETTH, nine TANMH, 23 MA and 33 MwA patients were included in this study. Analysis of seven groups consisting of 18 individual trigger factors showed that only two groups and five individual trigger factors were significantly different between groups. Hunger and odour were significantly more common in MA, MwA and TANMH patients. Foods were a significant precipitant factor for headache in MA patients. Head and neck movements were important trigger factors in ETTH. In prodrome phase only one out of three groups differed significantly between headache types. Migraine and TANMH patients reported significantly more general signs and symptoms. Analysis of aura signs and symptoms showed that only two out of six groups were significantly more frequent in MA and TANMH patients. Visual aura symptoms were more frequent in MA and TANMH groups, where sensorial auras were reported to be the most frequent in the MA group. Our results showed that different type of headaches share common prodrome and aura signs and symptoms as well as the same trigger factors. We suggest that similar trigger factors may trigger similar mechanisms and may cause common preheadache signs and symptoms in all headache types.
Introduction
Headache pathophysiology is one of the major study areas of headache investigators. Increasing experience and knowledge of the clinical features of primary headaches and mechanisms of pain in different types of headaches help clinicians to diagnose and treat headaches better. Extensive research is carried on both the clinical pathophysiology and the basic science of headache. Tension-type headache (TTH) and migraine are the most common primary headaches. One-year prevalance estimates for migraine and episodic-tension type headache (ETTH) are 16.4% and 25.5%, respectively, for Turkey (1). The high prevalence rates and socioeconomic impact of primary headaches stimulate investigators to a better understanding of headache pathophysiology. Currently, there are two different concepts concerning the clinical pathophysiology of TTH and migraine. One of these concepts accepts the two headache types as separate entities and the other as the opposite ends of a continuum (2–7).
Those who accept TTH and migraine as separate entities suggest that epidemiological differences such as age and sex distribution, age at onset and risk factors are all in favour of different pathophysiological mechanisms and against the continuum hypothesis (2, 8–10).
According to the continuum hypothesis, ETTH is at the mild end and migraine is at the severe end of a continuum of varying severity. Supporting evidence for this hypothesis has come from a number of different studies investigating epidemiological data, clinical features, biology and pain mechanisms of headache (11–15).
Pre-headache phases include aura and premonitory signs and symptoms. Prodrome phase is used synonymously for premonitory signs. As the prodrome phase is also widely accepted and easily understandable, we preferred to use this term rather than premonitory signs. There are still inadequate data about the preheadache phases of different types of headaches and preheadache phases may shed light on the pathophysiology of headache. Prodrome phase signs and symptoms may play an important role in the diagnosis of different type of headache. This may also help early differential diagnosis between different headache types and initiation of convenient therapy as soon as possible. Early initiation of convenient therapy can prevent chronic sensitization of the central nervous system.
The International Classification of Headache Disorders, First Edition (ICHD-1) did not require the headache of migraine with aura (MA) to fulfil the headache characteristics of migraine without aura (MwA) (16). The headache characteristics of MA have been classified in the International Classification of Headache Disorders, Second Edition (ICHD-2) as migraine headache and non-migraine headache (17)
In this study, we aimed to investigate the preheadache phases and trigger factors of ETTH, MA, MwA and typical aura with non-migraine headache (TANMH). The aim was to achieve a better understanding of the clinical pathophysiology of different headache types.
Methods
All patients were recruited randomly from the out-patients headache clinic. Patients who received a diagnosis of ETTH, MA, MwA and TANMH according to ICHD-2 were included in this study. Patients who suffered from more than one headache type were excluded in order to prevent misinterpretation of the symptoms. All patients were informed about the aims and procedure of the study.
Questionnaires, including prodrome and aura signs and symptoms, as well as trigger factors, were answered by all patients (Tables 1–3). Patients were questioned again with the same questionnaire by a headache expert. In the case of an inconsistency between patients’ and physician's answers, the final decision was made by agreement between patient and physician.
