Abstract
Migraine is a complex neurobiological disorder characterized by diverse phenotypes and unpredictable therapy outcomes. Digital phenotyping (DP), defined as the real-time collection of behavioral and physiological data in natural environments to identify individual phenotypes, represents a promising approach with the potential to enhance clinicians’ ability to identify migraine subtypes. Additionally, DP offers new insights into the intricate neurobiological and behavioral patterns, as well as environmental influences, associated with each phase of a migraine attack, including potential triggers, pre-attack symptoms, the characteristics of an attack and response to treatment. Moreover, a DP-based approach has the potential to revolutionize migraine research and clinical trials by enabling more personalized, patient-centred diagnostics and tailored acute and preventive treatments. Despite the limited literature available and the heterogeneity of study designs, migraine DP may lay the groundwork for future digital twin models and the discovery of digital biomarkers for diagnosis, therapy optimization and outcome evaluation. Furthermore, DP could serve as a predictive marker for migraine attacks, empowering patients to monitor their condition and adopt a proactive approach to treatment. Integrating DP into migraine studies could also contribute to the development of an updated international migraine classification that incorporates neurobiological and psychosocial factors alongside clinical symptomatology. To fully realize its potential in migraine research and care, experts should prioritize collaboration with artificial intelligence (AI) specialists, data scientists and medical engineers. Establishing a multidisciplinary ecosystem will be essential to developing robust and clinically meaningful DP tools for migraine research. This review aims to show the current landscape of both active and passive DP methodologies, which leverage smartphones, wearable biosensors and AI-driven analytics to capture real-time physiological, cognitive and environmental data, at the same time as pointing to the future ahead of migraine DP.
This is a visual representation of the abstract.
Introduction
Migraine is one of the most misdiagnosed, undertreated, and stigmatized disorders, affecting over one billion people worldwide and imposing a significant burden on individuals, families and society (1). Despite significant advancements in understanding its pathophysiology, migraine remains a complex and heterogeneous condition with considerable variability in symptoms, triggers and treatment responses among individuals (2). Conventional diagnostic methods rely solely on self-reported symptoms, which can be subjective and do not fully account for migraine's neurobiological and psychosocial complexity (3). Additionally, poor treatment outcomes likely arise from the high phenotypic variability of migraine, further complicating diagnosis and therapy. As a result, there is growing interest in leveraging digital phenotyping (DP) to improve migraine characterization and management (4–6).
DP refers to the real-time collection and analysis of behavioral and physiological data using digital devices, including smartphones, wearable technology and biosensors (7). It can provide insights into various factors, such as the environment (e.g. atmospheric pressure, exposure to environmental stressors like noise, bright light and temperature changes), lifestyle (identifying potential migraine triggers), mental state (e.g. tracking mood changes, assessing exposure to mental stress and quantifying cognitive burden during an attack) and physiological metrics (e.g. heart rate variability, blood glucose levels, electrolyte fluctuations and neural activity). By integrating these data points, DP captures a comprehensive picture of an individual's health, maximizing the potential of precision medicine (8). In migraine research, DP offers an unprecedented opportunity to objectively quantify symptom patterns, environmental influences, physiological and behavioral changes associated with attacks. This could lead to the identification of digital biomarkers and predictive markers, providing deeper insights into migraine pathophysiology and enabling more personalized treatment strategies (5).
This narrative review explores the current landscape of DP in migraine, focusing on its applications, challenges and future directions. Various digital tools, including wearable biosensors, mobile health applications and AI-driven analytics, are explored, highlighting their potential to revolutionize migraine diagnosis, monitoring and personalized treatment. Additionally, this review addresses the limitations and ethical considerations associated with integrating migraine DP into research and clinical practice.
Search strategy and selection criteria
The literature was reviewed with a structured search strategy. Search strategy combined information of two main terms (i.e. migraine and digital phenotyping), with possible variations). PubMed was searched for the terms, using database-specific variations, in the period between their inception and 31b March 2025. The search strings for PubMed were: (migraine[Title/Abstract] AND digital phenotyping[Title/Abstract] OR phenotyp*[Title/Abstract]) and (migraine[Title/Abstract] AND wearable[Title/Abstract] OR mHealth[Title/Abstract]). The first search string yielded 1271 studies. The second search string identified 12525 results. Publications were considered eligible for inclusion in this review if they were in the English language and related to DP of migraine patients. Additionally, other important publications, in our opinion, that were not identified through the search, were considered for this narrative review. Papers published in the last ten years were prioritized for this review article.
The DP concept in clinical neuroscience
DP is an emerging approach in clinical neuroscience that is at the forefront of mental health research (9). It leverages continuous, real-time data collection from digital devices to provide insights into disease patterns, symptom fluctuations and individualized treatment responses (10). This methodology captures behavioral, physiological and cognitive markers through passive and active monitoring as part of an evidence-based medicine strategy, offering a more objective and dynamic assessment of neurological disorders compared to conventional clinical evaluations (11).