Trigger factors and results

ETTH, Episodic tension-type headache; TANMH, typical aura with non-migraine headache; MA, migraine with aura; MwA, migraine without aura;
Prodrome phase signs and symptoms, and results

ETTH, Episodic tension-type headache; MA, migraine with aura; MwA, migraine without aura; TANMH, typical aura with non-migraine headache;
Aura signs and symptoms, and results

ETTH, Episodic tension-type headache; MA, migraine with aura; MwA, migraine without aura; TANMH, typical aura with non-migraine headache;
Some migraine patients reported associating visual or sensory symptoms within an hour before or during the headache. However, the duration of these symptoms was less than 5 min, which prevented us from defining them as an aura. Most patients reported that these symptoms lasted for only 1 or 2 min. Therefore, we preferred to call them aura-like symptoms and included them in the MwA group. We also compared them with aura symptoms of MA and TANMH patients.
One-way
Results
A total of 96 patients, 31 ETTH, 23 MA, 33 MwA and nine TANMH, were included in this study. All patients were headache free during the study. Mean age was 35.1 ± 10.1 years in the ETTH group, 40 ± 11.1 and 36.7 ± 10 years in the MA and MwA groups, respectively, and 34.7 ± 9.8 years in the TANMH group. Twenty-four, 21, 29 and nine patients were female in the ETTH, MA, MwA and TANMH groups, respectively. There were no significant differences in mean age and sex between groups. Comparisons between groups and results are given in Figs 1–3 and Tables 1–3.
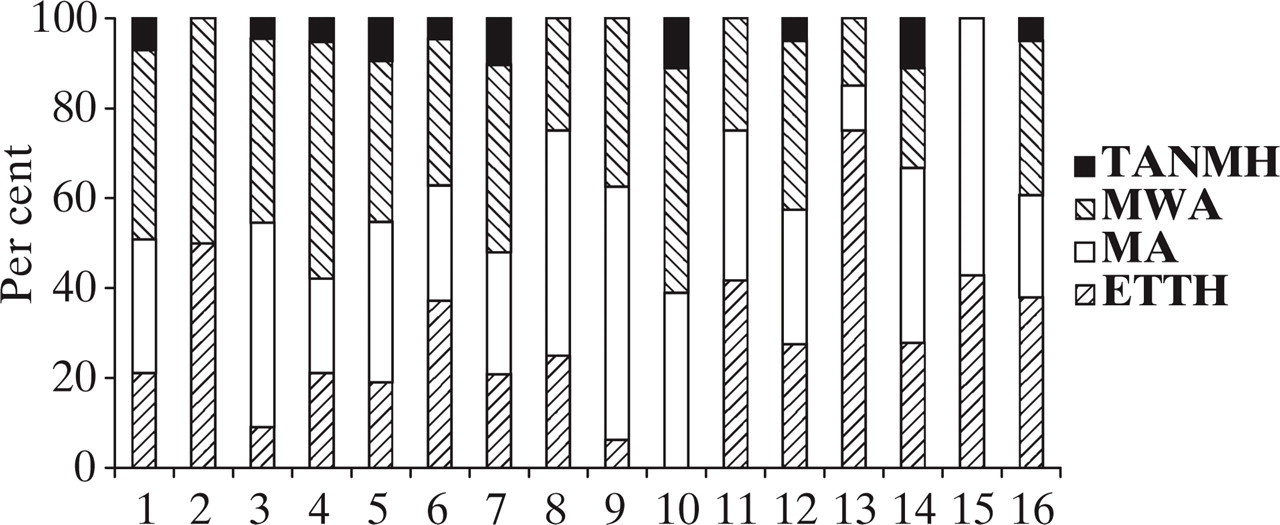
Trigger factors. 1, Hunger; 2, alcohol; 3, foods; 4, oversleep; 5, sleeplessness; 6, sleep pattern changes; 7, menstruation; 8, menapause; 9, bright light; 10, odours; 11, altitude; 12, weather change; 13, head and neck movements; 14, exercise; 15, sexual intercourse; 16, stress–anxiety.

Prodrome signs and symptoms. 1, Depression; 2, hyperactivity; 3, euphoria; 4, restlessness; 5, vertigo; 6, photophobia; 7, phonophobia; 8, dizziness; 9, concentration difficulty; 10, hyperosmia; 11, yawning; 12, neck stiffness; 13, food craving; 14, anorexia; 15, feeling cold; 16, slow movements; 17, urinating.
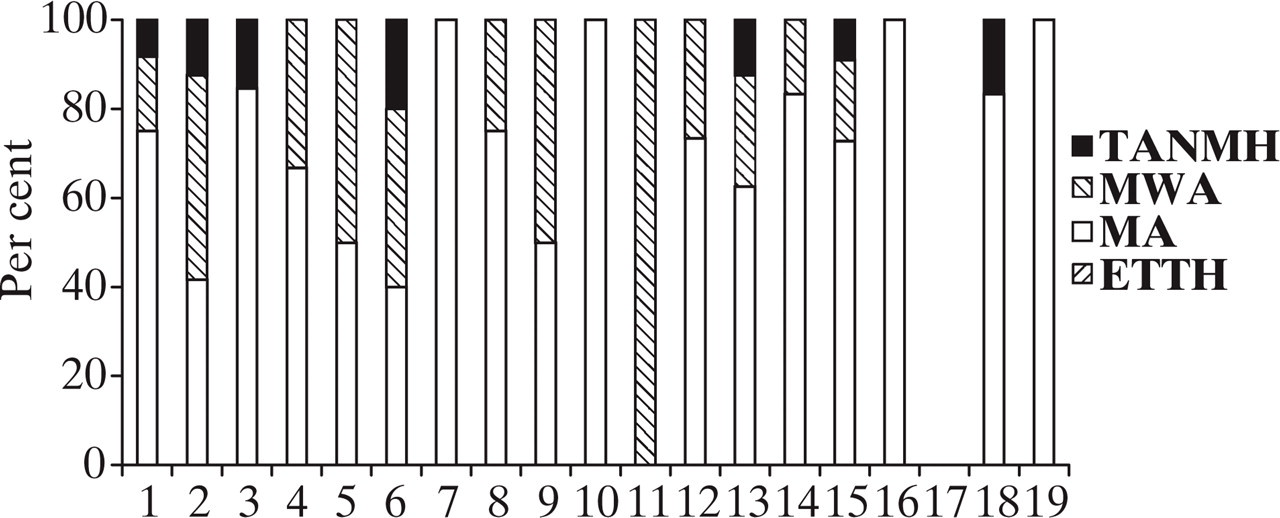
Aura signs and symptoms. 1, Scotoma; 2, photopsia; 3, fortification spectra; 4, bright ribbons; 5, geometric figures; 6, blurred vision; 7, zig-zag lines; 8, micropsia/macropsia; 9, metamorphopsia; 10, zooming objects; 11, mosaic vision; 12, paraesthesia; 13, olfactory hallucinations; 14, ataxia; 15, dysarthria; 16, jamais vu; 17, deja vu; 18, detailed dreams; 19, trance-like status.
Analysis of trigger factors revealed only two (diet and head and neck movements) out of seven groups of trigger factors that differed significantly between headache types. As a group, diet significantly precipitated headache in MA patients. Hunger and odour were significantly more common trigger factors in the MA, MwA and TANMH groups compared with the ETTH group. Neither trigger factor differed significantly between MA, MwA and TANMH. Foods played a significantly more common role in precipitating MA. As a trigger factor, sleeplessness showed the same features as foods. Head and neck movements were important trigger factors in precipitating ETTH and differed significantly from other groups. Only five out of 18 trigger factors differed significantly between headache groups. The remaining 11 were similar in all groups. Menstruation, which is known as a specific trigger factor for migraine, also appeared to be an important trigger factor for ETTH and TANMH. Nearly one-third of ETTH and more than half of TANMH patients reported menstruation as a trigger factor. Frequency of menstruation in migraine was nearly two-fold when compared with ETTH. However, this was not significant. Pregnancy and oral conraceptive intake could not be evaluated due to inadequate patient numbers.
Evaluation of prodrome phase signs and symptoms revealed that one (general) out of three groups showed a statistically significant difference. Prodrome phase general signs and symptoms were more frequent in MA, MwA and TANMH than in ETTH. None of these three headache groups differed from the others. When all prodrome phase signs and symptoms were compared between groups, only three (feeling cold, photo- and phonophobia) out of 24 showed significant differences between groups. All general signs and symptoms were compared individually and feeling cold was the only symptom revealing a difference between groups. Feeling cold was more frequent in the MwA group. Photo- and phonophobia, from the neurogenic signs and symptoms group, were more common in MA, MwA and TANMH. Psychogenic signs and symptoms were similar in all headache groups. All patients experienced similar prodrome phase signs and symptoms. Only minor differences were reported by the patients.