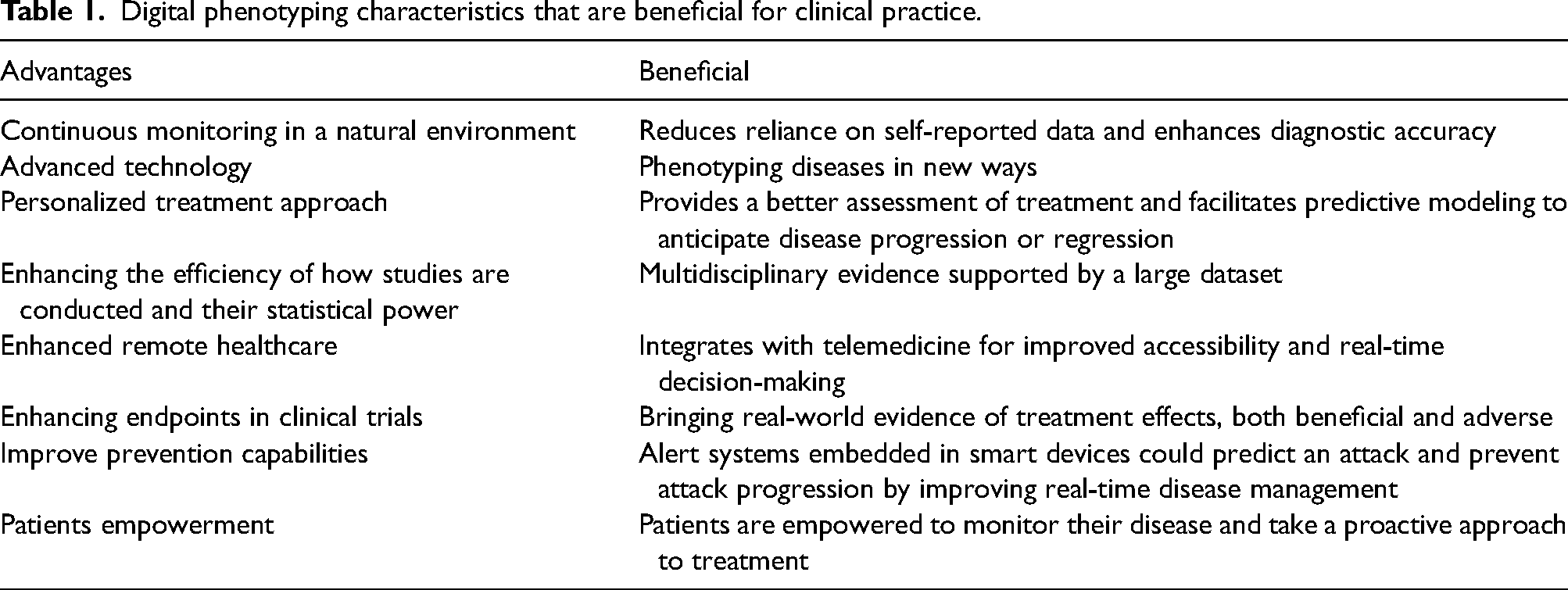
DP has the potential to revolutionize the diagnosis and monitoring of neurological conditions such as migraine (5), generalized anxiety disorder (12), schizophrenia (13), major depression (14), mood disorders (15), sleep disorders (16), epilepsy (17), Parkinson's disease (18), amyotrophic lateral sclerosis (19), and chronic low back pain (20) by integrating multimodal data sources. Moreover, smartphones, wearables and biosensors enable real-world, longitudinal tracking of disease manifestations, bridging the gap between clinic-based assessments and patients’ everyday experiences. This approach is particularly valuable for conditions characterized by episodic attacks or fluctuating symptoms, where intermittent clinical visits may fail to capture the full spectrum of disease activity (21). The advantages of DP over conventional clinical assessments are summarized in Table 1.
Digital phenotyping characteristics that are beneficial for clinical practice.
Data collected through DP methods are categorized into two types: (1) active data, which require participant engagement, typically collected via surveys and (2) passive data (e.g. those data collected by using wearable devices for recording neural activation and artificial intelligence (AI)-derived metrics), collected without any action from the participant (Figure 1) (22).
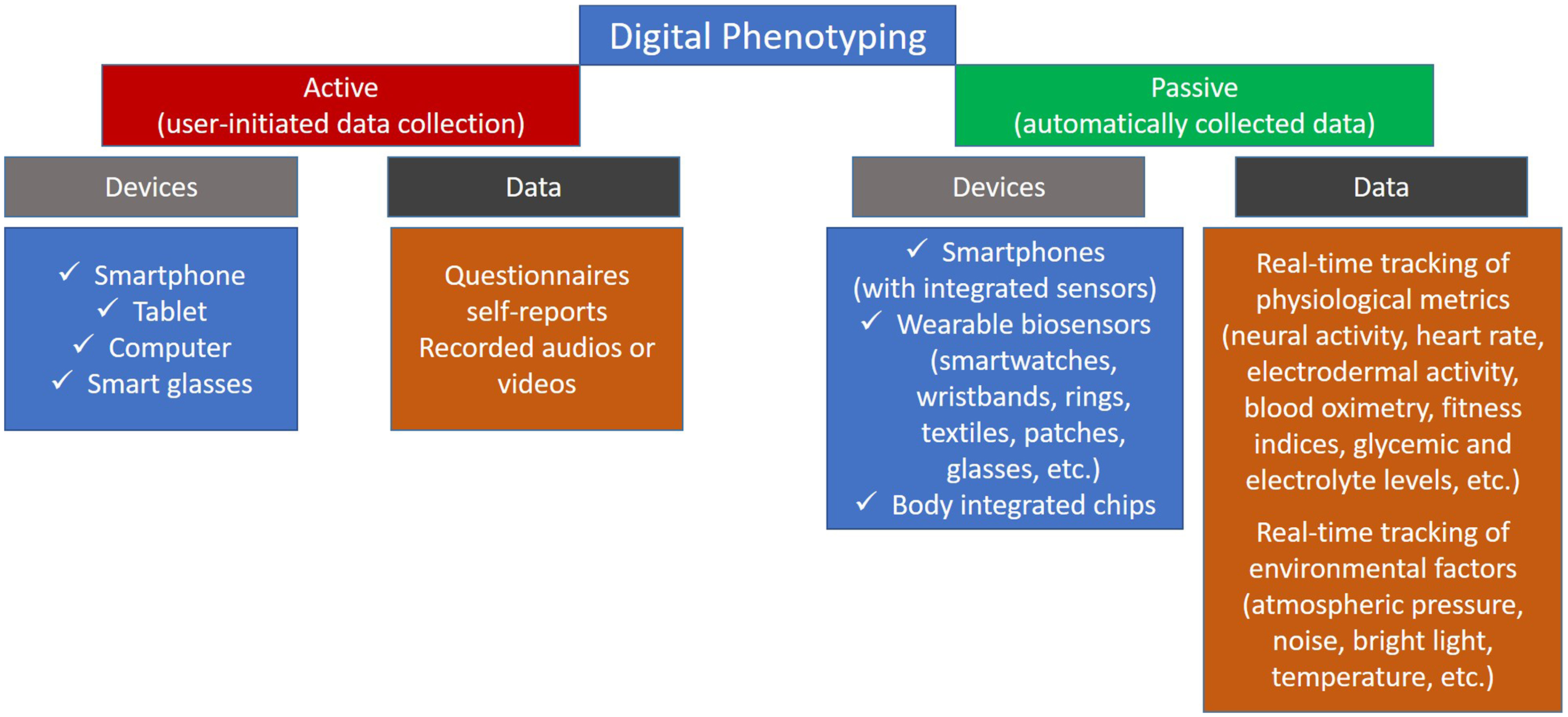
Examples of data collected by active and passive digital phenotyping tools.
Active DP
Active DP involves patient participation, including completing electronic diaries, performing cognitive tasks or providing symptom ratings via mobile health applications (23). This method enriches patient-reported data at the same time as complementing passive monitoring, enabling a more personalized diagnostic and therapeutic approach for clinicians (24). Additionally, these findings may support researchers and decision-makers in developing research strategies and promoting more standardized methodological studies.
It is worth noting that active DP is still an evolving field, with new digital health applications for neurological and mental disorders emerging daily in web-based spaces. Additionally, smartphones have become an integral part of daily life, providing 24/7 access to health applications without geographical limitations, making them particularly valuable for individuals in remote areas (25). Moreover, AI-based data analysis is advancing rapidly, enabling clinicians to leverage modern technologies for clinical decision-making and information exchange among colleagues involved in patient diagnosis and treatment (26). Furthermore, active DP offers patients new tools for self-understanding and behavioral insight, such as identifying daily patterns, recognizing how certain behaviors influence symptom progression and detecting attack triggers, comprising an approach that is particularly important for individuals with migraine. Therefore, DP could foster better collaboration among clinicians, patients and their environments at the same time as deepening the understanding of their complex interactions (9).
Passive DP
Passive DP involves the continuous, real-time collection of dense physiological, behavioral and environmental data without active user input (27). Wearable devices have advanced significantly in recent years, providing high-quality physiological data of clinical relevance that can be leveraged for passive DP (10). These devices now range from traditional wrist-worn monitors to patches, headbands, glasses and even implantable biosensors integrated into medical devices (28). Each of these devices is designed for a specific medical application, measuring particular biological or biochemical functions (29).
A National Institutes of Health working group concluded that wearables “herald a new era in healthcare delivery with the potential to transform many aspects of clinical care” (30). Moreover, wearable medical technologies are advancing faster than the healthcare systems that are beginning to adopt them. Recent breakthroughs in miniaturization, materials science and wireless communication have significantly improved the functionality, comfort and accessibility of wearable biosensors (29). Therefore, emerging devices and technologies should be continuously evaluated for their utility in DP, given the wealth of information they can provide (10).