Analysis of aura signs and symptoms showed that they were the most prominent feature of MA, as expected. Nine TANMH patients reported aura signs and symptoms within an hour before or during the headache. All of these patients’ headache characteristics were similar to ETTH. Two (visual and sensory) out of six aura groups differed significantly between headache groups. Visual auras were more frequent in the MA and TANMH groups. Presence of visual aura-like symptoms in MwA patients created a difference between the MwA and ETTH groups. Photopsia was significantly more common in MA and TANMH. When MwA patients’ aura-like photopsia symptoms were taken into account, this group also differed from the ETTH group and there were no differences between the MA, MwA and TANMH groups. Sensory auras significantly associated with MA and paraesthesia were more frequent in MWA patients.
Discussion
Pathophysiology is still one of the controversial issues in headache. The similar clinical features of ETTH, MA and MwA have arised the question of whether TTH and migraine share a similar pathophysiology (10, 18). The continuum severity hypothesis suggests that ETTH is at the mild and MA at the severe end of the evolving headache (3–6).
ETTH has some common clinical features with migraine. Numerous studies investigating biological markers, clinical features and trigger factors of headache have supported this idea (10–15). Recently, Cady et al. reported that TTH patients, especially if they had associating migraine symptoms, but not migraine according to ICHD-1, responded very well to sumatriptan treatment (11, 12). Sumatriptan is a 5-HT1B/1D agonist, specifically used in migraine therapy. Their findings supplied further evidence for the continuum hypothesis. All these data suggest that ETTH and migraine may share a similar clinical pathophysiology.
In our study, we investigated the preheadache phases and trigger factors in ETTH, MA, MwA and TANMH patients. Among seven groups of trigger factors, only two groups, diet and head and neck movements, showed significant differences between headache groups. Most trigger factors were similar in all headache types. Only five out of 18 trigger factors differed significantly between groups. Even most common trigger factors were the same. The most frequent trigger factors for migraine were stress–anxiety and hunger. For ETTH, head and neck movements followed stress–anxiety. Interestingly, none of TANMH patients reported head and neck movements as a trigger factor and this was similar to migraine groups. Although secular, Turkey is an islamic country. The alcohol consumption rate is therefore much lower than in the USA and Europe. Among women it is even lower. Most of the patients in this study were female and middle-class. Very few women drink alcohol among this socioeconomic class. Therefore, alcohol appeared not to be an important trigger factor in precipitating migraine headache in our study. Our results showed that menstruation was a common trigger factor for all headache types. Although more frequent in migraine groups, this did not reach statistical significance. In the future, the role of menstruation in precipitating headache in ETTH patients should be investigated carefully. These results obviously show that similar factors may precipitate different types of headaches, and are in accordance with other studies (19–23). Most of the trigger factors were similar for all headaches. Although some trigger factors were included as a diagnostic criterion in ICHD-2, we believe that one must be cautious about trigger factors in the diagnosis of headache.
In the prodrome phase, as a group, general signs and symptoms significantly differed between groups. Prodrome phase general signs and symptoms were significantly more common in MA, MwA and TANMH groups, but no difference was observed between these three groups. When compared individually, all general signs and symptoms, except feeling cold, were similar in all groups. Although prodrome phase neurogenic signs and symptoms, as a group, did not differ between headache groups, photophobia and phonophobia were significantly more frequent in the MA, MwA and TANMH groups. There was no statistically significant difference between these groups. Vingen and Stovner reported phonophobia and photophobia in episodic and chronic TTH patients, during both the headache and headache-free periods (13). Our results were similar to their findings and showed that ETTH patients may experience many prodrome phase signs and symptoms, even photophobia and phonophobia. The frequency of prodrome phase psychogenic signs and symptoms did not differ between groups. Therefore, we suggest that similar mechanisms may be involved in the prodrome phases of different types of headache. Furthermore, altough TANMH patients shared headache characteristics of TTH, they showed similar features with MA and MwA patients during the prodrome phase. To our knowledge, this is the first study comparing prodrome phase signs and symptoms between headache types. Future studies investigating the prodrome phase may reveal sufficient data in different types of headache to help include prodrome phase signs and symptoms into the diagnostic criteria of primary headaches. This would lead to early diagnosis of primary headaches and improvement of treatment strategies.
Any headache following aura was accepted as migraine according to the ICHD-1 (16). In ICHD-2 a new headache type, TANMH, was defined (17). Diagnostic criteria for TANMH included the characteristics of the aura, but not headache. Any headache type, other than migraine, following aura has been accepted as sufficient to be classified under this heading. Our TANMH patients had typical headache characteristics of TTH. Recently, Silberstein et al. reported cluster headache with aura (24). This shows that any type of headache may follow aura. Further evidence for this hypothesis came from Cady et al. (6). They reported that mild headache following aura had the clinical characteristics of TTH and may lack migrainous symptoms. Therefore, we classified these patients under the heading of TANMH according to ICHD-2. Two TANMH patients reported fortification spectra, which is characteristic of MA. Our data strongly support Cady et al.'s suggestion concerning non-migrainous headache following aura and the continuum hypothesis (6).
In the aura phase, visual aura signs and symptoms were significantly more common in MA and TANMH patients. All 23 patients in the MA group and all but one patient in the TANMH group reported visual signs and symptoms. MwA patients reported aura-like visual symptoms and that was significantly more common compared with the ETTH group. Photopsia was the most frequent visual aura symptom in TANMH and MwA patients and the second most frequent in MA patients. MA patients reported significantly more sensory aura symptoms. Paraesthesia was the only sensory aura symptom showing a slight difference in the MA group. Our results showed that MwA patients may experience aura-like symptoms and if not questionned carefully this may cause overdiagnosis of MA. In the aura phase, TANMH patients showed similar features to MA patients. Except for the visual and sensory aura groups, other aura groups showed similar features between the ETTH and MA, MwA and TANMH groups.
This study showed that all headaches might be precipitated by similar trigger factors and might have similar prodrome signs and symptoms. They may even have the same aura symptoms. These findings suggest that similar trigger factors may cause similar prodrome and aura signs and symptoms, and similar mechanisms may be involved in the headache process. Do similar pathophysiological mechanisms which might be involved in the prodrome phase, triggered by similar trigger factors, differentiate during the aura and headache period in different headache types? We think that patients with TANMH may help investigation of this subject. Similarly, MwA patients reported aura-like symptoms. These findings may suggest that similar pathophysiological mechanisms are also involved during aura and evolving headache phases. MwA patients reported aura-like symptoms which are shorter in duration than the aura. Following aura, TANMH patients reported headache characteristics of TTH. ETTH patients mostly share the same trigger factors and prodrome phase signs and symptoms with MA and MwA patients. All of these data may supply further evidence for the continuum hypothesis and clinical pathophysiology. Patients who suffer from TANMH and show headache characteristics of TTH may form a bridge between ETTH and MA. MwA patients who reported aura-like symptoms may be placed between MwA and MA in the continuum hypothesis.
In different studies Cady et al. reported that ETTH patients showing migrainous characteristics had responded well to sumatriptan and MA patients with mild headache had shown headache characteristics of TTH; they suggested that TTH and migraine might share a similar clinical pathophysiology (6, 11, 12). Our findings about the pre-headache phases and trigger factors support their suggestions.
This study has some limitations. Study groups included different numbers of patients. This may have affected the results. Also, in some parameters patient numbers were small and this may have influenced statistical analysis. All patients were headache free during the study. This may have caused poor recall of the accompanying symptoms. Headache diaries for recording the symptoms and signs might have been better for the purposes of this study. Nevertheless, we believe that our study revealed valuable data and filled a gap in the knowledge of the preheadache phases of headache.
Our results showed that different types of headache may share common prodrome and aura signs and symptoms, as well as the same trigger factors. These findings suggest that similar trigger factors trigger similar mechanisms and may cause common preheadache signs and symptoms in all headache types. We suggest that our findings support the continuum hypothesis and that all headaches may come from the same origin. The question is, what creates the difference between the headache characteristics of MA and TANMH (with TTH characteristics in our case) following the aura period? Is it just the difference of an evolving headache at different stages?
In future, similar studies in larger groups may help a better identification of pre-headache phases of TTH and migraine and the underlying pathophysiological mechanisms.
Footnotes
Acknowledgements
We are grateful to Professor Dr Mustafa Ertas for his important contributions.