One of the major challenges in the widespread adoption of wearable biosensors in healthcare is extracting meaningful, actionable health information from the large volume of data. However, this challenge can be addressed through AI-driven algorithms capable of automatically processing and interpreting data to generate evidence-based health insights (29). Additionally, for wearables to be effectively implemented in clinical practice, methods must be developed to integrate wearable-derived data into electronic health records meaningfully. This integration should be a priority because incorporating biosensing data into electronic health records can improve treatment outcomes. Furthermore, AI integrated into DP tools can support the development of predictive models that enable precise prognosis and more personalized therapeutic decisions (26). However, further research is needed to evaluate wearable biosensing's efficacy and external validity in extrapolating digital biomarkers for neurological disorders (31). Ultimately, passive DP has the potential to uncover novel connections between phenotypes and genetics (32).
Researchers and clinicians can advance data-driven precision medicine tailored to individual patients by applying passive DP to clinical neuroscience. As the field continues to evolve, further AI and machine learning integration will enhance its predictive capabilities, ultimately transforming neurological care.
Digital tools for migraine phenotyping
The diagnostic criteria for migraine in the International Classification of Headache Disorders, 3rd edition (ICHD-3) rely on patient-reported symptoms (33). which can be subjective and inconsistent. DP has the potential to enhance diagnostic accuracy by providing continuous, objective data on headache patterns and other migraine-related factors, including environmental triggers and physiological metrics across the prodrome, aura, headache and postdrome phases. Furthermore, DP in migraine holds promise for revolutionizing disease subtyping through multidimensional data analysis, enabling precision medicine approaches, and facilitating the discovery of novel digital biomarkers and treatment strategies. These advancements include AI-driven chatbot guidance to help patients avoid triggers, timely medication reminders to abort attacks, and even the identification of new drug targets for specific migraine subtypes when DP is integrated with neuroimaging and multiomics studies (3,5,34,35).
DP relies on a range of technological tools designed to capture real-time physiological and behavioral data, offering deeper insights into disease patterns, triggers and treatment responses. These tools include mobile health applications and wearable biosensors powered by AI-driven analytics.
Digital surveys for tracking migraine characteristics
The immense amount of data generated and collected through smartphones and personal devices presents an opportunity to obtain continuous, reliable insights into individual behavior (36) and to map the unique patterns of a migraine blueprint. Selecting an appropriate digital application for tracking migraine patterns is particularly important, as almost every migraine patient will eventually be encouraged to maintain a migraine diary (37). The adoption of electronic diaries, facilitated by these mobile applications, offers several advantages over traditional paper-based headache logs. Digital formats reduce the burden of manual record-keeping and minimize the risk of recall bias, allowing for more accurate and comprehensive data collection. Smartphone applications such as Migraine Buddy and N1-Headache are widely used because they offer a convenient and effective way for patients to track symptoms, potential triggers, medication use, and attack severity in real time, enhancing personalized management by connecting patients with their clinicians through a mobile application (38,39). Moreover, these applications have potential applications beyond personal tracking, including outcome measurement in clinical trials(40) and behavioral interventions as adjunctive treatments in migraine management (41,42). Beyond standard tracking applications, Ecological Momentary Assessment (EMA), a method of real-time surveying that evaluates symptom fluctuations in their environmental context, can serve as a valuable tool for assessing prodromal functioning in relation to the interictal state. EMA can capture data on physical symptoms, cognitive-affective functioning, external factors, therapy effects and symptom characterization in migraine patients (43–47). Another promising aspect of smartphone-based DP is the potential clinical value of metadata generated during data collection. For example, such metadata may provide insights into cognitive functions, including attention, processing speed and working memory (48).
Despite the significant advantages of migraine-tracking applications, such as the ability to collect large, longitudinal datasets, they still face limitations, including challenges in phenotypic clarity (38) and privacy concerns (49). Additionally, AI-driven pattern recognition for identifying personalized triggers and predicting attack likelihood is still evolving, and validation studies remain limited. Addressing these limitations in future applications will be essential for fully harnessing the potential of active DP and integrating it effectively with passive DP methods.
Wearable biosensors and physiological monitoring of tracking migraine characteristics
To select an appropriate device or, in some cases, multiple devices used in combination (Figure 2), clinicians and researchers must first determine which data will be most useful for addressing each intended purpose (11). Although multidomain wearable biosensors capable of capturing continuous, high-quality data may be ideal from a research perspective, several practical considerations and trade-offs must be accounted for in clinical settings. These include device comfort for long-term use, patient compliance, the need for calibration, storage requirements for large datasets, battery life and the overall availability of the technology. However, advancements in technology, computing and materials science have made wearable devices more compact, reliable, user-friendly and cost-effective (29,50). Additionally, although most wearable biosensors were initially limited to biophysical measurements, the next generation of devices now enables biochemical monitoring of sweat, saliva, tears, and interstitial fluid (51). This capability is particularly valuable in neurological diseases such as migraine, where these devices can serve as effective tools for identifying novel biomarkers (52).

Integration of multiple digital phenotyping tools, including neural activation, body movement, stress level, and environmental tracking, using real-time artificial intelligence (AI)-driven analytics (Edge AI and/or Cloud AI).
In the field of migraine, a wide range of wearable biosensing technologies holds great potential for passive DP (Table 2). Despite their promise to revolutionize migraine management, these digital tools remain largely underutilized in both research and clinical practice. To begin with, wearable biosensors could significantly enhance clinical trials in migraine by enabling continuous physiological and behavioral monitoring outside of clinical settings. Devices such as smartwatches and electroencephalography (EEG)-integrated headbands can record biometric data relevant to migraine pathophysiology (5). For example, the potential of combining somatosensory evoked potentials, psychometric assessment scores and machine learning could provide valuable information about episodic migraine progression toward migraine chronification (53). On the other hand, abnormal heart rate variability has been linked to autonomic dysfunction in chronic migraine patients (54), yet passive DP studies in this area are lacking. Furthermore, poor sleep quality and circadian rhythm disruptions are well-known migraine triggers (55); in such cases, wearable sleep monitors could aid in the DP of sleep patterns and migraine subtyping. One of the progresses in this field is the ability to monitor changes in biomedical signal patterns during the nights preceding migraine attacks (56,57) and forecast attacks based on preictal headache diary entries and simple physiological measurements using wearables (58). Furthermore, researchers who used wearable activity tracking devices and daily short questionnaires found that, on migraine days, patients slept more, had reduced physical activity and had lower maximum heart rates (59). In addition, Pagan et al.(60) created individual models for predicting migraine attacks based on data collected from a multi-sensor network and the N4SID state-space algorithm, achieving a positive predictive value of 100%. However, it is important to note that the prediction accuracy of photoplethysmographic and galvanic skin response sensors remains limited due to a relatively high false positive rate. As a result, other types of wearable biosensing, such as EEG devices, may be more suitable for migraine DP and attack forecasting (61). Additionally, wrist-worn actigraphy can effectively capture reduced movement during the ictal phase, reflecting attack severity (62), whereas ankle-worn actigraphy may help identify restless legs syndrome. All the above-mentioned represent real-world applications of DP in migraine, demonstrating the feasibility and translational potential of DP beyond theory. Further innovations in this field include microneedle-equipped patches that enable pain-free insertion into the skin, allowing real-time monitoring of lactate, glucose, cortisol, electrolytes and alcohol levels in interstitial fluid (58,63). These biosensors provide invaluable data for evaluating attack triggers and optimizing migraine management. Moreover, wearable biosensors for non-invasive, personalized reproductive hormone monitoring (64) may be particularly useful for DP in pure menstrual and menstrually related migraine. Passive DP could also facilitate a deeper understanding of the aura phase (65–67), offering valuable insights into the ictal phase and allowing comparisons with interictal findings in patients with varying levels of aura complexity (68–73).
Types of wearable biosensing potentially used in passive digital phenotyping of migraine.

DP = digital phenotyping; EEG = electroencephalography.
Mapping DP modalities to ictal migraine phases and aligning DP with ICHD-3 migraine classification
DP can be aligned with the temporal dynamics of a migraine attack. For example, passive DP using wrist-worn actigraphy and heart rate variability can detect early signs of physiological arousal during the prodrome phase (58). In addition, smart patches and biosensor chips may detect changes in calcitonin gene-related peptide, glucose and cortisol levels in interstitial fluid (58,63), potentially identifying the prodrome even before the onset of subjective symptoms. Such early detection could prompt patients to take abortive medication prior to the headache phase, thereby preventing or mitigating the migraine attack, as demonstrated in previous studies (74,75). EEG-based devices used during the aura phase can capture cortical spreading depression-like activity and perceptual distortions (76), offering valuable insights into aura complexity. These devices can also provide real-time alerts to help patients anticipate and prepare for upcoming symptoms. The headache phase can be assessed through a combination of self-reports (active DP) and galvanic skin response (passive DP). Passive DP may also help objectively quantify symptoms such as pain intensity, quality, location and duration, as well as associated features including photophobia, phonophobia, nausea and sensitivity to physical activity. In the postdrome phase, reduced movement and impaired cognition may be measurable via actigraphy and smartphone-based metadata (e.g. typing latency) (62). Table 3 illustrates how various DP modalities can be mapped to ictal migraine phases for optimal signal acquisition.
Digital phenotyping modalities in mapping ictal migraine phases.

DP = digital phenotyping; EEG = electroencephalography.
ICHD-3 provides clinical criteria to distinguish among migraine types, including episodic versus chronic migraine, and migraine with or without aura (33). DP can complement this classification by providing continuous physiological data, such as interictal autonomic instability or EEG markers of aura, which may enable the more precise differentiation of migraine phenotypes. For example, patients with migraine with aura may exhibit distinct cortical excitability patterns detectable through wearable EEG devices, whereas chronic migraine may be associated with prolonged reductions in movement and altered sleep rhythms, measurable via actigraphy and EEG wearables. In addition, active DP can comprehensively capture symptoms across all migraine subtypes, including hemiplegic, dysphasic, vestibular and menstrual-related variants. This could enhance diagnostic accuracy and support a more personalized treatment approach.
Technological, ethical and educational limitations in DP of migraine
Achieving the above-mentioned goals in migraine DP is currently hindered by a non-personalized, pseudo-innovative approach to developing new technologies, as well as limited educational opportunities for headache specialists regarding active and passive DP. Additionally, education should also be extended to those designing DP applications, helping them improve their designs to align with both migraine patients’ expectations and clinicians’ needs.
Beyond these challenges, DP raises ethical and data security concerns. However, clinical and research practices must evolve alongside advancements in wearable technology, ensuring innovation is not stifled at the same time as maximizing benefits for migraine patients. Privacy-by-default principles should be embedded into health information technology infrastructures, including pseudonymization and safeguards for data traceability and portability, setting a standard for both research and clinical practice (77,78). Furthermore, conducting transparent research and clinical trials, obtaining informed consent, maintaining continuous communication with patients regarding data collection and findings, and disseminating results to the public and policymakers will foster trust in DP methodologies (8).
In parallel, responsible data sharing, adhering to privacy, transparency, consent, accountability and fairness principles (77), should be encouraged, along with multicentric and multidisciplinary collaboration in both active and passive migraine DP. This will also help address broader ethical concerns such as equity, ensuring all patients have access to the best available migraine management. Additionally, the reliability of DP devices should be externally validated (10,79). It is important to emphasize that financial barriers remain a challenge even with the inclusion of wearables for DP (80) and may further exacerbate disparities if not addressed equitably (81). Therefore, ensuring insurance coverage or providing DP devices and tools will not only enhance equity, but also contribute to a more comprehensive understanding of migraine, leveraging a diverse range of digital data currently unavailable (10). This, in turn, will enable a precision medicine approach and therapy optimization, ultimately improving patient outcomes and reducing treatment costs.
It is also important to recognize that every step between data collection and AI-driven analysis introduces potential biases (82). Given that deep learning algorithms often function as “black boxes”, integrating explainable AI into DP devices is crucial to ensure transparency in decision-making (83). Additionally, educating headache specialists on how AI algorithms analyze collected data and generate insights will enable them to fully harness modern technology in addressing complex cases of migraine profiling and management. At the same time, it is essential to educate patients on how to use prescribed DP devices correctly to obtain reliable data. Moreover, guidance should be provided on how to interpret the presented data because some patients may experience increased anxiety upon seeing figures that deviate from the norm (9). Finally, DP should serve as a supplementary tool for delivering state-of-the-art migraine care rather than replacing patient-clinician communication.
Future perspectives
Advances in both active and passive DP (84) will drive the development of new recommendations for applications and devices that are more personalized for migraine patients. To accelerate this progress, there is an urgent need for headache specialists to engage in large-scale collaborations integrating DP to advance toward precision treatment in the field of migraine (5).
Large multicentric DP initiatives can provide detailed insights into migraine subtypes, their interplay with triggers, and real-time responses to acute and preventive treatments (85–87). These studies will deepen our understanding of migraine pathophysiology and pave the way for personalized treatment strategies. Additionally, they can contribute to the development of predictive models for migraine treatment in clinical settings (88), facilitating optimized therapy and faster response times, particularly for individuals with chronic migraine or severe, treatment-resistant attacks. Future research must also address the requirements for diverse data collection in migraine DP. Establishing standardized protocols for data collection, harmonization and machine learning-driven analysis should be a priority for international societies involved in migraine research and management (89).
Technological miniaturization will soon enable the integration of multiple biosensors onto a single chip embedded in the human body (90,91). These biosensors could target specific biomarkers, such as calcitonin gene-related peptide concentrations or particular changes in brain waveforms, allowing for rich AI-driven multiparametric analyses (5). Additionally, integrating multiple data sources from both diurnal and nocturnal recordings could enhance predictive accuracy and guide acute migraine treatment strategies (5). Furthermore, patient and clinician access to real-time, wirelessly transmitted data will be ensured through secure AI-edge solutions (92) and advanced blockchain-based standards (93), maintaining strict confidentiality of patient information. This technology will not only facilitate remote health monitoring, but also streamline the broader healthcare system. Integrating DP into telemedicine platforms will enable continuous tracking of symptom progression, treatment efficacy, tolerability and adherence, reducing the need for frequent in-person visits. This approach is especially beneficial for chronic migraine patients who require long-term treatment adjustments and close follow-up. Moreover, DP could take a proactive role in migraine management by alerting patients to impending attacks, allowing them to implement preventive measures such as avoiding specific triggers, modifying behavior (9), or initiating acute therapy before the full onset of an attack with a careful monitoring to avoid consequences like medication overuse headache (94). This proactive strategy has the potential to reduce migraine severity and frequency at the same time as decreasing hospital readmissions and associated healthcare costs.
The integration of DP into migraine research and clinical practice will also enable the development of extensive data repositories for “digital twins”. A digital twin is a novel concept in health research that aims to create a virtual replica of a patient to optimize real-time treatment decisions (95). In this context, each new migraine patient could be assigned a digital twin, modeled after the characteristics of their closest cluster group (Figure 3), using AI-driven platforms that learn from both personal DP data and population-level data. This would enable individualized treatment optimization, ensuring that patients receive therapies based on their predicted response and prognosis (96). Consequently, precision medicine will allow clinicians to tailor decision-making and stratify migraine patients according to their likelihood of treatment response (5,97). Ultimately, these advancements will usher in an era of true precision medicine and patient-centred care, augmenting the capabilities of healthcare professionals at the same time as empowering patients to take an active role in managing their condition through DP-driven digital twins (8). However, the accuracy and reliability of digital twins depend critically on the quality of the data used to train these models. AI-powered wearable devices equipped with next-generation biosensors will be essential for ensuring the collection of high-quality, comprehensive data, further strengthening the digital twin approach. Additionally, digital twin decisions must remain explainable so that clinicians, patients and policymakers can understand the rationale behind AI-generated recommendations.
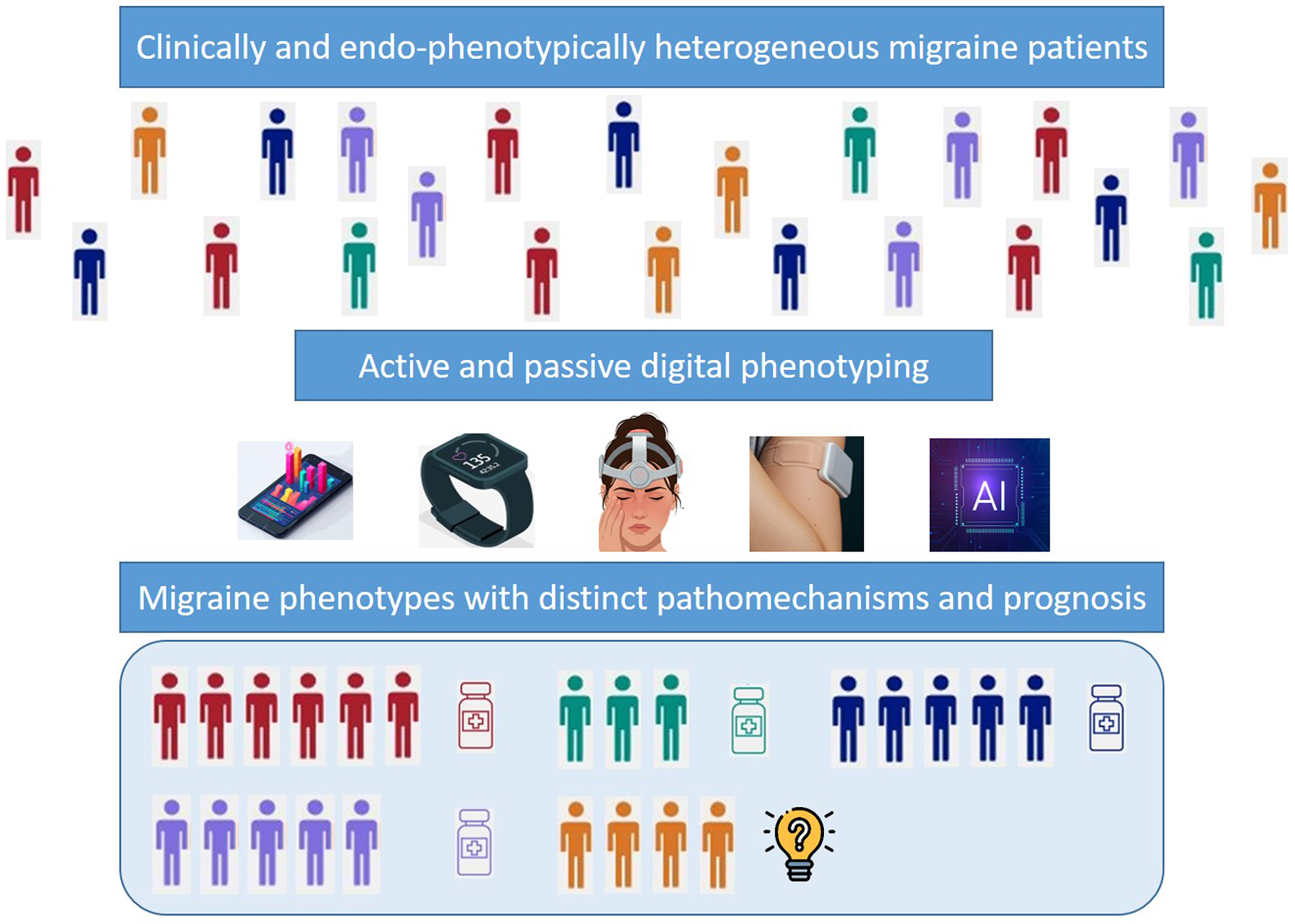
Schematic representation of digital phenotyping tools integrated with digital twin models to achieve precision medicine in migraine management. This approach also enables the identification of patients with poor treatment prognosis, allowing for early treatment optimization and the introduction of innovative therapies.
One of the key areas where migraine DP is expected to drive significant advancements is clinical trials. The integration of smart technologies and AI-driven models specifically designed for migraine will enhance the validity of findings and ensure patient-centred outcomes. This approach will be particularly valuable in pragmatic trials, including head-to-head comparisons, evaluations of combination therapies, assessments of long-term treatment effectiveness, investigations of residual migraine burden and studies on the impact of preventive treatments in altering disease progression (98–100). Rather than relying on retrospective headache diaries, researchers will be able to utilize real-time digital biomarkers to assess the efficacy of acute treatments. Additionally, AI-powered analytics will improve patient stratification, ensuring that clinical trials target the most relevant populations and enhance the success rate of novel treatments. However, to fully realize these benefits, standardization and validation of digital biomarkers in migraine research must be prioritized.
Finally, AI-driven predictive models embedded in passive DP devices could serve a dual role in migraine management. For example, DP could be combined with personal AI multi-agents that each handle different aspects of patient health, providing a unified, proactive care approach, leading to an enhanced individualized approach and reshaping migraine management. Agentic AI represents the next stage of AI evolution, enabling systems to autonomously plan, make decisions, execute tasks and learn from past interactions (101). Furthermore, integrating biosensors with neuromodulation devices could lead to groundbreaking advancements in both acute and preventive migraine treatments. However, although digital therapeutics, such as personal AI agents for migraine management and wearable neuromodulatory devices with integrated biosensing, can be powerful, it is essential to ensure that these technologies meet key criteria before widespread adoption. Specifically, they must be: (1) evidence-based (developed based on clinical trial findings and tailored to both clinicians’ and patients’ needs, rather than being driven by wellness trends); (2) accessible (available to a broad patient population); (3) ethical (designed with strong ethical frameworks to protect patient privacy and autonomy); and (4) integrated into healthcare (recognized as part of comprehensive migraine management guidelines and used alongside other treatments). Although numerous technical and regulatory challenges remain (102), addressing these issues will be crucial for the successful adoption of wearable biosensors in migraine DP.
Conclusions
DP offers a modern approach to improving migraine management. To fully realize its potential, interdisciplinary collaboration among migraine experts, AI and data scientists, and medical engineers is essential. This collaboration will ensure the development of robust and clinically meaningful DP tools for migraine research. By integrating these digital tools into clinical trials and, subsequently, into clinical practice, researchers and clinicians can establish more precise, data-driven approaches to migraine management, enhancing early attack detection, personalizing acute and preventive therapy and ultimately improving patient outcomes. In the future, DP could also contribute to the development of a new international migraine classification system that incorporates neurobiological and psychosocial factors alongside clinical symptomatology.
In the meantime, international headache societies and funding agencies should prioritize research on migraine DP and explore how it can become a standard clinical tool in migraine management. Additionally, investing in DP education for early-career clinicians specializing in migraine research and treatment will be crucial in bridging the gap between DP's transformative potential and its current applications in research and clinical practice.
Public health relevance
DP is transforming migraine research by enabling continuous, objective data collection through wearable biosensing devices and smart applications. The integration of active and passive data sources provides comprehensive insights into migraine triggers, symptom patterns, and real-time treatment responses. AI-driven analytics embedded in devices for DP is paving the way for personalized, precision medicine approaches tailored to individual migraine phenotypes. Multidisciplinary collaboration among clinicians, data scientists and engineers is essential for advancing DP tools and optimizing migraine management.
Footnotes
Acknowledgments
IP is supported by the Ministry of Science, Technological Development and Innovation, Republic of Serbia (contract number: 451-03-136/2025-03/200146). Furthermore, Figures 2 and ![]() are partially generated with the Gemini 2.0 Flash tool to improve the visual quality of schemes.
are partially generated with the Gemini 2.0 Flash tool to improve the visual quality of schemes.
Declaration of conflicting interests
The author declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: IP serves as Head of Imaging Section of the SN Comprehensive Clinical Medicine journal and as the Guest Editor in The Journal of Headache and Pain.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Data availability
No datasets were generated or analyzed during the current study.
